Abstract
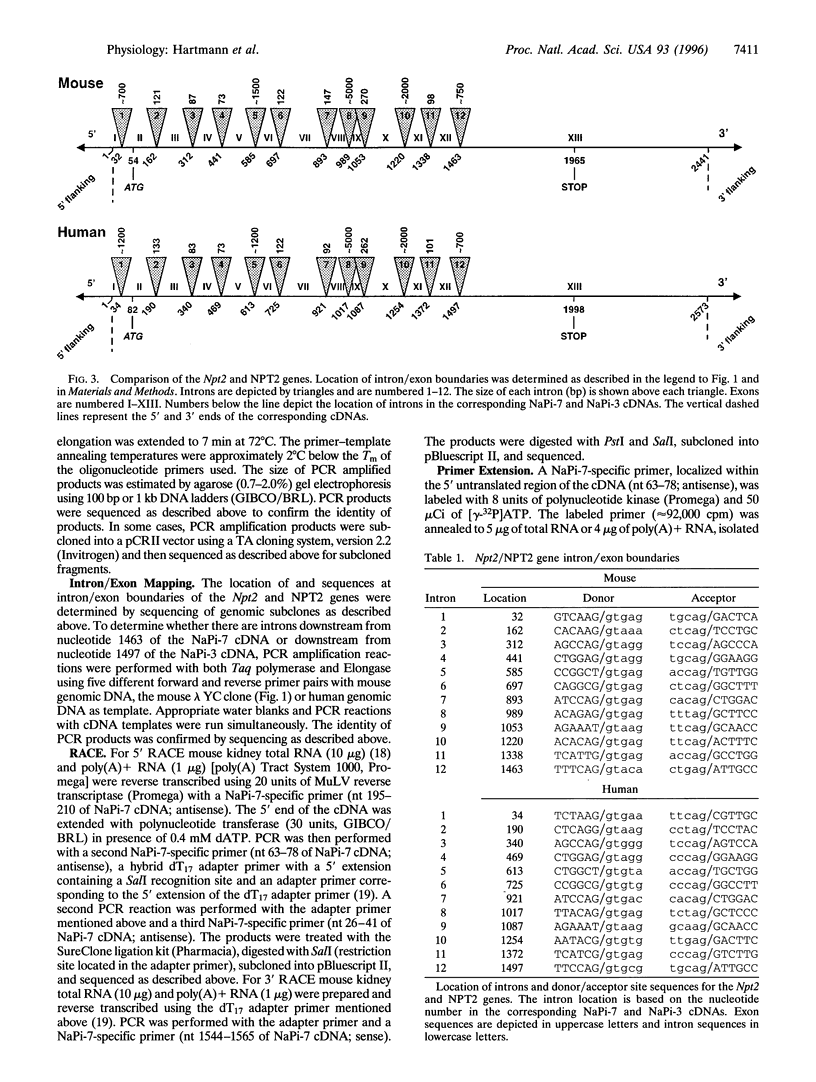
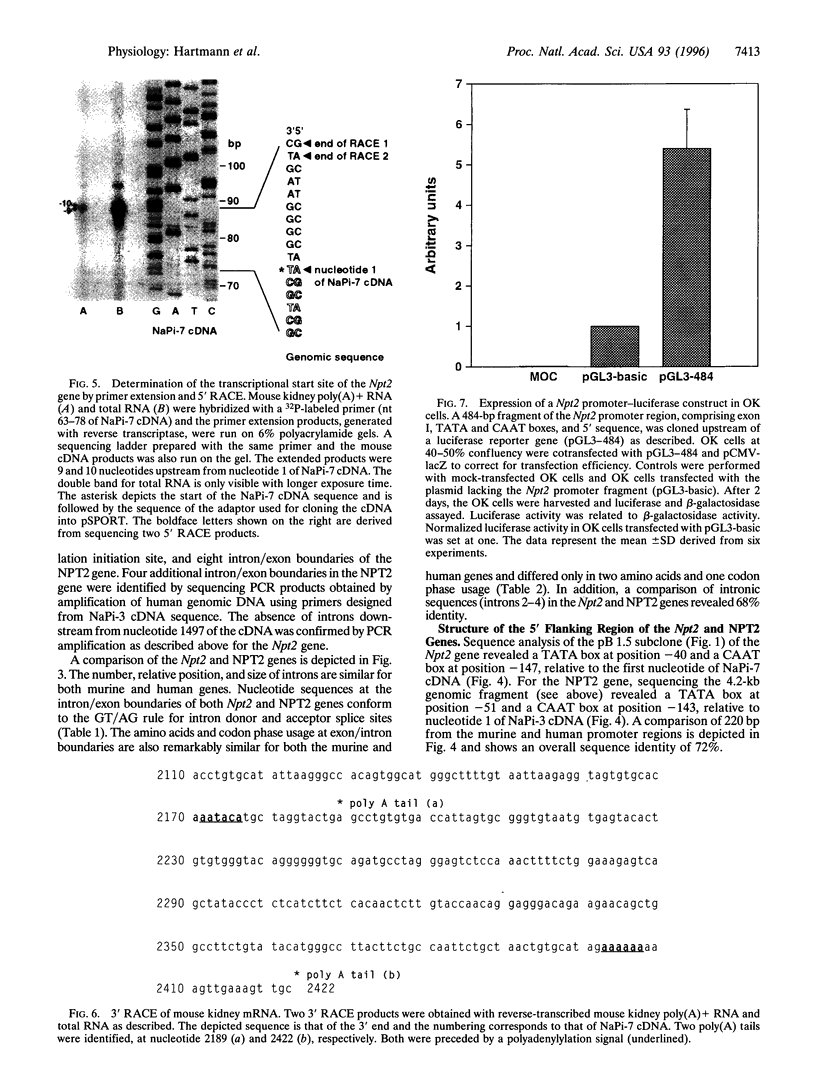
Na+-phosphate (Pi) cotransport across the renal brush border membrane is the rate limiting step in the overall reabsorption of filtered Pi. Murine and human renal-specific cDNAs (NaPi-7 and NaPi-3, respectively) related to this cotransporter activity (type II Na+-Pi cotransporter) have been cloned. We now report the cloning and characterization of the corresponding mouse (Npt2) and human (NPT2) genes. The genes were cloned by screening mouse genomic and human chromosome 5-specific libraries, respectively. Both genes are approximately 16 kb and are comprised of 13 exons and 12 introns, the junctions of which conform to donor and acceptor site consensus sequences. Putative CAAT and TATA boxes are located, respectively, at positions -147 and -40 of the Npt2 gene and -143 and -51 of the NPT2 gene, relative to nucleotide 1 of the corresponding cDNAs. The translation initiation site is within exon 2 of both genes. The first 220 bp of the mouse and human promoter regions exhibit 72% identity. Two transcription start sites (at positions -9 and - 10 with respect to nucleotide 1 of NaPi-7 cDNA) and two polyadenylylation signals were identified in the Npt2 gene by primer extension, 5' and 3' rapid amplification of cDNA ends (RACE). A 484-bp 5' flanking region of the Npt2 gene, comprising the CAAT box, TATA box, and exon 1, was cloned upstream of a luciferase reporter gene; this construct significantly stimulated luciferase gene expression, relative to controls, when transiently transfected into OK cells, a renal cell line expressing type II Na+ -Pi cotransporter activity. The present data provide a basis for detailed analysis of cis and trans elements involved in the regulation of Npt2/NPT2 gene transcription and facilitate screening for mutations in the NPT2 gene in patients with autosomally inherited disorders of renal Pi reabsorption.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bertran J., Werner A., Stange G., Markovich D., Biber J., Testar X., Zorzano A., Palacin M., Murer H. Expression of Na(+)-independent amino acid transport in Xenopus laevis oocytes by injection of rabbit kidney cortex mRNA. Biochem J. 1992 Feb 1;281(Pt 3):717–723. doi: 10.1042/bj2810717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caverzasio J., Bonjour J. P. Growth factors and renal regulation of phosphate transport. Pediatr Nephrol. 1993 Dec;7(6):802–806. doi: 10.1007/BF01213364. [DOI] [PubMed] [Google Scholar]
- Cramer W. A., Engelman D. M., Von Heijne G., Rees D. C. Forces involved in the assembly and stabilization of membrane proteins. FASEB J. 1992 Dec;6(15):3397–3402. doi: 10.1096/fasebj.6.15.1464373. [DOI] [PubMed] [Google Scholar]
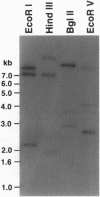
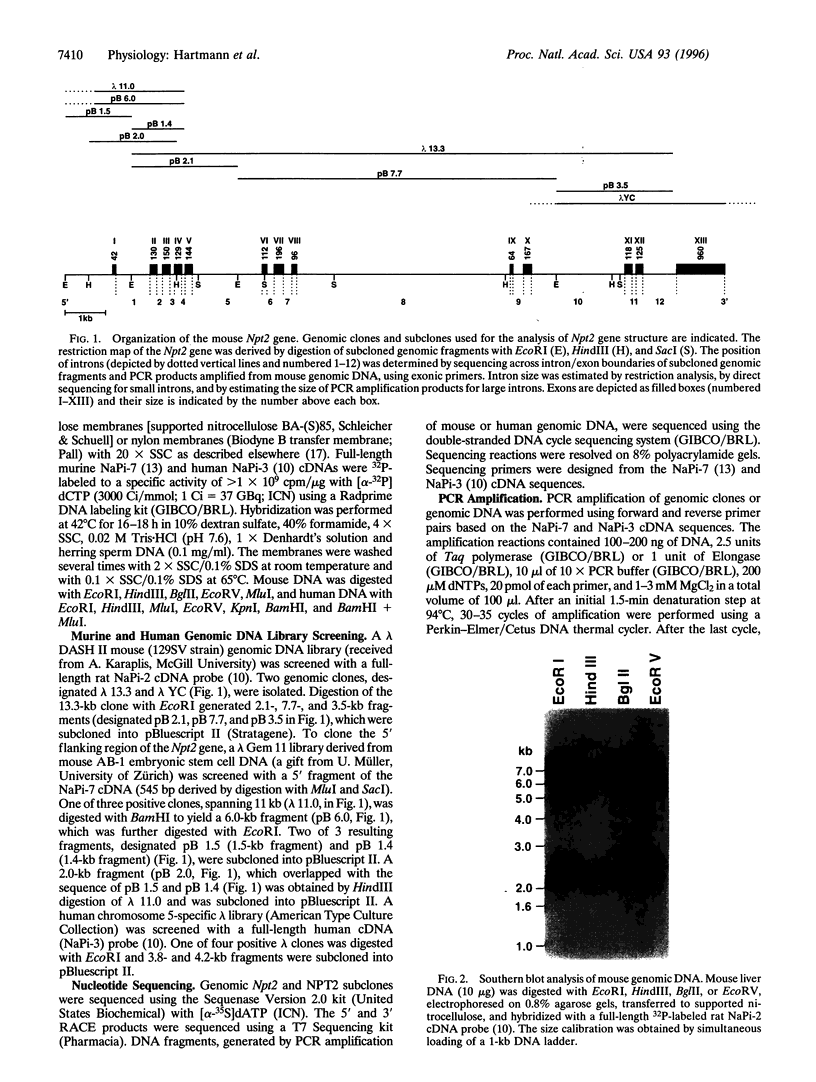
- Custer M., Lötscher M., Biber J., Murer H., Kaissling B. Expression of Na-P(i) cotransport in rat kidney: localization by RT-PCR and immunohistochemistry. Am J Physiol. 1994 May;266(5 Pt 2):F767–F774. doi: 10.1152/ajprenal.1994.266.5.F767. [DOI] [PubMed] [Google Scholar]
- Gmaj P., Murer H. Cellular mechanisms of inorganic phosphate transport in kidney. Physiol Rev. 1986 Jan;66(1):36–70. doi: 10.1152/physrev.1986.66.1.36. [DOI] [PubMed] [Google Scholar]
- Hartmann C. M., Wagner C. A., Busch A. E., Markovich D., Biber J., Lang F., Murer H. Transport characteristics of a murine renal Na/Pi-cotransporter. Pflugers Arch. 1995 Sep;430(5):830–836. doi: 10.1007/BF00386183. [DOI] [PubMed] [Google Scholar]
- Hartmann E., Rapoport T. A., Lodish H. F. Predicting the orientation of eukaryotic membrane-spanning proteins. Proc Natl Acad Sci U S A. 1989 Aug;86(15):5786–5790. doi: 10.1073/pnas.86.15.5786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein P., Kanehisa M., DeLisi C. The detection and classification of membrane-spanning proteins. Biochim Biophys Acta. 1985 May 28;815(3):468–476. doi: 10.1016/0005-2736(85)90375-x. [DOI] [PubMed] [Google Scholar]
- Kos C. H., Tihy F., Econs M. J., Murer H., Lemieux N., Tenenhouse H. S. Localization of a renal sodium-phosphate cotransporter gene to human chromosome 5q35. Genomics. 1994 Jan 1;19(1):176–177. doi: 10.1006/geno.1994.1034. [DOI] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Magagnin S., Werner A., Markovich D., Sorribas V., Stange G., Biber J., Murer H. Expression cloning of human and rat renal cortex Na/Pi cotransport. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):5979–5983. doi: 10.1073/pnas.90.13.5979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malmström K., Murer H. Parathyroid hormone inhibits phosphate transport in OK cells but not in LLC-PK1 and JTC-12.P3 cells. Am J Physiol. 1986 Jul;251(1 Pt 1):C23–C31. doi: 10.1152/ajpcell.1986.251.1.C23. [DOI] [PubMed] [Google Scholar]
- Mizgala C. L., Quamme G. A. Renal handling of phosphate. Physiol Rev. 1985 Apr;65(2):431–466. doi: 10.1152/physrev.1985.65.2.431. [DOI] [PubMed] [Google Scholar]
- Murer H. Homer Smith Award. Cellular mechanisms in proximal tubular Pi reabsorption: some answers and more questions. J Am Soc Nephrol. 1992 Jun;2(12):1649–1665. doi: 10.1681/ASN.V2121649. [DOI] [PubMed] [Google Scholar]
- Murer H., Werner A., Reshkin S., Wuarin F., Biber J. Cellular mechanisms in proximal tubular reabsorption of inorganic phosphate. Am J Physiol. 1991 May;260(5 Pt 1):C885–C899. doi: 10.1152/ajpcell.1991.260.5.C885. [DOI] [PubMed] [Google Scholar]
- Sorribas V., Markovich D., Hayes G., Stange G., Forgo J., Biber J., Murer H. Cloning of a Na/Pi cotransporter from opossum kidney cells. J Biol Chem. 1994 Mar 4;269(9):6615–6621. [PubMed] [Google Scholar]
- Tenenhouse H. S., Werner A., Biber J., Ma S., Martel J., Roy S., Murer H. Renal Na(+)-phosphate cotransport in murine X-linked hypophosphatemic rickets. Molecular characterization. J Clin Invest. 1994 Feb;93(2):671–676. doi: 10.1172/JCI117019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turk E., Martín M. G., Wright E. M. Structure of the human Na+/glucose cotransporter gene SGLT1. J Biol Chem. 1994 May 27;269(21):15204–15209. [PubMed] [Google Scholar]
- Verri T., Markovich D., Perego C., Norbis F., Stange G., Sorribas V., Biber J., Murer H. Cloning of a rabbit renal Na-Pi cotransporter, which is regulated by dietary phosphate. Am J Physiol. 1995 Apr;268(4 Pt 2):F626–F633. doi: 10.1152/ajprenal.1995.268.4.F626. [DOI] [PubMed] [Google Scholar]