Abstract
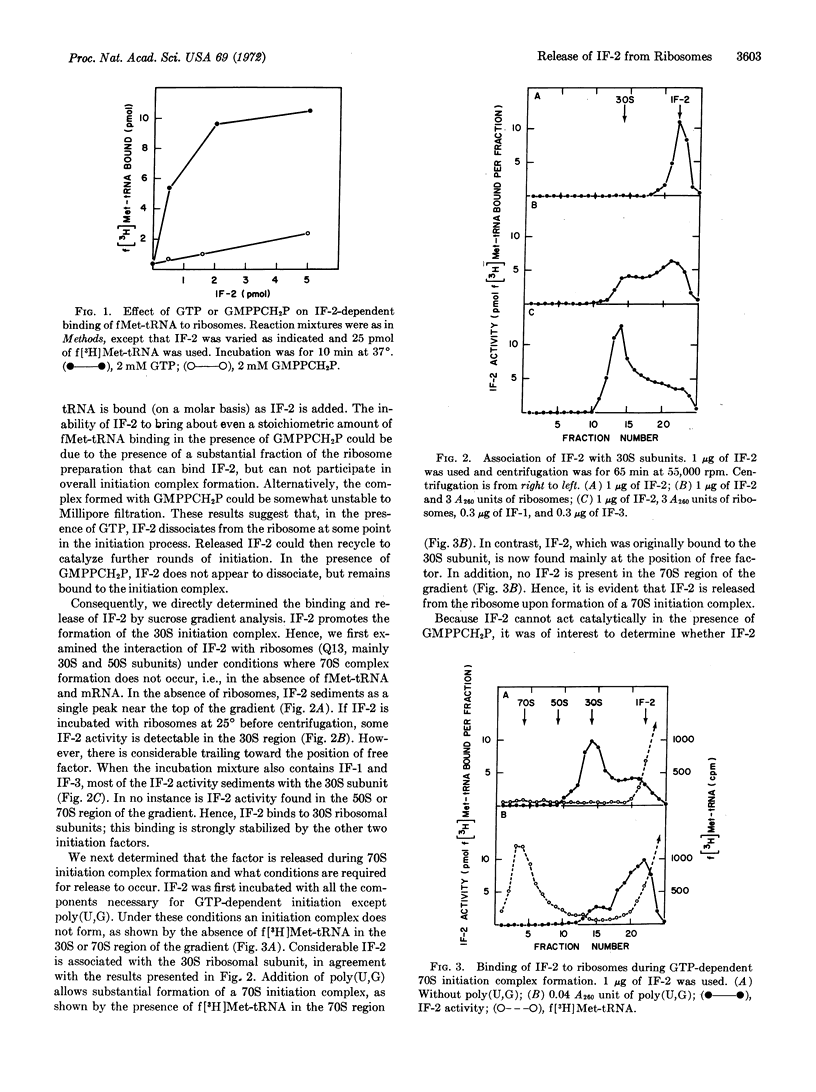
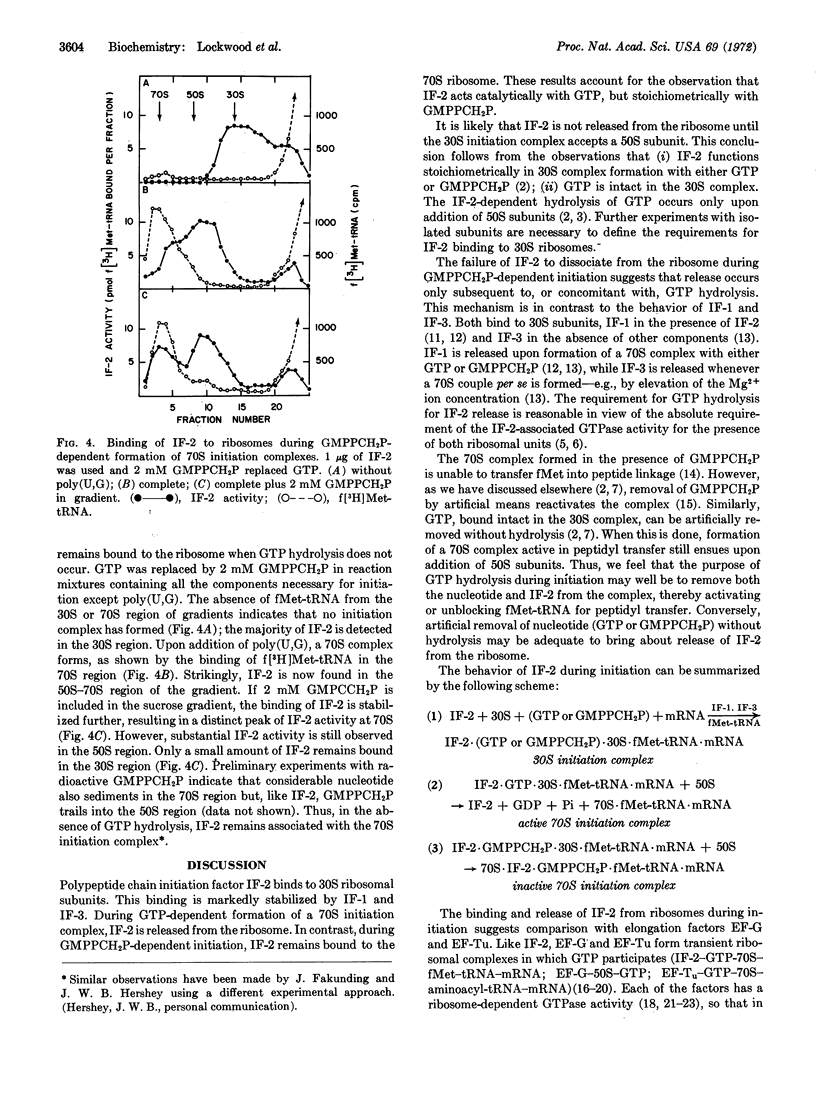
Polypeptide chain initiation factor IF-2 binds to 30S ribosomal subunits. This binding is enhanced by IF-1 and IF-3. During GTP-dependent formation of a 70S initiation complex, IF-2 is released from the ribosome. During 70S initiation complex formation dependent on the methylene analogue of GTP, GMPPCH2P, IF-2 is not released, but remains bound to the 70S ribosome. This result suggests that IF-2 release requires GTP hydrolysis. In agreement with this presumed requirement, IF-2 functions catalytically with GTP, but stoichiometrically with GMPPCH2P, in bringing about 70S initiation complex formation.
Keywords: protein synthesis, E. coli, GTP hydrolysis, ribosomes, fMet-tRNA
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bodley J. W., Lin L. Interaction of E. coli G factor with the 50S ribosomal subunit. Nature. 1970 Jul 4;227(5253):60–61. doi: 10.1038/227060a0. [DOI] [PubMed] [Google Scholar]
- Bodley J. W., Zieve F. J., Lin L., Zieve S. T. Studies on translocation. 3. Conditions necessary for the formation and detection of a stable ribosome-G factor-guanosine diphosphate complex in the presence of fusidic acid. J Biol Chem. 1970 Nov 10;245(21):5656–5661. [PubMed] [Google Scholar]
- Brot N., Spears C., Weissbach H. The formation of a complex containing ribosomes, transfer factor G and A guanosine nucleotide. Biochem Biophys Res Commun. 1969 Mar 31;34(6):843–848. doi: 10.1016/0006-291x(69)90257-5. [DOI] [PubMed] [Google Scholar]
- Dubnoff J. S., Lockwood A. H., Maitra U. Biochemical characterization and messenger specificity of polypeptide chain initiation factors from Escherichia coli. Arch Biochem Biophys. 1972 Apr;149(2):528–540. doi: 10.1016/0003-9861(72)90352-9. [DOI] [PubMed] [Google Scholar]
- Dubnoff J. S., Lockwood A. H., Maitra U. Studies on the role of guanosine triphosphate in polypeptide chain initiation in Escherichia coli. J Biol Chem. 1972 May 10;247(9):2884–2894. [PubMed] [Google Scholar]
- Dubnoff J. S., Maitra U. Characterization of the ribosome-dependent guanosine triphosphatase activity of polypeptide chain initiation factor IF 2. J Biol Chem. 1972 May 10;247(9):2876–2883. [PubMed] [Google Scholar]
- Gordon J. Hydrolysis of guanosine 5'-triphosphate associated wh binding of aminoacyl transfer ribonucleic acid to ribosomes. J Biol Chem. 1969 Oct 25;244(20):5680–5686. [PubMed] [Google Scholar]
- Hershey J. W., Dewey K. F., Thach R. E. Purification and properties of initiation factor f-1. Nature. 1969 Jun 7;222(5197):944–947. doi: 10.1038/222944a0. [DOI] [PubMed] [Google Scholar]
- Kolakofsky D., Dewey K. F., Hershey J. W., Thach R. E. Guanosine 5'-triphosphatase activity of initiation factor f2. Proc Natl Acad Sci U S A. 1968 Nov;61(3):1066–1070. doi: 10.1073/pnas.61.3.1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolakofsky D., Dewey K., Thach R. E. Purification and properties of initiation factor f2. Nature. 1969 Aug 16;223(5207):694–697. doi: 10.1038/223694a0. [DOI] [PubMed] [Google Scholar]
- Kolakofsky D., Ohta T., Thach R. E. Junction of the 50S ribosomal subunit with the 30S initiation complex. Nature. 1968 Oct 19;220(5164):244–247. doi: 10.1038/220244a0. [DOI] [PubMed] [Google Scholar]
- Lockwood A. H., Hattman S., Dubnoff J. S., Maitra U. Formation and properties of T factor complexes. J Biol Chem. 1971 May 10;246(9):2936–2947. [PubMed] [Google Scholar]
- Lucas-Lenard J., Haenni A. L. Requirement of granosine 5'-triphosphate for ribosomal binding of aminoacyl-SRNA. Proc Natl Acad Sci U S A. 1968 Feb;59(2):554–560. doi: 10.1073/pnas.59.2.554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucas-Lenard J. Protein biosynthesis. Annu Rev Biochem. 1971;40:409–448. doi: 10.1146/annurev.bi.40.070171.002205. [DOI] [PubMed] [Google Scholar]
- NIRENBERG M., LEDER P. RNA CODEWORDS AND PROTEIN SYNTHESIS. THE EFFECT OF TRINUCLEOTIDES UPON THE BINDING OF SRNA TO RIBOSOMES. Science. 1964 Sep 25;145(3639):1399–1407. doi: 10.1126/science.145.3639.1399. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y., Lipmann F. The interrelationship between guanosine triphosphatase and amino acid polymerization. Arch Biochem Biophys. 1966 Sep 26;116(1):344–351. doi: 10.1016/0003-9861(66)90040-3. [DOI] [PubMed] [Google Scholar]
- Ravel J. M., Shorey R. L., Shive W. The composition of the active intermediate in the transfer of aminoacyl-RNA to ribosomes. Biochem Biophys Res Commun. 1968 Jul 11;32(1):9–14. doi: 10.1016/0006-291x(68)90418-x. [DOI] [PubMed] [Google Scholar]
- Sabol S., Ochoa S. Ribosomal binding of labelled initiation factor F3. Nat New Biol. 1971 Dec 22;234(51):233–236. doi: 10.1038/newbio234233a0. [DOI] [PubMed] [Google Scholar]
- Shorey R. L., Ravel J. M., Garner C. W., Shive W. Formation and properties of the aminoacyl transfer ribonucleic acid-guanosine triphosphate-protein complex. J Biol Chem. 1969 Sep 10;244(17):4555–4564. [PubMed] [Google Scholar]
- Skoultchi A., Ono Y., Waterson J., Lengyel P. Peptide chain elongation. Cold Spring Harb Symp Quant Biol. 1969;34:437–454. doi: 10.1101/sqb.1969.034.01.050. [DOI] [PubMed] [Google Scholar]
- Skoultchi A., Ono Y., Waterson J., Lengyel P. Peptide chain elongation; indications for the binding of an amino acid polymerization factor, guanosine 5'-triphosphate--aminoacyl transfer ribonucleic acid complex to the messenger-ribosome complex. Biochemistry. 1970 Feb 3;9(3):508–514. doi: 10.1021/bi00805a009. [DOI] [PubMed] [Google Scholar]
- Thach R. E., Hershey J. W., Kolakofsky D., Dewey K. F., Remold-O'Donnell E. Purification and properties of initiation factors F1 and F2. Cold Spring Harb Symp Quant Biol. 1969;34:277–284. doi: 10.1101/sqb.1969.034.01.033. [DOI] [PubMed] [Google Scholar]
- Thach S. S., Thach R. E. 1 molecule of guanosine triphosphate is present in each 30S initiation complex. Nat New Biol. 1971 Feb 17;229(7):219–221. doi: 10.1038/newbio229219a0. [DOI] [PubMed] [Google Scholar]



