Abstract
The effectiveness of noninvasive treatment for central nervous system (CNS) diseases is generally limited by the poor access of therapeutic agents into the CNS. Most CNS drugs cannot permeate into the brain parenchyma because of the blood-brain barrier (BBB), and overcoming this has become one of the most significant challenges in the development of CNS therapeutics. Rapid advances in nanotechnology have provided promising solutions to this challenge. This review discusses the latest applications of dendrimers in the treatment of CNS diseases with an emphasis on brain tumors. Dendrimer-mediated drug delivery, imaging, and diagnosis are also reviewed. The toxicity, biodistribution, and transport mechanisms in dendrimer-mediated delivery of CNS therapeutic agents bypassing or crossing the BBB are also discussed. Future directions and major challenges of dendrimer-mediated delivery of CNS therapeutic agents are included.
Keywords: Dendrimer, drug delivery, central nervous system, blood−brain barrier, brain targeting
Central nervous system (CNS) diseases or disorders currently represent 11% of the global burden of disease, which is expected to rise to 14% by 2020, becoming one of the largest and fastest growing areas of unmet medical need. Over 1.5 billion people worldwide, including over 100 million people in the U.S., are suffering from the CNS diseases.1−3 There are many types of CNS diseases, including Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, head trauma, brain cancer, epilepsy, and stroke. Among these, brain cancer has been given great attention because of its common association with life-threatening risks. To date, these CNS diseases are devastating, and the current treatments are inadequate and expensive.1 Fewer new drugs have been approved for CNS disorders than for other diseases because of their extended development periods, increased drug-development costs, and higher risk of clinical failure as well as the incomplete understanding of the pathophysiology and requirements for delivery to the CNS.4 One of the most important challenges is the difficulty in delivering drugs to the CNS because of the existence of peripheral barriers, particularly the blood–brain barrier (BBB). Most drugs, nucleic acids, protein imaging agents, and other macromolecules are restricted from entry to the brain by the BBB.5,6 About 98% of small-molecule drugs fail to cross the BBB, whereas no large-molecule drugs cross the BBB except for a few natural peptides and proteins, such as insulin, which are specifically designed to do so. Therefore, it is essential to find ways to improve the delivery of therapeutics to the CNS by bypassing or crossing the BBB in an efficient, safe, and site-specific manner.
Nanomedicine has shown great potential for the treatment of many CNS diseases. Nanomedicine is the biomedical and pharmaceutical application of nanotechnology for making nanocarriers of therapeutics and imaging agents, nanoelectronic biosensors, and nanodevices with nanostructures. A number of nanocarrier delivery systems, including dendrimers,7−10 liposomes,11,12 polymeric micelles,13,14 linear polymers,15,16 quantum dots,17,18 and iron oxide nanoparticles,19,20 have been developed and have demonstrated promising properties in CNS drug delivery. Among these, much attention has been paid to dendrimers because of their advantages, which include (1) the ability to maintain drug levels in a therapeutically desirable range, (2) increased half-life, (3) increased solubility, stability, and permeability of drugs, (4) the capability to deliver a variety of drugs, (5) reduced macrophage uptake, (6) targeting ability, (7) facile passage across biological barriers by transcytosis, (8) rapid cellular entry, (9) improved delivery efficiency, and (10) reduced side effects by targeted delivery.5,6,21 In particulate, this review focuses on dendrimer-mediated CNS drug-delivery systems, providing a basic understanding of dendrimer advances in brain-specific drug delivery, imaging, and diagnosis. The review is divided into four major parts. The first gives a background on the effect of the BBB on drug penetration into the CNS; the second focuses on dendrimers recently developed and engineered for brain-specific drug delivery, imaging, and diagnosis; the third addresses the biodistribution and toxicity of dendrimers; and the fourth discusses the major challenges and future directions of dendrimer-mediated CNS drug delivery.
Blood–Brain Barrier
The BBB is composed of the capillary endothelial cells that line the cerebral microvessels and surrounding perivascular elements, including the basal lamina, pericytes, astrocyte end-feet, and interneurons.22 The structure of the BBB has been well-characterized. First, the human brain contains approximately 100 billion capillaries, which possess a net surface area of approximately 20 m2.5,6 Despite this extremely large surface area, the permeability of many substances is low because of the tight anatomical structure of tight junctions. The brain capillary endothelial cells form tight junctions by the interaction of various transmembrane proteins. These tight junctions of the BBB separate the brain from the rest of the body, which effectively protects the brain from toxic substances.5 The tight junctions of the BBB also lead to transendothelial electrical resistance (TEER), which is responsible for the permeability of small ions through the tight junctions between brain capillary endothelial cells.23 The value of TEER is mainly dependent on the abundance and complexity of tight junctions between these cells, and it can be as high as 8000 Ω cm2.24 Second, the BBB possesses a wide array of efflux transporters and serves as an enzymatic barrier.25 The efflux transporters, such as P-glycoprotein and P-glycoprotein multiple drug resistance (MDR)-related protein and ATP-binding cassette (ABC), clear waste products from the brain to the blood and prevent the entry of potentially neurotoxic compounds from the blood to the brain.25,26 Enzymes such as γ-glutamyl transpeptidase (γ-GTP), alkaline phosphatase, and aromatic acid decarboxylase are often polarized between the luminal and abluminal membrane surface of brain endothelial cells and are capable of metabolizing drug and nutrients.27,28 Third, the brain capillary endothelial cells have a large amount of mitochondria and possess low pinocytotic activity, which can decrease the passage of antiretroviral molecules even further.5 Essentially, the CNS is anatomically sealed from the rest of the body. Only highly hydrophobic molecules with a molecular weight of less than a 400 to 500 Da can passively diffuse across the brain capillary endothelial cells,29 whereas more than 98% of low-molecular-weight and nearly 100% of high-molecule-weight drugs cannot passively cross the BBB.30
The BBB, however, does not completely hamper the passage of all molecules into the brain. To maintain proper brain function, the BBB allows the passage of necessary nutrients and endogenous components, including glucose, amino acids, essential fatty acids, vitamins, minerals, electrolytes, and even cells to the CNS.31 These nutrients and endogenous components are effectively transported into the brain by numerous saturatable transporters or channels expressed at the BBB. Indeed, the BBB serves as a regulatory membrane to maintain the homeostasis of the brain.
Knowledge of the properties of the BBB has inspired scientists to develop effective drug-delivery systems for the CNS. In fact, the transport of most drugs to the brain from the periphery attempts to follow the paracellular aqueous pathway, transcellular lipophilic pathway, transport proteins, receptor-mediated transcytosis, or adsorption-mediated transcytosis.2,5 Few CNS drugs can be structurally modified to mimic endogenous, normally impermeable molecules (e.g., glucose) to take advantage of their transport proteins at the BBB.32 Other CNS drugs including high-molecular-weight biological therapeutic peptides, proteins, and genes may enter the brain via nanoparticles as carriers. To date, adsorptive-mediated and receptor-mediated transcytosis are two major routes for nanoparticle delivery across the BBB.33,34 Adsorptive-mediated transcytosis relies on the potential for binding and uptake of cationic molecules to the luminal surface of the brain capillary endothelial cells followed by exocytosis at the abluminal surface. Cationic proteins (e.g., hexamethylenediamine) and cell-penetrating peptides (e.g., Tat-derived peptides and Syn-B vectors) are commonly used for brain drug delivery via adsorptive-mediated transcytosis.33,35 Receptor-mediated transcytosis has been attracting more attention because of its high specificity. The key for this approach is to identify a unique receptor that is specifically expressed by the CNS; theses include the transferrin,36,37 insulin,38−40 insulin-like growth factor,41 low-density lipoprotein receptor-related protein 1 and 2,42−44 and diphtheria toxin receptors,45,46 which are highly expressed by brain capillaries and are the most investigated receptors to deliver therapeutics to the brain via receptor-mediated transcytosis.
Dendrimers for CNS Drug Delivery, Imaging, and Diagnosis
Dendrimers
Dendrimers exhibit a highly branched, 3D architecture and comprise an initiator core, several interior layers composed of repeating units, and multiple active surface terminal groups. The branches and surface groups of dendrimers increase exponentially in number with the generation (G) of the dendrimers, whereas the diameter of dendrimers increases by about 1 nm with the generation, as shown in Figure 1.47,48 Dendrimers possess very low polydispersity and high functionality. The presence of numerous surface groups and a hydrophobic core allows for a high drug payload and multifunctionality.21,49 Dendrimers have been recognized to be one of the most versatile compositionally and structurally controlled nanoscale building blocks for drug delivery. Dendrimers possess the ability to facilitate the transport of therapeutics across various cell membranes or biological barriers via an endocytosis-mediated cellular internalization. The modulation of tight junction proteins such as occludin and actin is one of the main reasons for the enhancement of the intracellular uptake, but such an event is reversible depending on the concentration, generation, and surface charge of the dendrimers.50,51
Figure 1.
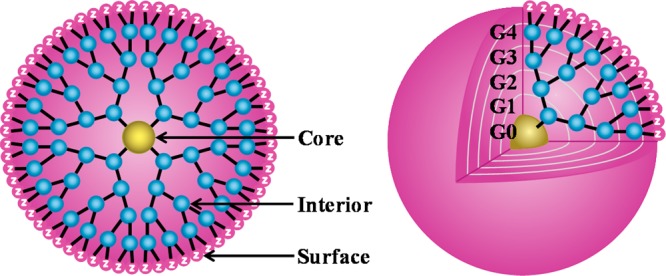
Schematic presentation of dendrimers as a nanoscaffold with a core, interior, and surface. Abbreviations: G, generation; Z, surface group for host–guest interactions and functionalization.
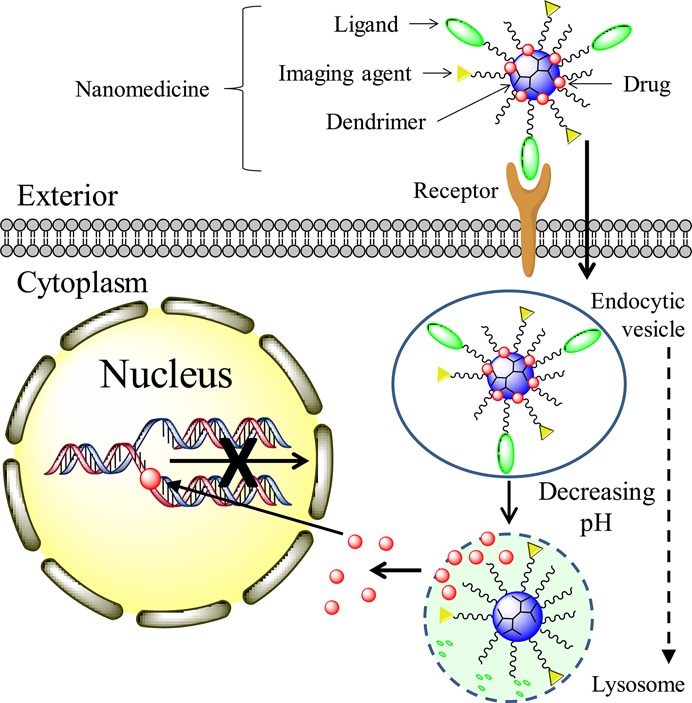
Surface groups and the molecular mass of dendrimers determine the dynamics of cellular entry.50 The mechanism of a dendrimer-mediated drug-delivery system is illustrated in Figure 2. The internalization of dendrimers occurs mainly through a clathrin- and caveolae-mediated energy-dependent endocytosis and partly through marcopinocytosis.52,53 Dendrimers can then function as a proton sponge to facilitate the escape from endosomes and lysosomes. The proton sponge mechanism occurs because dendrimers contain a large number of secondary and tertiary amines with a pKa at or below physiological pH. These amines enable the adsorbtion of protons released from ATPase and subsequently cause osmotic swelling and rupture of the endosome membrane to release the entrapped dendrimers.54
Figure 2.
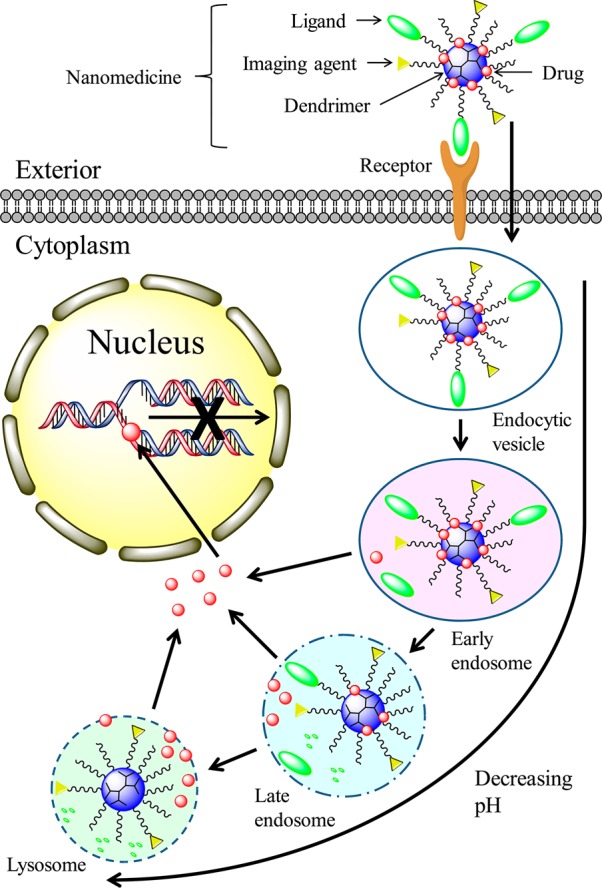
Mechanism of dendrimer intracellular delivery of therapeutics such as DOX and CPT. (1) Dendrimer nanomedicine is attracted to the cells by an electrostatic difference; (2) Ligand–receptor-mediated endocytosis occurs, and dendrimer nanomedicine is internalized into the cells; (3) reduction of the pH value from the endocytic vesicle to the lysosome triggers therapeutics to be cleaved from the dendrimer carrier and released into the cytoplasm; (5) released therapeutics diffuse into the nucleus, intercalate the DNA strand, and break the DNA chain to prevent its replication.
A variety of compositionally differentiated dendrimers have been exploited extensively for drug delivery and imaging, including poly(aminoamine) (PAMAM), poly(etherhydroxylamine) (PEHAM), and poly(propyleneimine) (PPI) dendrimers.21 Among these, PAMAM dendrimers have been the most investigated because of their unique structures and properties.50 A full-generation PAMAM dendrimer is a polycationic dendrimer that expresses primary amines on the surface, whereas a half-generation PAMAM dendrimer is a polyanionic dendrimer that expresses carboxylic acids on the surface. Polycationic dendrimers are able to form compacted polyplexes with nucleic acids at physiological pH, which can be used for gene therapeutics delivery,55,56 whereas polyanionic dendrimers have multiple anionic charges that are ideal for cationic drugs or for reversible coordination to platinum complexes.57 Both polycationic and polyanionic dendrimers possess extremely high serosal transfer rates, but polyanionic dendrimers are generally less toxic than polycationic dendrimers.58 Table 1 summarizes the key studies and findings related to the use of both full- and half-generation PAMAM dendrimers for CNS delivery of therapeutics. These findings suggest that favorable drug pharmacokinetics and high efficacy have been achieved using these dendrimer nanocarriers.
Table 1. List of PAMAM Dendrimers Used for CNS Drug Delivery.
| generation | linkage | drugs delivered | ligand(s) | major findings and comments | ref |
|---|---|---|---|---|---|
| G4 | MAL-PEG5000-NHS | doxorubicin | transferrin (Tf) and wheat germ agglutinin (WGA) | reduced nonspecific uptake by the normal cells; enhanced transport across the BBB by 2–3-fold compared to free drug; completely breaks avascular C6 glioma spheroids in vitro | (59) |
| MAL-PEG2000-NHS | doxorubicin | transferrin (Tf) and tamosifen (TAM) | pH-triggered DOX release under weak acidic conditions; enhanced transport across the BBB; reduced tumor volume of the avascular C6 glioma spheroids in vitro | (10) | |
| NHS-PEG3400-MAL | pEGFP-N2 plasmid | lactoferrin (Lf) | enhanced brain uptake and transfection efficiency in vivo by 2.2- and 2.3-fold, respectively, compared to that of PAMAM-PEG-Tf | (60) | |
| triglycine (GGG) | Quantum dots (Qdots) and YFP siRNA plasmid | epidermal growth factor (EGF) | intracellularly localized in an EGFR-dependent manner with minimal stimulation of the EGFR signaling pathway; enhanced nucleic acid delivery compared to trans-IT | (73) | |
| G5 | NHS-PEG3400-MAL | pEGFP-N2 plasmid | rabies virus glycoprotein (RVG29) | enhanced in vivo biodistribution in the brain | (53) |
| NHS-PEG3400-MAL | pEGFP-N2 plasmid | transferrin (Tf) | ecnhanced brain uptake and brain transfection efficiency in vivo by 2.25- and 2-fold, respectively, compared to unmodified nanoparticles | (61) | |
| NHS-PEG3400-MAL | pEGFP-N2 plasmid | angiopep-2 | enhanced brain uptake and transfection efficiency in vivo compared to the unmodified nanoparticles | (52) | |
| NHS-PEG3500-MAL | pORF-TRAIL plasmid | angiopep-2 | enhanced in vivo biodistribution in the brain, especially the tumor site; extended the median survival time of brain-tumor-bearing mice from 49 to 61 days | (9) | |
| NHS-PEG3400-MAL | pORF-TRAIL plasmid | chlorotoxin (CTX) | enhanced in vivo biodistribution in the brain, especially for glioma; extended the median survival time of brain-tumor-bearing mice from 49 to 59.5 days | (8) | |
| G4.5 | NH2-PEG3400-MAL | opioid peptide DPDPE | transferrin receptor monoclonal antibody OX26 | enhanced permeability through buccal mucosa by multiple fold | (71) |
Dendrimers for CNS Drug Delivery
The synthesis of PAMAM dendrimer nanocarriers suitable for CNS delivery of therapeutics involves four steps, as illustrated in Figure 3.8−10,52,53,59−61 First, dendrimers are modified with spacers or linkages on the surface to improve biocompatibility, buffering capacity, half-life, and drug-release kinetics. Second, specific ligands are conjugated to the surface-modified dendrimers to target to the BBB or tumor and to facilitate transportation across the BBB. Third, drug or gene therapeutics are bioconjugated or complexed with the surfaced-modified dendrimers. Notice that steps 2 and 3 can be switched depending on the particular circumstances. Fourth, imaging agents can be covalently conjugated onto the dendrimer nanocarriers, which allows for the tracking of biodistribution and provides tools for in vivo imaging and diagnosis. The details of dendrimer nanocarriers are illustrated in the following sections.
Figure 3.
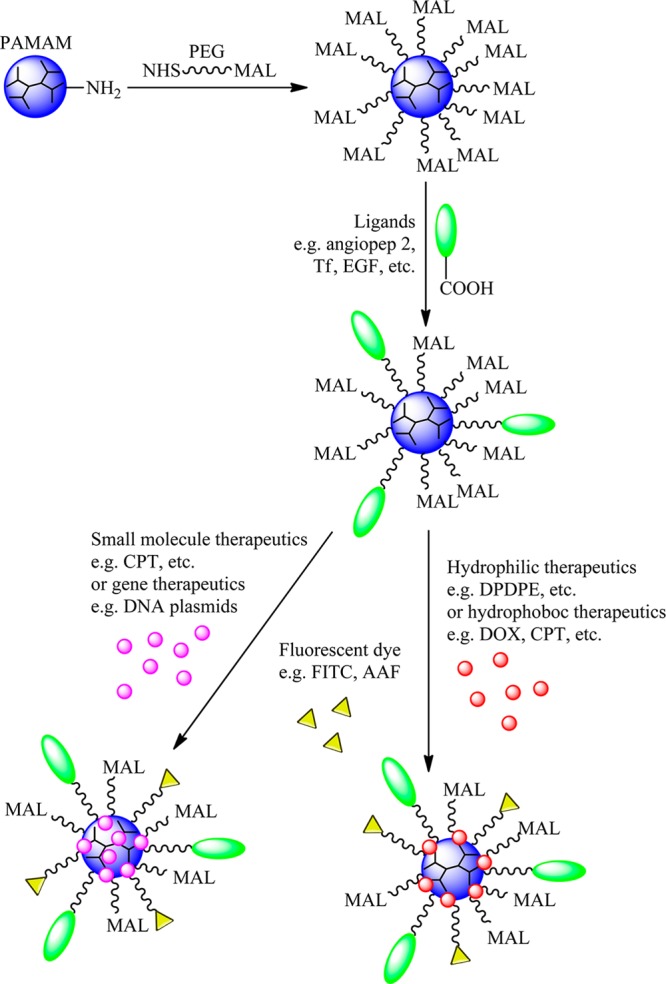
Dendrimer platform for CNS delivery of therapeutics and imaging reagents. (1) Full-generation PAMAM dendrimer is reacted with NHS-PEG-MAL to express MAL on the surface. (2) CNS ligands are specifically reacted with MAL of the dendrimer to form a PAMAM-PEG-ligand nanoparticle. (3) Hydrophobic or gene therapeutics can be entrapped inside of the dendrimer core via hydrophobicity or electrostatics. (4) Hydrophilic or hydrophobic therapeutics can also be covalent conjugated onto dendrimer surface. (5) Imaging reagents are reacted with MAL of the dendrimer to form a PAMAM-PEG-drug-ligands-imaging reagent nanoparticle. Abbreviation: NHS, N-hydroxysuccinimide; MAL, maleimide; PEG, poly(ethylene glycol); Tf, transferrin; EGF, epidermal growth factor; DOX, doxorubicin; FITC, fluorescein isothiocyanate; AAF, N-acetyl-2-aminofluorene; CPT, camptothecin.
Application of Spacers and Linkages to Dendrimer Nanocarriers
Step 1 is to introduce an appropriate spacer to the dendrimer. Ester and amide bonds are usually employed in the coupling reaction because such bonds can be hydrolyzed by endosomal or lysosomal enzymes inside cells.62,63 Poly(ethylene glycol) (PEG) is commonly introduced into the dendrimer structure to improve biocompatibility, reduce immunogenicity, increase water solubility, prolong half-life, and enhance structural stability.63,64 By applying functionalized N-hydroxysuccinimide-PEG-maleimide (NHS-PEG-MAL), the NHS groups can specifically react with the primary amine on the polycationic PAMAM dendrimer, and the MAL groups can specifically react with sulfhydryl groups from ligands under near neutral conditions to form stable thioether linkages. This approach has been widely explored for full-generation PAMAM dendrimers. The overall positive net charge provides buffering capacity to attract plasmids for gene delivery. However, the buffering capacity of the PEGylated dendrimer (PAMAM-PEG) can be impaired because of the occupation of the primary amine groups on the surface, which may result in unsatisfactory transfection efficiency.65 It has been reported that a stable bis-aryl hydrazone (BAH) can compensate for the loss of the amines on the surface and provide an additional site for protonation.66 BAH can serve as a linkage between PEG and the dendrimer to enhance the buffering capacity. Despite the probable beneficial effect of using BAH linkage, its ability to improve gene delivery to the CNS, if any, remains to be elucidated. Another sophisticated linkage using N-acetyl cysteine (NAC) has been employed to PAMAM dendrimers (PAMAM-NAC), which proved to be 16 times more efficacious than the drug alone. The carboxyl terminus of NAC was linked to the amino terminus of PAMAM dendrimers via a cleavable disulfide linkage using N-succinimidyl 3-(2-pyridyldithio)-propionate (SPDP). Two key features of this linkage are that (1) PAMAM-NAC possesses the ability to avoid plasma protein binding and thereby enhance bioavailability and (2) PAMAM-NAC enables rapid in vivo release of NAC in its active form using disulfide-exchange reactions with endogenous intracellular glutathione.67 It has been shown that PAMAM-NAC suppresses neuroinflammation and leads to a significant improvement in motor function in a rabbit model of cerebral palsy. The effectiveness of the PAMAM-NAC treatment, administered in the postnatal period for a prenatal insult, provides a window of opportunity for the treatment of cerebral palsy in humans after birth.68
Employment of Target Ligands with Dendrimer Nanocarriers
Step 2 is to couple specific BBB-targeting ligands onto PAMAM-PEG. The thiol groups of the ligands can specifically react with the terminal MAL groups of the PEGylated dendrimer, forming PAMAM-PEG-ligand. A variety of ligands have been developed for CNS-targeted dendrimer drug-delivery systems. A detailed description of the currently used ligands is given as follows.
Transferrin family ligands are well-known for brain targeting because of the high expression of transferrin receptor on brain capillary endothelial surfaces and many malignant cells.10,59,61 Lactoferrin (Lf), a single-chain iron-binding glycoprotein,60 HAIYPRH (T7), a transferrin receptor-specific peptide,69 and OX26, a monoclonal antibody against the transferrin receptor70,71 all belong to the transferrin family and have been explored for their ability to facilitate BBB transport. For instance, it has been shown that the brain capillary endothelial cells (BCECs) uptake of PAMAM-PEG-Lf was concentration-dependent manner. The brain uptake of PAMAM-PEG-Tf was 2.1-fold and PAMAM-PEG-Lf was 4.6-fold compared to that of PAMAM-PEG in Balb/c mice 2 h after intravenous administration.60
Wheat germ agglutinin (WGA), one of the lectins, is a newly discovered brain-targeting ligand, which possesses a strong affinity for the cerebral capillary endothelium and malignant tumor cells but a low affinity for normal tissues.59 For instance, WGA exhibited the highest binding strength and specificity in a study of the lectin-binding pattern of two typical BBB mimicking cells (human ECV304 and porcine brain microvascular endothelial cells) at the monolayer cell level.72
Tamoxifen (TAM), known as an estrogen receptor modulator, is able to reverse multidrug resistance (MDR) by directly inhibiting the action of the transmembrane P-170 glycoprotein pump to prevent drug efflux across the lipid bilayer component of membranes in human cells.10 It has been shown that the transportation ratios were 4.62% for PAMAM-PEG-DOX, 4.93% for PAMAM-PEG-DOX-Tf, and 6.06% for PAMAM-PEG-DOX-Tf-TAM, respectively, using an in vitro BBB model, suggesting that TAM could improve BBB transport.10
Epidermal growth factor (EGF) targets the epidermal growth factor receptor (EGFR), which is overexpressed in multiple human solid tumors, including cancers of the head and neck, lung, breast, colon, and brain. EGF and anti-EGFR antibodies such as Cetuximab have been utilized as targeting ligands to enhance selectively the cellular uptake of antitumor drug-delivery systems.73
Chlorotoxin (CTX) is a small 36 amino acid peptide purified from the venom of the giant Israeli scorpion (Leiurus quinquestriatus). CTX exhibits a high affinity for the membrane-bound matrix metalloproteinase-2 (MMP-2) endopeptidase, which is preferentially upregulated in gliomas, medulloblastomas, and other tumors of neuroectodermal origin.74,75 CTX possesses high specificity and binding affinity to the vast majority of glioma tumors, which has been used to target brain tumors.8
Rabies virus glycoprotein (RVG29) is a 29 amino acid peptide derived from rabies virus glycoprotein that has the ability to bind to nicotinic acetylcholine receptor (nAchR). The a7 subunit of nAchR is widely expressed in the brain including in capillary endothelial cells.53 RVG29 has been used as a brain-targeted ligand to deliver siRNA into the brain via nAchR-mediated transcytosis.76
Angiopep-2 targets low-density lipoprotein receptor-related protein (LRP), which is ubiquitously expressed in the CNS.77 Angiopep-2 has been shown to have an excellent ability to cross the BBB and to possess a higher brain transcytosis capacity and more parenchymal accumulation than transferrin, lactoferrin, extendin-4, avidin, and PYY, which was demonstrated using in situ brain perfusion in mice.9,52
Introduction of Therapeutics into Dendrimer Nanocarriers
Step 3 is to encapsulate or conjugate therapeutics to the PAMAM-PEG-ligands. Therapeutic drugs, such as doxorubicin (DOX) and camptothecin (CPT), are anticancer drugs that can intercalate the DNA strand and break the DNA chain to prevent it from being able to replicate. However, the use of these two drugs has been severely limited by their extremely low solubility in water and unexpected toxicity to normal healthy cells. Full-generation PAMAM dendrimers possess (1) large numbers of primary amines on their surface, which can interact electrostatically with the carboxyl group in the CPT molecules, (2) empty internal cavities, which can keep CPT molecules entrapped, and (3) tertiary amines in the internal cavities, which can interact with the atoms of the CPT-lactone molecules by forming hydrogen bonds. This PAMAM/CPT complex can significantly enhance the solubility and oral absorption of CPT.78,79
Other than passive hydrolysis or specific enzymatic release, more sophisticated strategies have been developed to improve the drug-release kinetics. The drug can be released from the carrier by pH-controlled release or light-controlled release. As a common approach, pH-controlled release can be employed to deliver and release drug to the tumor site effectively by incorporating an acid-sensitive linkage between the drugs and PAMAM dendrimers. It has been reported that the extracellular pH in most tumors (pH 6.5–7.2) is lower than in normal tissues. During the process of endocytosis, the pH in internalized vesicles drops gradually from 6.2 to 6.5 in the early endosomes to 5.5 in late endosomes and finally to 4.5 in lysosomes. Thus, acid-triggered release can be achieved either in extracellular fluid around the tumor site or in lysosomes/endosomes.80,81 As a common acid-sensitive linkage, cis-aconityl linkage has been introduced to between DOX and PAMAM dendrimers.80−83 DOX-release kinetics from PAMAM-PEG-DOX conjugates followed an acid-triggered manner and increased with the increasing degree of PEGylation. Pharmacokinetic and biodistribution studies indicated that increasing the degree of PEGylation could reduce liver and splenic accumulation, prolong the circulation time, and enhance the tumor accumulation of the conjugates.83 PAMAM-PEG-DOX conjugates produced via a N-(β-maleimidopropionic acid) hydrazide (BMPH) linkage also exhibited a comparatively fast drug release under weak acidic conditions but maintained a stable state under physiological conditions.10 A pH-sensitive diblock copolymer of poly(methacryloyl sulfadimethoxine) (PSD) and poly(ethylene glycol) (PEG) modified by lactose (LA-PEG-b-PSD) can interact electrostatically with PAMAM dendrimer and DOX to form LA-PEG-b-PSD/PAMAM/DOX complexes. These complexes can distinguish a small difference in pH values and exhibit selective targeting and cytotoxicity against HepG2 cells in vitro.84 As another example, PAMAM-CPT conjugates produced via a succinic acid–glycine linkage displayed minimal release of CPT in PBS and cell culture media but effectively inhibited the growth of colorectal carcinoma cells.85
For light-controlled release, a photochemical internalization (PCI) was invented for site-specific delivery of membrane-impermeable macromolecules from endocytic vesicles into the cytosol.86 Photocleavable linkers, such as o-nitrobenzyl (ONB), coumarin, xanthene, and benzophenone, enable controlled drug release.87−89 The drug-release kinetics is determined by the light wavelength, exposure time, substitution patterns of the photolabile linkage core, and pH value of the media. For instance, PAMAM-methotrexate (MTX) conjugates produced via an ONB linkage could fully inhibit the growth of cancer cells in a UV-light-controlled manner.87
All of these strategies raise the possibility of controlling drug-release kinetics in tumor models, but their effects on the enhancement of drug delivery to the CNS remain to be elucidated. There are some issues that need to be clarified, including but not limited to the following. One needs to consider the limitation of these acid-sensitive linkages in drug delivery to the CNS. Tumors, including those of the brain, provide a relatively acidic microenvironment. Given such a low pH microenvironment, drugs can be rapidly released from the dendrimer carriers to the tumor site using these acid-sensitive linkages, leading to a higher chance of exposing the drug to the brain tumor. However, this approach may not be ideal for the treatment of other CNS diseases, such as neurodegenerative diseases, because the pH remains neutral in these diseases. This disadvantage of acid-sensitive linkages can be overcome using light-sensitive linkages, which provide a universal platform to release drug once light is applied. Nonetheless, such UV exposure could be an independent risk factor for the CNS. The overall safety of using a light-sensitive linkage in dendrimer drug delivery remains to be elucidated by further studies.
RNA interference (RNAi) is a post-transcriptional gene-silencing mechanism that offers selective and specific silencing of target genes.90 Over the past few years, RNAi has been rapidly developed as an effective therapeutic for cancer therapy. However, the bioavailability of RNAi therapeutics can be severely reduced by the following barriers. (1) RNAi encounters RNA-degrading ribonucleases present in the extracellular fluids during transport, (2) RNAi may be absorbed nonspecifically by the liver, accessible mucosal surfaces, or body cavities, (3) RNAi by itself has no specificity toward target cells, and (4) RNAi hardly enters the cell because of the presence of negative charges on the cell surface.50 Dendrimers are well-established as gene-delivery vehicles to address these barriers.56,91 Both PAMAM and PPI dendrimers possess cationic primary amine groups at the surface that can participate in the RNAi-binding process and increase the cellular uptake of RNAi by transforming the entire complex into nanoscale polyplexes.91
For instance, it has been reported that most glial cells express the agonist of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) receptor but undetectable levels of the antagonist of the TRAIL receptor, whereas normal cells express the antagonist of the TRAIL receptor.92 Therefore, TRAIL can rapidly induce apoptosis only in tumorigenic or transformed cells but not in normal cells.93 The pORF-TRAIL plasmid was adopted as a therapeutic gene and delivered by PAMAM-PEG-ligand into the brain of mice to achieve a dual-targeting effect.8,9 A co-delivery system of DOX and pORF-TRAIL via PAMAM-PEG-T7 induced apoptosis of tumor cells in vitro and inhibited tumor growth in mice bearing Bel-7402 xenografts more efficiently, which led to a significant improvement of the antitumor effect.69 However, the efficacy of this co-delivery system in mice bearing brain tumors remains to be elucidated.
A triblock PAMAM-PEG-poly-l-lysine (PAMAM-PEG-PLL) nanocarrier was designed for the delivery of small-interfering RNA (siRNA).94 In this system, the tertiary amine groups in the PAMAM dendrimer work as a proton sponge for the endosomal escape and cytoplasmic delivery of siRNA. PEG renders stability against nuclease and protects siRNA in human plasma. PLL provides primary amines to form polyplexes with siRNA through electrostatic interactions and also to enhance the penetration. The PAMAM-PEG-PLL nanocarrier exhibited excellent stability in human plasma and effective enhancement of cellular uptake and transfection efficiency. Another interesting modular platform, dendriworm, was developed to deliver EGFR siRNA for brain cancer treatment.95 PAMAM dendrimer generation 4 with a cystamine core was first reduced to dendrons and then coupled to near-infrared-dye-labeled iron oxide nanoworms via a heterobifunctional linker, N-succinimidyl 3-(2-pyridyldithio)-propionate (SPDP). SPDP serves as a reducible linker for the rapid removal of dendrons from the conjugates in the reducing intracellular environment and improvement of siRNA delivery and diffusion inside the cell. The obtained dendriworms displayed a 16–25 mV zeta potential and a 80–110 nm hydrodynamic diameter. In vivo investigation suggested that dendriworms were able to diffuse into brain tumors, deliver siRNA into the tumor cells, and lead to significant gene silencing in vivo.95
Endosomal escape is partly determined by the uptake of dendrimers or dendriworms into endosomes, which is dependent on primary amines on the surface of dendrimers that are positively charged in serum. Addition of more negatively charged siRNAs neutralizes the positively charged primary amines and therefore reduces the number of dendrimers or dendriworms that can load into the endosomes. Addition of siRNAs may also affect the number of buffering amines per endosome available for inducing endosomal escape. Indeed, a higher feeding ratio of siRNAs to dendrimers or dendriworms can significantly reduce endosomal escape, resulting in the failure of siRNA delivery.95
In addition to their application in brain tumors, dendrimers have been intensively investigated in neurodegenerative diseases, especially in cerebral palsy and Alzheimer’s disease. After subarachnoid administration of fluorescein-labeled neutral PAMAM dendrimers generation 4 without target ligands in a rabbit model of cerebral palsy, these dendrimers were localized in inactivated microglia and astrocytes that are responsible for neuroinflammation. These dendrimers were even found in newborn rabbits with maternal inflammation-induced cerebral palsy, which was far from the site of injection.96 Such in vivo biodistribution of dendrimers can advance the targeted delivery of therapeutics in Alzheimer’s diseases.
According to the amyloid cascade hypothesis, amyloid peptide aggregation is closely related to the onset and development of Alzheimer’s disease. β-amyloid (Aβ) peptides play a central role in triggering neuronal damage in cerebral β-amyloidosis and most particularly in familial and sporadic Alzheimer’s disease. Because soluble Aβ peptide oligomers and intermediates in the assembly of Aβ fibrils are more neurotoxic than the end product of the fibrils, possible action against the toxic effects of amyloid peptides is to block the formation of the toxic oligomers, to disrupt their structure, or to lock the amyloid into nontoxic fibrils.
Cationic phosphorus-containing dendrimers of generations 3 and 4 were able to interact with the Aβ1–28 fragment and MAP-Tau protein aggregation processes.97 Twenty seven terminal morpholine groups decorating the gallic acid–triethylene glycol dendrimer (GATG-G3-Mor) significantly reduced the toxicity of Aβ, most likely by speeding up the process of fibril formation and then lowering the amount of prefibrillar forms in the system.98 PAMAM and phosphorus dendrimers have been shown to be able to modulate amyloid formation. Maltose-decorated PPI dendrimers of generations 4 (PPI-G4-Mal) and 5 (PPI-G5-Mal) possessed nontoxicity to PC12 and SH-5YSY neuroblastoma cells and had the capacity to interfere with Aβ1–40 fibrilization. Such interactions exhibited a generation dependence: PPI-G5-Mal blocked amyloid fibril formation, generating granular nonfibrillar amorphous aggregates, whereas PPI-G4-Mal generated clumped fibrils at low dendrimer–peptide ratios and amorphous aggregates at high ratios.99,100 Sialic acid-conjugated PAMAM dendrimers were able to attenuate Aβ toxicity at micromolar concentrations that were approximately 3 orders of magnitude lower than the soluble sialic acid.101,102 It has been reported that PAMAM dendrimers can perturb the aggregation of the peptides more than PPI dendrimers do.103
Labeling Dendrimer Nanocarriers
Step 4 is to label PAMAM-PEG-ligands-drug. To track the in vitro and in vivo distribution, PAMAM-PEG-ligands-drug can be further labeled by fluorescent dyes, including 4,4-difluoro-5,7-dimethyl-4-bora-3a,4a-diaza-s-indacene-3-propionic acid, sulfosuccinimidyl ester, sodium salt (BODIPY fluorophore),8 ethidium monoazide bromide (EMA),9,69 5-(aminoacetamido) fluorescein (AAF),71 and Quantum dots (Qdots).73
Dendrimers for CNS Imaging and Diagnosis
CNS imaging is an important tool for studying and monitoring the structure and functional changes in the brain and spinal cord. Advanced imaging can lead to a better understanding of the effect of cellular damage on CNS function and can help to improve the precision of neurological procedures.104,105 To date, surgical resection remains the main treatment for brain tumors. The success of brain tumor removal during surgery is highly dependent on the surgeon’s ability to differentiate tumor from normal tissues using subjective criteria that are not easily quantifiable. A recent interesting report revealed that Cy5-labeled free activatable cell-penetrating peptides (ACPPs) conjugated to PAMAM dendrimers delineated the margin between tumor and adjacent tissue and thereby improved the precision of tumor resection in mouse xenograft models.106,107 Because the brain is the most complex organ in human body, the precision of brain tumor resection becomes more crucial and relevant than ever. It is of interest and the utmost urgency to investigate further dendrimer application in brain tumor imaging. In addition, the complete excision of a malignant brain tumor is also challenged by its infiltrative nature. Contrast-enhanced magnetic resonance imaging (MRI) is widely used for defining brain tumors in the clinic, but its ability to be used in tumor visualization is limited by the transient circulation lifetime, nonspecificity, and poor BBB permeability of the commercially available MRI contrast agents. Interest in nanoparticles has grown for the development of new imaging and diagnostic agents for the assessment of brain function and diagnosis of CNS disorders and diseases using structural imaging techniques, such as MRI, computed tomography (CT), positron emission tomography (PET), magnetoencephalography (MEG), and optical imaging.105 Nanoparticles have been developed that have better body-compartment distribution and tissue targeting than standard contrast agents.108 Theoretically, nanoparticles used in drug delivery can be applied to deliver imaging agents and diagnostic molecules. Consequently, increasing attention has been paid to the development of nanoparticles that can fulfill multiple functions including therapy, imaging, and diagnosis.
In addition to the extensive studies of dendrimers in drug delivery, the development of dendrimer-mediated CNS imaging and diagnosis has attracted considerable attention.109,110 PAMAM-PEG-T7 was explored to deliver MRI contrast agents to liver and early brain glioma tumors.111 PAMAM-PEG-T7 was conjugated with diethylene triamine pentaacetic acid (DTPA) and further chelated gadolinium (Gd) to yield Gd-DTPA-PAMAM-PEG-T7. The MRI results showed that Gd-DTPA-PAMAM-PEG-T7 could selectively identify liver cancer but not early glioma, suggesting that this nanoscaled MRI contrast agent might allow for selective and efficient diagnosis of tumors without the natural barrier.111 To overcome this natural barrier, a two-component targeted nanoprobe was developed on the basis of PAMAM dendrimer (G5) labeled with MR/optical imaging reporters and tumor vasculature-targeted cyclic (RGDyK) peptides and angiopep-2 peptides.112 It has been reported that brain tumor cells highly express both αVβ3 integrin and LRP receptor.113,114 This nanoprobe first targets the αVβ3 integrin expressed in the tumor vasculature. Second, the increased local concentration of the nanoprobe facilitates the association between angiopep-2 peptides and LRP receptors on the vascular endothelial cells and further accelerates the BBB transversing of the nanoprobe via LRP receptor-mediated endocytosis. In vivo imaging studies illustrated that this nanoprobe could efficiently cross the intact BBB in normal mice and precisely delineate the boundary of the orthotopic U87MG human glioblastoma xenograft with a high target-to-background signal ratio.112 Another macromolecular MRI contrast agent was developed on the basis of dendrigraft poly-l-lysines (DGLs) modified with chlorotoxin (CTX) and a tumor-specific ligand and loaded with Gd-DTPA as a contrast agent. The MRI results showed that both signal intensity and duration were significantly enhanced in tumor-bearing nude mice treated with CTX-modified contrast agent compared to those treated with an unmodified counterpart and a commercial control.7
Ion imaging is another powerful methodology to assess fundamental biological processes in live cells. However, this approach is limited by the efficiency of some ion-sensing probes and the fast leakage from cells. A dendrimer-based nanoparticle was developed to achieve better intracellular retention of fluorescent probes and to perform prolonged fluorescence imaging of intracellular ion dynamics.115 A sodium dye, CoreNa Green, was encapsulated within a PEGylated PAMAM dendrimer (G5) to generate a sodium-sensitive nanoprobe. This nanoprobe is very stable and possesses high sodium sensitivity and selectivity. This nanoprobe homogenously filled the entire cell volume and remained for a long duration without detectable alterations of functional cellular properties when it was loaded in neurons in live brain tissue. The same principle can be applied to other existing fluorescent dyes, generating new applications for live fluorescent imaging.115
In addition to their straightforward assembly, dendrimer-based nanoprobes are promising for the noninvasive visualization of brain tumors with uncompromised BBB, providing the possibility for real-time optical-image-guided brain tumor resection during surgery. Thus, dendrimer-based nanoprobes could become an attractive tool for CNS imaging and diagnosis.
Toxicity and Biodistribution of Dendrimers
A systematic evaluation of dendrimer toxicokinetics regarding their adsorption, distribution, metabolism, and excretion is required to address the safety concerns associated with the use of those dendrimers clinically. A number of factors can potentially impact dendrimer toxicokinetics, such as their size, chemistry of the core, and surface property.58,116 Of special concern is the effect of these factors on the integrity of the BBB and on the acute or long-term toxicity to the CNS. The in vivo and clinical data of the toxic effects of dendrimers on the CNS are still scarce and hence call for considerable attention to study the systematic toxicity of dendrimers intended for brain-specific drug delivery, imaging, and diagnosis.
Generally, NH2-terminated dendrimers exhibit concentration- and usually generation-dependent cytotoxicity.58 In vitro cytotoxicity assessment indicated that the 90% inhibitory concentration (IC90) was 1 nM (∼7 ng/mL) for PAMAM dendrimer generation 3, 10 μM (∼280 μg/mL) for PAMAM dendrimer generation 5, and 100 nM (∼9 mg/mL) for PAMAM dendrimer generation 7.117 The order of polymer cytotoxicity was ranked as poly(l-lysine) (PLL) = polyethyleneimine (PEI) > dextran > PAMAM generation 3, according to lactate dehydrogenase (LDH) release, cell viability, and morphology evaluations in L929 mouse fibroblasts.118,119 Although the cytotoxicity of dendrimers is dependent on the generation, it is most strongly influenced by the nature of the dendrimer surface. It has been shown that anionic,120 PEGylated,120 OH-terminated,121 COOH-terminated,122 and melamine-based120 dendrimers are much less cytotoxic than NH2-terminated dendrimers because of shielding of the internal cationic charges by surface modification. However, surface modification of C12,123 lysine, and arginine124 on PAMAM dendrimer generation 4 led to an increased toxicity compared to native the PAMAM dendrimer because of increased hydrophobicity, charge density, and molecular weight. In addition, the cytotoxicity of dendrimers is also dependent on the chemistry of the core. It has been shown that PAMAM dendrimers can induce hemolytic activity in a generation-dependent manner. However, this hemolytic activity could be diminished by dendrimer surface modification with PEG.122 In fact, increasing the generation and surface modification with biocompatible terminal groups like PEG are widely used to reduce cytotoxicity of dendrimers.58
In brain tumor drug-delivery and imaging applications, drug or imaging contrast agents have to be delivered into the CNS before renal and hepatic clearance occurs. Thus, it is important to consider dendrimer biodistribution in the context of toxicokinetics. Biodistribution of parenterally administered dendrimers has been widely studied,125 particularly in relation to the development of dendrimer-based imaging agents and drug-delivery systems. Generally, smaller generation dendrimers are subjected to rapid renal elimination within 1 h. Dendrimers with charged (cationic or anionic) or hydrophobic surfaces are rapidly cleared from the circulation, particularly by the liver. Several strategies have been applied to prevent rapid clearance, including unique surface modifications and nanosize control. Dendrimers with a hydrophilic surface (e.g., −OH terminal or PEGylated dendrimers) possess the ability to escape rapid clearance.58,122 Therefore, in addition to their influence on drug-release patterns, surface or property modifications of dendrimers modulate their biodistribution, organ-specific location, circulation duration, renal clearance, and associated toxicity.
With the rapidly increasing interest in the use of dendrimers as vehicles for CNS drug delivery, a growing number of targeting ligands to the BBB or CNS have been explored as described earlier. The concept is appealing, and evidence of targeting is easy to establish in vitro, but it has proved more difficult in vivo. For instance, PEGylated PAMAM dendrimers of generation 5 attached with targeting ligands of chlorotoxin (CTX) (PAMAM-PEG-CTX) produced a significantly higher brain distribution after intravenous (i.v.) injection in vivo than native PAMAM dendrimers. However, the biodistribution study still showed a significant accumulation of PAMAM-PEG-CTX in the other organs, such as the liver and kidney.8 Similar results were observed in PAMAM-PEG-Angiopep,52 PAMAM-PEG-T7,69 and PAMAM-PEG-Tf.61 In turn, intratumoral (i.t.) administration of dendrimer–drug nanoparticles is not ideal, but it is a practical treatment for life-threatening disseminated metastatic disease. The accumulation of boronated dendrimers conjugated with EGF (BD-EGF) in the liver and spleen after i.t. administration was significantly lower (0.1% of injected dose/g) than the accumulation after i.v. injection (1.3% of injected dose/g). The accumulation in EGFR-positive gliomas after i.t. administration in 48 h was significantly higher (16.3% of injected dose/g) than after i.v. injection (1.3% of injected dose/g).126,127
Major Challenges and Future Directions
Despite these promising results, this field is still in its infancy. Several issues need be addressed before dendrimers can be widely used in a clinical setting involving the CNS. First, in-depth and comprehensive toxicological studies of brain-targeting dendrimers with and without therapeutics are required. Because CNS diseases require chronic treatment, the chronic and cumulative effects of dendritic medicine on brain tissues need to be clarified in addition to the acute toxicity. Although the in vitro cytotoxicity and blood compatibility of dendrimers have been extensively studied,58,122 few studies have been conducted to evaluate the toxicity of dendrimers in the CNS. A recent study indicates that PAMAM generation 4 induced slow neural cell apoptosis in the submicromolar concentration range and low microglia activation in brain tissue after 1 week.123 Therefore, detailed in vivo toxicity examination of dendrimers are important and can facilitate the design of tailored dendrimer-mediated CNS drug-delivery systems.
Second, targeted delivery is still an active research area for the application of dendrimers in CNS drug delivery. Several current targets such as LDL receptors, insulin receptor, and transferrin receptor have been also found in other tissues, suggesting that they are not specific. More specific and efficient targets need to be identified to facilitate the development of safer and more effective dendrimer delivery systems for use in the CNS.
Third, developing a noninvasive and safe alternative drug administration to substitute i.v. or i.t. administration is highly preferred because of their associated poor patient compliance. With the significant increase in the number of CNS drug prescriptions worldwide, the societal burden of health-care services and the risk of cross-contamination of i.v. injections will continuously increase. New drug-administration routes, such as oral, transbuccal mucosal/sublingual, or nasal, for dendrimer-mediated CNS drug-delivery systems need to be developed without compromising the efficacy of i.v. administration. To date, only a few studies have been conducted for developing these alternative routes. The study of PEGylated PAMAM dendrimer generation 4.5 conjugated with OX26 and labeled with AAF makes it possible to administer CNS dendrimer-mediated therapeutics via the transbuccal mucosal route.71
Dendrimers are capable of improving the solubility, biodistribution, and efficacy of a number of therapeutics as well as being used as imaging and diagnostic molecules in animal models bearing brain tumors. These therapeutics can be conjugated to the surface via tumor-labile (pH sensitive) linkers or encapsulated noncovalently into the structure via electrostatics. PEGylation of the dendrimer surface can prolong its circulation time and reduce its toxicity. BBB- or CNS-targeting ligand modification of the dendrimer surface can improve the rate and duration of drug delivery to brain tumor cells prior to the clearance of the remaining drug-delivery system. Imaging and diagnostic agents incorporated into dendrimers can help with the evaluation of brain tumors and dendrimer-associated CNS toxicity. However, challenges still exist regarding the deeper toxicological studies, specific targeting, and a noninvasive alternative drug administration methods. The ultimate goal of dendrimer-mediated CNS drug-delivery systems is to engineer the dendrimers to be safe and to enable their long-term use without the accumulation of adverse effects.
Acknowledgments
This work was supported in part by a National Science Foundation CAREER award (CBET0954957). We acknowledge the advice of Dr. Hu Yang in the Department of Biomedical Engineering at Virginia Commonwealth University.
The authors declare no competing financial interest.
References
- Barchet T. M.; Amiji M. M. (2009) Challenges and opportunities in CNS delivery of therapeutics for neurodegenerative diseases. Expert Opin. Drug Delivery 6, 211–225. [DOI] [PubMed] [Google Scholar]
- Yang H. (2010) Nanoparticle-mediated brain-specific drug delivery, imaging, and diagnosis. Pharm. Res. 27, 1759–1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tiwari S. B.; Amiji M. M. (2006) A review of nanocarrier-based CNS delivery systems. Curr. Drug Delivery 3, 219–232. [DOI] [PubMed] [Google Scholar]
- DiNunzio J. C.; Williams R. O. 3rd (2008) CNS disorders–current treatment options and the prospects for advanced therapies. Drug Dev. Ind. Pharm. 34, 1141–1167. [DOI] [PubMed] [Google Scholar]
- Nowacek A.; Gendelman H. E. (2009) NanoART, neuroAIDS and CNS drug delivery. Nanomedicine 4, 557–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong H. L.; Wu X. Y.; Bendayan R. (2012) Nanotechnological advances for the delivery of CNS therapeutics. Adv. Drug Delivery Rev. 64, 686–700. [DOI] [PubMed] [Google Scholar]
- Huang R.; Han L.; Li J.; Liu S.; Shao K.; Kuang Y.; Hu X.; Wang X.; Lei H.; Jiang C. (2011) Chlorotoxin-modified macromolecular contrast agent for MRI tumor diagnosis. Biomaterials 32, 5177–5186. [DOI] [PubMed] [Google Scholar]
- Huang R.; Ke W.; Han L.; Li J.; Liu S.; Jiang C. (2011) Targeted delivery of chlorotoxin-modified DNA-loaded nanoparticles to glioma via intravenous administration. Biomaterials 32, 2399–2406. [DOI] [PubMed] [Google Scholar]
- Huang S.; Li J.; Han L.; Liu S.; Ma H.; Huang R.; Jiang C. (2011) Dual targeting effect of angiopep-2-modified, DNA-loaded nanoparticles for glioma. Biomaterials 32, 6832–6838. [DOI] [PubMed] [Google Scholar]
- Li Y.; He H.; Jia X.; Lu W. L.; Lou J.; Wei Y. (2012) A dual-targeting nanocarrier based on poly(amidoamine) dendrimers conjugated with transferrin and tamoxifen for treating brain gliomas. Biomaterials 33, 3899–3908. [DOI] [PubMed] [Google Scholar]
- Cardoso A. L.; Simoes S.; de Almeida L. P.; Plesnila N.; Pedroso de Lima M. C.; Wagner E.; Culmsee C. (2008) Tf-lipoplexes for neuronal siRNA delivery: A promising system to mediate gene silencing in the CNS. J. Controlled Release 132, 113–123. [DOI] [PubMed] [Google Scholar]
- Gaillard P. J.; Appeldoorn C. C.; Rip J.; Dorland R.; van der Pol S. M.; Kooij G.; de Vries H. E.; Reijerkerk A. (2012) Enhanced brain delivery of liposomal methylprednisolone improved therapeutic efficacy in a model of neuroinflammation. J. Controlled Release 164, 364–369. [DOI] [PubMed] [Google Scholar]
- Zhang P.; Hu L.; Yin Q.; Zhang Z.; Feng L.; Li Y. (2012) Transferrin-conjugated polyphosphoester hybrid micelle loading paclitaxel for brain-targeting delivery: Synthesis, preparation and in vivo evaluation. J. Controlled Release 159, 429–434. [DOI] [PubMed] [Google Scholar]
- Shao K.; Wu J.; Chen Z.; Huang S.; Li J.; Ye L.; Lou J.; Zhu L.; Jiang C. (2012) A brain-vectored angiopep-2 based polymeric micelles for the treatment of intracranial fungal infection. Biomaterials 33, 6898–6907. [DOI] [PubMed] [Google Scholar]
- Zolotarskaya O. Y.; Wagner A. F.; Beckta J. M.; Valerie K.; Wynne K. J.; Yang H. (2012) Synthesis of water-soluble camptothecin-polyoxetane conjugates via click chemistry. Mol. Pharmaceutics 9, 3403–3408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang G. P.; Zeng J. M.; Gao S. J.; Ma Y. X.; Shi L.; Li Y.; Too H. P.; Wang S. (2003) Polyethylene glycol modified polyethylenimine for improved CNS gene transfer: Effects of PEGylation extent. Biomaterials 24, 2351–2362. [DOI] [PubMed] [Google Scholar]
- Gao X.; Chen J.; Chen J.; Wu B.; Chen H.; Jiang X. (2008) Quantum dots bearing lectin-functionalized nanoparticles as a platform for in vivo brain imaging. Bioconjugate Chem. 19, 2189–2195. [DOI] [PubMed] [Google Scholar]
- Bonoiu A.; Mahajan S. D.; Ye L.; Kumar R.; Ding H.; Yong K. T.; Roy I.; Aalinkeel R.; Nair B.; Reynolds J. L.; Sykes D. E.; Imperiale M. A.; Bergey E. J.; Schwartz S. A.; Prasad P. N. (2009) MMP-9 gene silencing by a quantum dot-siRNA nanoplex delivery to maintain the integrity of the blood brain barrier. Brain Res. 1282, 142–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montagne A.; Gauberti M.; Macrez R.; Jullienne A.; Briens A.; Raynaud J. S.; Louin G.; Buisson A.; Haelewyn B.; Docagne F.; Defer G.; Vivien D.; Maubert E. (2012) Ultra-sensitive molecular MRI of cerebrovascular cell activation enables early detection of chronic central nervous system disorders. NeuroImage 63, 760–770. [DOI] [PubMed] [Google Scholar]
- Farrell B. T.; Hamilton B. E.; Dosa E.; Rimely E.; Nasseri M.; Gahramanov S.; Lacy C. A.; Frenkel E. P.; Doolittle N. D.; Jacobs P. M.; Neuwelt E. A. (2013) Using iron oxide nanoparticles to diagnose CNS inflammatory diseases and PCNSL. Neurology 81, 256–263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menjoge A. R.; Kannan R. M.; Tomalia D. A. (2010) Dendrimer-based drug and imaging conjugates: design considerations for nanomedical applications. Drug Discovery Today 15, 171–185. [DOI] [PubMed] [Google Scholar]
- Abbott N. J.; Ronnback L.; Hansson E. (2006) Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 7, 41–53. [DOI] [PubMed] [Google Scholar]
- Gaillard P. J.; de Boer A. G. (2000) Relationship between permeability status of the blood–brain barrier and in vitro permeability coefficient of a drug. Eur. J. Pharm. Sci. 12, 95–102. [DOI] [PubMed] [Google Scholar]
- Smith Q. R.; Rapoport S. I. (1986) Cerebrovascular permeability coefficients to sodium, potassium, and chloride. J. Neurochem. 46, 1732–1742. [DOI] [PubMed] [Google Scholar]
- Liu X.; Chen C.; Smith B. J. (2008) Progress in brain penetration evaluation in drug discovery and development. J. Pharmacol. Exp. Ther. 325, 349–356. [DOI] [PubMed] [Google Scholar]
- van R. I.; Cakir-Tascioglu S.; Hennink W. E.; Storm G.; Schiffelers R. M.; Mastrobattista E. (2011) In vivo methods to study uptake of nanoparticles into the brain. Pharm. Res. 28, 456–471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brownson E. A.; Abbruscato T. J.; Gillespie T. J.; Hruby V. J.; Davis T. P. (1994) Effect of peptidases at the blood brain barrier on the permeability of encephalin. J. Pharmacol. Exp. Ther. 270, 675–680. [PubMed] [Google Scholar]
- Matter K.; Balda M. S. (2003) Holey barrier: Claudins and the regulation of brain endothelial permeability. J. Cell Biol. 161, 459–460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardridge W. M. (2005) The blood-brain barrier: Bottleneck in brain drug development. NeuroRx 2, 3–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardridge W. M. (2002) Drug and gene delivery to the brain: The vascular route. Neuron 36, 555–558. [DOI] [PubMed] [Google Scholar]
- Simpson I. A.; Carruthers A.; Vannucci S. J. (2007) Supply and demand in cerebral energy metabolism: The role of nutrient transporters. J. Cereb. Blood Flow Metab. 1766–1791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biddlestone-Thorpe L.; Marchi N.; Guo K.; Ghosh C.; Janigro D.; Valerie K.; Yang H. (2012) Nanomaterial-mediated CNS delivery of diagnostic and therapeutic agents. Adv. Drug Delivery Rev. 64, 605–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herve F.; Ghinea N.; Scherrmann J. M. (2008) CNS delivery via adsorptive transcytosis. AAPS J. 10, 455–472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones A. R.; Shusta E. V. (2007) Blood-brain barrier transport of therapeutics via receptor-mediation. Pharm. Res. 24, 1759–1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumling Iii J. P.; Silva G. A. (2012) Targeting the brain: Advances in drug delivery. Curr. Pharm. Biotechnol. 13, 2417–2426. [DOI] [PubMed] [Google Scholar]
- Friden P. M.; Walus L. R.; Musso G. F.; Taylor M. A.; Malfroy B.; Starzyk R. M. (1991) Anti-transferrin receptor antibody and antibody-drug conjugates cross the blood-brain barrier. Proc. Natl. Acad. Sci. U.S.A. 88, 4771–4775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi N.; Boado R. J.; Pardridge W. M. (2000) Antisense imaging of gene expression in the brain in vivo. Proc. Natl. Acad. Sci. U.S.A. 97, 14709–14714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu D.; Yang J.; Pardridge W. M. (1997) Drug targeting of a peptide radiopharmaceutical through the primate blood-brain barrier in vivo with a monoclonal antibody to the human insulin receptor. J. Clin. Invest. 100, 1804–1812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y.; Schlachetzki F.; Pardridge W. M. (2003) Global non-viral gene transfer to the primate brain following intravenous administration. Mol. Ther. 7, 11–18. [DOI] [PubMed] [Google Scholar]
- Boado R. J.; Zhang Y.; Zhang Y.; Pardridge W. M. (2007) Genetic engineering, expression, and activity of a fusion protein of a human neurotrophin and a molecular Trojan horse for delivery across the human blood-brain barrier. Biotechnol. Bioeng. 97, 1376–1386. [DOI] [PubMed] [Google Scholar]
- Urayama A.; Grubb J. H.; Sly W. S.; Banks W. A. (2004) Developmentally regulated mannose 6-phosphate receptor-mediated transport of a lysosomal enzyme across the blood-brain barrier. Proc. Natl. Acad. Sci. U.S.A. 101, 12658–12663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreuter J.; Ramge P.; Petrov V.; Hamm S.; Gelperina S. E.; Engelhardt B.; Alyautdin R.; von Briesen H.; Begley D. J. (2003) Direct evidence that polysorbate-80-coated poly(butylcyanoacrylate) nanoparticles deliver drugs to the CNS via specific mechanisms requiring prior binding of drug to the nanoparticles. Pharm. Res. 20, 409–416. [DOI] [PubMed] [Google Scholar]
- Benchenane K.; Berezowski V.; Ali C.; Fernandez-Monreal M.; Lopez-Atalaya J. P.; Brillault J.; Chuquet J.; Nouvelot A.; MacKenzie E. T.; Bu G.; Cecchelli R.; Touzani O.; Vivien D. (2005) Tissue-type plasminogen activator crosses the intact blood-brain barrier by low-density lipoprotein receptor-related protein-mediated transcytosis. Circulation 111, 2241–2249. [DOI] [PubMed] [Google Scholar]
- Michaelis K.; Hoffmann M. M.; Dreis S.; Herbert E.; Alyautdin R. N.; Michaelis M.; Kreuter J.; Langer K. (2006) Covalent linkage of apolipoprotein E to albumin nanoparticles strongly enhances drug transport into the brain. J. Pharmacol. Exp. Ther. 317, 1246–1253. [DOI] [PubMed] [Google Scholar]
- Anderson P.; Pichichero M. E.; Insel R. A. (1985) Immunogens consisting of oligosaccharides from the capsule of Haemophilus influenzae type b coupled to diphtheria toxoid or the toxin protein CRM197. J. Clin. Invest. 76, 52–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang P.; Xue Y.; Shang X.; Liu Y. (2010) Diphtheria toxin mutant CRM197-mediated transcytosis across blood–brain barrier in vitro. Cell. Mol. Neurobiol. 30, 717–725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (2011) Designing Dendrimers (Campagna S., Ceroni P., and Puntoriero F., Eds.) John Wiley & Sons, Inc., Hoboken, NJ. [Google Scholar]
- Tomalia D. A., Christensen J. B., and Boas U. (2012) Dendrimers, Dendrons, and Dendritic Polymers: Discovery, Applications, and the Future, Cambridge University Press, Cambridge, UK. [Google Scholar]
- Imae T. (2012) Physicochemical properties of dendrimers and dendrimer complexes, in Dendrimer-Based Drug Delivery Systems (Cheng Y., Ed.) pp 55–92, John Wiley & Sons, Inc., Hoboken, NJ. [Google Scholar]
- Xu L., Yeudall W. A., and Yang H. (2013) Dendrimer-based RNA interference delivery for cancer therapy, in Tailored Polymer Architectures for Pharmaceutical and Biomedical Applications, pp 197–213, American Chemical Society, Washington, DC. [Google Scholar]
- Sadekar S.; Ghandehari H. (2012) Transepithelial transport and toxicity of PAMAM dendrimers: Implications for oral drug delivery. Adv. Drug Delivery Rev. 64, 571–588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ke W.; Shao K.; Huang R.; Han L.; Liu Y.; Li J.; Kuang Y.; Ye L.; Lou J.; Jiang C. (2009) Gene delivery targeted to the brain using an angiopep-conjugated polyethyleneglycol-modified polyamidoamine dendrimer. Biomaterials 30, 6976–6985. [DOI] [PubMed] [Google Scholar]
- Liu Y.; Huang R.; Han L.; Ke W.; Shao K.; Ye L.; Lou J.; Jiang C. (2009) Brain-targeting gene delivery and cellular internalization mechanisms for modified rabies virus glycoprotein RVG29 nanoparticles. Biomaterials 30, 4195–4202. [DOI] [PubMed] [Google Scholar]
- Sonawane N. D.; Szoka F. C. Jr.; Verkman A. S. (2003) Chloride accumulation and swelling in endosomes enhances DNA transfer by polyamine-DNA polyplexes. J. Biol. Chem. 278, 44826–44831. [DOI] [PubMed] [Google Scholar]
- Perez-Martinez F. C.; Ocana A. V.; Perez-Carrion M. D.; Cena V. (2012) Dendrimers as vectors for genetic material delivery to the nervous system. Curr. Med. Chem. 19, 5101–5108. [DOI] [PubMed] [Google Scholar]
- Santander-Ortega M. J., Uchegbu I. F., and Schätzlein A. G. (2012) Dendrimer-based gene delivery systems: Administration routes and in vivo evaluation, in Dendrimer-Based Drug Delivery Systems (Cheng Y., Ed.) pp 329–354, John Wiley & Sons, Inc., Hoboken, NJ. [Google Scholar]
- Kirkpatrick G. J.; Plumb J. A.; Sutcliffe O. B.; Flint D. J.; Wheate N. J. (2011) Evaluation of anionic half generation 3.5–6.5 poly(amidoamine) dendrimers as delivery vehicles for the active component of the anticancer drug cisplatin. J. Inorg. Biochem. 105, 1115–1122. [DOI] [PubMed] [Google Scholar]
- Duncan R.; Izzo L. (2005) Dendrimer biocompatibility and toxicity. Adv. Drug Delivery Rev. 57, 2215–2237. [DOI] [PubMed] [Google Scholar]
- He H.; Li Y.; Jia X. R.; Du J.; Ying X.; Lu W. L.; Lou J. N.; Wei Y. (2011) PEGylated poly(amidoamine) dendrimer-based dual-targeting carrier for treating brain tumors. Biomaterials 32, 478–487. [DOI] [PubMed] [Google Scholar]
- Huang R.; Ke W.; Liu Y.; Jiang C.; Pei Y. (2008) The use of lactoferrin as a ligand for targeting the polyamidoamine-based gene delivery system to the brain. Biomaterials 29, 238–246. [DOI] [PubMed] [Google Scholar]
- Huang R. Q.; Qu Y. H.; Ke W. L.; Zhu J. H.; Pei Y. Y.; Jiang C. (2007) Efficient gene delivery targeted to the brain using a transferrin-conjugated polyethyleneglycol-modified polyamidoamine dendrimer. FASEB J. 21, 1117–1125. [DOI] [PubMed] [Google Scholar]
- Kolhe P.; Khandare J.; Pillai O.; Kannan S.; Lieh-Lai M.; Kannan R. M. (2006) Preparation, cellular transport, and activity of polyamidoamine-based dendritic nanodevices with a high drug payload. Biomaterials 27, 660–669. [DOI] [PubMed] [Google Scholar]
- Kono K.; Kojima C.; Hayashi N.; Nishisaka E.; Kiura K.; Watarai S.; Harada A. (2008) Preparation and cytotoxic activity of poly(ethylene glycol)-modified poly(amidoamine) dendrimers bearing adriamycin. Biomaterials 29, 1664–1675. [DOI] [PubMed] [Google Scholar]
- Guillaudeu S. J.; Fox M. E.; Haidar Y. M.; Dy E. E.; Szoka F. C.; Frechet J. M. (2008) PEGylated dendrimers with core functionality for biological applications. Bioconjugate Chem. 19, 461–469. [DOI] [PubMed] [Google Scholar]
- Pack D. W.; Hoffman A. S.; Pun S.; Stayton P. S. (2005) Design and development of polymers for gene delivery. Nat. Rev. Drug Discovery 4, 581–593. [DOI] [PubMed] [Google Scholar]
- Yuan Q.; Yeudall W. A.; Yang H. (2010) PEGylated polyamidoamine dendrimers with bis-aryl hydrazone linkages for enhanced gene delivery. Biomacromolecules 11, 1940–1947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurtoglu Y. E.; Navath R. S.; Wang B.; Kannan S.; Romero R.; Kannan R. M. (2009) Poly(amidoamine) dendrimer-drug conjugates with disulfide linkages for intracellular drug delivery. Biomaterials 30, 2112–2121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kannan S.; Dai H.; Navath R. S.; Balakrishnan B.; Jyoti A.; Janisse J.; Romero R.; Kannan R. M. (2012) Dendrimer-based postnatal therapy for neuroinflammation and cerebral palsy in a rabbit model. Sci. Transl. Med. 4, 130–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han L.; Huang R.; Li J.; Liu S.; Huang S.; Jiang C. (2011) Plasmid pORF-hTRAIL and doxorubicin co-delivery targeting to tumor using peptide-conjugated polyamidoamine dendrimer. Biomaterials 32, 1242–1252. [DOI] [PubMed] [Google Scholar]
- Gosk S.; Vermehren C.; Storm G.; Moos T. (2004) Targeting anti-transferrin receptor antibody (OX26) and OX26-conjugated liposomes to brain capillary endothelial cells using in situ perfusion. J. Cereb. Blood Flow Metab. 1193–1204. [DOI] [PubMed] [Google Scholar]
- Yuan Q.; Fu Y.; Kao W. J.; Janigro D.; Yang H. (2011) Transbuccal delivery of CNS therapeutic nanoparticles: Synthesis, characterization, and in vitro permeation studies. ACS Chem. Neurosci. 2, 676–683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plattner V. E.; Germann B.; Neuhaus W.; Noe C. R.; Gabor F.; Wirth M. (2010) Characterization of two blood-brain barrier mimicking cell lines: distribution of lectin-binding sites and perspectives for drug delivery. Int. J. Pharm. 387, 34–41. [DOI] [PubMed] [Google Scholar]
- Yuan Q.; Lee E.; Yeudall W. A.; Yang H. (2010) Dendrimer-triglycine-EGF nanoparticles for tumor imaging and targeted nucleic acid and drug delivery. Oral Oncol. 46, 698–704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deshane J.; Garner C. C.; Sontheimer H. (2003) Chlorotoxin inhibits glioma cell invasion via matrix metalloproteinase-2. J. Biol. Chem. 278, 4135–4144. [DOI] [PubMed] [Google Scholar]
- Lyons S. A.; O’Neal J.; Sontheimer H. (2002) Chlorotoxin, a scorpion-derived peptide, specifically binds to gliomas and tumors of neuroectodermal origin. Glia 39, 162–173. [DOI] [PubMed] [Google Scholar]
- Kumar P.; Wu H.; McBride J. L.; Jung K. E.; Kim M. H.; Davidson B. L.; Lee S. K.; Shankar P.; Manjunath N. (2007) Transvascular delivery of small interfering RNA to the central nervous system. Nature 448, 39–43. [DOI] [PubMed] [Google Scholar]
- Bu G.; Maksymovitch E. A.; Nerbonne J. M.; Schwartz A. L. (1994) Expression and function of the low density lipoprotein receptor-related protein (LRP) in mammalian central neurons. J. Biol. Chem. 269, 18521–18528. [PubMed] [Google Scholar]
- Cheng Y.; Li M.; Xu T. (2008) Potential of poly(amidoamine) dendrimers as drug carriers of camptothecin based on encapsulation studies. Eur. J. Med. Chem. 43, 1791–1795. [DOI] [PubMed] [Google Scholar]
- Sadekar S.; Thiagarajan G.; Bartlett K.; Hubbard D.; Ray A.; McGill L. D.; Ghandehari H. (2013) Poly(amido amine) dendrimers as absorption enhancers for oral delivery of camptothecin. Int. J. Pharm. 456, 175–185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu S.; Hong M.; Zhang L.; Tang G.; Jiang Y.; Pei Y. (2010) PEGylated PAMAM dendrimer-doxorubicin conjugates: In vitro evaluation and in vivo tumor accumulation. Pharm. Res. 27, 161–174. [DOI] [PubMed] [Google Scholar]
- Zhu S.; Qian L.; Hong M.; Zhang L.; Pei Y.; Jiang Y. (2011) RGD-modified PEG-PAMAM-DOX conjugate: In vitro and in vivo targeting to both tumor neovascular endothelial cells and tumor cells. Adv. Mater. 23, H84–H89. [DOI] [PubMed] [Google Scholar]
- Zhang L.; Zhu S.; Qian L.; Pei Y.; Qiu Y.; Jiang Y. (2011) RGD-modified PEG-PAMAM-DOX conjugates: In vitro and in vivo studies for glioma. Eur. J. Pharm. Biopharm. 79, 232–240. [DOI] [PubMed] [Google Scholar]
- Zhu S.; Hong M.; Tang G.; Qian L.; Lin J.; Jiang Y.; Pei Y. (2010) Partly PEGylated polyamidoamine dendrimer for tumor-selective targeting of doxorubicin: The effects of PEGylation degree and drug conjugation style. Biomaterials 31, 1360–1371. [DOI] [PubMed] [Google Scholar]
- Liu D.; Hu H.; Zhang J.; Zhao X.; Tang X.; Chen D. (2011) Drug pH-sensitive release in vitro and targeting ability of polyamidoamine dendrimer complexes for tumor cells. Chem. Pharm. Bull. 59, 63–71. [DOI] [PubMed] [Google Scholar]
- Thiagarajan G.; Ray A.; Malugin A.; Ghandehari H. (2010) PAMAM-camptothecin conjugate inhibits proliferation and induces nuclear fragmentation in colorectal carcinoma cells. Pharm. Res. 27, 2307–2316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai P. S.; Lou P. J.; Peng C. L.; Pai C. L.; Yen W. N.; Huang M. Y.; Young T. H.; Shieh M. J. (2007) Doxorubicin delivery by polyamidoamine dendrimer conjugation and photochemical internalization for cancer therapy. J. Controlled Release 122, 39–46. [DOI] [PubMed] [Google Scholar]
- Choi S. K.; Verma M.; Silpe J.; Moody R. E.; Tang K.; Hanson J. J.; Baker J. R. Jr. (2012) A photochemical approach for controlled drug release in targeted drug delivery. Bioorg. Med. Chem. 20, 1281–1290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuta T.; Wang S. S.; Dantzker J. L.; Dore T. M.; Bybee W. J.; Callaway E. M.; Denk W.; Tsien R. Y. (1999) Brominated 7-hydroxycoumarin-4-ylmethyls: Photolabile protecting groups with biologically useful cross-sections for two photon photolysis. Proc. Natl. Acad. Sci. U.S.A. 96, 1193–1200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goard M.; Aakalu G.; Fedoryak O. D.; Quinonez C.; St Julien J.; Poteet S. J.; Schuman E. M.; Dore T. M. (2005) Light-mediated inhibition of protein synthesis. Chem. Biol. 12, 685–693. [DOI] [PubMed] [Google Scholar]
- Jackson A. L.; Linsley P. S. (2010) Recognizing and avoiding siRNA off-target effects for target identification and therapeutic application. Nat. Rev. Drug Discovery 9, 57–67. [DOI] [PubMed] [Google Scholar]
- Kesharwani P.; Gajbhiye V.; Jain N. K. (2012) A review of nanocarriers for the delivery of small interfering RNA. Biomaterials 33, 7138–7150. [DOI] [PubMed] [Google Scholar]
- Pan G.; Ni J.; Wei Y. F.; Yu G.; Gentz R.; Dixit V. M. (1997) An antagonist decoy receptor and a death domain-containing receptor for TRAIL. Science 277, 815–818. [DOI] [PubMed] [Google Scholar]
- Wiley S. R.; Schooley K.; Smolak P. J.; Din W. S.; Huang C. P.; Nicholl J. K.; Sutherland G. R.; Smith T. D.; Rauch C.; Smith C. A.; et al. (1995) Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3, 673–682. [DOI] [PubMed] [Google Scholar]
- Patil M. L.; Zhang M.; Minko T. (2011) Multifunctional triblock nanocarrier (PAMAM-PEG-PLL) for the efficient intracellular siRNA delivery and gene silencing. ACS Nano 5, 1877–1887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agrawal A.; Min D. H.; Singh N.; Zhu H.; Birjiniuk A.; von M. G.; Harris T. J.; Xing D.; Woolfenden S. D.; Sharp P. A.; Charest A.; Bhatia S. (2009) Functional delivery of siRNA in mice using dendriworms. ACS Nano 3, 2495–2504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai H.; Navath R. S.; Balakrishnan B.; Guru B. R.; Mishra M. K.; Romero R.; Kannan R. M.; Kannan S. (2010) Intrinsic targeting of inflammatory cells in the brain by polyamidoamine dendrimers upon subarachnoid administration. Nanomedicine 5, 1317–1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasiak T.; Ionov M.; Nieznanski K.; Nieznanska H.; Klementieva O.; Granell M.; Cladera J.; Majoral J. P.; Caminade A. M.; Klajnert B. (2012) Phosphorus dendrimers affect Alzheimer’s (Abeta1–28) peptide and MAP-Tau protein aggregation. Mol. Pharmaceutics 9, 458–469. [DOI] [PubMed] [Google Scholar]
- Klajnert B.; Wasiak T.; Ionov M.; Fernandez-Villamarin M.; Sousa-Herves A.; Correa J.; Riguera R.; Fernandez-Megia E. (2012) Dendrimers reduce toxicity of Abeta 1–28 peptide during aggregation and accelerate fibril formation. Nanomedicine 8, 1372–1378. [DOI] [PubMed] [Google Scholar]
- Klementieva O.; Aso E.; Filippini D.; Benseny-Cases N.; Carmona M.; Juves S.; Appelhans D.; Cladera J.; Ferrer I. (2013) Effect of poly(propylene imine) glycodendrimers on beta-amyloid aggregation in vitro and in APP/PS1 transgenic mice, as a model of brain amyloid deposition and Alzheimer’s disease. Biomacromolecules 14, 3570–3580. [DOI] [PubMed] [Google Scholar]
- Klementieva O.; Benseny-Cases N.; Gella A.; Appelhans D.; Voit B.; Cladera J. (2011) Dense shell glycodendrimers as potential nontoxic anti-amyloidogenic agents in Alzheimer’s disease. Amyloid-dendrimer aggregates morphology and cell toxicity. Biomacromolecules 12, 3903–3909. [DOI] [PubMed] [Google Scholar]
- Patel D.; Henry J.; Good T. (2006) Attenuation of beta-amyloid induced toxicity by sialic acid-conjugated dendrimeric polymers. Biochim. Biophys. Acta 1760, 1802–1809. [DOI] [PubMed] [Google Scholar]
- Patel D. A.; Henry J. E.; Good T. A. (2007) Attenuation of b-amyloid induced toxicity by sialic acid-conjugated dendrimers: Role of sialic acid attachment. Brain Res. 1161, 95–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klajnert B.; Cangiotti M.; Calici S.; Majoral J. P.; Caminade A. M.; Cladera J.; Bryszewska M.; Ottaviani M. F. (2007) EPR study of the interactions between dendrimers and peptides involved in Alzheimer’s and prion diseases. Macromol. Biosci. 7, 1065–1074. [DOI] [PubMed] [Google Scholar]
- Misgeld T.; Kerschensteiner M. (2006) In vivo imaging of the diseased nervous system. Nat. Rev. Neurosci. 7, 449–463. [DOI] [PubMed] [Google Scholar]
- Nunes A.; Al-Jamal K. T.; Kostarelos K. (2012) Therapeutics, imaging and toxicity of nanomaterials in the central nervous system. J. Controlled Release 161, 290–306. [DOI] [PubMed] [Google Scholar]
- Nguyen Q. T.; Olson E. S.; Aguilera T. A.; Jiang T.; Scadeng M.; Ellies L. G.; Tsien R. Y. (2010) Surgery with molecular fluorescence imaging using activatable cell-penetrating peptides decreases residual cancer and improves survival. Proc. Natl. Acad. Sci. U.S.A. 107, 4317–4322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang T.; Olson E. S.; Nguyen Q. T.; Roy M.; Jennings P. A.; Tsien R. Y. (2004) Tumor imaging by means of proteolytic activation of cell-penetrating peptides. Proc. Natl. Acad. Sci. U.S.A. 101, 17867–17872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kateb B.; Chiu K.; Black K. L.; Yamamoto V.; Khalsa B.; Ljubimova J. Y.; Ding H.; Patil R.; Portilla-Arias J. A.; Modo M.; Moore D. F.; Farahani K.; Okun M. S.; Prakash N.; Neman J.; Ahdoot D.; Grundfest W.; Nikzad S.; Heiss J. D. (2011) Nanoplatforms for constructing new approaches to cancer treatment, imaging, and drug delivery: What should be the policy?. NeuroImage 54, S106–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai H., Shen M., and Shi X. (2012) Dendrimer-based medical nanodevices for magnetic resonance imaging applications, in Dendrimer-Based Drug Delivery Systems (Cheng Y., Ed.) pp 463–478, John Wiley & Sons, Inc., Hoboken, NJ. [Google Scholar]
- Peng C., and Shi X. (2012) Dendrimer-related nanoparticle system for computed tomography imaging, in Dendrimer-Based Drug Delivery Systems (Cheng Y., Ed.) pp 479–500, John Wiley & Sons, Inc., Hoboken, NJ. [Google Scholar]
- Han L.; Li J.; Huang S.; Huang R.; Liu S.; Hu X.; Yi P.; Shan D.; Wang X.; Lei H.; Jiang C. (2011) Peptide-conjugated polyamidoamine dendrimer as a nanoscale tumor-targeted T1 magnetic resonance imaging contrast agent. Biomaterials 32, 2989–2998. [DOI] [PubMed] [Google Scholar]
- Yan H.; Wang L.; Wang J.; Weng X.; Lei H.; Wang X.; Jiang L.; Zhu J.; Lu W.; Wei X.; Li C. (2012) Two-order targeted brain tumor imaging by using an optical/paramagnetic nanoprobe across the blood brain barrier. ACS Nano 6, 410–420. [DOI] [PubMed] [Google Scholar]
- Demeule M.; Regina A.; Che C.; Poirier J.; Nguyen T.; Gabathuler R.; Castaigne J. P.; Beliveau R. (2008) Identification and design of peptides as a new drug delivery system for the brain. J. Pharmacol. Exp. Ther. 324, 1064–1072. [DOI] [PubMed] [Google Scholar]
- Schottelius M.; Laufer B.; Kessler H.; Wester H. J. (2009) Ligands for mapping alphavbeta3-integrin expression in vivo. Acc. Chem. Res. 42, 969–980. [DOI] [PubMed] [Google Scholar]
- Lamy C. M.; Sallin O.; Loussert C.; Chatton J. Y. (2012) Sodium sensing in neurons with a dendrimer-based nanoprobe. ACS Nano 6, 1176–1187. [DOI] [PubMed] [Google Scholar]
- Li Y., Zhao L., and Cheng Y. (2012) Improving the biocompatibility of dendrimers in drug delivery, in Dendrimer-Based Drug Delivery Systems (Cheng Y., Ed.) pp 207–237, John Wiley & Sons, Inc., Hoboken, NJ. [Google Scholar]
- Roberts J. C.; Bhalgat M. K.; Zera R. T. (1996) Preliminary biological evaluation of polyamidoamine (PAMAM) starburst dendrimers. J. Biomed. Mater. Res. 30, 53–65. [DOI] [PubMed] [Google Scholar]
- Fischer D.; Bieber T.; Li Y.; Elsasser H. P.; Kissel T. (1999) A novel non-viral vector for DNA delivery based on low molecular weight, branched polyethylenimine: effect of molecular weight on transfection efficiency and cytotoxicity. Pharm. Res. 16, 1273–1279. [DOI] [PubMed] [Google Scholar]
- Fischer D.; Li Y.; Ahlemeyer B.; Krieglstein J.; Kissel T. (2003) In vitro cytotoxicity testing of polycations: Influence of polymer structure on cell viability and hemolysis. Biomaterials 24, 1121–1131. [DOI] [PubMed] [Google Scholar]
- Chen H. T.; Neerman M. F.; Parrish A. R.; Simanek E. E. (2004) Cytotoxicity, hemolysis, and acute in vivo toxicity of dendrimers based on melamine, candidate vehicles for drug delivery. J. Am. Chem. Soc. 126, 10044–10048. [DOI] [PubMed] [Google Scholar]
- Lee J. H.; Lim Y. B.; Choi J. S.; Lee Y.; Kim T. I.; Kim H. J.; Yoon J. K.; Kim K.; Park J. S. (2003) Polyplexes assembled with internally quaternized PAMAM-OH dendrimer and plasmid DNA have a neutral surface and gene delivery potency. Bioconjugate Chem. 14, 1214–1221. [DOI] [PubMed] [Google Scholar]
- Malik N.; Wiwattanapatapee R.; Klopsch R.; Lorenz K.; Frey H.; Weener J. W.; Meijer E. W.; Paulus W.; Duncan R. (2000) Dendrimers: Relationship between structure and biocompatibility in vitro, and preliminary studies on the biodistribution of 125I-labelled polyamidoamine dendrimers in vivo. J. Controlled Release 65, 133–148. [DOI] [PubMed] [Google Scholar]
- Albertazzi L.; Gherardini L.; Brondi M.; Sulis Sato S.; Bifone A.; Pizzorusso T.; Ratto G. M.; Bardi G. (2013) In vivo distribution and toxicity of PAMAM dendrimers in the central nervous system depend on their surface chemistry. Mol. Pharmacol. 10, 249–260. [DOI] [PubMed] [Google Scholar]
- Choi J. S.; Nam K.; Park J. Y.; Kim J. B.; Lee J. K.; Park J. S. (2004) Enhanced transfection efficiency of PAMAM dendrimer by surface modification with l-arginine. J. Controlled Release 99, 445–456. [DOI] [PubMed] [Google Scholar]
- Lesniak W. G.; Mishra M. K.; Jyoti A.; Balakrishnan B.; Zhang F.; Nance E.; Romero R.; Kannan S.; Kannan R. M. (2013) Biodistribution of fluorescently labeled PAMAM dendrimers in neonatal rabbits: Effect of neuroinflammation. Mol. Pharmaceutics 10, 4560–4571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capala J.; Barth R. F.; Bendayan M.; Lauzon M.; Adams D. M.; Soloway A. H.; Fenstermaker R. A.; Carlsson J. (1996) Boronated epidermal growth factor as a potential targeting agent for boron neutron capture therapy of brain tumors. Bioconjugate Chem. 7, 7–15. [DOI] [PubMed] [Google Scholar]
- Yang W.; Barth R. F.; Wu G.; Bandyopadhyaya A. K.; Thirumamagal B. T.; Tjarks W.; Binns P. J.; Riley K.; Patel H.; Coderre J. A.; Ciesielski M. J.; Fenstermaker R. A. (2004) Boronated epidermal growth factor as a delivery agent for neutron capture therapy of EGF receptor positive gliomas. Appl. Radiat. Isot. 61, 981–985. [DOI] [PubMed] [Google Scholar]


