Abstract
A 61-year-old man presented to our hospital with hypercalcemia and elevated C reactive protein (CRP). Evaluation revealed renal cell carcinoma (RCC) with metastasis to lung, bone, and brain. He underwent partial resection of the right kidney and a left nephrectomy. Histopathologic findings of resected tumors were consistent with clear cell RCC. Whole-brain irradiation was performed for management of brain metastasis. Postoperatively, he was treated with molecularly targeted therapy using a mammalian target of rapamycin inhibitor. Approximately 14 months later, he suffered an episode of upper gastrointestinal bleeding with secondary anemia and melena. Upper gastrointestinal endoscopy revealed a distinctly protruding lesion in the gastric body. Biopsy of the gastric lesion showed metastatic clear cell RCC. He underwent partial gastrectomy. His postoperative course was uneventful. However, 4 months after surgery, he died from brain metastasis. Metastatic RCC to the stomach, although rare, should be suspected in any patient with a history of RCC who presents with gastrointestinal symptoms.
Key words: Gastric metastasis, Renal cell carcinoma, Metastatic tumor
The occurrence of metastases to the stomach from various neoplasms is not common. In the autopsy series, the stomach has been reported as a metastatic site in 0.2% to 0.7% of cases.1–3 Lung cancer, breast cancer, and malignant melanoma were reported most often as primary tumors associated with gastric metastasis.1,4
In the literature, gastric metastases from renal cell carcinoma (RCC) have only rarely been described,5–22 occurring in 0.2% of RCCs in the clinical setting.5 The present report describes an unusual case of a 61-year-old man who presented with gastrointestinal bleeding due to gastric metastasis from RCC. The clinical characteristics, therapy, and outcomes of gastric metastasis from RCC are reviewed.
Case Report
A 61-year-old man presented to our hospital with hypercalcemia and elevated C reactive protein (CRP). He complained of gross hematuria. Examination by ultrasound and computed tomography (CT) showed bilateral RCC as well as metastases to the lung, bone, and brain. TNM classification was T3aN0M1, and the clinical stage was IV. He was admitted to our hospital and treated by partial resection of the right kidney. One month later, radiotherapy for brain metastasis was performed (30 Gy for whole brain, including metastasis). Ten days after the end of radiotherapy, a left nephrectomy was performed. The histopathologic finding of tumors from both kidneys was consistent with clear cell carcinoma. The patient's postoperative course was uneventful, and he was subsequently treated with molecularly targeted therapy using temsirolimus, a mammalian target of rapamycin (mTOR) inhibitor.
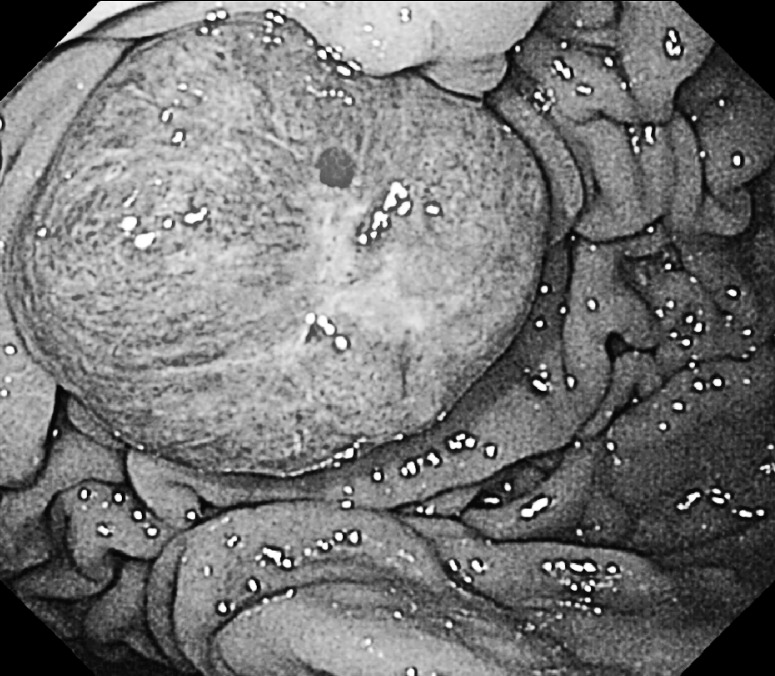
Two years later, he developed melena and severe anemia. Gastric endoscopy revealed a distinctly protruding lesion (diameter, 2 cm), with a central depression, at the greater curvature of the middle third of the gastric body (Fig. 1). The surface of the tumor bled easily upon contact with the endoscope. Microscopic examination of hematoxylin and eosin–stained biopsy specimens of the lesion revealed characteristics of RCC. Thus, a diagnosis of gastric metastasis from RCC was made. Abdominal CT showed no lymphadenopathy around the stomach or metastatic liver tumors. The patient's performance states were quite well, and anemia progressed repeatedly despite blood transfusion. Therefore, surgery was scheduled.
Fig. 1.
Gastric endoscopy reveals a solitary, distinctly protruding lesion, with a central depression, at the greater curvature of the middle gastric body.
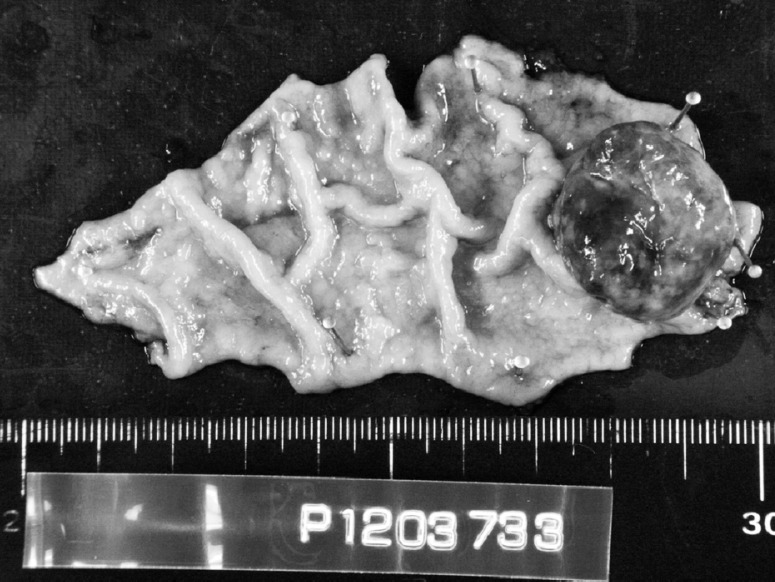
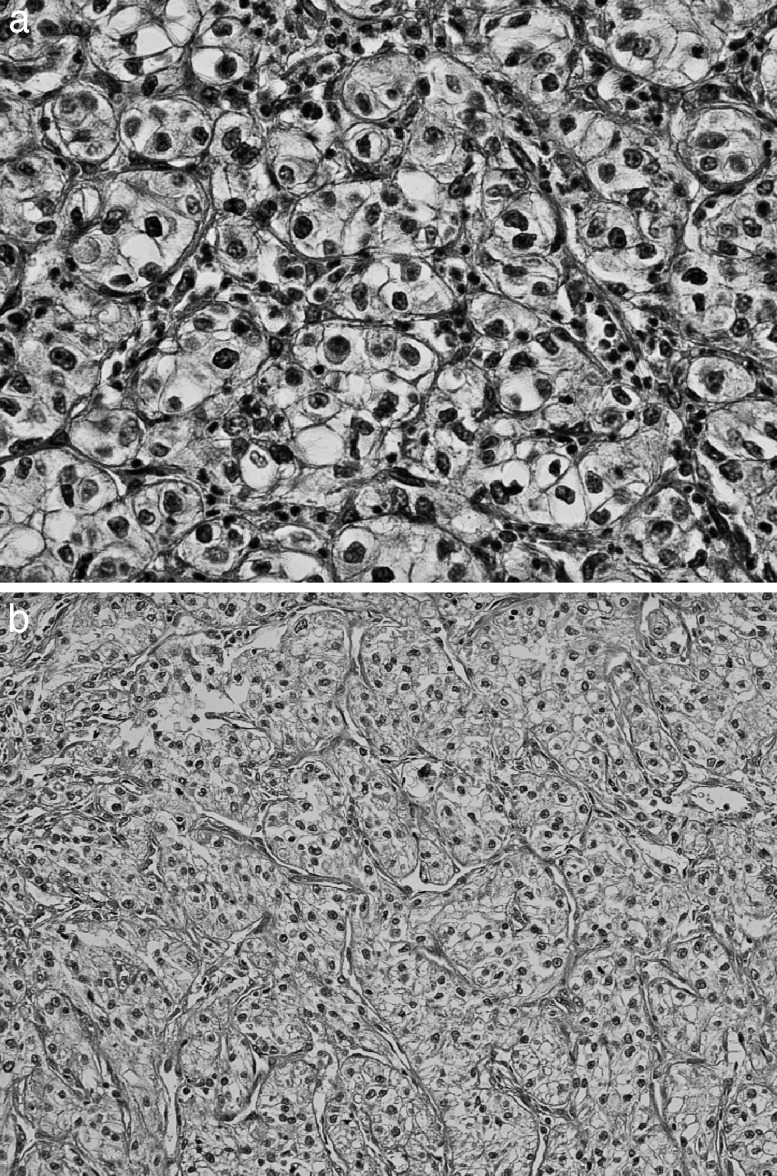
The patient underwent partial resection of the stomach. No peritoneal or liver metastases were found. Macroscopically, the tumor was approximately 2.5 × 2.5 cm in diameter with a central depression and was located at the greater curvature of the gastric body (Fig. 2). Microscopic examination of the resected specimen showed that the gastric tumor was metastasis of RCC (Fig. 3a and 3b). The surgical margin was free of tumor.
Fig. 2.
Macroscopically, the tumor is approximately 2.5 × 2.5 cm in diameter with a central depression.
Fig. 3.
The microscopic appearance of the primary (a) and gastric metastatic tumor (b). Both tumors show typical histologic findings of clear cell carcinoma (hematoxylin and eosin stain; a, ×400; b, ×200).
The patient's postoperative course was uneventful, and he was treated with everolimus, an mTOR inhibitor. However, he died 4 months after surgery because of the progression of brain metastasis.
Discussion
Metastatic tumors of the stomach are rare, with an incidence of 0.2% to 0.7%.1–3 Though RCC shows a propensity to metastasize to lung, bone, and liver,5 metastasis from RCC to the stomach is extremely rare.
The gross appearance of gastric metastasis from RCC usually consists of a polypoid submucosal-like tumor with a central depression. The interval from the diagnosis of primary tumor to the diagnosis of metastatic tumor depends on the primary lesion. Gastric metastasis from lung cancer and malignant melanoma is most commonly diagnosed within 2 years after the diagnosis of the primary tumor.4 On the other hand, gastric metastasis from RCC can be a late event; in one study, the average period from RCC surgery to the diagnosis of metastasis was approximately 7 years.15 Therefore, metastasis from RCC to the stomach may be a slow process.
Patients with RCC with gastric metastasis usually have metastatic lesions in other organ systems as well, and the prognosis for such patients is poor. The impact of resection of the metastatic gastric tumor on survival remains unclear, but the chance of survival may improve if no additional metastatic lesions, aside from the gastric metastasis, are present. In the present case, surgical resection of the gastric lesion might have prevented further bleeding and improved the patient's quality of life until he died of brain metastasis 4 months later. Therefore, we conclude that surgical resection may be useful for the management of gastric metastasis with bleeding.
Namikawa et al23 reported that unfavorable prognostic factors in patients with gastric metastasis from RCC included a grossly protruding type of gastric tumor, the presence of additional metastasis to the other organs, and development of gastric metastasis in <6.3 years from therapy for RCC. By contrast, the number and size of tumors in the stomach had no significant impact on survival. In the present case, a protruding type of gastric metastasis was diagnosed at 1.5 years after the diagnosis of RCC, and the patient died 4 months after the last surgery.
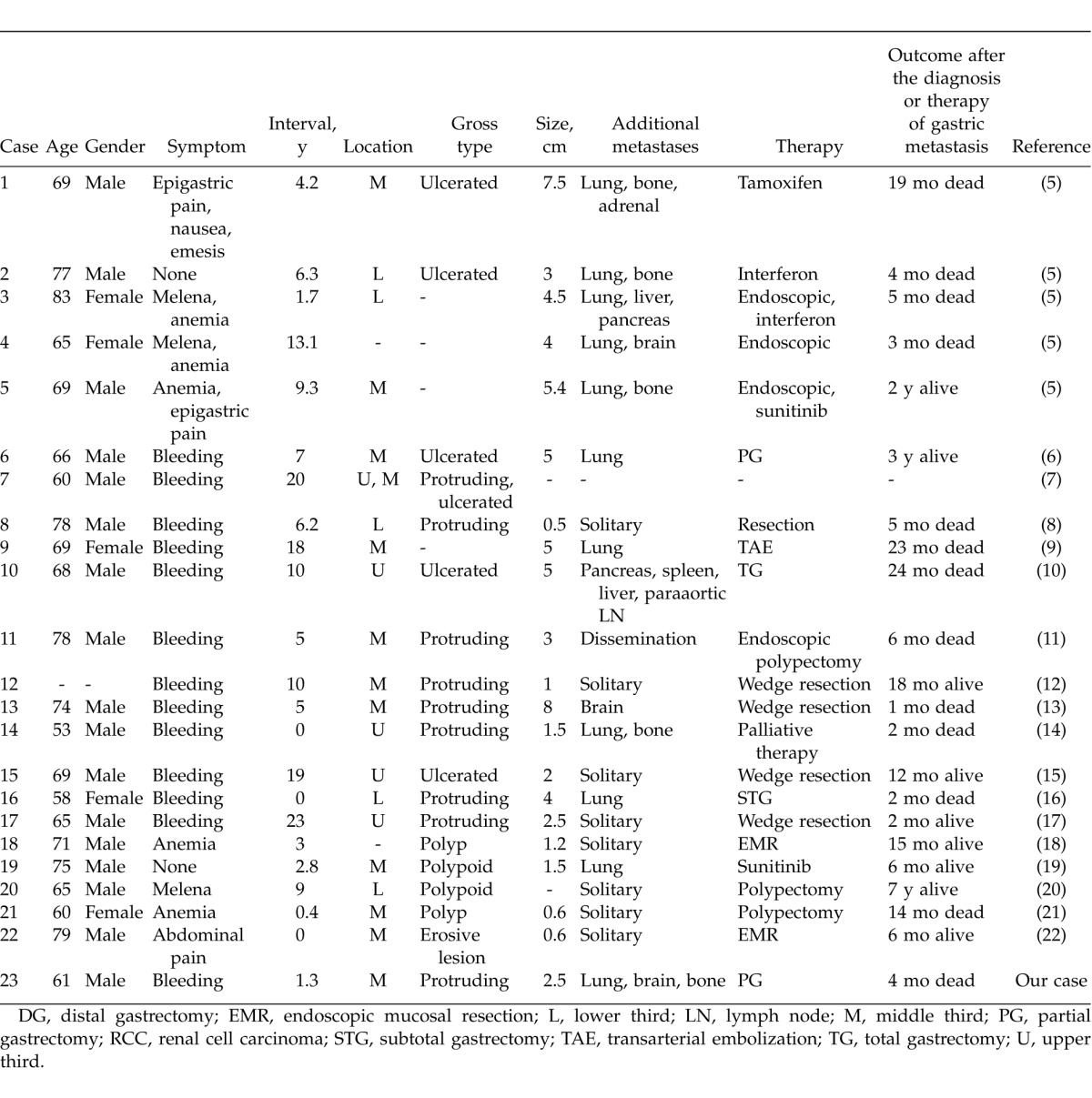
Review of the English literature in the PubMed database with the key words “renal cell carcinoma” and “gastric metastasis” identified 22 patients (Table 1).6–9,11–14,16–22 The mean age of the patients was 68.7 years (age range, 53–83 years; 17 men, 5 women). The median time from the diagnosis of RCC to the detection of gastric metastasis was 7.6 years (range, 0–23 years). Signs and symptoms included gastrointestinal bleeding, melena, anemia, and hematemesis in 19 cases (82.6%). Abdominal pain was observed in 3 cases (13.0%), and 2 patients (8.7%) were asymptomatic. The tumors were located in the middle third of the stomach in 12 cases (52.2%), the upper third in 5 cases (21.7%), and the lower third in 5 cases (21.7%). Grossly, a polypoid lesion was more common than an ulcerated lesion. Concomitant tumor spread to other organs was seen in 14 cases (60.9%), and gastric metastasis without metastasis to other organs was seen in 8 cases (34.8%). Treatment consisted of surgical resection in 9 cases (39.1%); endoscopic polypectomy in 8 cases (34.8%); chemotherapy, including interferon, tamoxifen, or mTOR inhibitors, in 5 cases (21.7%); arterial embolization in 1 case (4.3%); and palliative therapy in 1 case (4.3%). The median survival time was 6 months (range, 1–84 months) after treatment of gastric metastasis.
Table 1.
Reported patients with gastric metastasis from RCC
Molecularly targeted therapies, including sorafenib, temsirolimus, and bevacizumab, are effective for the management of advanced or metastatic RCC.10,24,25 The optimal treatment for gastric metastasis from RCC remains controversial. Namikawa et al23 reported that patients with solitary metastasis arising from RCC had good outcomes following treatment compared with those with multiple metastases. I suggest that surgical resection is the preferred treatment for solitary metastasis and symptomatic disease such as bleeding to help preserve quality of life.
Footnotes
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-commercial License which permits use, distribution, and reproduction in any medium, provided the original work is properly cited, the use is non-commercial and is otherwise in compliance with the license. See: http://creativecommons.org/licenses/by-nc/3.0
References
- 1.Green LK. Hematogenous metastases to the stomach. A review of 67 cases. Cancer. 1990;65(7):1596–600. doi: 10.1002/1097-0142(19900401)65:7<1596::aid-cncr2820650724>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 2.Higgins PM. Pyloric obstructions due to a metastatic deposit from carcinoma of the bronchus. Can J Surg. 1962;5((Oct)):438–441. [PubMed] [Google Scholar]
- 3.Davis GH, Zollinger RW. Metastatic melanoma of the stomach. Am J Surg. 1960;99((Jan)):94–96. doi: 10.1016/0002-9610(60)90258-0. [DOI] [PubMed] [Google Scholar]
- 4.De Palma GD, Masone S, Rega M, Simeoli I, Donisi M, Addeo P, et al. Metastatic tumors to the stomach: clinical and endoscopic features. World J Gastroenterol. 2006;12(45):7326–7328. doi: 10.3748/wjg.v12.i45.7326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pollheimer MJ, Hinterleitner TA, Pollheimer VS, Schlemmer A, Langner C. Renal cell carcinoma metastatic to the stomach: single-centre experience and literature review. BJU Int. 2008;102(3):315–319. doi: 10.1111/j.1464-410X.2008.07617.x. [DOI] [PubMed] [Google Scholar]
- 6.Mascarenhas B, Konety B, Rubin JT. Recurrent metastatic renal cell carcinoma presenting as a bleeding gastric ulcer after a complete response to high-dose interleukin-2 treatment. Urology. 2001;57(1):168. doi: 10.1016/s0090-4295(00)00877-3. [DOI] [PubMed] [Google Scholar]
- 7.Kok Wee L, Shyu RY, Sheu LF, Hsieh TY, Yan JC, Chen PJ. Metastatic renal cell cancer. Gastrointest Endosc. 2004;60(2):265. doi: 10.1016/s0016-5107(04)01542-1. [DOI] [PubMed] [Google Scholar]
- 8.Kobayashi O, Murakami H, Yoshida T, Cho H, Yoshikawa T, Tsuburaya A, et al. Clinical diagnosis of metastatic gastric tumors: clinicopathologic findings and prognosis of nine patients in a single cancer center. World J Surg. 2004;28(6):548–551. doi: 10.1007/s00268-004-7216-8. [DOI] [PubMed] [Google Scholar]
- 9.Lamb GW, Moss J, Edwards R, Aitchison M. Case report: octreotide as an adjunct to embolisation in the management of recurrent bleeding upper gastrointestinal metastases from primary renal cell cancer. Int Urol Nephrol. 2005;37(4):691–693. doi: 10.1007/s11255-005-0251-z. [DOI] [PubMed] [Google Scholar]
- 10.Hudes G, Carducci M, Tomczak P, Dutcher J, Figlin R, Kapoor A, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med. 2007;356(22):2271–2281. doi: 10.1056/NEJMoa066838. [DOI] [PubMed] [Google Scholar]
- 11.Pezzoli A, Matarese V, Boccia S, Simone L, Gullini S. Gastrointestinal bleeding from gastric metastasis of renal cell carcinoma, treated by endoscopic polypectomy. Endoscopy. 2007;39((suppl 1)):E52. doi: 10.1055/s-2006-945127. [DOI] [PubMed] [Google Scholar]
- 12.Saidi RF, Remine SG. Isolated gastric metastasis from renal cell carcinoma 10 years after radical nephrectomy. J Gastroenterol Hepatol. 2007;22(1):143–144. doi: 10.1111/j.1440-1746.2006.04335.x. [DOI] [PubMed] [Google Scholar]
- 13.Yamamoto D, Hamada Y, Okazaki S, Kawakami K, Kanzaki S, Yamamoto C, et al. Metastatic gastric tumor from renal cell carcinoma. Gastric Cancer. 2009;12(3):170–173. doi: 10.1007/s10120-009-0519-6. [DOI] [PubMed] [Google Scholar]
- 14.Kibria R, Sharma K, Ali SA, Rao P. Upper gastrointestinal bleeding revealing the stomach metastases of renal cell carcinoma. J Gastrointest Cancer. 2009;40((1–2)):51–54. doi: 10.1007/s12029-009-9074-y. [DOI] [PubMed] [Google Scholar]
- 15.Sugasawa H, Ichikura T, Ono S, Tsujimoto H, Hiraki S, Sakamoto N, et al. Isolated gastric metastasis from renal cell carcinoma 19 years after radical nephrectomy. Int J Clin Oncol. 2010;15(2):196–200. doi: 10.1007/s10147-010-0025-1. [DOI] [PubMed] [Google Scholar]
- 16.Tiwari P, Tiwari A, Vijay M, Kumar S, Kundu AK. Upper gastro-intestinal bleeding—rare presentation of renal cell carcinoma. Urol Ann. 2010;2(3):127–129. doi: 10.4103/0974-7796.68864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dayes LA, Rouhe SA, Barnes RW. Excision of multiple intracranial metastatic hypernephroma. Report of a case with a 7-year survival. J Neurosurg. 1977;46(4):533–535. doi: 10.3171/jns.1977.46.4.0533. [DOI] [PubMed] [Google Scholar]
- 18.Siriwardana HP, Harvey MH, Kadirkamanathan SS, Tang B, Kamel D, Radzioch R. Endoscopic mucosal resection of a solitary metastatic tumor in the stomach: a case report. Surg Laparosc Endosc Percutan Tech. 2012;22(3):e132–e134. doi: 10.1097/SLE.0b013e318247d13c. [DOI] [PubMed] [Google Scholar]
- 19.Garcia-Campelo R, Quindos M, Vazquez DD, Lopez MR, Carral A, Calvo OF, et al. Renal cell carcinoma: complete pathological response in a patient with gastric metastasis of renal cell carcinoma. Anticancer Drugs. 2010;21((suppl 1)):S13–S15. doi: 10.1097/01.cad.0000361530.51675.60. [DOI] [PubMed] [Google Scholar]
- 20.Eslick GD, Kalantar JS. Gastric metastasis in renal cell carcinoma: a case report and systematic review. J Gastrointest Cancer. 2011;42(4):296–301. doi: 10.1007/s12029-010-9165-9. [DOI] [PubMed] [Google Scholar]
- 21.Xu J, Latif S, Wei S. Metastatic renal cell carcinoma presenting as gastric polyps: a case report and review of the literature. Int J Surg Case Rep. 2012;3(12):601–604. doi: 10.1016/j.ijscr.2012.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim MY, Jung HY, Choi KD, Song HJ, Lee JH. Kim do H et al. Solitary synchronous metastatic gastric cancer arising from t1b renal cell carcinoma: a case report and systematic review. Gut Liver. 2012;6(3):388–394. doi: 10.5009/gnl.2012.6.3.388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Namikawa T, Munekage M, Kitagawa H, Okabayashi T, Kobayashi M, Hanazaki K. Metastatic gastric tumors arising from renal cell carcinoma: clinical characteristics and outcomes of this uncommon disease. Oncol Lett. 2012;4(4):631–636. doi: 10.3892/ol.2012.807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356(2):125–134. doi: 10.1056/NEJMoa060655. [DOI] [PubMed] [Google Scholar]
- 25.Escudier B, Pluzanska A, Koralewski P, Ravaud A, Bracarda S, Szczylik C, et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet. 2007;370(9605):2103–2111. doi: 10.1016/S0140-6736(07)61904-7. [DOI] [PubMed] [Google Scholar]