Abstract
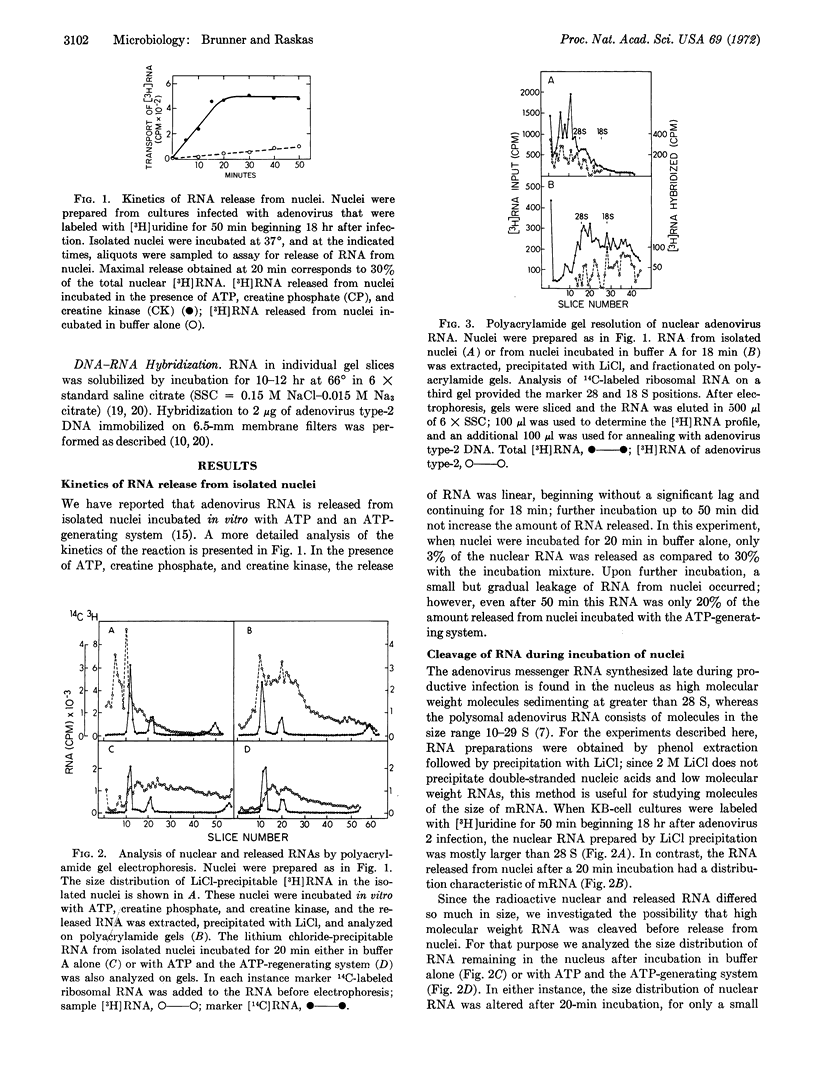
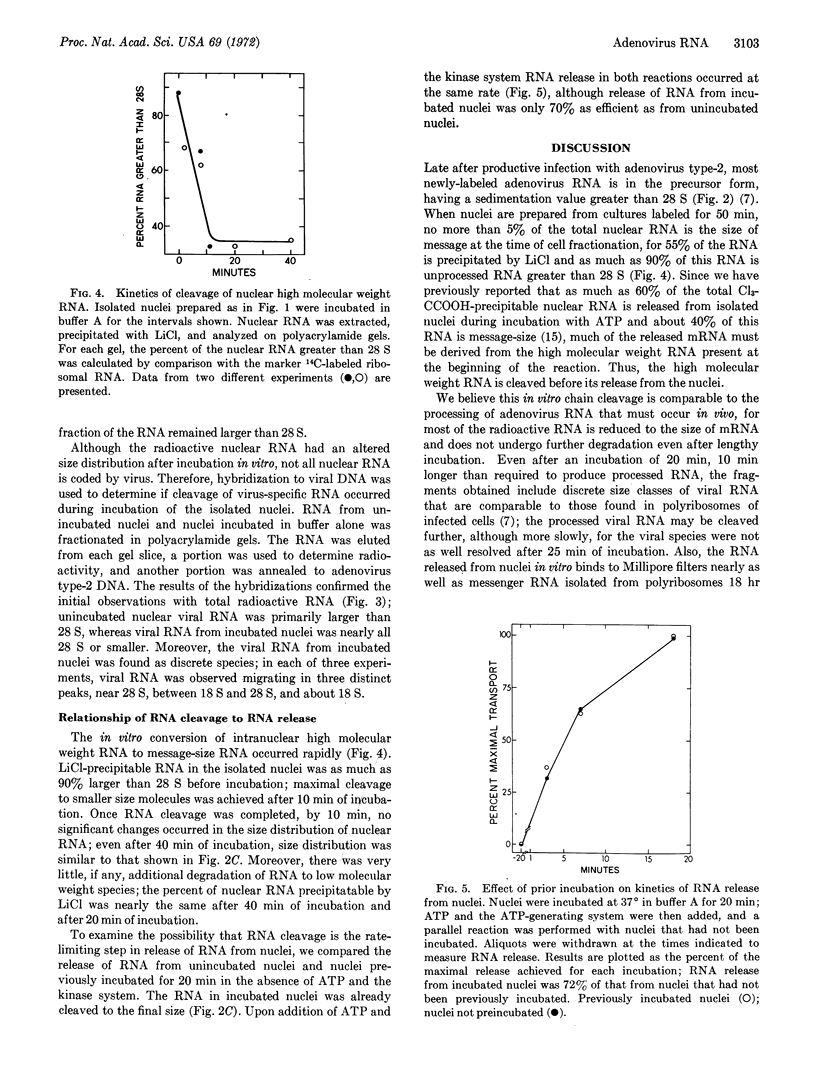
Nuclei isolated from cultured human cells (KB) infected with adenovirus type-2 were used to study events occurring during the adenosine triphosphate-dependent release of viral RNA. When cells were labeled with [3H]uridine for 50 min beginning 18 hr after infection, most nuclear viral messenger RNA was in the form of high molecular weight precursors with sedimentation coefficients greater than 28 S. When nuclei were incubated in the presence of ATP and an ATP-generating system, labeled RNA the size of viral mRNA (10-29 S) was released. Cleavage of viral RNA precursor molecules of high molecular weight occurred during incubation of nuclei. The change in size of viral RNA did not require exogenous ATP and occurred before release of RNA from nuclei. This RNA cleavage seems comparable to processing of mRNA in vivo, for discrete viral species were produced. Cleavage was completed after 10 min of incubation but was not the rate-limiting step in the release reaction, which required about 20 min for completion.
Keywords: KB cells, mRNA, ATP
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aronson A. I. Degradation products and a unique endonuclease in heterogeneous nuclear RNA in sea urchin embryos. Nat New Biol. 1972 Jan 12;235(54):40–44. doi: 10.1038/newbio235040a0. [DOI] [PubMed] [Google Scholar]
- Attardi G., Parnas H., Hwang M. I., Attardi B. Giant-size rapidly labeled nuclear ribonucleic acid and cytoplasmic messenger ribonucleic acid in immature duck erythrocytes. J Mol Biol. 1966 Sep;20(1):145–182. doi: 10.1016/0022-2836(66)90123-9. [DOI] [PubMed] [Google Scholar]
- Baltimore D. Purification and properties of poliovirus double-stranded ribonucleic acid. J Mol Biol. 1966 Jul;18(3):421–428. doi: 10.1016/s0022-2836(66)80034-7. [DOI] [PubMed] [Google Scholar]
- Darnell J. E., Wall R., Tushinski R. J. An adenylic acid-rich sequence in messenger RNA of HeLa cells and its possible relationship to reiterated sites in DNA. Proc Natl Acad Sci U S A. 1971 Jun;68(6):1321–1325. doi: 10.1073/pnas.68.6.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edmonds M., Vaughan M. H., Jr, Nakazato H. Polyadenylic acid sequences in the heterogeneous nuclear RNA and rapidly-labeled polyribosomal RNA of HeLa cells: possible evidence for a precursor relationship. Proc Natl Acad Sci U S A. 1971 Jun;68(6):1336–1340. doi: 10.1073/pnas.68.6.1336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S. Y., Mendecki J., Brawerman G. A polynucleotide segment rich in adenylic acid in the rapidly-labeled polyribosomal RNA component of mouse sarcoma 180 ascites cells. Proc Natl Acad Sci U S A. 1971 Jun;68(6):1331–1335. doi: 10.1073/pnas.68.6.1331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liau M. C., Craig N. C., Perry R. P. The production of ribosomal RNA from high molecular weight precursors. I. Factors which influence the ability of isolated nucleoili to process 45-S RNA. Biochim Biophys Acta. 1968 Nov 20;169(1):196–205. doi: 10.1016/0005-2787(68)90020-8. [DOI] [PubMed] [Google Scholar]
- Lindberg U., Darnell J. E. SV40-specific RNA in the nucleus and polyribosomes of transformed cells. Proc Natl Acad Sci U S A. 1970 Apr;65(4):1089–1096. doi: 10.1073/pnas.65.4.1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirault Marc Edouard, Scherrer Klaus. in vitro processing of HeLa cell preribosomes by a nucleolar endoribonuclease. FEBS Lett. 1972 Feb 1;20(2):233–238. doi: 10.1016/0014-5793(72)80803-2. [DOI] [PubMed] [Google Scholar]
- Parsons J. T., Gardner J., Green M. Biochemical studies on adenovirus multiplication, XIX. Resolution of late viral RNA species in the nucleus and cytoplasm. Proc Natl Acad Sci U S A. 1971 Mar;68(3):557–560. doi: 10.1073/pnas.68.3.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons J. T., Green M. Biochemical studies on adenovirus multiplication. 18. Resolution of early virus-specific RNA species in Ad 2 infected and transformed cells. Virology. 1971 Jul;45(1):154–162. doi: 10.1016/0042-6822(71)90122-x. [DOI] [PubMed] [Google Scholar]
- Penman S., Smith I., Holtzman E. Ribosomal RNA synthesis and processing in a particulate site in the HeLa cell nucleus. Science. 1966 Nov 11;154(3750):786–789. doi: 10.1126/science.154.3750.786. [DOI] [PubMed] [Google Scholar]
- Philipson L., Wall R., Glickman G., Darnell J. E. Addition of polyadenylate sequences to virus-specific RNA during adenovirus replication. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2806–2809. doi: 10.1073/pnas.68.11.2806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raskas H. J. Release of adenovirus messenger RNA from isolated nuclei. Nat New Biol. 1971 Sep 29;233(39):134–136. doi: 10.1038/newbio233134a0. [DOI] [PubMed] [Google Scholar]
- Raskas H. J., Thomas D. C., Green M. Biochemical studies on adenovirus multiplication. XVII. Ribosome synthesis in uninfected and infected KB cells. Virology. 1970 Apr;40(4):893–902. doi: 10.1016/0042-6822(70)90135-2. [DOI] [PubMed] [Google Scholar]
- Scherrer K., Marcaud L., Zajdela F., London I. M., Gros F. Patterns of RNA metabolism in a differentiated cell: a rapidly labeled, unstable 60S RNA with messenger properties in duck erythroblasts. Proc Natl Acad Sci U S A. 1966 Nov;56(5):1571–1578. doi: 10.1073/pnas.56.5.1571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soeiro R., Vaughan M. H., Warner J. R., Darnell J. E., Jr The turnover of nuclear DNA-like RNA in HeLa cells. J Cell Biol. 1968 Oct;39(1):112–118. doi: 10.1083/jcb.39.1.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stévenin J., Mandel P., Jacob M. Relationship between nuclear giant-size dRNA and microsomal dRNA of rat brain. Proc Natl Acad Sci U S A. 1969 Feb;62(2):490–497. doi: 10.1073/pnas.62.2.490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vesco C., Penman S. The fractionation of nuclei and the integrity of purified nucleoli in HeLa cells. Biochim Biophys Acta. 1968 Nov 20;169(1):188–195. doi: 10.1016/0005-2787(68)90019-1. [DOI] [PubMed] [Google Scholar]
- Wall R., Darnell J. E. Presence of cell and virus specific sequences in the same molecules of nuclear RNA from virus transformed cells. Nat New Biol. 1971 Jul 21;232(29):73–76. doi: 10.1038/newbio232073a0. [DOI] [PubMed] [Google Scholar]
- Warner J. R., Soeiro R., Birnboim H. C., Girard M., Darnell J. E. Rapidly labeled HeLa cell nuclear RNA. I. Identification by zone sedimentation of a heterogeneous fraction separate from ribosomal precursor RNA. J Mol Biol. 1966 Aug;19(2):349–361. doi: 10.1016/s0022-2836(66)80009-8. [DOI] [PubMed] [Google Scholar]



