Abstract
Sperm become most sensitive to cold shock when cooled from 37ºC to 5ºC at rates that are too fast or too slow; cold shock increases the susceptibility to oxidative damage due to its influence on reactive oxygen species (ROS) production ([1]. ROS are significant stress factors that are generated during cooling and low temperature storage, and may be a main cause of decreased motility and fertility upon warming. ROS have been shown to change cellular function through the disruption of the sperm plasma membrane and through damage to proteins and DNA. The objective of this study was to determine which cryopreservation rates result in the lowest degree of oxidative damage and greatest sperm quality. In the rhesus model it has not been determined whether suprazero cooling or subzero freezing rates causes a significant amount of ROS damage to sperm. Semen samples were collected from male rhesus macaques, washed, and resuspended in TEST-yolk cryopreservation buffer to 100 x 106 sperm/mL. Sperm were frozen in 0.5mL straws at four different combinations of suprazero and subzero rates. Three different suprazero rates were used between 22ºC and 0ºC: 0.5ºC/min (Slow), 45ºC/min (Medium), and 93ºC/min (Fast). These suprazero rates were used in combination with two different subzero rates for temperatures 0ºC to −110ºC: 42ºC/min (Medium) and 87ºC/min (Fast). The different freezing groups were as follows: Slow-Med (SM), Slow-Fast (SF), Med-Med (MM), and Fast-Fast (FF). Flow cytometry was used to detect lipid peroxidation (LPO), a result of ROS generation. Motility was evaluated using a computer assisted sperm motion analyzer. The MM and FF treated sperm had less viable (P < 0.0001) and motile sperm (P < 0.001) than the SM, SF, or fresh sperm. Sperm exposed to MM and FF treatments demonstrated significantly higher oxidative damage than SM, SF, or fresh sperm (P < 0.05). The SM and SF treated sperm showed decreased motility, membrane integrity, and LPO compared to fresh semen (P<0.001). Slow cooling from room temperature promotes higher membrane integrity and motility post thaw, compared to medium or fast cooling rates. Cells exposed to similar cooling rates with differing freezing rates were not different in motility and membrane integrity, whereas comparison of cells exposed to differing cooling rates with similar freezing rates indicated significant differences in motility, membrane integrity, and LPO. These data suggest that sperm quality appears to be more sensitive to the cooling, rather than freezing rate and highlight the role of the suprazero cooling rate in post thaw sperm quality.
Keywords: Rhesus, sperm, cryopreservation, lipid peroxidation
1. INTRODUCTION
Optimization of cryopreservation techniques is essential for genetic preservation due to large intra-species variability and the unavoidable occurrence of cryoinjury that leads to subfertility upon thawing. The cooling process alone disturbs normal chemical and physical cellular conditions, therefore upsetting homeostasis and metabolism. More specifically, cellular injuries due to low temperatures are a result of cold shock-induced osmotic and oxidative changes, which are consequences of thermotropic phase transitions of membranes [2, 3]. Osmotic and oxidative changes induce an upregulation of reactive oxygen species (ROS; [4, 5] and shift cellular oxidative balance in favor of pro-oxidant forces.
The rate of cooling is a major determining factor of cell survival, in which maximum survival occurs at intermediate rates that are not too fast or too slow [6, 7]. Sperm survival depends on appropriate interactions between cooling and freezing rates, and osmotic balance. Particularly, sperm survival relies on transfer of cells through thermotropic phase transitions using the most opportune cooling rate in order to allow for proper osmotic adjustments. Addition of cryoprotective agents (CPA), as well as the cooling process alone, disturbs osmotic balance. Alterations to the intra- and extracellular osmolyte concentrations result in a stimulation of mechanisms that regulate sperm cell volume and osmolarity, and this encourages a state of osmotic stress. Osmotic stress is attributed to differences in the relative permeabilities of CPA’s and water, as well as temperature dependence of these permeabilities [8–10]. These findings highlight the importance of cryopreservation rate in optimal cooling response of sperm.
Osmotic stress has been shown to trigger ROS production and increases likelihood of oxidative stress [11]. ROS, such as superoxide anion and hydrogen peroxide, are significant stress factors that are generated during semen cooling and cryopreservation, and may be a major cause of sublethal damage to sperm. ROS have been shown to disrupt the sperm plasma membrane, induce lipid peroxidation (LPO), alter proteins and DNA, and decrease motility and fertility. ROS production has been shown to be maximal at suprazero temperatures rather than below 0ºC, suggesting most of the oxidative stress endured by sperm occurs during suprazero cooling [12]. Therefore discovery of cooling rates that result in the least likelihood of a pro-oxidant condition is crucial to optimal sperm function post thaw.
Cryopreservation damage to sperm has not been well studied in a valuable non-human primate model. In the rhesus model, it has not been determined whether cellular oxidative balance is most sensitive to a deviation from optimal suprazero cooling or optimal subzero freezing rates. The objective of this study was to determine whether suboptimal cooling or freezing rates are more likely to lead to a disruption in oxidative balance and thus increase cellular damage and reduce sperm quality.
2. MATERIALS AND METHODS
2.1. Chemicals and Media
All chemicals were purchased from Sigma-Aldrich Co (St. Louis, MO) with the following exceptions: NaCl (Fisher, Fair Lawn, NJ, USA) and KCl (Fisher). BODIPY581/591 C11 (4,4-difluro-5-(4-phenyl-1,3-butadienyl)-4-bora-3a,4a-dia- za-s-indacene-3-undecanoic acid) and propidium iodide (PI) was provided by Invitrogen (Carlsbad, CA). 0.5mL semen straws were provided by IMV Technologies (Maple Grove, MN, USA).
A Biggers, Whitten, and Whittingham (BWW) medium with 21mM HEPES buffer and 4mM sodium bicarbonate was used for sperm collections. This media consisted of 89.83mM NaCl, 4.78mM KCl, 1.19mM MgSO4·7H2O, 1.19mM KH2PO4, 1.7mM CaCl2·2H2O, 5.55mM glucose, 0.25mM sodium pyruvate, 11%(v/v) DL-Lactic acid syrup, 21.5mM sodium lactate, and 1% Penicillin/Streptomycin. A TEST-Yolk medium was used for rhesus semen cryopreservation and consisted of 4.325g of TES, 1.27 g of Tris, and 1g dextrose in 100 mL of distilled water with 20% egg yolk, with a pH of 7.4 and 350mOsm/kg [13].
2.2. Animal Handling, Semen Collection, and Sperm Preparation
Male rhesus monkeys (Macaca mulatta) were individually housed at the California National Primate Research Center, maintained in accordance with the NIH Guide for the Care and Use of Laboratory Animals, and under the approval of the University of California, Davis, Animal Care and Use Committee. Semen samples were obtained by electroejaculation from three males under chair restraint as described previously [14]. Semen was collected into a 50mL centrifuge tube containing 5 mL of BWW medium containing 1 mg/mL polyvinylalcohol (BWW/PVA). Following collection, semen samples were further diluted in 5 mL BWW/PVA, the coagulum was removed, and the suspension was evaluated for initial motility and sperm concentration.
2.3. Sperm Motility Evaluation
Fresh, pre-freeze, and post thaw sperm motility characteristics were measured with computer-assisted sperm analysis (CASA) using HTM Ceros (Version 12.2 g; Hamilton Thorne Biosciences, Beverly, MA, USA). At least 200 cells in a minimum of five fields were evaluated on a pre-warmed slide. Slides were maintained at 37ºC through use of a heated slide holder (Hamiliton Thorne Research, Beverly, MA, USA). The following instrument settings were used for CASA analysis: frame rate, 60 Hz; frames acquired, 30; minimum contrast, 80; minimum cell size, 4 pixels; static VAP cutoff, 20 μm/s; static VSL cutoff, 10 μm/s; progressive VAP threshold, 25 μm/s; progressive STR threshold, 80%; static intensity limits, 0.6 – 1.4; static size limits, 0.6 – 2.31; and static elongation limits 0–80. Percent total motility and percent progressive (forward) motility were determined.
2.4. Flow cytometry
Flow cytometry was performed using a FACScan flow cytometer (Becton Dickinson, Franklin Lakes, NJ. USA) with a 488 nm excitation laser. Emission measurements were made using 530/30 band pass (green/FL-1), 585/42 band pass (red/FL-2) and >670 long pass (far red/FL-3) filters. Forward scatter and side scatter measurements were taken to generate a scatter plot, which was used to gate for sperm cells only, excluding any larger contaminating cells. All data was acquired and analyzed using CellQuest Pro software (Becton Dickinson) with a total of 10,000 events collected per sample.
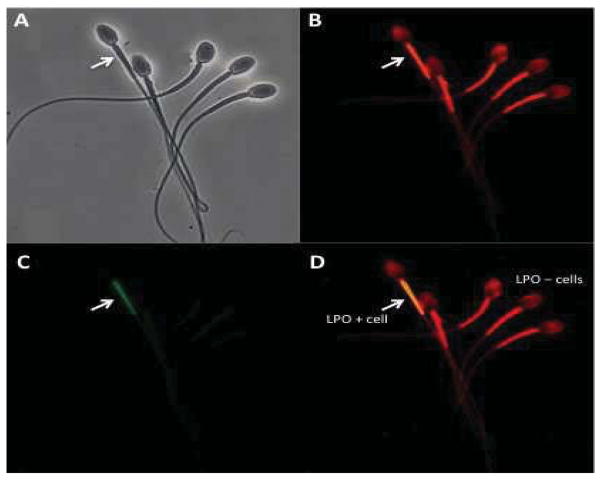
Lipid peroxidation (LPO) was determined using the lipid-based fluorophore BODIPY581/591 C11. BODIPY581/591 C11 readily integrates into biological membranes and reacts to free radical attack with a spectral emission shift from red to green, which can be monitored and quantified by flow cytometry. Red fluorescence represents overall incorporation of the non-oxidized probe into the cell; Green fluorescence represents oxidization of membrane-incorporated probe (Fig. 1).
Figure 1.
Representative fluorescent images of rhesus sperm labeled with membrane lipid probe, BODIPY581/591 C11. The images were obtained at 100x magnification in a Zeiss Axio Imager M1 microscope. (A) phase-contrast image of rhesus sperm. Red fluorescence represents overall incorporation of the non-oxidized probe into the cell (B and D); Green fluorescence represents oxidization of membrane incorporated probe (C); Merged images in which areas of LPO (lipid peroxidation) appear yellow (D). Arrows indicate one sperm with high LPO as distinguished from sperm with low oxidation state.
Post thaw sperm were washed once at 300xg for 8 min to remove cryopreservation medium, labeled with BODIPY581/591 C11 at 20 x 106 cells/mL at a final concentration of 5μM, and allowed to incubate for 30 min at 37ºC in BWW/PVA. The cells were washed once at 300xg for 8 min to remove excess probe and then treated with iron promoter (80μM ferrous sulphate) in BWW/PVA for 15 min. Immediately after incubation with fluorophore and iron promoter, labeled cells were measured. As it is possible for dead cells to undergo LPO, the membrane integrity probe PI was added 5 min prior to flow cytometry, so that dead lipid-peroxidized cells could be discerned from membrane-intact lipid-peroxidized cells. Cell Quest software was used to differentiate and quantify dot-plot sperm populations.
2.5. Cryopreservation
Cryopreservation of semen samples was performed by slow addition of glycerol-based cryoprotectant TEST-yolk medium with gentle continuous shaking. Semen was loaded into 0.5mL straws at 100 x 106 cells/mL and allowed a brief room temperature equilibration period (20–22ºC). A Styrofoam box was filled to a depth of 4 cm with liquid nitrogen (LN2) and a 10, 4 or 0.5 cm thick foam ‘raft’ was floated on top of the LN2 for 10 min. In preliminary studies, we determined the cooling rate at the top of each raft using thermocouples (Omega, Stamford, CT, USA) and Omegasoft® Temp Monitor analysis software (Omega) based on raft thickness and distance from the liquid vapor interface. Straws were then placed on top of the ‘raft’ and exposed to 4 different cooling-freezing rates: slow-medium (SM; 0.5ºC/min–45ºC/min), slow-fast (SF; 0.5ºC/min–93ºC/min), medium-medium (MM; 45ºC/min–42ºC/min), and fast-fast (FF; 93ºC/min–87ºC/min).
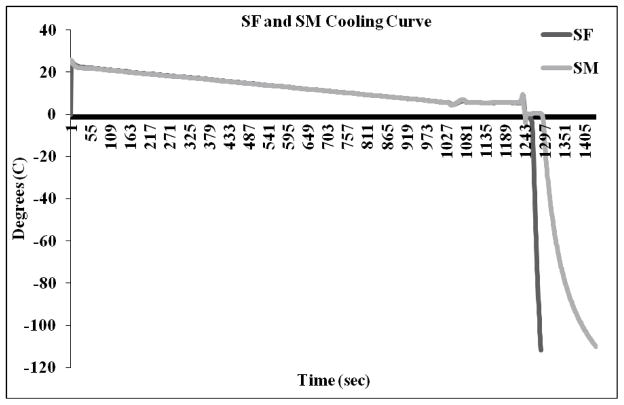
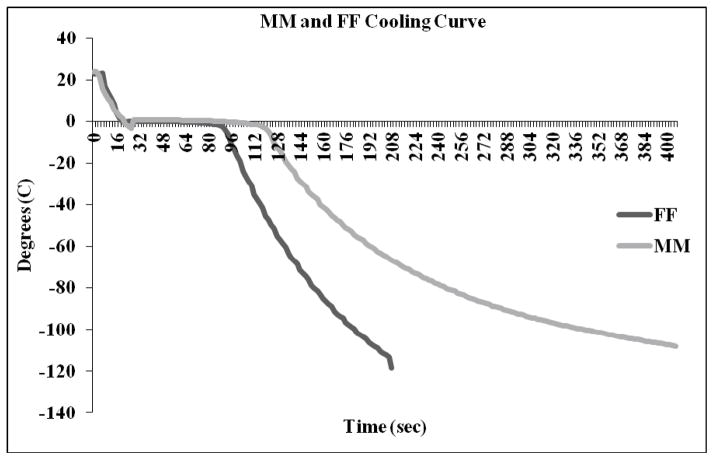
The Styrofoam box lid was replaced, the samples remained suspended in the vapor for 1 to 5 min until straws reached −110ºC, and then the straws were plunged into LN2 (−196ºC) for at least 15 min or longer before transfer to a large LN2 dewar for long term storage. Semen samples were kept at −196ºC for at least 24 hr before analysis. Upon sample evaluations, duplicate semen straws were thawed for 30 s in a 37ºC water bath and resuspended to 50 x106 cells/mL, in which motility and flow cytometric assessments were performed immediately after thawing. The cooling rate of straws while on the ‘raft’ was measured using a data logger thermocouple (Omega) with the monitor wire inserted into a 0.5-mL straw filled with cryopreservation medium; typical curves are shown in Figs. 2 and 3. Average rates of 0.5ºC, 42–45ºC, and 87–93ºC were achieved for the 10, 4, and 0.5 cm rafts, respectively.
Figure 2.
Thermocouple measurements of cooling and freezing rates of various foam raft holders floating on the surface of liquid nitrogen. The slow initial suprazero cooling rate combined with medium subzero rate (SM) averaged 0.5ºC/min above zero and 45ºC/min below zero. The slow initial suprazero cooling rate combined with fast subzero rate (SF) rate averaged 0.5ºC/min above zero and 93ºC/min below zero.
Figure 3.
Thermocouple measurements of cooling and freezing rates of various foam raft holders floating on the surface of liquid nitrogen. The medium initial suprazero cooling rate combined with medium subzero rate (MM) rate averaged 45ºC/min above and 42ºC/min below zero. The fast initial suprazero cooling rate combined with fast subzero rate (FF) rate averaged 93ºC/min above and 87ºC/min below zero.
2.6. Statistical analyses
This experiment was repeated three times each for three rhesus males. The effect of cryopreservation treatment was analyzed using one-way ANOVA and comparisons among treatment groups were made using a student’s t-test. Analysis of sperm quality parameters in response to sample day and male were insignificant and were removed from the overall model (n=9). Model fit was assessed using graphical analysis of residuals and a Shapiro-Wilk test for normality. Analysis was performed using JMP statistical software for Mac (SAS Institute, Cary, NC, USA) and a level of significance of P < 0.05 was used to assess significance.
3. RESULTS
3.1 Cooling rate mediates membrane integrity of cells post thaw
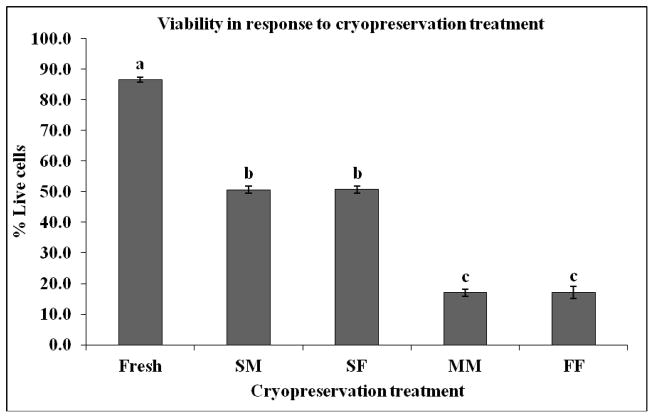
Overall, the percentage of membrane-intact sperm was reduced by 60.8% as a result of cryopreservation (P < 0.0001; Fig 4.). More specifically, the percentage of membrane-intact cells exposed to SM, SF, MM, and FF cryopreservation treatments was reduced from 86.5% fresh membrane-intact cells by 41.4%, 41.4%, 80.3%, 80.1% respectively. Slow supra zero cooling treatments resulted in approximately 50% membrane-intact cells, which was greater than 17% membrane-intact for medium and fast rates (P < 0.0001; Fig 5).
Figure 4.
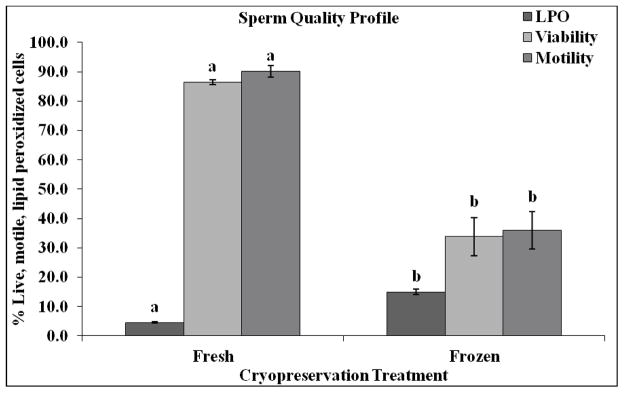
Comparison of rhesus sperm quality parameters between fresh and frozen sperm. Sperm were exposed to five different cryopreservation treatments: Fresh, slow initial suprazero cooling rate combined with medium subzero rate (SM), slow initial suprazero cooling rate combined with fast subzero rate (SF), medium initial suprazero cooling rate combined with medium subzero rate (MM), and fast initial suprazero cooling rate combined with fast subzero rate (FF). Bars bearing different superscripts differ significantly. Sperm cell quality in response to cryopreservation was different from fresh controls; frozen sperm demonstrated significantly different motility, membrane integrity, and LPO (P < 0.0001).
Figure 5.
Membrane integrity measured by propidium iodide (PI) exclusion for rhesus sperm exposed to five different cryopreservation treatments: Fresh, slow initial suprazero cooling rate combined with medium subzero rate (SM), slow initial suprazero cooling rate combined with fast subzero rate (SF), medium initial suprazero cooling rate combined with medium subzero rate (MM), and fast initial suprazero cooling rate combined with fast subzero rate (FF). Slow supra zero cooling resulted in more membrane-intact sperm cells compared to medium and fast rates. Cellular response to all cryopreservation treatments was different from fresh controls. Bars bearing different superscripts differ significantly (P < 0.0001).
The percentage of membrane-intact cells post thaw was greatly influenced by supra zero cooling rate rather than sub zero freezing rate, as membrane integrity of post-thaw sperm proved to be unaffected by the freezing rate used. Suprazero cooling demonstrated a treatment effect on sperm membrane integrity; exposure of cells to cooling treatments other than a slow rate resulted in a reduction in the percentage of membrane-intact cells post thaw (Fig 5).
3.2 Cooling rate determines motility post thaw
As expected, a significant loss of motility was observed between fresh and frozen samples, 59.9% total motility (TM) loss and 71.1% progressive motility (PM) loss in cryopreserved samples (P < 0.001; Fig. 6). Total motility was reduced from 90.1% in fresh controls to 36.1% in cryopreserved samples; PM was reduced from 61.4% in fresh controls to 17.7% in cryopreserved samples. Specifically, slow cooling from ambient to 5ºC resulted in approximately 55.8% TM and 29.6% PM after thaw, which was higher than the medium or fast cooling rates (P < 0.001; Fig. 7). Medium and fast cooling resulted in 23.3% and 16.6% TM, respectively (P = 0.28). Likewise, PM for medium and fast cooling rates were substantially decreased and resulted in 8.9% and 5.2%, respectively (P = 0.54).
Figure 6.
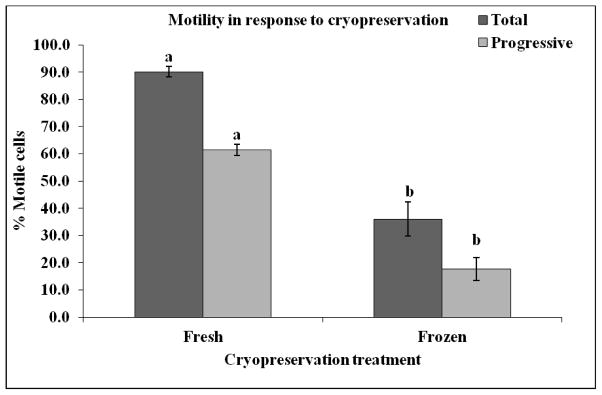
Comparison of total motility (TM) and progressive motility (PM) between fresh and frozen sperm. Sperm were exposed to five different cryopreservation treatments: Fresh, slow initial suprazero cooling rate combined with medium subzero rate (SM), slow initial suprazero cooling rate combined with fast subzero rate (SF), medium initial suprazero cooling rate combined with medium subzero rate (MM), and fast initial suprazero cooling rate combined with fast subzero rate (FF). All treatments except for fresh were grouped and subsequently averaged into a “frozen” category for analysis. Bars bearing different superscripts differ significantly; TM was different from PM across all treatments (P < 0.05) and frozen sperm demonstrated significantly reduced total and progressive motility compared to fresh semen (P < 0.0001).
Figure 7.
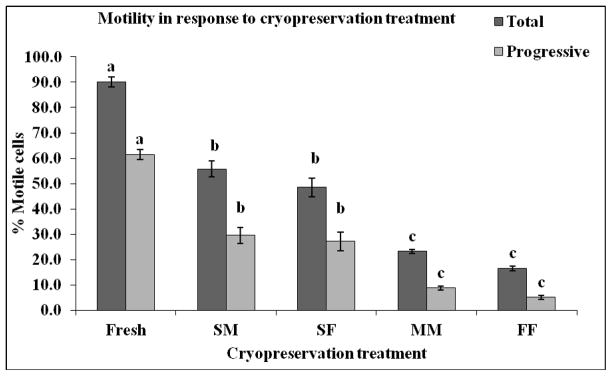
Total (TM) and progressive motility (PM) response of rhesus sperm exposed to five different cryopreservation treatments: Fresh, slow initial suprazero cooling rate combined with medium subzero rate (SM), slow initial suprazero cooling rate combined with fast subzero rate (SF), medium initial suprazero cooling rate combined with medium subzero rate (MM), and fast initial suprazero cooling rate combined with fast subzero rate (FF). Medium and fast supra zero cooling rates resulted in decreased TM and PM post thaw compared to slow treatments. All cryopreservation treatments were different from fresh controls (P < 0.0001); TM was different from PM across all treatments (P < 0.05). Bars bearing different superscripts differ significantly (P < 0.001).
Post thaw TM and PM were influenced by the suprazero cooling rate, rather than the subzero freezing rate (Fig. 7). Comparison of slow cooling treatments with differing freezing rates demonstrates little to no effect of the freezing rate on motility (Fig. 7). Exposure of cells to SM and SF cryopreservation treatments resulted in 55.8%/29.6% and 48.6%/27.2%, respectively for TM/PM characteristics. Comparison of treatments containing different cooling rates but similar freezing rates indicated a notable influence of cooling rate on sperm motility. The TM percentage of sperm in SM and MM was reduced from 90.1% fresh TM by 38% and 74.1%, respectively; TM percentage of sperm in SF and FF was reduced by 46% and 81.5%, respectively (Fig. 7). Deviation from slow cooling negatively influenced motility of rhesus sperm post thaw.
3.3. Cooling rate influences membrane lipid peroxidation
The dual-staining system utilizing BODIPY581/591 C11 plus PI identified sperm populations with lipid oxidation, as well as dead cell populations. Flow cytometric assessment of sperm incubated with previously mentioned fluorophores mirrored what was observed microscopically (Fig. 1).
Consequently, cryopreservation of sperm resulted in a 226% increase in oxidative membrane damage compared to their fresh counterparts (P < 0.001; Fig. 4). Sperm exposed to Fresh, SM, SF, MM, and FF treatments demonstrated 4.61%, 12.49%, 12.96%, 17.24%, and 17.43% membrane lipid peroxidation, respectively. LPO was most influenced by the suprazero cooling rate, not the subzero freezing rate (Fig. 8). Sperm exposed to treatments different in cooling rates but similar in freezing rates demonstrated differences in LPO compared to sperm exposed to the same cooling rates with differing freezing rates (P < 0.05; Fig. 8). Deviation from a slow cooling rate resulted in increased LPO, whereas a variation in subzero freezing rate demonstrated no differences in LPO response. These data suggest degree of oxidative stress is linked to variation in the suprazero cooling stage of cryopreservation (Fig 8).
Figure 8.
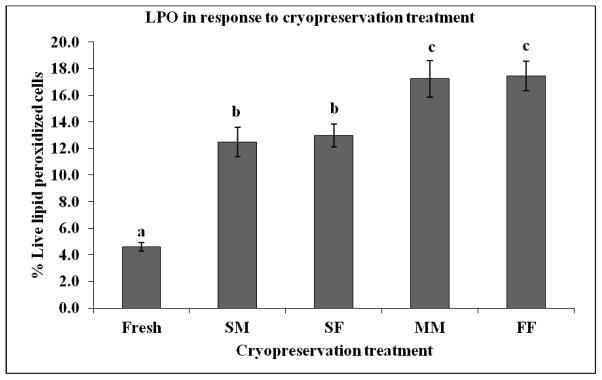
Lipid peroxidation (LPO) measured by BODIPY581/591 C11 for rhesus sperm exposed to five different cryopreservation treatments: Fresh, slow initial suprazero cooling rate combined with medium subzero rate (SM), slow initial suprazero cooling rate combined with fast subzero rate (SF), medium initial suprazero cooling rate combined with medium subzero rate (MM), and fast initial suprazero cooling rate combined with fast subzero rate (FF)using flow cytometery. Frozen sperm resulted in increased LPO compared to fresh controls (P<0.001). Slow supra zero cooling resulted in less LPO compared to medium or fast rates. Bars bearing different superscripts differ significantly (P < 0.05).
4. DISCUSSION
In this study, we evaluated the effects of different cryopreservation rates on sperm membrane integrity, motility, and membrane lipid peroxidation in the rhesus macaque model and whether the supra zero cooling or sub zero freezing stages of cryopreservation differed in their influence on post thaw quality of sperm. The results from this study confirm the previously reported negative effects of cryopreservation on sperm (Fig. 4; [15], and indicate relationships of membrane integrity, motility, and oxidative activity with the suprazero cooling stage of low temperature storage. In support of the current work, another report demonstrated a substantial portion of membrane changes after boar sperm cryopreservation may be attributed to cooling to 5ºC rather than to the freezing–thawing [16]. Membrane lipid phase changes as a result of cooling may be responsible for the strong influence of the suprazero cooling treatment on ROS activity and thus the increased oxidative damage seen in frozen versus fresh samples. In addition, rapid cooling through thermotropic phase transitions stimulates lipid loss from membranes [17, 18], and inappropriate lipid/protein reorganizations [19, 20] within the sperm membranes.
Others have reported on the effects of different cooling rates on sperm in many species including human [21–23] and rhesus monkey [24]. Particularly, rhesus monkey sperm are able to tolerate a wide range of cooling rates (5–400 ºC/min) in the presence of permeable CPA’s and egg yolk [24]. In the present study, it was determined that a slow cooling rate is optimal and necessary to maintain membrane integrity and motility, and limit oxidative damage to sperm. The 0.5ºC/min cooling rate optimized the freeze/thaw response of rhesus macaque sperm and thus allowed the cells to sufficiently dehydrate, evade high salt solution effects, and avoid intracellular ice formation. Cooling rates of 45ºC/min and 93ºC/min did not support the greatest cell survival and quality. However a similar, yet more intermediate, cooling rate of 17–34ºC/min has demonstrated optimal cryo-survival using a programmable freezer [25].
Optimal cryopreservation rates for any particular species, male, or ejaculate can vary depending on the CPA choice, freezing method, and thawing rate [24]. The present work indicates a positive interaction of slow cooling with glycerol, whereas the inverse was true for glycerol and medium or fast rates. Other CPA’s, like ethylene glycol, utilized in cryopreservation have proven effective for rhesus sperm exposed to slow and rapid cooling [24], which advocates further exploration of other potential cryopreservation rates with ethylene glycol and other CPA’s. Medium to fast cooling rates result in decreased sperm quality likely as a result of limited CPA exposure, insufficient dehydration and thus intracellular ice formation. A clearly optimal subzero rate was not indicated and therefore no obvious effect of freezing rate on post thaw quality of sperm. This observation supports the belief that sperm are relatively insensitive to the rate of cooling below zero [26]. It is probable that ROS activity in temperatures below zero is limited due to profound temperature depression into a sub-thermal condition not conducive to biological or molecular activity.
Motility is critical in facilitating sperm ascent of the female reproductive tract to the site of fertilization and also is required for fertilization [27]. Unfortunately, only about 60% of the motile population of a frozen ejaculate is recovered upon thawing (Fig. 7). Changes in plasma membrane structure and integrity appear to be an important component associated with reduced motility of frozen–thawed sperm [28]. Specifically, cold shock-induced modifications are characterized by metabolic damage, which may negatively influence motility [29], as well as non-specific bilayer faults [30, 31] that allow for inappropriate loss and gain of vital intracellular constituents. The latter may include but are not limited to calcium ions, nucleotides, antioxidants, and enzymes. Loss of adenine nucleotides depletes ATP stores and without ATP, motility ceases.
An intracellular calcium accumulation has been reported during low temperature storage and this influx can be attributed to modified plasma, mitochondrial, and nuclear membrane interactions that introduce leakage and ion pump dysfunction. Calcium is a major mediator of sperm function, in which Ca2+ ATPase pumps maintain low ion concentration to allow correct signaling by calcium influxes, which are needed to initiate motility, hyperactivation, capacitation, acrosome reaction, and therefore fertilization in vivo [32]. Cooling-induced calcium release triggered by compromised membranes may cause premature motility activation and subsequent depletion of ATP stores necessary to maintain motility post thaw. Intracellular calcium increase is also linked to loss of membrane integrity [32] and ROS production [33]. Based on our results, it is probable that slow transit through lipid phase transition temperatures achieved through slow cooling rates resulted in the least amount of membrane damage and thus leakage of cellular components essential to sperm function.
Modified membrane interactions due to cooling may also promote increased ROS production that readily increases the probability of LPO to the intrinsically unstable sperm membrane. The sperm quality profile (Fig. 4) indicates an inverse relationship between LPO and motility, as well as LPO and membrane integrity, suggesting that oxidative damage may be one of the causes of decreased sperm function upon thawing. LPO is most significantly seen along the sperm midpiece [34], and may be responsible for motility loss due to the proximity of the damage to energy-generating mitochondria. In addition to its negative effects on motility, ROS generation and peroxidation of sperm membrane can cause midpiece abnormalities, and reduce the ability of sperm–oocyte fusion [35]. Oxidative damage is caused by intra- and extracellular ROS production and this cooling-induced oxidative imbalance, like osmotic stress, has been linked to reduced post thaw membrane integrity, motility, and membrane integrity [36].
Due to a limited antioxidant reserve and polyunsaturated-rich membranes, sperm are susceptible to LPO during cryopreservation and upon thawing; lipid peroxidation causes considerable mechanical stress to cell membranes [37]. Increased LPO in cryopreserved sperm cells compared to fresh sperm has been demonstrated [38], as well as a sudden burst of nitric oxide and superoxide radicals upon thawing [15]. This ROS burst may be linked to increased LPO, not only due to their damaging nature individually, but also due to their ability to combine with each other to form highly reactive and damaging peroxynitrite (ONOO-). At low concentrations ONOO- induces capacitation [39], perhaps contributing to the “cryo-capacitated” state seen in cryopreserved sperm; at high concentrations, ONOO- excessively nitrosylates tyrosine residues, affecting sperm function [40].
In general, loss of motility and fertilizing ability of cryopreserved sperm can be attributed to faulty membranes and LPO of the plasma membrane [41, 42]. Most of the previously and specifically described consequences stem from membrane damages caused by cold shock-induced membrane phase changes and the natural susceptibility of sperm membranes to peroxidation.
5. Conclusions
The results presented here provide evidence for differences in oxidative activity and cell sensitivity between suprazero and subzero cryopreservation environments and highlight the influence of suprazero cooling rates on sperm membrane oxidative damage and post thaw cell quality. We also provide possible explanations for the decreased sperm cell quality post thaw. We suggest that optimization of cryopreservation methods in pre-freeze supra zero conditions may yield improvement in cryopreservation survival of sperm. Several cooling rates have been tested to cryopreserve sperm cells, but cryopreservation progress depends on a better cryobiological understanding of sperm cells that can inspire development of methods for effective membrane stabilization and protection.
Acknowledgments
This work was supported by the National Center for Research Resources (NCRR) by R01RR016581 to SM. The authors wish to thank Dr. Victoria Burruel for technical assistance.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Bucak MN, Atessahin A, Yuce A. Effect of anti-oxidants and oxidative stress parameters on ram semen after the freeze-thawing process. Small Rum Res. 2008;77:89. [Google Scholar]
- 2.Drobnis EZ, Crowe LM, Berger T, Anchordoguy TJ, Overstreet JW, Crowe JH. Cold shock damage is due to lipid phase transitions in cell membranes: a demonstration using sperm as a model. J Exp Zool. 1993;265:432–7. doi: 10.1002/jez.1402650413. [DOI] [PubMed] [Google Scholar]
- 3.Ricker JV, Linfor JJ, Delfino WJ, Kysar P, Scholtz EL, Tablin F, et al. Equine sperm membrane phase behavior: the effects of lipid-based cryoprotectants. Biol Reprod. 2006;74:359–65. doi: 10.1095/biolreprod.105.046185. [DOI] [PubMed] [Google Scholar]
- 4.McCarthy MJ, Meyers SA. Antioxidant treatment in the absence of exogenous lipids and proteins protects rhesus macaque sperm from cryopreservation-induced cell membrane damage. Theriogenol. 2011;76:168–76. doi: 10.1016/j.theriogenology.2011.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baumber J, Ball BA, Linfor JJ. Assessment of the cryopreservation of equine spermatozoa in the presence of enzyme scavengers and antioxidants. Am J Vet Res. 2005;66:772–9. doi: 10.2460/ajvr.2005.66.772. [DOI] [PubMed] [Google Scholar]
- 6.Aurich C. Factors affecting the plasma membrane function of cooled-stored stallion spermatozoa. Anim Reprod Sci. 2005;89:65–75. doi: 10.1016/j.anireprosci.2005.06.025. [DOI] [PubMed] [Google Scholar]
- 7.Sarason RL, VandeVoort CA, Mader DR, Overstreet JW. The use of nonmetal electrodes in electroejaculation of restrained but unanesthetized macaques. J Med Primatol. 1991;20:122–5. [PubMed] [Google Scholar]
- 8.Gilmore JA, Liu J, Gao DY, Critser JK. Determination of optimal cryoprotectants and procedures for their addition and removal from human spermatozoa. Hum Reprod. 1997;12:112–8. doi: 10.1093/humrep/12.1.112. [DOI] [PubMed] [Google Scholar]
- 9.Gilmore JA, Mcgann LE, Liu J, Gao DY, Peter AT, Kleinhans FW, et al. Effect of Cryoprotectant Solutes on Water Permeability of Human Spermatozoa. Biol Reprod. 1995;53:985–95. doi: 10.1095/biolreprod53.5.985. [DOI] [PubMed] [Google Scholar]
- 10.Gao DY, Mazur P, Kleinhans FW, Watson PF, Noiles EE, Critser JK. Glycerol Permeability of Human Spermatozoa and Its Activation-Energy. Cryobiol. 1992;29:657–67. doi: 10.1016/0011-2240(92)90068-d. [DOI] [PubMed] [Google Scholar]
- 11.McCarthy MJ, Baumber J, Kass PH, Meyers SA. Osmotic Stress Induces Oxidative Cell Damage to Rhesus Macaque Spermatozoa. Biol Reprod. 2010;82:644–51. doi: 10.1095/biolreprod.109.080507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang AW, Zhang H, Ikemoto I, Anderson DJ, Loughlin KR. Reactive oxygen species generation by seminal cells during cryopreservation. Urology. 1997;49:921–5. doi: 10.1016/s0090-4295(97)00070-8. [DOI] [PubMed] [Google Scholar]
- 13.Tollner TL, VandeVoort CA, Overstreet JW, Drobnis EZ. Cryopreservation of spermatozoa from cynomolgus monkeys (Macaca fascicularis) J Reprod Fertil. 1990;90:347–52. doi: 10.1530/jrf.0.0900347. [DOI] [PubMed] [Google Scholar]
- 14.Sarason RL, Vandevoort CA, Mader DR, Overstreet JW. The Use of Nonmetal Electrodes in Electroejaculation of Restrained but Unanesthetized Macaques. J Med Primatol. 1991;20:122–5. [PubMed] [Google Scholar]
- 15.Chatterjee S, Gagnon C. Production of reactive oxygen species by spermatozoa undergoing cooling, freezing, and thawing. Mol Reprod Dev. 2001;59:451–8. doi: 10.1002/mrd.1052. [DOI] [PubMed] [Google Scholar]
- 16.Kim S, Lee YJ, Kim YJ. Changes in sperm membrane and ROS following cryopreservation of liquid boar semen stored at 15 degrees C. Anim Reprod Sci. 2011;124:118–24. doi: 10.1016/j.anireprosci.2011.01.014. [DOI] [PubMed] [Google Scholar]
- 17.Darin-Bennett A, Poulos A, White IG. Effect of Cold Shock and Freeze-Thawing on Release of Phospholipids by Ram, Bull, and Boar Spermatozoa. Aust J Biol Sci. 1973;26:1409–20. doi: 10.1071/bi9731409. [DOI] [PubMed] [Google Scholar]
- 18.Watson PF. The roles of lipid and protein in the protection of ram spermatozoa at 5 degrees C by egg-yolk lipoprotein. J Reprod Fertil. 1981;62:483–92. doi: 10.1530/jrf.0.0620483. [DOI] [PubMed] [Google Scholar]
- 19.Amann RP. Cryopreservation of sperm. Encyclopedia Reprod. 1999;1:773–83. [Google Scholar]
- 20.Hammerstedt RH, Graham JK, Nolan JP. Cryopreservation of mammalian sperm: what we ask them to survive. J Androl. 1990;11:73–88. [PubMed] [Google Scholar]
- 21.Henry MA, Noiles EE, Gao DY, Mazur P, Critser JK. Cryopreservation of Human Spermatozoa. 4. The Effects of Cooling Rate and Warming Rate on the Maintenance of Motility, Plasma-Membrane Integrity, and Mitochondrial-Function. Fertil Steril. 1993;60:911–8. [PubMed] [Google Scholar]
- 22.Morris GJ. Rapidly cooled human sperm: no evidence of intracellular ice formation. Hum Reprod. 2006;21:2075–83. doi: 10.1093/humrep/del116. [DOI] [PubMed] [Google Scholar]
- 23.Morris GJ, Goodrich M, Acton E, Fonseca F. The high viscosity encountered during freezing in glycerol solutions: Effects on cryopreservation. Cryobiol. 2006;52:323–34. doi: 10.1016/j.cryobiol.2006.01.003. [DOI] [PubMed] [Google Scholar]
- 24.Dong QX, Hill D, VandeVoort CA. Interactions among pre-cooling, cryoprotectant, cooling, and thawing for sperm cryopreservation in rhesus monkeys. Cryobiol. 2009;59:268–74. doi: 10.1016/j.cryobiol.2009.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sumigama S, Meyers S. Cooling rate affects rhesus monkey sperm survival. J Med Primatol. 2012;41:278–83. doi: 10.1111/j.1600-0684.2012.00548.x. [DOI] [PubMed] [Google Scholar]
- 26.Benson JD, Woods EJ, Walters EM, Critser JK. The cryobiology of spermatozoa. Theriogenol. 2012;78:1682–99. doi: 10.1016/j.theriogenology.2012.06.007. [DOI] [PubMed] [Google Scholar]
- 27.Aitken RJ. Development of in vitro tests of human sperm function: A diagnostic tool and model system for toxicological analyses. Toxicology in vitro : an international journal published in association with BIBRA. 1990;4:560–9. doi: 10.1016/0887-2333(90)90116-b. [DOI] [PubMed] [Google Scholar]
- 28.Sion B, Janny L, Boucher D, Grizard G. Annexin V binding to plasma membrane predicts the quality of human cryopreserved spermatozoa. Int J Androl. 2004;27:108–14. doi: 10.1046/j.1365-2605.2003.00457.x. [DOI] [PubMed] [Google Scholar]
- 29.Parks JE, Arion JW, Foote RH. Lipids of plasma membrane and outer acrosomal membrane from bovine spermatozoa. Biol Reprod. 1987;37:1249–58. doi: 10.1095/biolreprod37.5.1249. [DOI] [PubMed] [Google Scholar]
- 30.De Leeuw FE, Chen HC, Colenbrander B, Verkleij AJ. Cold-induced ultrastructural changes in bull and boar sperm plasma membranes. Cryobiol. 1990;27:171–83. doi: 10.1016/0011-2240(90)90009-s. [DOI] [PubMed] [Google Scholar]
- 31.Holt WV, North RD. The Role of Membrane-Active Lipids in the Protection of Ram Spermatozoa during Cooling and Storage. Gamete Res. 1988;19:77–89. doi: 10.1002/mrd.1120190108. [DOI] [PubMed] [Google Scholar]
- 32.Guthrie HD, Welch GR, Theisen DD, Woods LC., 3rd Effects of hypothermic storage on intracellular calcium, reactive oxygen species formation, mitochondrial function, motility, and plasma membrane integrity in striped bass (Morone saxatilis) sperm. Theriogenol. 2011;75:951–61. doi: 10.1016/j.theriogenology.2010.10.037. [DOI] [PubMed] [Google Scholar]
- 33.Cadenas E, Boveris A. Enhancement of hydrogen peroxide formation by protophores and ionophores in antimycin-supplemented mitochondria. Biochem J. 1980;188:31–7. doi: 10.1042/bj1880031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Neild DM, Brouwers JFHM, Colenbrander B, Aguero A, Gadella BM. Lipid peroxide formation in relation to membrane stability of fresh and frozen thawed stallion spermatozoa. Mol Reprod Dev. 2005;72:230–8. doi: 10.1002/mrd.20322. [DOI] [PubMed] [Google Scholar]
- 35.Kim JG, Parthasarathy S. Oxidation and the spermatozoa. Seminars in Reproductive Endocrinology. 1998;16:235–9. doi: 10.1055/s-2007-1016283. [DOI] [PubMed] [Google Scholar]
- 36.Bansal AK, Bilaspuri GS. Impacts of oxidative stress and antioxidants on semen functions. Vet Med International 2010. 2010 doi: 10.4061/2011/686137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yousef MI, Abdallah GA, Kamel KI. Effect of ascorbic acid and Vitamin E supplementation on semen quality and biochemical parameters of male rabbits. Anim Reprod Sci. 2003;76:99–111. doi: 10.1016/s0378-4320(02)00226-9. [DOI] [PubMed] [Google Scholar]
- 38.Ferrusola CO, Fernandez LG, Morrell JM, Sandoval CS, Garcia BM, Rodriguez-Martinez H, et al. Lipid Peroxidation, Assessed with BODIPY-C11, Increases after Cryopreservation of Stallion Spermatozoa, is Stallion-dependent and is Related to Apoptotic-like Changes. Reprod Dom Anim. 2009;44:88. doi: 10.1530/REP-08-0484. [DOI] [PubMed] [Google Scholar]
- 39.Herrero MB, de Lamirande E, Gagnon C. Tyrosine nitration in human spermatozoa: a physiological function of peroxynitrite, the reaction product of nitric oxide and superoxide. Mol Human Reprod. 2001;7:913–21. doi: 10.1093/molehr/7.10.913. [DOI] [PubMed] [Google Scholar]
- 40.Vignini A, Nanetti L, Buldreghini E, Moroni C, Ricciardo-Lamonica G, Mantero F, et al. The production of peroxynitrite by human spermatozoa may affect sperm motility through the formation of protein nitrotyrosine. Fertil Steril. 2006;85:947–53. doi: 10.1016/j.fertnstert.2005.09.027. [DOI] [PubMed] [Google Scholar]
- 41.Aitken RJ, Wingate JK, De Iuliis GN, McLaughlin EA. Analysis of lipid peroxidation in human spermatozoa using BODIPY C-11. Mol Human Reprod. 2007;13:203–11. doi: 10.1093/molehr/gal119. [DOI] [PubMed] [Google Scholar]
- 42.Almeida J, Ball BA. Effect of alpha-tocopherol and tocopherol succinate on lipid peroxidation in equine spermatozoa. Anim Reprod Sci. 2005;87:321–37. doi: 10.1016/j.anireprosci.2004.12.004. [DOI] [PubMed] [Google Scholar]