Abstract
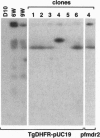
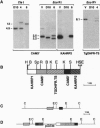
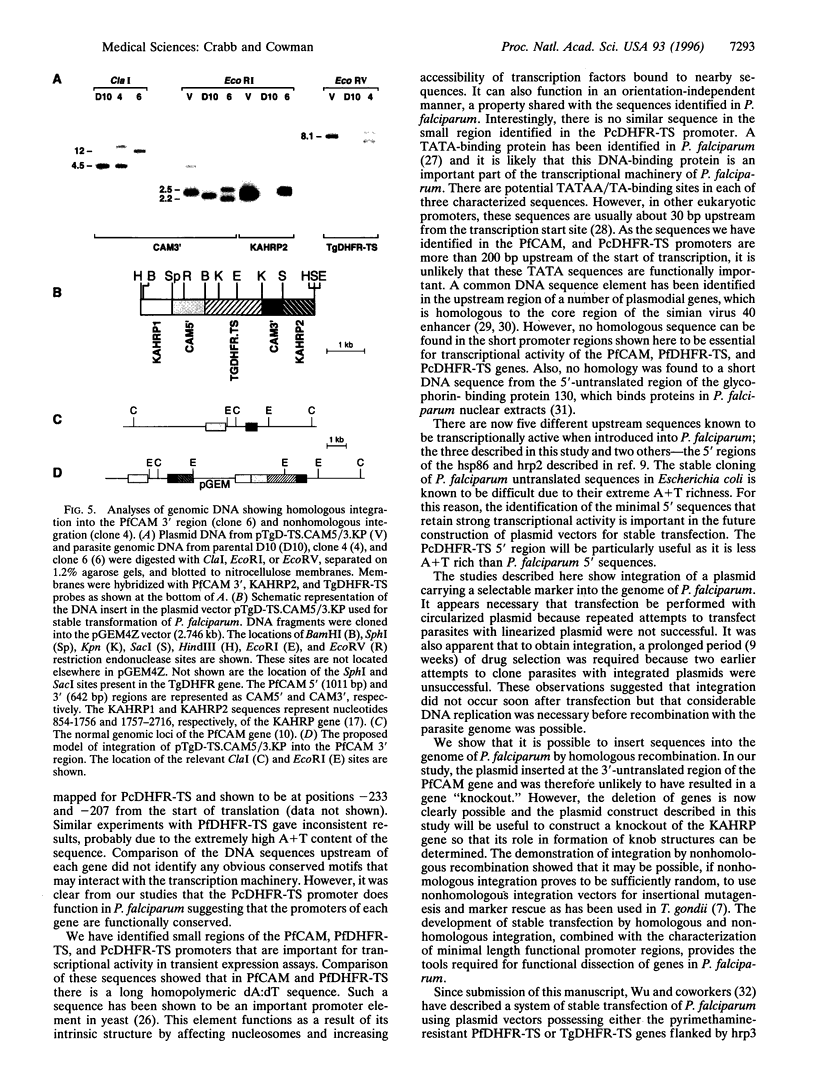
Genetic studies of the protozoan parasite Plasmodium falciparum have been severely limited by the inability to introduce or modify genes. In this paper we describe a system of stable transfection of P. falciparum using a Toxoplasma gondii dihydrofolate reductase-thymidylate synthase gene, modified to confer resistance to pyrimethamine, as a selectable marker. This gene was placed under the transcriptional control of the P. falciparum calmodulin gene flanking sequences. Transfected parasites generally maintained plasmids episomally while under selection; however, parasite clones containing integrated forms of the plasmid were obtained. Integration occurred by both homologous and nonhomologous recombination. In addition to the flanking sequence of the P. falciparum calmodulin gene, the 5' sequences of the P. falciparum and P. chabaudi dihydrofolate reductase-thymidylate synthase genes were also shown to be transcriptionally active in P. falciparum. The minimal 5' sequence that possessed significant transcriptional activity was determined for each gene and short sequences containing important transcriptional control elements were identified. These sequences will provide considerable flexibility in the future construction of plasmid vectors to be used for the expression of foreign genes or for the deletion or modification of P. falciparum genes of interest.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bzik D. J., Li W. B., Horii T., Inselburg J. Molecular cloning and sequence analysis of the Plasmodium falciparum dihydrofolate reductase-thymidylate synthase gene. Proc Natl Acad Sci U S A. 1987 Dec;84(23):8360–8364. doi: 10.1073/pnas.84.23.8360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu G., Vollrath D., Davis R. W. Separation of large DNA molecules by contour-clamped homogeneous electric fields. Science. 1986 Dec 19;234(4783):1582–1585. doi: 10.1126/science.3538420. [DOI] [PubMed] [Google Scholar]
- Coppel R. L., Bianco A. E., Culvenor J. G., Crewther P. E., Brown G. V., Anders R. F., Kemp D. J. A cDNA clone expressing a rhoptry protein of Plasmodium falciparum. Mol Biochem Parasitol. 1987 Aug;25(1):73–81. doi: 10.1016/0166-6851(87)90020-x. [DOI] [PubMed] [Google Scholar]
- Corcoran L. M., Forsyth K. P., Bianco A. E., Brown G. V., Kemp D. J. Chromosome size polymorphisms in Plasmodium falciparum can involve deletions and are frequent in natural parasite populations. Cell. 1986 Jan 17;44(1):87–95. doi: 10.1016/0092-8674(86)90487-3. [DOI] [PubMed] [Google Scholar]
- Cowman A. F., Galatis D. Plasmodium falciparum: the calmodulin gene is not amplified or overexpressed in chloroquine resistant or sensitive isolates. Exp Parasitol. 1991 Oct;73(3):269–275. doi: 10.1016/0014-4894(91)90098-h. [DOI] [PubMed] [Google Scholar]
- Cowman A. F., Lew A. M. Antifolate drug selection results in duplication and rearrangement of chromosome 7 in Plasmodium chabaudi. Mol Cell Biol. 1989 Nov;9(11):5182–5188. doi: 10.1128/mcb.9.11.5182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowman A. F., Morry M. J., Biggs B. A., Cross G. A., Foote S. J. Amino acid changes linked to pyrimethamine resistance in the dihydrofolate reductase-thymidylate synthase gene of Plasmodium falciparum. Proc Natl Acad Sci U S A. 1988 Dec;85(23):9109–9113. doi: 10.1073/pnas.85.23.9109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruz A., Beverley S. M. Gene replacement in parasitic protozoa. Nature. 1990 Nov 8;348(6297):171–173. doi: 10.1038/348171a0. [DOI] [PubMed] [Google Scholar]
- Donald R. G., Roos D. S. Stable molecular transformation of Toxoplasma gondii: a selectable dihydrofolate reductase-thymidylate synthase marker based on drug-resistance mutations in malaria. Proc Natl Acad Sci U S A. 1993 Dec 15;90(24):11703–11707. doi: 10.1073/pnas.90.24.11703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foote S. J., Cowman A. F. The mode of action and the mechanism of resistance to antimalarial drugs. Acta Trop. 1994 Mar;56(2-3):157–171. doi: 10.1016/0001-706x(94)90061-2. [DOI] [PubMed] [Google Scholar]
- Iyer V., Struhl K. Poly(dA:dT), a ubiquitous promoter element that stimulates transcription via its intrinsic DNA structure. EMBO J. 1995 Jun 1;14(11):2570–2579. doi: 10.1002/j.1460-2075.1995.tb07255.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly J. M., Ward H. M., Miles M. A., Kendall G. A shuttle vector which facilitates the expression of transfected genes in Trypanosoma cruzi and Leishmania. Nucleic Acids Res. 1992 Aug 11;20(15):3963–3969. doi: 10.1093/nar/20.15.3963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemp D. J., Cowman A. F., Walliker D. Genetic diversity in Plasmodium falciparum. Adv Parasitol. 1990;29:75–149. doi: 10.1016/s0065-308x(08)60105-0. [DOI] [PubMed] [Google Scholar]
- Kim K., Soldati D., Boothroyd J. C. Gene replacement in Toxoplasma gondii with chloramphenicol acetyltransferase as selectable marker. Science. 1993 Nov 5;262(5135):911–914. doi: 10.1126/science.8235614. [DOI] [PubMed] [Google Scholar]
- Lambros C., Vanderberg J. P. Synchronization of Plasmodium falciparum erythrocytic stages in culture. J Parasitol. 1979 Jun;65(3):418–420. [PubMed] [Google Scholar]
- Lanzer M., de Bruin D., Ravetch J. V. A sequence element associated with the Plasmodium falciparum KAHRP gene is the site of developmentally regulated protein-DNA interactions. Nucleic Acids Res. 1992 Jun 25;20(12):3051–3056. doi: 10.1093/nar/20.12.3051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanzer M., de Bruin D., Ravetch J. V. Transcription mapping of a 100 kb locus of Plasmodium falciparum identifies an intergenic region in which transcription terminates and reinitiates. EMBO J. 1992 May;11(5):1949–1955. doi: 10.1002/j.1460-2075.1992.tb05248.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeBowitz J. H., Coburn C. M., McMahon-Pratt D., Beverley S. M. Development of a stable Leishmania expression vector and application to the study of parasite surface antigen genes. Proc Natl Acad Sci U S A. 1990 Dec;87(24):9736–9740. doi: 10.1073/pnas.87.24.9736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAndrew M. B., Read M., Sims P. F., Hyde J. E. Characterisation of the gene encoding an unusually divergent TATA-binding protein (TBP) from the extremely A+T-rich human malaria parasite Plasmodium falciparum. Gene. 1993 Feb 28;124(2):165–171. doi: 10.1016/0378-1119(93)90390-o. [DOI] [PubMed] [Google Scholar]
- Peterson D. S., Walliker D., Wellems T. E. Evidence that a point mutation in dihydrofolate reductase-thymidylate synthase confers resistance to pyrimethamine in falciparum malaria. Proc Natl Acad Sci U S A. 1988 Dec;85(23):9114–9118. doi: 10.1073/pnas.85.23.9114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pologe L. G., Ravetch J. V. A chromosomal rearrangement in a P. falciparum histidine-rich protein gene is associated with the knobless phenotype. 1986 Jul 31-Aug 6Nature. 322(6078):474–477. doi: 10.1038/322474a0. [DOI] [PubMed] [Google Scholar]
- Robson K. J., Jennings M. W. The structure of the calmodulin gene of Plasmodium falciparum. Mol Biochem Parasitol. 1991 May;46(1):19–34. doi: 10.1016/0166-6851(91)90195-c. [DOI] [PubMed] [Google Scholar]
- Rubio J. P., Cowman A. F. Plasmodium falciparum: the pfmdr2 protein is not overexpressed in chloroquine-resistant isolates of the malaria parasite. Exp Parasitol. 1994 Sep;79(2):137–147. doi: 10.1006/expr.1994.1073. [DOI] [PubMed] [Google Scholar]
- Ruiz i Altaba A., Ozaki L. S., Gwadz R. W., Godson G. N. Organization and expression of the Plasmodium knowlesi circumsporozoite antigen gene. Mol Biochem Parasitol. 1987 Apr;23(3):233–245. doi: 10.1016/0166-6851(87)90030-2. [DOI] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Trager W., Jensen J. B. Human malaria parasites in continuous culture. Science. 1976 Aug 20;193(4254):673–675. doi: 10.1126/science.781840. [DOI] [PubMed] [Google Scholar]
- Triglia T., Stahl H. D., Crewther P. E., Scanlon D., Brown G. V., Anders R. F., Kemp D. J. The complete sequence of the gene for the knob-associated histidine-rich protein from Plasmodium falciparum. EMBO J. 1987 May;6(5):1413–1419. doi: 10.1002/j.1460-2075.1987.tb02382.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiley S. R., Kraus R. J., Mertz J. E. Functional binding of the "TATA" box binding component of transcription factor TFIID to the -30 region of TATA-less promoters. Proc Natl Acad Sci U S A. 1992 Jul 1;89(13):5814–5818. doi: 10.1073/pnas.89.13.5814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y., Kirkman L. A., Wellems T. E. Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc Natl Acad Sci U S A. 1996 Feb 6;93(3):1130–1134. doi: 10.1073/pnas.93.3.1130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y., Sifri C. D., Lei H. H., Su X. Z., Wellems T. E. Transfection of Plasmodium falciparum within human red blood cells. Proc Natl Acad Sci U S A. 1995 Feb 14;92(4):973–977. doi: 10.1073/pnas.92.4.973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Dijk M. R., Janse C. J., Waters A. P. Expression of a Plasmodium gene introduced into subtelomeric regions of Plasmodium berghei chromosomes. Science. 1996 Feb 2;271(5249):662–665. doi: 10.1126/science.271.5249.662. [DOI] [PubMed] [Google Scholar]
- van Dijk M. R., Waters A. P., Janse C. J. Stable transfection of malaria parasite blood stages. Science. 1995 Jun 2;268(5215):1358–1362. doi: 10.1126/science.7761856. [DOI] [PubMed] [Google Scholar]