Abstract
Bone and immune system are functionally interconnected. Immune and bone cells derive from same progenitors in the bone marrow, they share a common microenvironment and are being influenced by similar mediators. The evidence on increased bone resorption associated with inappropriate activation of T cells such as during inflammation, is well established. However, the molecular mechanisms beyond this clinical observation have begun to be intensively studied with the advancement of osteoimmunology. Now days, we have firm evidence on the influence of numerous proinflammatory cytokines on bone cells, with the majority of data focused on osteoclasts, the bone resorbing cells. It has been shown that some proinflammatory cytokines could possess osteoclastogenic and/or anti-osteoclastogenic properties and can target osteoclasts directly or via receptor activator of nuclear factor κB (RANK)/RANK ligand(RANKL)/osteoprotegerin (OPG) system. Several studies have reported opposing data regarding (anti)osteoclastogenic properties of these cytokines.
Therefore, the first part of this review is summarizing current evidence on the influence of pro-inflammatory cytokines on osteoclasts and thus on bone resorption. In the second part, the evidence on the role of pro-inflammatory cytokines in osteoporosis and osteoarthritis is reviewed to show that unravelling the mechanisms beyond such complex bone diseases, is almost impossible without considering skeletal and immune systems as an indivisible integrated system.
Keywords: osteoimmunology, cytokines, osteoclasts, RANKL, osteoporosis
Introduction
It is now well established that bone and immune cells are functionally connected. Diverse interactions between bone and immune cells occur within the bone microenvironment. Bone marrow serves as a site for priming naive T cells, recruitment of effector T cells and their proliferation (1,2). Bone and immune cells share the same progenitors residing in bone marrow and are being affected by the same cytokines, influencing hematopoiesis, local immune responses and bone cells as well. In addition, different immune cells such as macrophages, B lymphocytes, mast cells, natural killer cells (NK), etc. have been shown to influence bone cells as well. However, the most powerful players in this regulation are activated T cells. Following successful antigen-specific activation in secondary lymphoid tissues, T cells begin to produce numerous pro-inflammatory cytokines such as interleukin (IL)-2, -4, -5, -6 and -8, tumor necrosis factor-α (TNF-α), transforming growth factor-β (TGF-β), granulocyte-macrophage colony stimulating factor (GM-CSF), interferon γ (IFN-γ), etc. (3). These pro-inflammatory cytokines are soluble mediators with pleiotropic effects on different types of cells. Interestingly, they affect bone cells within a particular bone microenvironment. The pro-inflammatory cytokines can be classified either based on the primary source of their production, i.e. the type of immune cell or their specific targets in bone, i.e. bone cells and thus the process they affect, i.e. bone resorption or formation. Although these cytokines can influence both, the bone forming cells (osteoblasts) and the bone resorbing cells (osteoclasts), the majority of existing data is focused on the influence of immune cells on osteoclasts and thus on bone resorption. Nevertheless we have to take in account that pro-inflammatory cytokines such as TNF-α and IL-17, released from activated T cells, can target osteoblasts as well. The result of such influence is the increased production of pro-inflammatory cytokines and RANKL in osteoblasts and thus the contribution of these cells to bone resorption (4). Depending on their influence on osteoclast differentiation, activation or survival, the pro-inflammatory cytokines can roughly be divided into osteoclastogenic, if they stimulate osteoclasts, or anti-osteoclastogenic, if they inhibit these unique bone-resorbing cells. Initially, the interplay between immune cells and osteoclasts has been observed in clinical settings of increased bone resorption following glucocorticoid therapy and in typical inflammatory bone diseases, such as rheumatoid arthritis (RA) and periodontal disease. The cross-talk between immune and bone cells via cytokines, chemokines, growth factors, signaling molecules and transcription factors, has begun to be intensively studied with the onset of osteoimmunology. This terminus was first mentioned in 2000, when Arron pointed out the importance of studying both, the skeletal and immune system, as an integrated entity (5). The milestone for osteoimmunology however, dates far before that, already in 1972, when Horton et al. found a new soluble factor in the supernatant from cultures of stimulated human peripheral blood leukocytes that was capable to trigger in vitro bone resorption (6). More than 20 years later, four different groups of researchers independently characterized the receptor activator of nuclear factor κB ligand (RANKL), an essential molecule that controls osoteoclastogenesis and thus bone resorption. The first two groups have found strong upregulation of RANKL in immune cells such as antigen-stimulated T cells and in mature dendritic cells (7,8) and the second two groups in bone marrow stromal cells (9,10). These findings pinpointed the important role of RANKL within both, skeletal and immune system, leading to first speculations that both systems might be functionally connected. However the main stimulus giving rise to osteoimmunology was the study of Takayanagi et al. in 2000, where they showed in vitro bone protecting action of IFN-γ (11). Every time our T cells are activated, they release RANKL, the osteoclast-stimulating molecule inducing bone resorption. Interestingly, IFN-γ binds to its receptor on osteoclasts, degrades RANKL signaling and thus inhibits the activation of osteoclasts and protects our bones from being resorbed. This cytokine is produced predominantly by NK and natural killer T (NKT) cells involved in the innate immune response, and by CD4+ Th1 and CD8+ cytotoxic T lymphocyte (CTL) effector T cells, once antigen-specific immunity develops (12).
As already mentioned the role of pro-inflammatory cytokines has been most extensively studied in RA (13). With the advancement of osteoimmunology the involvement of these soluble factors in the pathogenesis of bone diseases that have traditionally not been considered as inflammatory, such as osteoporosis (OP) and osteoarthritis (OA), has also been suggested (14,15).
The scope of this review is to summarize the current evidence on the influence of pro-inflammatory cytokines on osteoclasts and thus on the process of bone resorption. In the first part, the bone microenvironment, involving osteoclasts, osteoblasts and their connection to immune cells, will be addressed, while in the second part the osteoclastogenic, anti-osteoclastogenic and mixed (dual) effects of pro-inflammatory cytokines will be reviewed. Finally, up to date evidence on the role of pro-inflammatory cytokines in OP and OA will be summarized, with the aim to show that researching such complex bone disorders is almost impossible without considering skeletal and immune system in an integrated way.
Bone microenvironment
The interplay between bone and immune cells
Bone and immune cells are very similar meaning they have common progenitors in bone marrow, they require common receptors for cell signaling, and immune cells have shown the ability to differentiate into osteoclast as well.
Osteoclasts, macrophages and dendritic cells all derive from the same progenitors, i.e monocyte-macrophage lineage (16). Besides RANKL and macrophage colony stimulating factor (M-CSF), which are essential for commitment of the common precursor to the osteoclast lineage and survival of generated osteoclasts, numerous cytokines are also able to influence osteoclast differentiation and/or function (17). RANKL, when signaling via its receptor activator of nuclear factor κB (RANK) on osteoclasts, requires expression and activation of costimulatory molecules, containing immunoreceptor tyrosine-based activation motif (ITAM) domains. ITAMs are important components of numerous immune receptors. They are also present in DNAX adaptor protein 12 (DAP12) and Fc receptor γ (FcRγ) adapter molecules. The receptors that associate with DAP12 are the triggering receptor expressed by myeloid cells-2 (TREM-2) and the signal regulatory protein β1 (SIRP β1) (18). Receptors that associate with FcRγ adapters in myeloid cells are the osteoclast associated receptor (OSCAR) and the paired Ig-like receptor A (PIR-A). Mice lacking DAP12 and FcRγ are severely osteopetrotic (18,19). Interestingly, OSCAR can partially rescue the abnormal cytoskeleton of the osteoclasts in DAP12-/-mice (20) and was recently shown to bind to fibrillar collagen and promote osteoclastogenesis (21). These data suggest OSCAR as important immunoreceptor in osteoclastogenesis.
Furthermore, for osteoclast precursors to turn into mature resorbing osteoclasts, the interaction between members of the immunoglobulin super-family, namely the type-1 membrane glycoprotein CD200 and its receptor (CD200R), are needed. Osteoclast numbers decreased and the bone mass was increased in CD200 deficient mice (22). During different phases of their development, osteoclasts express specific molecules such as RANK, calcitonin receptor, β3 integrin, OSCAR, cathepsin K and tartrate-resistant acid phosphatase (TRAP), which are involved in osteoclast differentiation, function and survival (23).
Immune cells have shown the ability to differentiate into osteoclasts as well. Dendritic cells, which are professional antigen presenting cells able to efficiently activate naïve T lymphocytes, can differentiate into osteoclasts in vitro during their early stage of development (24). This process is stimulated by pro-inflammatory cytokines IL-1β and TNF-α and is inhibited in the presence of IFN-α (25). Moreover, osteoclasts from myeloma patients contain nuclei with translocated chromosomes of myeloma B-cell clone origin, suggesting that B-lymphoid lineage cells might also give rise to osteoclasts (26).
Osteoblasts, the bone forming cells, originate from bone marrow-residing multipotent mesenchymal stem cells. Osteoblasts are one of the major sources of RANKL and in this manner they control bone resorption. Interestingly, they influence immune cells as well. Osteoblasts are critical regulators of the hematopoietic stem cells (HSC), located in a specialized structure within bone marrow, the niche, from where immune and other blood cells derive (27). This regulation occurs via annexin II, an osteoblast derived protein. It has been shown that bone marrow of annexin II-deficient mice contains decreased number of hematopoetic stem cells (28). HSC can remain dormant or replicate to either self-renew or differentiate into multipotent progenitor cells capable of differentiating into lymphoid or myeloid precursors. In addition, the production of IL-10 by osteoblasts has also been shown to promote the self-renewal of HSC in their bone marrow located niches (29). Primary osteoblast lineage cells have been shown to produce MCSF, granulocyte colony stimulating factor (G-CSF), GM-CSF, IL-1, IL-6, lymphotoxin, TGF-β, TNF-α, leukemia inhibitory factor (LIF), and stem cell factor (SCF), which play a role in hematopoiesis and many of them are involved in osteoclast development as well (30).
RANK/RANKL/OPG system
Discovery of the molecular triad RANK/RANKL/osteoprotegerin (OPG) and characterization of their central role in the interplay between immune and bone cells, have contributed significantly to the emergence of the osteoimmunology (31). RANKL, a member of the TNF superfamily, is a potent stimulator of both, osteoclast formation and their bone-resorbing activity (31,32). Upon binding to its receptor, RANK located on osteoclasts, RANKL signaling increases differentiation and activation of the osteoclasts resulting in expression of osteoclast specific molecules. Although it has been proposed for a long time that the main source of RANKL are stromal and osteoblastic cells, a very recent study by Nakashima et al. proved that the main RANKL production site resides within osteocytes (33). OPG is a secreted TNF receptor super-family member acting as a decoy receptor molecule for RANKL, thereby counteracting its osteoclastogenic activity. It is produced by a variety of cells, including stromal cells, B lymphocytes and dendritic cells (34). RANK or RANKL knockout mice have significant osteopetrosis with no osteoclasts. In contrast, animals lacking OPG develop severe OP due to a response to enhanced osteoclastogenesis (35). Mutations in humans causing overexpression of RANK are associated with familial expansile osteolysis and expansile skeletal hyper-phosphatasia (36,37). Anti-RANKL specific antibody, denosumab has been recently approved in clinical practice for the treatment of OP (38).
Numerous osteoclastogenic factors including hormones, cytokines and growth factors, that are produced physiologically or excessively during inflammation, are now believed to exert their primary activity on osteoclasts by modulating the RANK/RANKL/OPG axis (39). Upon activation T cells produce both, RANKL and pro-inflammatory cytokines and can thus influence osteoclastogenesis (40–42).
The influence of pro-inflammatory cytokines on osteoclasts
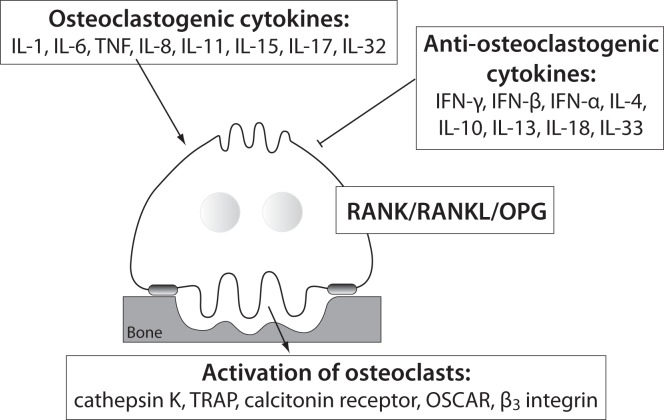
Several systemic and local factors are known to influence osteoclasts and their bone resorption ability. In a physiologic state, the osteoclast activity is highly balanced by those factors. In pathological conditions, such as excessive activation of the immune system, their activity becomes deregulated due to additional pro-inflammatory cytokines, produced mainly by activated T cells (43). As already mentioned, the pro-inflammatory cytokines are osteoclastogenic if they have shown stimulation of osteoclast differentiation and/or activity, or anti-osteoclastogenic, if their ability to inhibit osteoclasts has been proved (44). Based on the current evidence, pro-inflammatory cytokines such as IL-1, IL-6, IL-8, IL-11, IL-17, TNF-α etc. are considered osteoclastogenic, and IL-4, IL-10, IL-13, IL-18, IFN-γ, IFN-β etc. anti-osteoclastogenic. Th17, a special subtype of T helper cells, producing IL-17 and RANKL, are classified as osteoclastogenic, while the classical Th1 and Th2 cells are considered anti-osteoclastogenic, due to their production of IFN-γ (Th1) and IL-4 (Th2) (45,46). Some of the pro-inflammatory cytokines, such as IL-7, IL-12, IL-23, and IL-6 and TGF-β as well, have been shown to possess dual, osteoclastogenic and anti-osteoclastogenic properties. It seems that their net effect depends on the specific pathophysiological condition of bone in which they are being studied in vivo, while in experiments in vitro it depends on developmental stage of osteoclasts (43,47,48). The determination of their exact role in bone microenvironment is even more difficult, as synergistic and antagonizing effects on osteoclasts of these cytokines have been observed (49–56). As already mentioned, the pro-inflammatory cytokines can target osteoclasts directly or indirectly by modulating the RANK/RANKL/OPG system. The osteoclastogenic effect of these cytokines can be assessed by determining the expression of osteoclast-specific genes encoding RANK, calcitonin receptor, β3 integrin, OSCAR, cathepsin K and TRAP, being involved in osteoclast differentiation, bone-resorbing activity and survival (23). The schematic representation of the influence of the pro-inflammatory cytokines on osteoclast is shown in Figure 1.
Figure 1.
Different influences of pro-inflammatory cytokines on osteoclasts.
IL - interleukin; TNF- tumor necrosis factor; IFN - interferon; RANK - receptor activator of nuclear factor κB; RANKL - RANK ligand; OPG - osteoprotegerin; TRAP - tartrate-resistant acid phosphatase; OSCAR - osteoclast associated immunoreceptor.
Cytokines with stimulating effects on osteoclasts (osteoclastogenic cytokines)
Mechanisms of action of osteoclastogenic pro-inflammatory cytokines, the evidence on their opposite effects, and synergy or antagonism with other cytokines are summarized in Table 1.
Table 1.
Osteoclastogenic cytokines.
| Cytokine | Mechanism of osteoclastogenic action | Evidence on direct effect on osteoclasts | Synergy/Antagonism with other pro-inflammatory cytokines | Evidence on opposite/anti-osteoclastogenic effect |
|---|---|---|---|---|
| IL-1 | ↑ RANKL in stromal cells (54,172,173) | (174) | synergy with TNF-α (54), IL-6 and TNF-α (53) and PGE2(175) | |
| TNF-α | ↑ RANKL dependent osteoclastogenesis (176), ↑ RANKL (172,173) | (53,176–179) | synergy with IL-1 (54), with IL-1 and IL-6 (53), with RANKL (176) | |
| IL-6 | ↑ RANKL and OPG (180) | (181) | synergy with IL-1 and TNF-α (53,182), with TNF-α (183), with PGE2(184) | (48) |
| IL-8 | ↑ RANKL(78) | (78,185) | ↑ NO (104) | |
| IL-11 | ↑ RANKL/OPG (186) | (181) | antagonism with IL-6 (187) | |
| IL-15 | ↑ differentiation of OC (188) | synergy with TNF-α (188) | ||
| IL-17 | ↑ RANKL (189) | (190) | synergy with TNF-α and IL-1 (71–73), synergy with PGE2(189) | (74) |
| IL-32 | ↑ NFATc1, OSCAR and cathepsin K (191) | ↑ release of IL-4 and IFN-γ (191) |
IL - interleukin; TNF-α – tumor necrosis factor α; RANKL – receptor activator of nuclear factor κB ligand; OPG – osteoprotegerin; Th17 – IL-17 producing T helper cells; NFATc1 – nuclear factor of activated T cells; OSCAR – osteoclast associated immunoreceptor; PGE2 – prostaglandin E2; NO – nitric oxide; OC – osteoclast.
IL-1, existing in a form of IL-1α and IL-1β polypeptides, is produced by macrophages and is a potent stimulator of bone resorption, both in vitro and in vivo (57–61). Similarly to IL-1, TNF is also represented as a family of two related polypeptides α and β that are both potent stimulators of osteoclastogenesis (60,62,63). TNF-α is produced by a variety of immune cells, primarily by macrophages, but also by NK cells, mast cells and T (Th1) and B lymphocytes. Anti-TNF-α antibody is used for the treatment of RA (12). IL-6, produced by T cells (Th2), dendritic cells and macrophages, is a pivotal cytokine in acute and chronic inflammation. Interestingly, stromal cells and osteoblasts can also produce nanomolar quantities of IL-6 in response to TGF-β, IL-1 and TNF-α (64). The blocking of IL-6 signaling is effective in controlling RA symptoms (65), as well as in vivo and in vitro blocking of osteoclast formation (66). Interestingly, data on direct inhibition of osteoclast differentiation by IL-6 are also available (48). IL-11 is produced in bone marrow stromal cells and is required for normal bone remodeling (67). In vitro studies have shown that it stimulates osteoclast formation and bone resorption (68). Mice being deficient in specific IL-11 receptor have increased trabecular bone mass due to decreased bone resorption (69). IL-17 exists as a family of six distinctive cytokines (IL-17A - F) that are produced by a distinct lineage of T helper lymphocytes, the Th17 cells and are able to exert high osteoclastogenic activity (70). In a synergy with TNF-α and IL-1, IL-17 has been causatively implied in the bone destruction observed in RA (71,72). IL-17A inhibition in an antigen-induced arthritis model, prevented joint and bone destruction and decreased the levels of RANKL, IL-1β, and TNF-α within pathologic lesions (73). Recent study however showed, that the osteoclastogenic effect of IL-17A depends on its concentration, as only high concentrations of IL-17A were able to suppress osteoclastogenesis in vitro (74). Like IL-17, IL-15 can also be produced by T lymphocytes, although its main sources are monocytes/macrophages and dendritic cells (75) and is involved in bone destruction in RA as well (76). Mice deficient in IL-15Rα, the specific IL-15 receptor subunit have increased bone mineral density (BMD) and decreased numbers of osteoclasts (77). IL-8 belongs to a group of chemokines and is, unlike other osteoclastogenic cytokines that we have described up to this point, produced not only by T cells and macrophages but also by osteoclasts themselves. It has been shown to play an osteoclastogenic role in metastatic bone disease (78). IL-32 is a relatively newly described cytokine showing typical properties of a pro-inflammatory mediator, as it stimulates TNF-α, IL-1β and IL-8 production. It exerts complex osteoclastogenic effects, such as promoting osteoclast differentiation but not maturation of osteoclasts (79).
Cytokines with inhibiting effects on osteoclasts (anti-osteoclastogenic cytokines)
The mechanisms of action of anti-osteoclastogenic pro-inflammatory cytokines, the evidence on their functional effects, as well as synergy or antagonism with other cytokines, are summarized in Table 2.
Table 2.
Anti-osteoclastogenic cytokines.
| Cytokine | Mechanism of anti-osteoclastogenic action | Evidence on direct effect on osteoclasts | Synergy/Antagonism with other pro-inflammatory cytokines | Evidence on opposite/osteoclastogenic effect |
|---|---|---|---|---|
| IFN-γ | ↓ RANK signaling (11) ↓ cathepsin K (192) ↓ 1,25-(OH)2 D3, PTH and IL-1 mediated osteoclastogenesis (193) |
↓ TRAF6 (11) ↑ osteoclasts’ apoptosis by Fas/Fas ligand signals (194) ↓ RANKL-induced osteoclasts formation (195) |
synergy with IL-12 (125) antagonism with TNF-α (194) |
(43,80,81) |
| IFN-β | negative feedback regulation of osteoclasts via RANKL induced c-Fos signaling (85) ↑ RANKL induced NO release (196) ↑ expression of CXCL11 (84) |
(85) | ||
| IFN-α | ↓ TRAP and c-Fos (89) | |||
| IL-4 | ↓ NFATc1 expression via STAT6 (197) ↓ NFATc1 and c-Fos expression (198) ↑ OPG and ↓ RANKL via STAT6 (199–201) ↑ T cell surface molecules (202) |
(202, 203) | ↓TNF-α signaling, ↑IL-1 (203) | |
| IL-10 | ↑ NO (104) ↑ OPG and ↓ RANKL expression (105) |
↓ expression of NFATc1 (101) ↓ expression of NFATc1, c-Fos and c-Jun downstream RANK signaling (102) ↓ transcription of TREM2 downstream RANK signaling (103) |
||
| IL-13 | ↑OPG and ↓ RANKL via STAT6 (199–201) | |||
| IL-18 | T cells independent mechanism (110) ↑ apoptosis of osteoclasts with ↑ NO production (108) ↑ OPG from stromal cells (109) ↑ GM-CSF from T cells (107) ↑ IFN-γ from T cells (56) |
synergy with IL-12 (50,55,110) | ↑ RANKL from T cells (204) | |
| IL-33 | ↓ osteoclastogenesis (112,113) |
IFN - interferon; IL - interleukin; RANK - receptor activator of nuclear factor κB; OPG - osteoprotegerin; PTH - parathyroid hormone; TNF-α - tumor necrosis factor α; NO - nitric oxide; CXCL11; - chemokine ligand 11; TRAP - tartrate-resistant acid phosphatase; NFATc1 - nuclear factor of activated T cells; TRAF6 - TNF receptor associated factor 6; STAT6 - signal transducer and activator of transcription 6; GM-CSF - granulocyte macrophage colony stimulating factor; TREM2 - triggering receptor expressed by myeloid cells 2.
IFN-γ, a type II interferon, which is produced by Th1 lymphocytes and NK cells, is a potent inducer of antigen presentation and therefore of T cell activation. As previously mentioned, the discovery of IFN-γ anti-osteoclastogenic effect in vitro has initiated development of osteoimmunology (11). In vivo effects of IFN-γ on bone tissue are more difficult to categorize and often indicate its opposite, osteoclastogenic effect as well (43,80,81). It seems that IFN-γ functions as an anti-osteoclastogenic cytokine in physiological conditions of bone turnover as proven by lower BMD in nude and IFN-γ receptor deficient mice (43,81). However, in pathological bone turnover such as in postmenopausal OP, inflammation or bacterial infection, the net effect of IFN-γ is biased towards bone resorption via antigen driven T cell activation and RANKL production (43). In rats, intraperitoneal IFN-γ injections induced osteopenia (82), while in humans this cytokine has been reported to be efficacious in the treatment of osteopetrosis as it restores bone resorption (83).
Comparing to IFN-γ, there are fewer studies published on the effects of type I interferons, IFN-α and IFN-β. However, the existing data on the anti-osteoclastogenic activity of IFN-β are quite uniform (84). Namely, mice that are deficient in IFN-β exhibit severe osteopenia accompanied by enhanced osteoclastogenesis (85). In order to suppress the anti-osteoclastogenic effects of IFN-β, osteoclast precursors have been shown to upregulate the expression of the suppressors of cytokines signaling (SOCS) (86). Recently, the upregulation of IFN-β expression has been suggested as a basic molecular mechanism needed for the efficacy of vitamin D (1α,25(OH)2D3) used to treat human bone diseases (87). In animal models of RA, the application of IFN-β resulted in significantly reduced cartilage and bone destruction accompanied by downregulated expression of TNF-α and IL-6, as well as increased expression of IL-10 (88).
There are even much more limited data on IFN-α, showing its anti-osteoclastogenic effects in vitro (89). In vivo no effect of IFN-α on bone turnover could be detected in rats (90), however in mice models of systemic lupus erythematosus it was shown to decrease bone resorption (91). Children receiving IFN-α therapy due to chronic hepatitis B have been shown to have higher femoral BMD (92).
IL-4 and IL-13 are anti-osteoclastogenic cytokines, moreover they have shown influence on osteoblast as well (93,94). Transgenic mice overexpressing IL-4 develop an osteoporotic phenotype (95) that may result from both, the inhibition of osteoclast formation and their activity (96,97), as well as the inhibition of osteoblasts and bone formation (94). Both interleukins have also been shown to inhibit IL-1 stimulated bone resorption by decreasing the activity of cyclooxygenase-2 (COX-2) and thereby the production of prostaglandins (98). Additionally, IL-4 has the ability to inhibit osteoclastogenesis being physiologically stimulated by RANKL or pathologically by TNF-α (99).
IL-10 is produced by monocytes, T cells and B cells (100) and inhibits not only osteoclastogenesis (101–105) but also osteoblastogenesis and the onset of mineralization (106).
Besides IL-17 and IL-23, IL-18 has been also found to be increased in inflammation sites in RA (56). Although it belongs to the IL-1 superfamily, and is also structurally similar to IL-1, it has been shown to exert its anti-osteoclastogenic properties via various mechanisms (56,107–110). It is produced by macrophages and other cells, also osteoblastic ones. IL-18 overexpressing transgenic mice have decreased numbers of osteoclasts and decreased bone mass (111). IL-33 is another member of the IL-1 superfamily, and recent data suggest that similarly to IL-18, it has anti-osteoclastogenic effects (112,113).
Cytokines with dual (opposite) effects on osteoclasts
The mechanisms of action of pro-inflammatory cytokines with mixed, osteoclastogenic and anti-osteoclastogenic effects are summarized in Table 3.
Table 3.
Cytokines with osteoclastogenic and anti-osteoclastogenic properties.
| Cytokine | Mechanism of osteoclastogenic action | Mechanism of anti-osteoclastogenic action | Synergy/Antagonism with other pro-inflammatory cytokines |
|---|---|---|---|
| IL-7 | ↑ secretion of T cells derived osteoclastogenic cytokines and RANKL (205–207) RANKL independent (206) |
↓ of osteoclastogenesis acting directly on osteoclasts (116,117) | synergy in osteoclastogenesis with TNF-α and IL-1 (205,206), with TNF-α (120,207), with TNF-α and IFN-γ (121) |
| IL-12 | ↑ IL-1β and Th1 derived cytokines (122) | ↓ of osteoclastogenesis indirectly via T cells (50,125) ↓ of osteoclastogenesis acting directly on osteoclasts (208) |
synergy with IFN-γ (125), with IL-18 (50,55), with IL-18 in TNF-α mediated osteoclastogenesis (108,209) |
| IL-23 | ↑ Th17 (210) ↑ IL17 (127) ↑ RANKL (211,212) |
↓ fusion of osteoclasts precursors or survival of osteoclasts (213) ↓ osteoclast formation via T cells (128) |
synergy with IL-18 in blocking osteoclastogenesis (128) |
IFN - interferon; IL - interleukin; RANKL - receptor activator of nuclear factor κB ligand; TNF-α - tumor necrosis factor α; Th1 - type I helper T cells; Th17 - Th subpopulation expressing IL-17.
IL-7 is produced by stromal cells in red bone marrow and thymus, keratinocytes, follicular dendritic cells and other non-hematopoietic cells. The production of IL-7 was enhanced in ovariectomized (OVX) mice where it stimulated osteoclastogenesis (114). Another study found that in IL-7 deficient mice, as compared to wild-type animals, the numbers of osteoclasts increased and the trabecular bone volume decreased; however both types of mice displayed similar bone loss following OVX (115). Addition of IL-7 to murine bone marrow cells cultured in the presence of M-CSF and RANKL, inhibited osteoclast formation as well (116). Recently, mice overexpressing human IL-7 in their osteoblast lineage were found to have increased trabecular bone volume in vivo and decreased osteoclast formation in vitro (117). In thymectomized women the levels of IL-7 and RANKL were increased compared to controls, however their RANKL/OPG ratios and indices of bone metabolism were not affected (118). In humans, IL-7 has shown its osteoclastogenic nature in psoriatic arthritis (119) and in solid tumors bearing patients (120), while in those suffering from RA it was suggested to contribute to the perpetuation of Th1 and TNF-α mediated pro-inflammatory immune responses (121).
IL-12 is another controversial cytokine in terms of its anti-osteoclastogenic/osteoclastogenic effects (122). It is produced by myeloid cells, i.e. dendritic cells and macrophages and induces differentiation and activation of Th1 lymphocytes and their subsequent production of IFN-γ and GM-CSF (123,124). There are conflicting results on the involvement of IFN-γ in IL-12 mediated osteoclastogenesis inhibition as well (50,125).
IL-23 is produced by dendritic cells and macrophages. It plays a role in bone destruction in RA, as shown in mice models of RA, with animals being deficient for either IL-17 or IL-23 (70,126), and in rat models of collagen-induced arthritis, where animals were treated with anti-IL-23 antibodies (127). However, in non-pathological conditions IL-23 functions in an opposite, anti-osteoclastogenic way (128).
Transforming growth factor β
TGF-β, an ubiquitous growth factor, is not a probut an anti-inflammatory cytokine, playing an important and complex role in bone remodelling by influencing bone resorption, formation and the production of certain pro-inflammatory cytokines (47,49). It is produced by many different cells, including macrophages and exists in at least three isoforms, TGF-β1, TGF-β2 and TGF-β3. Recently, the role of TGF-ß1 in coupling bone formation and resorption has been suggested (129). Its effects on osteoclasts cultured in vitro greatly depend on numerous factors, such as: differentiation stage of the starting cell population, cell density, the presence or absence of serum in the cell culture medium, concentration of added TGF-ß1, and whether its effect was studied on isolated cells or in co-cultures. In vivo the presence of other cytokines such as IL-1, IL-6, TNF-α, IFN-γ, prostaglandin E2 (PGE2) etc. and environment as such determine the exact outcome of TGF-ß1 activity (47,51).
The pro-inflammatory cytokines and bone diseases
In many pathological conditions, activated immune cells such as T lymphocytes, macrophages and dendritic cells produce a variety of pro-inflammatory cytokines which in turn modulate osteoclast activity and bone resorption as already described above. In continuation we will provide actual data on the roles of certain pro-inflammatory cytokines in two common bone diseases, OP and OA.
The role of pro-inflammatory cytokines in osteoporosis
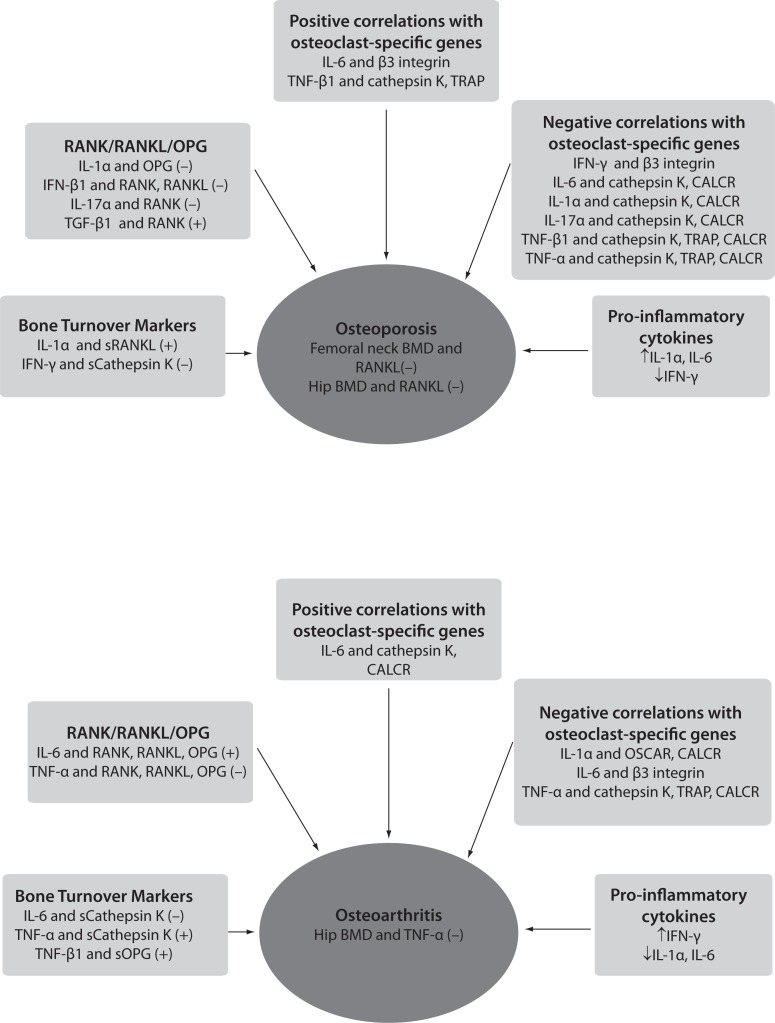
Osteoporosis has been traditionally considered an endocrine disease resulting mainly from the estrogens decline after menopause. This change affects bone remodeling, leading to impaired microarchitecture of bone tissue and consequently to higher risk of fractures. Since solely the endocrine point of view does not fully explain the pathogenesis of OP, the osteoimmunological approach suggests that activated T lymphocytes can contribute to menopausal changes in bone remodeling by producing pro-inflammatory cytokines such as TNF-α, IL-1, IL-6, IL-7 and IFN-γ (130,131). This statement has caused a paradigm shift in the pathogenesis of OP, which can now be also classified as an inflammatory disease (4, 15,132). The effects of pro-inflammatory cytokines have been initially studied in OVX mice which represent an experimental model of postmenopausal OP. Nude mice that are deficient in T cells were found to be resistant to bone loss following acute estrogen decline, artificially caused by OVX. It has been proposed that this was due to the lack of CD4+ subpopulation of T lymphocytes (130). Another mechanism of bone loss due to estrogen decline is the enhanced TNF-α production by activated T cells (133,134). Mice deficient in IL-1 receptor (135), TNF-α (133,136) or IL-6 (68) did not lose bone mass following OVX, while no difference in bone loss could be found between IL-7 deficient mice and the wild type animals (115). However, there are also studies showing the opposite effects. Namely, no influence on trabecular bone loss has been observed in mice lacking functional T lymphocytes after OVX (137), and additionally nude mice can develop OP as well (138). Studies in humans have shown higher T cell activity and increased levels of IL-1, TNF-α and IL-6 in post-menopausal osteoporotic women (130,139–143). Higher expression of RANKL/OPG and M-CSF in patients with fragility fracture has also been reported (144). Furthermore, increased serum levels of IL-6 were proved to account for up to 34% of the total variance of BMD change within the first few years after the onset of menopause (145). In Figure 2, the connections of the pro-inflammatory cytokines with OP phenotype in patients with low-energy femoral neck fractures are presented according to results of our recently published study (146). In this study we found higher expression of genes encoding for IL-1α, IL-6, RANKL and RANK in osteoporotic bone tissue. Additionally, we have found two genetic variants in IL-1α gene showing possible influence on osteoporotic phenotype, such as osteoporotic fracture (147). It has been suggested that activated T cells could contribute to enhanced osteoclastogenesis in two ways, first by increasing the production of bone resorbing cytokines, especially TNF-α and RANKL, and secondly, by increasing the numbers of osteoclast precursors (139,148). Bone cells isolated from untreated postmenopausal women showed higher mRNA expression for IL-1, TNF and IL-6 in comparison to those taken from women on estrogen replacement therapy (149). A blockade of both, TNF and IL-1 reduced bone resorption in postmenopausal osteoporotic women (150). Interestingly, Canadian multicentre osteoporosis study showed that treatment with COX-2 inhibitors was associated with higher BMD in postmenopausal women not taking estrogen (151). When looking at the extent of IFN-γ production, no differences between postmenopausal osteoporotic and healthy women were found (152). As already mentioned in case of IFN-γ, differences exist in effects of pro-inflammatory cytokines on osteoclasts depending on whether the bone is being remodelled under physiological or pathological conditions. In pathological conditions such as inflammation, infection and estrogen deficiency, Gao et al. have shown that the net balance between osteoclastogenic and anti-osteoclastogenic effect of IFN-γ is shifted towards osteoclastogenesis and bone resorption (43). This is due to predominant indirect effect of IFN-γ that presents enhanced antigen presentation by IFN-γ to T cells leading to T cells’ activation, proliferation and production of osteoclastogenic cytokines, such as RANKL and TNF-α. In vivo, this indirect (via RANKL) osteoclastogenic activity overcomes the direct suppressive action of IFN-γ on osteoclast precursors, leading to a net bone loss. Similarly, IL-6 does not seem to play an osteoclastogenic role under physiological conditions such as the estrogen-replete state (131).
Figure 2.
Schematic representation of correlations between pro-inflammatory cytokines, osteoclasts-specific genes, bone mineral density and levels of bone turnover markers in osteoporosis and osteoarthritis, according to results of Zupan et al. (146).
(−) - denotes negative correlation; (+) - denotes positive correlation; BMD - bone mineral density; RANK/RANKL/OPG - the most significant correlations of pro-inflammatory cytokines with RANK, RANKL and OPG genes are shown; sCathepsin K - cathepsin K serum levels; sOPG - OPG serum levels; sRANKL - RANKL serum levels; OSCAR - osteoclast-associated immunoglobulin-like receptor; CALCR - calcitonin receptor; TRAP - tartrate-resistant acid phosphatase.
Rodent models were used to show that TGF-β1 and TGF-β2 prevent bone loss following OVX (153) and that estrogen upregulates the expression of TGF-β (154). There is also evidence on the involvement of TGF-β into a mechanism by which estrogen deficiency upregulates the production of IFN-γ (155,156). In humans, long-term in vivo estrogen treatment has been shown to increase serum levels of TGF-β1 and TGF-β2 (157).
The role of pro-inflammatory cytokines in osteoarthritis
The role of pro-inflammatory cytokines such as IL-17, IL-1 and TNF-α in bone loss has been most intensively elucidated in RA and periodontal disease. In contrast to RA, OA has long been considered an age-related degenerative disease of cartilage with accompanying pathological changes in subchondral bone. Since the concept of synovial inflammation contributing to OA was suggested, numerous studies have shown that the inflamed synovium and activated synovial macrophages importantly promote the osteoarthritic pathology (14,158,159). These results provided the missing inflammatory component to the pathology of this disease. Unlike in OP, where the influence of pro-inflammatory cytokines has been focused on bone cells, the most intensively studied targets of these soluble factors in OA are synovial cells and chondrocytes. Synovial biopsy specimens taken from patients with early OA or RA are similar in cell morphology and cytokine spectra, although the percentage of macrophages, T and B lymphocytes, as well as the levels of TNF-α and IL-1, are generally lower in OA (160). TNF-α and IL-1 have been suggested as key players in pathogenesis of OA. Namely, they have been shown to promote their own production, induce synovial cells and chondrocytes to produce other pro-inflammatory cytokines such as IL-6 and IL-8, as well as to stimulate protease and prostaglandin production (158,161,162). Animal models and human studies of OA have shown decreased cartilage damage following anti-IL-1 antibody therapy or gene transfer of IL-1Ra, the IL-1 receptor antagonist (163,164). In mice with OA, being deficient in IL-1 the pathology of OA was reduced (162,165). However, another study in IL-1 deficient mice found the opposite results (166), indicating a more complex role of IL-1 in OA. In our opinion this disrepancy could be due to different models of OA in animals, whether IL-1 or just IL-1β was studied or similarly to IFN-γ, IL-1 could play dual role depending on the presence of other cytokines. Interestingly, IL-4, IL-10 and IL-13 that are classified as anti-osteoclastogenic cytokines according to their influence on bone, have fully proven their anti-inflammatory role in OA. They were increased in synovial fluid of OA patients and were shown to inhibit a number of pro-inflammatory cytokines, such as IL-1β, TNF-α, PGE2, etc. (161).
Synovial macrophages play an important role in perpetuating the production of pro-inflammatory cytokines and mediating osteophyte formation and fibrosis in subchondral osteoarthritic bone (167). The latter two processes may also be under control of TGF-β (168). Namely, the inhibition of TGF-β markedly reduced both, fibrosis and osteophyte formation. The impaired TGF-β signaling then leads to chondrocyte hypertrophy and results in osteoarthritic pathology of cartilage as well (168,169).
Data on the influence of pro-inflammatory cytokines on osteoarthritic bone are scarce. A recent study pinpointed the increased gene expression of TGF-β (144). Our own data have shown higher expression of TGF-β1 receptor and the anti-osteoclastogenic cytokine IFN-γ in bone tissue samples of osteoarthritic patients, and connections of the pro-inflammatory cytokines with BMD, serum levels of bone turnover markers and osteoclast-specific genes as shown in Figure 2 (146). Another study in primary hip osteoarthritic patients showed positive correlations between IL-8 and bone resorption marker CTX, C terminal telopeptide of collagen type I in both, serum and synovium (170). However most of the published data indicate that in human OA, RANKL, released from activated T cells and synoviocytes, is the principal mediator of bone destruction. Additional proof for that is the fact that RANKL inhibition does not diminish inflammation but effectively prevents bone loss at the site of inflammation (35). Also, in randomized controlled clinical trials no effect in preventing bone loss in patients with RA following IFN-γ treatment could be found (171).
Conclusion
Thanks to recent advances in osteoimmunology, remarkable progress has been made in elucidating dynamic cross-talk between bone and immune cells that takes place in a complex bone microenvironment. Osteoimmunology has postulated a new approach to studying bone diseases such as OP and OA that were traditionally not considered to be inflammatory. Data reviewed in this paper provide an insight into the influence of pro-inflammatory cytokines on osteoclasts and thus on the bone resorption. They clearly show that T cell-derived cytokines can exert different effects on osteoclasts very much depending on whether a study was performed in vitro or in vivo. A huge step forward to elucidation of interactions between immune and skeletal system has been made by using animal models such as OVX mice and rodent models of OA. However, further studies in humans will be critical for clearly understanding the roles of pro-inflammatory cytokines in bone loss with the aim of raising novel therapeutic strategies for these kinds of patients.
Footnotes
Potential conflict of interest
None declared.
References
- 1.Feuerer M, Beckhove P, Garbi N, Mahnke Y, Limmer A, Hommel M, et al. Bone marrow as a priming site for T-cell responses to blood-borne antigen. Nat Med. 2003;9:1151–7. doi: 10.1038/nm914. http://dx.doi.org/10.1038/nm914. [DOI] [PubMed] [Google Scholar]
- 2.Mazo IB, Honczarenko M, Leung H, Cavanagh LL, Bonasio R, Weninger W, et al. Bone marrow is a major reservoir and site of recruitment for central memory CD8+ T cells. Immunity. 2005;22:259–70. doi: 10.1016/j.immuni.2005.01.008. http://dx.doi.org/10.1016/j.immuni.2005.01.008. [DOI] [PubMed] [Google Scholar]
- 3.Delves PJ, Roitt IM. The immune system. First of two parts. N Engl J Med. 2000;343:37–49. doi: 10.1056/NEJM200007063430107. [DOI] [PubMed] [Google Scholar]
- 4.Mundy GR. Osteoporosis and inflammation. Nutr Rev. 2007;65:S147–51. doi: 10.1111/j.1753-4887.2007.tb00353.x. http://dx.doi.org/10.1301/nr.2007.dec.S147-S151. [DOI] [PubMed] [Google Scholar]
- 5.Arron JR, Choi Y. Bone versus immune system. Nature. 2000;408:535–6. doi: 10.1038/35046196. http://dx.doi.org/10.1038/35046196. [DOI] [PubMed] [Google Scholar]
- 6.Horton JE, Raisz LG, Simmons HA, Oppenheim JJ, Mergenhagen SE. Bone resorbing activity in supernatant fluid from cultured human peripheral blood leukocytes. Science. 1972;177:793–5. doi: 10.1126/science.177.4051.793. http://dx.doi.org/10.1126/science.177.4051.793. [DOI] [PubMed] [Google Scholar]
- 7.Anderson DM, Maraskovsky E, Billingsley WL, Dougall WC, Tometsko ME, Roux ER, et al. A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature. 1997;390:175–9. doi: 10.1038/36593. http://dx.doi.org/10.1038/36593. [DOI] [PubMed] [Google Scholar]
- 8.Wong BR, Josien R, Lee SY, Sauter B, Li HL, Steinman RM, et al. TRANCE (tumor necrosis factor [TNF]-related activation-induced cytokine), a new TNF family member predominantly expressed in T cells, is a dendritic cell-specific survival factor. J Exp Med. 1997;186:2075–80. doi: 10.1084/jem.186.12.2075. http://dx.doi.org/10.1084/jem.186.12.2075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lacey DL, Timms E, Tan HL, Kelley MJ, Dunstan CR, Burgess T, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93:165–76. doi: 10.1016/s0092-8674(00)81569-x. http://dx.doi.org/10.1016/S0092-8674(00)81569-X. [DOI] [PubMed] [Google Scholar]
- 10.Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci U S A. 1998;95:3597–602. doi: 10.1073/pnas.95.7.3597. http://dx.doi.org/10.1073/pnas.95.7.3597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Takayanagi H, Ogasawara K, Hida S, Chiba T, Murata S, Sato K, et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature. 2000;408:600–5. doi: 10.1038/35046102. http://dx.doi.org/10.1038/35046102. [DOI] [PubMed] [Google Scholar]
- 12.Delves PJ, Roitt IM. The immune system. Second of two parts. N Engl J Med. 2000;343:108–17. doi: 10.1056/NEJM200007133430207. [DOI] [PubMed] [Google Scholar]
- 13.Brennan FM, McInnes IB. Evidence that cytokines play a role in rheumatoid arthritis. J Clin Invest. 2008;118:3537–45. doi: 10.1172/JCI36389. http://dx.doi.org/10.1172/JCI36389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bondeson J, Blom AB, Wainwright S, Hughes C, Caterson B, van den Berg WB. The role of synovial macrophages and macrophage-produced mediators in driving inflammatory and destructive responses in osteoarthritis. Arthritis Rheum. 2010;62:647–57. doi: 10.1002/art.27290. http://dx.doi.org/10.1002/art.27290. [DOI] [PubMed] [Google Scholar]
- 15.Lencel P, Magne D. Inflammaging: the driving force in osteoporosis? Med Hypotheses. 2011;76:317–21. doi: 10.1016/j.mehy.2010.09.023. http://dx.doi.org/10.1016/j.mehy.2010.09.023. [DOI] [PubMed] [Google Scholar]
- 16.Naoyuki Takahashi NU, Kobayashi Yasuhiro, Takami Masamichi, Martin T John, Suda Tatsuo. Osteoclast Generation. In: Bilezikian JP, Raisz LG, Martin TJ, editors. Principles of bone biology. 3rd ed. San Diego, Calif: Academic Press/Elsevier; 2008. pp. 175–92. [Google Scholar]
- 17.Takayanagi H, Sato K, Takaoka A, Taniguchi T. Interplay between interferon and other cytokine systems in bone metabolism. Immunol Rev. 2005;208:181–93. doi: 10.1111/j.0105-2896.2005.00337.x. http://dx.doi.org/10.1111/j.0105-2896.2005.00337.x. [DOI] [PubMed] [Google Scholar]
- 18.Koga T, Inui M, Inoue K, Kim S, Suematsu A, Kobayashi E, et al. Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature. 2004;428:758–63. doi: 10.1038/nature02444. http://dx.doi.org/10.1038/nature02444. [DOI] [PubMed] [Google Scholar]
- 19.Mocsai A, Humphrey MB, Van Ziffle JA, Hu Y, Burghardt A, Spusta SC, et al. The immunomodulatory adapter proteins DAP12 and Fc receptor gamma-chain (FcRgamma) regulate development of functional osteoclasts through the Syk tyrosine kinase. Proc Natl Acad Sci U S A. 2004;101:6158–63. doi: 10.1073/pnas.0401602101. http://dx.doi.org/10.1073/pnas.0401602101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zou W, Zhu T, Craft CS, Broekelmann TJ, Mecham RP, Teitelbaum SL. Cytoskeletal dysfunction dominates in DAP12-deficient osteoclasts. J Cell Sci. 2010;123:2955–63. doi: 10.1242/jcs.069872. http://dx.doi.org/10.1242/jcs.069872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Barrow AD, Raynal N, Andersen TL, Slatter DA, Bihan D, Pugh N, et al. OSCAR is a collagen receptor that costimulates osteoclastogenesis in DAP12-deficient humans and mice. J Clin Invest. 2011;121:3505–16. doi: 10.1172/JCI45913. http://dx.doi.org/10.1172/JCI45913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cui W, Cuartas E, Ke J, Zhang Q, Einarsson HB, Sedgwick JD, et al. CD200 and its receptor, CD200R, modulate bone mass via the differentiation of osteoclasts. Proc Natl Acad Sci U S A. 2007;104:14436–41. doi: 10.1073/pnas.0702811104. http://dx.doi.org/10.1073/pnas.0702811104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Asagiri M, Takayanagi H. The molecular understanding of osteoclast differentiation. Bone. 2007;40:251–64. doi: 10.1016/j.bone.2006.09.023. http://dx.doi.org/10.1016/j.bone.2006.09.023. [DOI] [PubMed] [Google Scholar]
- 24.Alnaeeli M, Penninger JM, Teng YT. Immune interactions with CD4+ T cells promote the development of functional osteoclasts from murine CD11c+ dendritic cells. J Immunol. 2006;177:3314–26. doi: 10.4049/jimmunol.177.5.3314. [DOI] [PubMed] [Google Scholar]
- 25.Speziani C, Rivollier A, Gallois A, Coury F, Mazzorana M, Azocar O, et al. Murine dendritic cell transdifferentiation into osteoclasts is differentially regulated by innate and adaptive cytokines. Eur J Immunol. 2007;37:747–57. doi: 10.1002/eji.200636534. http://dx.doi.org/10.1002/eji.200636534. [DOI] [PubMed] [Google Scholar]
- 26.Andersen TL, Boissy P, Sondergaard TE, Kupisiewicz K, Plesner T, Rasmussen T, et al. Osteoclast nuclei of myeloma patients show chromosome translocations specific for the myeloma cell clone: a new type of cancer-host partnership? J Pathol. 2007;211:10–7. doi: 10.1002/path.2078. http://dx.doi.org/10.1002/path.2078. [DOI] [PubMed] [Google Scholar]
- 27.Yin T, Li L. The stem cell niches in bone. J Clin Invest. 2006;116:1195–201. doi: 10.1172/JCI28568. http://dx.doi.org/10.1172/JCI28568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jung Y, Wang J, Song J, Shiozawa Y, Havens A, Wang Z, et al. Annexin II expressed by osteoblasts and endothelial cells regulates stem cell adhesion, homing, and engraftment following transplantation. Blood. 2007;110:82–90. doi: 10.1182/blood-2006-05-021352. http://dx.doi.org/10.1182/blood-2006-05-021352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kang YJ, Yang SJ, Park G, Cho B, Min CK, Kim TY, et al. A novel function of interleukin-10 promoting self-renewal of hematopoietic stem cells. Stem Cells. 2007;25:1814–22. doi: 10.1634/stemcells.2007-0002. http://dx.doi.org/10.1634/stemcells.2007-0002. [DOI] [PubMed] [Google Scholar]
- 30.Mark C, Horowitz JAL. Local Regulators of Bone: IL-1, TNF, Lymphotoxin, Interferon-γ, the LIF/IL-6 Family, and Additional Cytokines. In: Bilezikian JP, Raisz LG, Martin TJ, editors. Principles of bone biology. 3rd ed. San Diego, Calif: Academic Press/Elsevier; 2008. pp. 1209–34. [Google Scholar]
- 31.Theill LE, Boyle WJ, Penninger JM. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annu Rev Immunol. 2002;20:795–823. doi: 10.1146/annurev.immunol.20.100301.064753. http://dx.doi.org/10.1146/annurev.immunol.20.100301.064753. [DOI] [PubMed] [Google Scholar]
- 32.Dougall WC, Glaccum M, Charrier K, Rohrbach K, Brasel K, De Smedt T, et al. RANK is essential for osteoclast and lymph node development. Genes Dev. 1999;13:2412–24. doi: 10.1101/gad.13.18.2412. http://dx.doi.org/10.1101/gad.13.18.2412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nakashima T, Hayashi M, Fukunaga T, Kurata K, Oh-Hora M, Feng JQ, et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat Med. 2011;17:1231–4. doi: 10.1038/nm.2452. http://dx.doi.org/10.1038/nm.2452. [DOI] [PubMed] [Google Scholar]
- 34.Yun TJ, Chaudhary PM, Shu GL, Frazer JK, Ewings MK, Schwartz SM, et al. OPG/FDCR-1, a TNF receptor family member, is expressed in lymphoid cells and is up-regulated by ligating CD40. J Immunol. 1998;161:6113–21. [PubMed] [Google Scholar]
- 35.Leibbrandt A, Penninger JM. RANK/RANKL: regulators of immune responses and bone physiology. Ann N Y Acad Sci. 2008;1143:123–50. doi: 10.1196/annals.1443.016. http://dx.doi.org/10.1196/annals.1443.016. [DOI] [PubMed] [Google Scholar]
- 36.Hughes AE, Ralston SH, Marken J, Bell C, MacPherson H, Wallace RG, et al. Mutations in TNFRSF11A, affecting the signal peptide of RANK, cause familial expansile osteolysis. Nat Genet. 2000;24:45–8. doi: 10.1038/71667. http://dx.doi.org/10.1038/71667. [DOI] [PubMed] [Google Scholar]
- 37.Whyte MP, Mumm S. Heritable disorders of the RANKL/OPG/RANK signaling pathway. J Musculoskelet Neuronal Interact. 2004;4:254–67. [PubMed] [Google Scholar]
- 38.Roux S. New treatment targets in osteoporosis. Joint Bone Spine. 2010;77:222–8. doi: 10.1016/j.jbspin.2010.02.004. http://dx.doi.org/10.1016/j.jbspin.2010.02.004. [DOI] [PubMed] [Google Scholar]
- 39.Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003;423:337–42. doi: 10.1038/nature01658. http://dx.doi.org/10.1038/nature01658. [DOI] [PubMed] [Google Scholar]
- 40.Horwood NJ, Kartsogiannis V, Quinn JM, Romas E, Martin TJ, Gillespie MT. Activated T lymphocytes support osteoclast formation in vitro. Biochem Biophys Res Commun. 1999;265:144–50. doi: 10.1006/bbrc.1999.1623. http://dx.doi.org/10.1006/bbrc.1999.1623. [DOI] [PubMed] [Google Scholar]
- 41.Kong YY, Feige U, Sarosi I, Bolon B, Tafuri A, Morony S, et al. Activated T cells regulate bone loss and joint destruction in adjuvant arthritis through osteoprotegerin ligand. Nature. 1999;402:304–9. doi: 10.1038/46303. http://dx.doi.org/10.1038/46303. [DOI] [PubMed] [Google Scholar]
- 42.Kotake S, Udagawa N, Hakoda M, Mogi M, Yano K, Tsuda E, et al. Activated human T cells directly induce osteoclastogenesis from human monocytes: possible role of T cells in bone destruction in rheumatoid arthritis patients. Arthritis Rheum. 2001;44:1003–12. doi: 10.1002/1529-0131(200105)44:5<1003::AID-ANR179>3.0.CO;2-#. http://dx.doi.org/10.1002/1529-0131(200105)44:5<1003::AID-ANR179>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 43.Gao Y, Grassi F, Ryan MR, Terauchi M, Page K, Yang X, et al. IFN-gamma stimulates osteoclast formation and bone loss in vivo via antigen-driven T cell activation. J Clin Invest. 2007;117:122–32. doi: 10.1172/JCI30074. http://dx.doi.org/10.1172/JCI30074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nakashima T, Kobayashi Y, Yamasaki S, Kawakami A, Eguchi K, Sasaki H, et al. Protein expression and functional difference of membrane-bound and soluble receptor activator of NF-kappaB ligand: modulation of the expression by osteotropic factors and cytokines. Biochem Biophys Res Commun. 2000;275:768–75. doi: 10.1006/bbrc.2000.3379. http://dx.doi.org/10.1006/bbrc.2000.3379. [DOI] [PubMed] [Google Scholar]
- 45.Takayanagi H, Kim S, Taniguchi T. Signaling crosstalk between RANKL and interferons in osteoclast differentiation. Arthritis Res. 2002;4(Suppl 3):S227–32. doi: 10.1186/ar581. http://dx.doi.org/10.1186/ar581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kotake S, Nanke Y, Yago T, Kawamoto M, Yamanaka H. Human osteoclastogenic T cells and human osteoclastology. Arthritis Rheum. 2009;60:3158–63. doi: 10.1002/art.24886. http://dx.doi.org/10.1002/art.24886. [DOI] [PubMed] [Google Scholar]
- 47.Janssens K, ten Dijke P, Janssens S, Van Hul W. Transforming growth factor-beta1 to the bone. Endocr Rev. 2005;26:743–74. doi: 10.1210/er.2004-0001. http://dx.doi.org/10.1210/er.2004-0001. [DOI] [PubMed] [Google Scholar]
- 48.Yoshitake F, Itoh S, Narita H, Ishihara K, Ebisu S. Interleukin-6 directly inhibits osteoclast differentiation by suppressing receptor activator of NF-kappaB signaling pathways. J Biol Chem. 2008;283:11535–40. doi: 10.1074/jbc.M607999200. http://dx.doi.org/10.1074/jbc.M607999200. [DOI] [PubMed] [Google Scholar]
- 49.Franchimont N, Rydziel S, Canalis E. Transforming growth factor-beta increases interleukin-6 transcripts in osteoblasts. Bone. 2000;26:249–53. doi: 10.1016/s8756-3282(99)00275-6. http://dx.doi.org/10.1016/S8756-3282(99)00275-6. [DOI] [PubMed] [Google Scholar]
- 50.Horwood NJ, Elliott J, Martin TJ, Gillespie MT. IL-12 alone and in synergy with IL-18 inhibits osteoclast formation in vitro. J Immunol. 2001;166:4915–21. doi: 10.4049/jimmunol.166.8.4915. [DOI] [PubMed] [Google Scholar]
- 51.Huang W, Drissi MH, O’Keefe RJ, Schwarz EM. A rapid multiparameter approach to study factors that regulate osteoclastogenesis: demonstration of the combinatorial dominant effects of TNF-alpha and TGF-ss in RANKL-mediated osteoclastogenesis. Calcif Tissue Int. 2003;73:584–93. doi: 10.1007/s00223-003-0059-8. http://dx.doi.org/10.1007/s00223-003-0059-8. [DOI] [PubMed] [Google Scholar]
- 52.Liu XH, Kirschenbaum A, Yao S, Levine AC. Cross-talk between the interleukin-6 and prostaglandin E(2) signaling systems results in enhancement of osteoclastogenesis through effects on the osteoprotegerin/receptor activator of nuclear factor-{kappa}B (RANK) ligand/RANK system. Endocrinology. 2005;146:1991–8. doi: 10.1210/en.2004-1167. http://dx.doi.org/10.1210/en.2004-1167. [DOI] [PubMed] [Google Scholar]
- 53.Ragab AA, Nalepka JL, Bi Y, Greenfield EM. Cytokines synergistically induce osteoclast differentiation: support by immortalized or normal calvarial cells. Am J Physiol Cell Physiol. 2002;283:C679–87. doi: 10.1152/ajpcell.00421.2001. [DOI] [PubMed] [Google Scholar]
- 54.Wei S, Kitaura H, Zhou P, Ross FP, Teitelbaum SL. IL-1 mediates TNF-induced osteoclastogenesis. J Clin Invest. 2005;115:282–90. doi: 10.1172/JCI23394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yamada N, Niwa S, Tsujimura T, Iwasaki T, Sugihara A, Futani H, et al. Interleukin-18 and interleukin-12 synergistically inhibit osteoclastic bone-resorbing activity. Bone. 2002;30:901–8. doi: 10.1016/s8756-3282(02)00722-6. http://dx.doi.org/10.1016/S8756-3282-(02)00722-6. [DOI] [PubMed] [Google Scholar]
- 56.Yamamura M, Kawashima M, Taniai M, Yamauchi H, Tanimoto T, Kurimoto M, et al. Interferon-gamma-inducing activity of interleukin-18 in the joint with rheumatoid arthritis. Arthritis Rheum. 2001;44:275–85. doi: 10.1002/1529-0131(200102)44:2<275::AID-ANR44>3.0.CO;2-B. http://dx.doi.org/10.1002/1529-0131(200102)44:2<275::AIDANR44>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 57.Dinarello CA. Interleukin-1 and interleukin-1 antagonism. Blood. 1991;77:1627–52. [PubMed] [Google Scholar]
- 58.Gowen M, Mundy GR. Actions of recombinant interleukin 1, interleukin 2, and interferon-gamma on bone resorption in vitro. J Immunol. 1986;136:2478–82. [PubMed] [Google Scholar]
- 59.Pfeilschifter J, Chenu C, Bird A, Mundy GR, Roodman GD. Interleukin-1 and tumor necrosis factor stimulate the formation of human osteoclastlike cells in vitro. J Bone Miner Res. 1989;4:113–8. doi: 10.1002/jbmr.5650040116. http://dx.doi.org/10.1002/jbmr.5650040116. [DOI] [PubMed] [Google Scholar]
- 60.Teitelbaum SL. Bone resorption by osteoclasts. Science. 2000;289:1504–8. doi: 10.1126/science.289.5484.1504. http://dx.doi.org/10.1126/science.289.5484.1504. [DOI] [PubMed] [Google Scholar]
- 61.Vargas SJ, Naprta A, Glaccum M, Lee SK, Kalinowski J, Lorenzo JA. Interleukin-6 expression and histomorphometry of bones from mice deficient in receptors for interleukin-1 or tumor necrosis factor. J Bone Miner Res. 1996;11:1736–44. doi: 10.1002/jbmr.5650111117. http://dx.doi.org/10.1002/jbmr.5650111117. [DOI] [PubMed] [Google Scholar]
- 62.Azuma Y, Kaji K, Katogi R, Takeshita S, Kudo A. Tumor necrosis factor-alpha induces differentiation of and bone resorption by osteoclasts. J Biol Chem. 2000;275:4858–64. doi: 10.1074/jbc.275.7.4858. http://dx.doi.org/10.1074/jbc.275.7.4858. [DOI] [PubMed] [Google Scholar]
- 63.Kitaura H, Zhou P, Kim HJ, Novack DV, Ross FP, Teitelbaum SL. M-CSF mediates TNF-induced inflammatory osteolysis. J Clin Invest. 2005;115:3418–27. doi: 10.1172/JCI26132. http://dx.doi.org/10.1172/JCI26132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ishimi Y, Miyaura C, Jin CH, Akatsu T, Abe E, Nakamura Y, et al. IL-6 is produced by osteoblasts and induces bone resorption. J Immunol. 1990;145:3297–303. [PubMed] [Google Scholar]
- 65.Fonseca JE, Santos MJ, Canhao H, Choy E. Interleukin-6 as a key player in systemic inflammation and joint destruction. Autoimmun Rev. 2009;8:538–42. doi: 10.1016/j.autrev.2009.01.012. http://dx.doi.org/10.1016/j.autrev.2009.01.012. [DOI] [PubMed] [Google Scholar]
- 66.Axmann R, Bohm C, Kronke G, Zwerina J, Smolen J, Schett G. Inhibition of interleukin-6 receptor directly blocks osteoclast formation in vitro and in vivo. Arthritis Rheum. 2009;60:2747–56. doi: 10.1002/art.24781. http://dx.doi.org/10.1002/art.24781. [DOI] [PubMed] [Google Scholar]
- 67.Hill PA, Tumber A, Papaioannou S, Meikle MC. The cellular actions of interleukin-11 on bone resorption in vitro. Endocrinology. 1998;139:1564–72. doi: 10.1210/endo.139.4.5946. http://dx.doi.org/10.1210/en.139.4.1564. [DOI] [PubMed] [Google Scholar]
- 68.Girasole G, Passeri G, Jilka RL, Manolagas SC. Interleukin-11: a new cytokine critical for osteoclast development. J Clin Invest. 1994;93:1516–24. doi: 10.1172/JCI117130. http://dx.doi.org/10.1172/JCI117130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sims NA, Jenkins BJ, Nakamura A, Quinn JM, Li R, Gillespie MT, et al. Interleukin-11 receptor signaling is required for normal bone remodeling. J Bone Miner Res. 2005;20:1093–102. doi: 10.1359/JBMR.050209. http://dx.doi.org/10.1359/JBMR.050209. [DOI] [PubMed] [Google Scholar]
- 70.Sato K, Suematsu A, Okamoto K, Yamaguchi A, Morishita Y, Kadono Y, et al. Th17 functions as an osteoclastogenic helper T cell subset that links T cell activation and bone destruction. J Exp Med. 2006;203:2673–82. doi: 10.1084/jem.20061775. http://dx.doi.org/10.1084/jem.20061775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Miossec P. Interleukin-17 in rheumatoid arthritis: if T cells were to contribute to inflammation and destruction through synergy. Arthritis Rheum. 2003;48:594–601. doi: 10.1002/art.10816. http://dx.doi.org/10.1002/art.10816. [DOI] [PubMed] [Google Scholar]
- 72.Van bezooijen RL, Farih-Sips HC, Papapoulos SE, Lowik CW. Interleukin-17: A new bone acting cytokine in vitro. J Bone Miner Res. 1999;14:1513–21. doi: 10.1359/jbmr.1999.14.9.1513. [DOI] [PubMed] [Google Scholar]
- 73.Koenders MI, Lubberts E, Oppers-Walgreen B, van den Bersselaar L, Helsen MM, Di Padova FE, et al. Blocking of interleukin-17 during reactivation of experimental arthritis prevents joint inflammation and bone erosion by decreasing RANKL and interleukin-1. Am J Pathol. 2005;167:141–9. doi: 10.1016/S0002-9440(10)62961-6. http://dx.doi.org/10.1016/S0002-9440(10)62961-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kitami S, Tanaka H, Kawato T, Tanabe N, Katono-Tani T, Zhang F, et al. IL-17A suppresses the expression of bone resorption-related proteinases and osteoclast differentiation via IL-17RA or IL-17RC receptors in RAW264.7 cells. Biochimie. 2010;92:398–404. doi: 10.1016/j.biochi.2009.12.011. http://dx.doi.org/10.1016/j.biochi.2009.12.011. [DOI] [PubMed] [Google Scholar]
- 75.Fehniger TA, Caligiuri MA. Interleukin 15: biology and relevance to human disease. Blood. 2001;97:14–32. doi: 10.1182/blood.v97.1.14. http://dx.doi.org/10.1182/blood.V97.1.14. [DOI] [PubMed] [Google Scholar]
- 76.Miranda-Carus ME, Benito-Miguel M, Balsa A, Cobo-Ibanez T, Perez de Ayala C, Pascual-Salcedo D, et al. Peripheral blood T lymphocytes from patients with early rheumatoid arthritis express RANKL and interleukin-15 on the cell surface and promote osteoclastogenesis in autologous monocytes. Arthritis Rheum. 2006;54:1151–64. doi: 10.1002/art.21731. http://dx.doi.org/10.1002/art.21731. [DOI] [PubMed] [Google Scholar]
- 77.Djaafar S, Pierroz DD, Chicheportiche R, Zheng XX, Ferrari SL, Ferrari-Lacraz S. Inhibition of T cell-dependent and RANKL-dependent osteoclastogenic processes associated with high levels of bone mass in interleukin-15 receptor-deficient mice. Arthritis Rheum. 2010;62:3300–10. doi: 10.1002/art.27645. http://dx.doi.org/10.1002/art.27645. [DOI] [PubMed] [Google Scholar]
- 78.Bendre MS, Montague DC, Peery T, Akel NS, Gaddy D, Suva LJ. Interleukin-8 stimulation of osteoclastogenesis and bone resorption is a mechanism for the increased osteolysis of metastatic bone disease. Bone. 2003;33:28–37. doi: 10.1016/s8756-3282(03)00086-3. http://dx.doi.org/10.1016/S8756-3282(03)00086-3. [DOI] [PubMed] [Google Scholar]
- 79.Kim SH, Han SY, Azam T, Yoon DY, Dinarello CA. Interleukin-32: a cytokine and inducer of TNFalpha. Immunity. 2005;22:131–42. doi: 10.1016/j.immuni.2004.12.003. http://dx.doi.org/10.1016/S1074-7613-(04)00380-2. [DOI] [PubMed] [Google Scholar]
- 80.Cenci S, Toraldo G, Weitzmann MN, Roggia C, Gao Y, Qian WP, et al. Estrogen deficiency induces bone loss by increasing T cell proliferation and lifespan through IFN-gamma-induced class II transactivator. Proc Natl Acad Sci U S A. 2003;100:10405–10. doi: 10.1073/pnas.1533207100. http://dx.doi.org/10.1073/pnas.1533207100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Vermeire K, Heremans H, Vandeputte M, Huang S, Billiau A, Matthys P. Accelerated collagen-induced arthritis in IFN-gamma receptor-deficient mice. J Immunol. 1997;158:5507–13. [PubMed] [Google Scholar]
- 82.Mann GN, Jacobs TW, Buchinsky FJ, Armstrong EC, Li M, Ke HZ, et al. Interferon-gamma causes loss of bone volume in vivo and fails to ameliorate cyclosporin A-induced osteopenia. Endocrinology. 1994;135:1077–83. doi: 10.1210/endo.135.3.8070349. http://dx.doi.org/10.1210/en.135.3.1077. [DOI] [PubMed] [Google Scholar]
- 83.Key LL, Jr, Rodriguiz RM, Willi SM, Wright NM, Hatcher HC, Eyre DR, et al. Long-term treatment of osteopetrosis with recombinant human interferon gamma. N Engl J Med. 1995;332:1594–9. doi: 10.1056/NEJM199506153322402. http://dx.doi.org/10.1056/NEJM199506153322402. [DOI] [PubMed] [Google Scholar]
- 84.Coelho LF, Magno de Freitas Almeida G, Mennechet FJ, Blangy A, Uze G. Interferon-alpha and -beta differentially regulate osteoclastogenesis: role of differential induction of chemokine CXCL11 expression. Proc Natl Acad Sci U S A. 2005;102:11917–22. doi: 10.1073/pnas.0502188102. http://dx.doi.org/10.1073/pnas.0502188102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Takayanagi H, Kim S, Matsuo K, Suzuki H, Suzuki T, Sato K, et al. RANKL maintains bone homeostasis through c-Fos-dependent induction of interferon-beta. Nature. 2002;416:744–9. doi: 10.1038/416744a. http://dx.doi.org/10.1038/416744a. [DOI] [PubMed] [Google Scholar]
- 86.Hayashi T, Kaneda T, Toyama Y, Kumegawa M, Hakeda Y. Regulation of receptor activator of NF-kappa B ligand-induced osteoclastogenesis by endogenous interferon-beta (INF-beta) and suppressors of cytokine signaling (SOCS). The possible counteracting role of SOCSs- in IFN-beta-inhibited osteoclast formation. J Biol Chem. 2002;277:27880–6. doi: 10.1074/jbc.M203836200. http://dx.doi.org/10.1074/jbc.M203836200. [DOI] [PubMed] [Google Scholar]
- 87.Sakai S, Takaishi H, Matsuzaki K, Kaneko H, Furukawa M, Miyauchi Y, et al. 1-Alpha, 25-dihydroxy vitamin D3 inhibits osteoclastogenesis through IFN-beta-dependent NFATc1 suppression. J Bone Miner Metab. 2009;27:643–52. doi: 10.1007/s00774-009-0084-4. http://dx.doi.org/10.1007/s00774-009-0084-4. [DOI] [PubMed] [Google Scholar]
- 88.van Holten J, Reedquist K, Sattonet-Roche P, Smeets TJ, Plater-Zyberk C, Vervoordeldonk MJ, et al. Treatment with recombinant interferon-beta reduces inflammation and slows cartilage destruction in the collagen-induced arthritis model of rheumatoid arthritis. Arthritis Res Ther. 2004;6:R239–49. doi: 10.1186/ar1165. http://dx.doi.org/10.1186/ar1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Avnet S, Cenni E, Perut F, Granchi D, Brandi ML, Giunti A, et al. Interferon-alpha inhibits in vitro osteoclast differentiation and renal cell carcinoma-induced angiogenesis. Int J Oncol. 2007;30:469–76. [PubMed] [Google Scholar]
- 90.Goodman GR, Dissanayake IR, Gorodetsky E, Zhou H, Ma YF, Jee WS, et al. Interferon-alpha, unlike interferon-gamma, does not cause bone loss in the rat. Bone. 1999;25:459–63. doi: 10.1016/s8756-3282(99)00182-9. http://dx.doi.org/10.1016/S8756-3282(99)00182-9. [DOI] [PubMed] [Google Scholar]
- 91.Mensah KA, Mathian A, Ma L, Xing L, Ritchlin CT, Schwarz EM. Mediation of nonerosive arthritis in a mouse model of lupus by interferon-alpha-stimulated monocyte differentiation that is nonpermissive of osteoclastogenesis. Arthritis Rheum. 2010;62:1127–37. doi: 10.1002/art.27312. http://dx.doi.org/10.1002/art.27312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gur A, Dikici B, Nas K, Bosnak M, Haspolat K, Sarac AJ. Bone mineral density and cytokine levels during interferon therapy in children with chronic hepatitis B: does interferon therapy prevent from osteoporosis? BMC Gastroenterol. 2005;5:30. doi: 10.1186/1471-230X-5-30. http://dx.doi.org/10.1186/1471-230X-5-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lind M, Deleuran B, Yssel H, Fink-Eriksen E, Thestrup-Pedersen K. IL-4 and IL-13, but not IL-10, are chemotactic factors for human osteoblasts. Cytokine. 1995;7:78–82. doi: 10.1006/cyto.1995.1010. http://dx.doi.org/10.1006/cyto.1995.1010. [DOI] [PubMed] [Google Scholar]
- 94.Okada Y, Morimoto I, Ura K, Nakano Y, Tanaka Y, Nishida S, et al. Short-term treatment of recombinant murine interleukin-4 rapidly inhibits bone formation in normal and ovariectomized mice. Bone. 1998;22:361–5. doi: 10.1016/s8756-3282(97)00296-2. http://dx.doi.org/10.1016/S8756-3282(97)00296-2. [DOI] [PubMed] [Google Scholar]
- 95.Lewis DB, Liggitt HD, Effmann EL, Motley ST, Teitelbaum SL, Jepsen KJ, et al. Osteoporosis induced in mice by overproduction of interleukin 4. Proc Natl Acad Sci U S A. 1993;90:11618–22. doi: 10.1073/pnas.90.24.11618. http://dx.doi.org/10.1073/pnas.90.24.11618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Nakano Y, Watanabe K, Morimoto I, Okada Y, Ura K, Sato K, et al. Interleukin-4 inhibits spontaneous and parathyroid hormone-related protein-stimulated osteoclast formation in mice. J Bone Miner Res. 1994;9:1533–9. doi: 10.1002/jbmr.5650091005. http://dx.doi.org/10.1002/jbmr.5650091005. [DOI] [PubMed] [Google Scholar]
- 97.Shioi A, Teitelbaum SL, Ross FP, Welgus HG, Suzuki H, Ohara J, et al. Interleukin 4 inhibits murine osteoclast formation in vitro. J Cell Biochem. 1991;47:272–7. doi: 10.1002/jcb.240470313. http://dx.doi.org/10.1002/jcb.240470313. [DOI] [PubMed] [Google Scholar]
- 98.Onoe Y, Miyaura C, Kaminakayashiki T, Nagai Y, Noguchi K, Chen QR, et al. IL-13 and IL-4 inhibit bone resorption by suppressing cyclooxygenase-2-dependent prostaglandin synthesis in osteoblasts. J Immunol. 1996;156:758–64. [PubMed] [Google Scholar]
- 99.Kitaura H, Nagata N, Fujimura Y, Hotokezaka H, Tatamiya M, Nakao N, et al. Interleukin-4 directly inhibits tumor necrosis factor-alpha-mediated osteoclast formation in mouse bone marrow macrophages. Immunol Lett. 2003;88:193–8. doi: 10.1016/s0165-2478(03)00082-8. http://dx.doi.org/10.1016/S0165-2478-(03)00082-8. [DOI] [PubMed] [Google Scholar]
- 100.Carter NA, Rosser EC, Mauri C. Interleukin-10 produced by B cells is crucial for the suppression of Th17/Th1 responses, induction of T regulatory type 1 cells and reduction of collagen-induced arthritis. Arthritis Res Ther. 2012;14:R32. doi: 10.1186/ar3736. http://dx.doi.org/10.1186/ar3736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Evans KE, Fox SW. Interleukin-10 inhibits osteoclastogenesis by reducing NFATc1 expression and preventing its translocation to the nucleus. BMC Cell Biol. 2007;8:4. doi: 10.1186/1471-2121-8-4. http://dx.doi.org/10.1186/1471-2121-8-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Mohamed SG, Sugiyama E, Shinoda K, Taki H, Hounoki H, Abdel-Aziz HO, et al. Interleukin-10 inhibits RANKL-mediated expression of NFATc1 in part via suppression of c-Fos and c-Jun in RAW264.7 cells and mouse bone marrow cells. Bone. 2007;41:592–602. doi: 10.1016/j.bone.2007.05.016. http://dx.doi.org/10.1016/j.bone.2007.05.016. [DOI] [PubMed] [Google Scholar]
- 103.Park-Min KH, Ji JD, Antoniv T, Reid AC, Silver RB, Humphrey MB, et al. IL-10 suppresses calcium-mediated co-stimulation of receptor activator NF-kappa B signaling during human osteoclast differentiation by inhibiting TREM-2 expression. J Immunol. 2009;183:2444–55. doi: 10.4049/jimmunol.0804165. http://dx.doi.org/10.4049/jimmunol.0804165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Sunyer T, Rothe L, Jiang X, Osdoby P, Collin-Osdoby P. Proinflammatory agents, IL-8 and IL-10, upregulate inducible nitric oxide synthase expression and nitric oxide production in avian osteoclast-like cells. J Cell Biochem. 1996;60:469–83. doi: 10.1002/(sici)1097-4644(19960315)60:4<469::aid-jcb4>3.0.co;2-q. http://dx.doi.org/10.1002/(SICI)1097-4644(19960315)60:4<469::AID-JCB4>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 105.Liu D, Yao S, Wise GE. Effect of interleukin-10 on gene expression of osteoclastogenic regulatory molecules in the rat dental follicle. Eur J Oral Sci. 2006;114:42–9. doi: 10.1111/j.1600-0722.2006.00283.x. http://dx.doi.org/10.1111/j.1600-0722.2006.00283.x. [DOI] [PubMed] [Google Scholar]
- 106.Van Vlasselaer P, Borremans B, Van Den Heuvel R, Van Gorp U, de Waal Malefyt R. Interleukin-10 inhibits the osteogenic activity of mouse bone marrow. Blood. 1993;82:2361–70. [PubMed] [Google Scholar]
- 107.Horwood NJ, Udagawa N, Elliott J, Grail D, Okamura H, Kurimoto M, et al. Interleukin 18 inhibits osteoclast formation via T cell production of granulocyte macrophage colony-stimulating factor. J Clin Invest. 1998;101:595–603. doi: 10.1172/JCI1333. http://dx.doi.org/10.1172/JCI1333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Kitaura H, Fujimura Y, Yoshimatsu M, Kohara H, Morita Y, Aonuma T, et al. IL-12- and IL-18-mediated, nitric oxide-induced apoptosis in TNF-alpha-mediated osteoclastogenesis of bone marrow cells. Calcif Tissue Int. 2011;89:65–73. doi: 10.1007/s00223-011-9494-0. http://dx.doi.org/10.1007/s00223-011-9494-0. [DOI] [PubMed] [Google Scholar]
- 109.Makiishi-Shimobayashi C, Tsujimura T, Iwasaki T, Yamada N, Sugihara A, Okamura H, et al. Interleukin-18 up-regulates osteoprotegerin expression in stromal/osteoblastic cells. Biochem Biophys Res Commun. 2001;281:361–6. doi: 10.1006/bbrc.2001.4380. http://dx.doi.org/10.1006/bbrc.2001.4380. [DOI] [PubMed] [Google Scholar]
- 110.Morita Y, Kitaura H, Yoshimatsu M, Fujimura Y, Kohara H, Eguchi T, et al. IL-18 inhibits TNF-alpha-induced osteoclastogenesis possibly via a T cell-independent mechanism in synergy with IL-12 in vivo. Calcif Tissue Int. 2010;86:242–8. doi: 10.1007/s00223-010-9335-6. http://dx.doi.org/10.1007/s00223-010-9335-6. [DOI] [PubMed] [Google Scholar]
- 111.Kawase Y, Hoshino T, Yokota K, Kuzuhara A, Nakamura M, Maeda Y, et al. Bone malformations in interleukin-18 transgenic mice. J Bone Miner Res. 2003;18:975–83. doi: 10.1359/jbmr.2003.18.6.975. http://dx.doi.org/10.1359/jbmr.2003.18.6.975. [DOI] [PubMed] [Google Scholar]
- 112.Keller J, Catala-Lehnen P, Wintges K, Schulze J, Bickert T, Ito W, et al. Transgenic over-expression of interleukin-33 in osteoblasts results in decreased osteoclastogenesis. Biochem Biophys Res Commun. 2012;417:217–22. doi: 10.1016/j.bbrc.2011.11.088. http://dx.doi.org/10.1016/j.bbrc.2011.11.088. [DOI] [PubMed] [Google Scholar]
- 113.Schulze J, Bickert T, Beil FT, Zaiss MM, Albers J, Wintges K, et al. Interleukin-33 is expressed in differentiated osteoblasts and blocks osteoclast formation from bone marrow precursor cells. J Bone Miner Res. 2011;26:704–17. doi: 10.1002/jbmr.269. http://dx.doi.org/10.1002/jbmr.269. [DOI] [PubMed] [Google Scholar]
- 114.Sato T, Watanabe K, Masuhara M, Hada N, Hakeda Y. Production of IL-7 is increased in ovariectomized mice, but not RANKL mRNA expression by osteoblasts/stromal cells in bone, and IL-7 enhances generation of osteoclast precursors in vitro. J Bone Miner Metab. 2007;25:19–27. doi: 10.1007/s00774-006-0723-y. http://dx.doi.org/10.1007/s00774-006-0723-y. [DOI] [PubMed] [Google Scholar]
- 115.Lee SK, Kalinowski JF, Jacquin C, Adams DJ, Gronowicz G, Lorenzo JA. Interleukin-7 influences osteoclast function in vivo but is not a critical factor in ovariectomy-induced bone loss. J Bone Miner Res. 2006;21:695–702. doi: 10.1359/jbmr.060117. http://dx.doi.org/10.1359/jbmr.060117. [DOI] [PubMed] [Google Scholar]
- 116.Lee SK, Kalinowski JF, Jastrzebski SL, Puddington L, Lorenzo JA. Interleukin-7 is a direct inhibitor of in vitro osteoclastogenesis. Endocrinology. 2003;144:3524–31. doi: 10.1210/en.2002-221057. http://dx.doi.org/10.1210/en.2002-221057. [DOI] [PubMed] [Google Scholar]
- 117.Aguila HL, Mun SH, Kalinowski J, Adams DJ, Lorenzo JA, Lee SK. Osteoblast specific overexpression of human interleukin-7 rescues the bone mass phenotype of interleukin-7 deficient female mice. J Bone Miner Res. 2012 doi: 10.1002/jbmr.1553. http://dx.doi.org/10.1002/jbmr.1553. [DOI] [PMC free article] [PubMed]
- 118.D’Amelio P, Grimaldi A, Bernabei P, Pescarmona GP, Isaia G. Immune system and bone metabolism: Does thymectomy influence postmenopausal bone loss in humans? Bone. 2006;39:658–65. doi: 10.1016/j.bone.2006.03.009. http://dx.doi.org/10.1016/j.bone.2006.03.009. [DOI] [PubMed] [Google Scholar]
- 119.Colucci S, Brunetti G, Cantatore FP, Oranger A, Mori G, Quarta L, et al. Lymphocytes and synovial fluid fibroblasts support osteoclastogenesis through RANKL, TNFalpha, and IL-7 in an in vitro model derived from human psoriatic arthritis. J Pathol. 2007;212:47–55. doi: 10.1002/path.2153. http://dx.doi.org/10.1002/path.2153. [DOI] [PubMed] [Google Scholar]
- 120.Roato I, Gorassini E, Brunetti G, Grano M, Ciuffreda L, Mussa A, et al. IL-7 modulates osteoclastogenesis in patients affected by solid tumors. Ann N Y Acad Sci. 2007;1117:377–84. doi: 10.1196/annals.1402.002. http://dx.doi.org/10.1196/annals.1402.002. [DOI] [PubMed] [Google Scholar]
- 121.van Roon JA, Glaudemans KA, Bijlsma JW, Lafeber FP. Interleukin 7 stimulates tumour necrosis factor alpha and Th1 cytokine production in joints of patients with rheumatoid arthritis. Ann Rheum Dis. 2003;62:113–9. doi: 10.1136/ard.62.2.113. http://dx.doi.org/10.1136/ard.62.2.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Queiroz-Junior CM, Silva MJ, Correa JD, Madeira MF, Garlet TP, Garlet GP, et al. A controversial role for IL-12 in immune response and bone resorption at apical periodontal sites. Clin Dev Immunol. 2010;2010:327417. doi: 10.1155/2010/327417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Hsieh CS, Macatonia SE, Tripp CS, Wolf SF, O’Garra A, Murphy KM. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science. 1993;260:547–9. doi: 10.1126/science.8097338. http://dx.doi.org/10.1126/science.8097338. [DOI] [PubMed] [Google Scholar]
- 124.Takayanagi H. Osteoimmunology: shared mechanisms and crosstalk between the immune and bone systems. Nat Rev Immunol. 2007;7:292–304. doi: 10.1038/nri2062. http://dx.doi.org/10.1038/nri2062. [DOI] [PubMed] [Google Scholar]
- 125.Nagata N, Kitaura H, Yoshida N, Nakayama K. Inhibition of RANKL-induced osteoclast formation in mouse bone marrow cells by IL-12: involvement of IFN-gamma possibly induced from non-T cell population. Bone. 2003;33:721–32. doi: 10.1016/s8756-3282(03)00213-8. http://dx.doi.org/10.1016/S8756-3282-(03)00213-8. [DOI] [PubMed] [Google Scholar]
- 126.Adamopoulos IE, Tessmer M, Chao CC, Adda S, Gorman D, Petro M, et al. IL-23 is critical for induction of arthritis, osteoclast formation, and maintenance of bone mass. J Immunol. 2011;187:951–9. doi: 10.4049/jimmunol.1003986. http://dx.doi.org/10.4049/jimmunol.1003986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Yago T, Nanke Y, Kawamoto M, Furuya T, Kobashigawa T, Kamatani N, et al. IL-23 induces human osteoclastogenesis via IL-17 in vitro, and anti-IL-23 antibody attenuates collagen-induced arthritis in rats. Arthritis Res Ther. 2007;9:R96. doi: 10.1186/ar2297. http://dx.doi.org/10.1186/ar2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Quinn JM, Sims NA, Saleh H, Mirosa D, Thompson K, Bouralexis S, et al. IL-23 inhibits osteoclastogenesis indirectly through lymphocytes and is required for the maintenance of bone mass in mice. J Immunol. 2008;181:5720–9. doi: 10.4049/jimmunol.181.8.5720. [DOI] [PubMed] [Google Scholar]
- 129.Tang Y, Wu X, Lei W, Pang L, Wan C, Shi Z, et al. TGF-beta1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat Med. 2009;15:757–65. doi: 10.1038/nm.1979. http://dx.doi.org/10.1038/nm.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Pacifici R. Estrogen deficiency, T cells and bone loss. Cell Immunol. 2008;252:68–80. doi: 10.1016/j.cellimm.2007.06.008. http://dx.doi.org/10.1016/j.cellimm.2007.06.008. [DOI] [PubMed] [Google Scholar]
- 131.Jilka RL, Hangoc G, Girasole G, Passeri G, Williams DC, Abrams JS, et al. Increased osteoclast development after estrogen loss: mediation by interleukin-6. Science. 1992;257:88–91. doi: 10.1126/science.1621100. http://dx.doi.org/10.1126/science.1621100. [DOI] [PubMed] [Google Scholar]
- 132.Gruber R. Cell biology of osteoimmunology. Wien Med Wochenschr. 2010;160:438–45. doi: 10.1007/s10354-010-0809-y. http://dx.doi.org/10.1007/s10354-010-0809-y. [DOI] [PubMed] [Google Scholar]
- 133.Roggia C, Gao Y, Cenci S, Weitzmann MN, Toraldo G, Isaia G, et al. Up-regulation of TNF-producing T cells in the bone marrow: a key mechanism by which estrogen deficiency induces bone loss in vivo. Proc Natl Acad Sci USA. 2001;98:13960–5. doi: 10.1073/pnas.251534698. http://dx.doi.org/10.1073/pnas.251534698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Cenci S, Weitzmann MN, Roggia C, Namba N, Novack D, Woodring J, et al. Estrogen deficiency induces bone loss by enhancing T-cell production of TNF-alpha. J Clin Invest. 2000;106:1229–37. doi: 10.1172/JCI11066. http://dx.doi.org/10.1172/JCI11066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Lorenzo JA, Naprta A, Rao Y, Alander C, Glaccum M, Widmer M, et al. Mice lacking the type I interleukin-1 receptor do not lose bone mass after ovariectomy. Endocrinology. 1998;139:3022–5. doi: 10.1210/endo.139.6.6128. http://dx.doi.org/10.1210/en.139.6.3022. [DOI] [PubMed] [Google Scholar]
- 136.Ammann P, Rizzoli R, Bonjour JP, Bourrin S, Meyer JM, Vassalli P, et al. Transgenic mice expressing soluble tumor necrosis factor-receptor are protected against bone loss caused by estrogen deficiency. J Clin Invest. 1997;99:1699–703. doi: 10.1172/JCI119333. http://dx.doi.org/10.1172/JCI119333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Lee SK, Kadono Y, Okada F, Jacquin C, Koczon-Jaremko B, Gronowicz G, et al. T lymphocyte-deficient mice lose trabecular bone mass with ovariectomy. J Bone Miner Res. 2006;21:1704–12. doi: 10.1359/jbmr.060726. http://dx.doi.org/10.1359/jbmr.060726. [DOI] [PubMed] [Google Scholar]
- 138.Li Y, Toraldo G, Li A, Yang X, Zhang H, Qian WP, et al. B cells and T cells are critical for the preservation of bone homeostasis and attainment of peak bone mass in vivo. Blood. 2007;109:3839–48. doi: 10.1182/blood-2006-07-037994. http://dx.doi.org/10.1182/blood-2006-07-037994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.D’Amelio P, Grimaldi A, Di Bella S, Brianza SZ, Cristofaro MA, Tamone C, et al. Estrogen deficiency increases osteoclastogenesis up-regulating T cells activity: a key mechanism in osteoporosis. Bone. 2008;43:92–100. doi: 10.1016/j.bone.2008.02.017. http://dx.doi.org/10.1016/j.bone.2008.02.017. [DOI] [PubMed] [Google Scholar]
- 140.Pacifici R, Brown C, Puscheck E, Friedrich E, Slatopolsky E, Maggio D, et al. Effect of surgical menopause and estrogen replacement on cytokine release from human blood mononuclear cells. Proc Natl Acad Sci U S A. 1991;88:5134–8. doi: 10.1073/pnas.88.12.5134. http://dx.doi.org/10.1073/pnas.88.12.5134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Pfeilschifter J, Koditz R, Pfohl M, Schatz H. Changes in proinflammatory cytokine activity after menopause. Endocr Rev. 2002;23:90–119. doi: 10.1210/edrv.23.1.0456. http://dx.doi.org/10.1210/er.23.1.90. [DOI] [PubMed] [Google Scholar]
- 142.Weitzmann MN, Pacifici R. Estrogen regulation of immune cell bone interactions. Ann N Y Acad Sci. 2006;1068:256–74. doi: 10.1196/annals.1346.030. http://dx.doi.org/10.1196/annals.1346.030. [DOI] [PubMed] [Google Scholar]
- 143.Pino AM, Rios S, Astudillo P, Fernandez M, Figueroa P, Seitz G, et al. Concentration of adipogenic and proinflammatory cytokines in the bone marrow supernatant fluid of osteoporotic women. J Bone Miner Res. 2010;25:492–8. doi: 10.1359/jbmr.090802. http://dx.doi.org/10.1359/jbmr.090802. [DOI] [PubMed] [Google Scholar]
- 144.D’Amelio P, Roato I, D’Amico L, Veneziano L, Suman E, Sassi F, et al. Bone and bone marrow pro-osteoclastogenic cytokines are up-regulated in osteoporosis fragility fractures. Osteoporos Int. 2011;22:2869–77. doi: 10.1007/s00198-010-1496-7. http://dx.doi.org/10.1007/s00198-010-1496-7. [DOI] [PubMed] [Google Scholar]
- 145.Scheidt-Nave C, Bismar H, Leidig-Bruckner G, Woitge H, Seibel MJ, Ziegler R, et al. Serum interleukin 6 is a major predictor of bone loss in women specific to the first decade past menopause. J Clin Endocrinol Metab. 2001;86:2032–42. doi: 10.1210/jcem.86.5.7445. http://dx.doi.org/10.1210/jc.86.5.2032. [DOI] [PubMed] [Google Scholar]
- 146.Zupan J, Komadina R, Marc J. The relationship between osteoclastogenic and anti-osteoclastogenic pro-inflammatory cytokines differs in human osteoporotic and osteoarthritic bone tissues. J Biomed Sci. 2012;19:28. doi: 10.1186/1423-0127-19-28. http://dx.doi.org/10.1186/1423-0127-19-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Zupan J, Pristovšek N, Mencej-Bedrač S, Komadina R, Preželj J, Marc J. Interleukin-1α gene variants influence bone mineral density and the risk of osteoporotic hip fractures in elderly Slovenian people. Clin Chem Lab Med. 2012;50:1379–85. doi: 10.1515/cclm-2011-0589. http://dx.doi.org/10.1515/cclm-2011-0589. [DOI] [PubMed] [Google Scholar]
- 148.Eghbali-Fatourechi G, Khosla S, Sanyal A, Boyle WJ, Lacey DL, Riggs BL. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women. J Clin Invest. 2003;111:1221–30. doi: 10.1172/JCI17215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Ralston SH, Russell RG, Gowen M. Estrogen inhibits release of tumor necrosis factor from peripheral blood mononuclear cells in postmenopausal women. J Bone Miner Res. 1990;5:983–8. doi: 10.1002/jbmr.5650050912. http://dx.doi.org/10.1002/jbmr.5650050912. [DOI] [PubMed] [Google Scholar]
- 150.Charatcharoenwitthaya N, Khosla S, Atkinson EJ, McCready LK, Riggs BL. Effect of blockade of TNF-alpha and interleukin-1 action on bone resorption in early postmenopausal women. J Bone Miner Res. 2007;22:724–9. doi: 10.1359/jbmr.070207. http://dx.doi.org/10.1359/jbmr.070207. [DOI] [PubMed] [Google Scholar]
- 151.Richards JB, Joseph L, Schwartzman K, Kreiger N, Tenenhouse A, Goltzman D. The effect of cyclooxygenase-2 inhibitors on bone mineral density: results from the Canadian Multicentre Osteoporosis Study. Osteoporos Int. 2006;17:1410–9. doi: 10.1007/s00198-006-0142-x. http://dx.doi.org/10.1007/s00198-006-0142-x. [DOI] [PubMed] [Google Scholar]
- 152.Zheng SX, Vrindts Y, Lopez M, De Groote D, Zangerle PF, Collette J, et al. Increase in cytokine production (IL-1 beta, IL-6, TNF-alpha but not IFN-gamma, GM-CSF or LIF) by stimulated whole blood cells in postmenopausal osteoporosis. Maturitas. 1997;26:63–71. doi: 10.1016/s0378-5122(96)01080-8. http://dx.doi.org/10.1016/S0378-5122(96)01080-8. [DOI] [PubMed] [Google Scholar]
- 153.Beaudreuil J, Mbalaviele G, Cohen-Solal M, Morieux C, De Vernejoul MC, Orcel P. Short-term local injections of transforming growth factor-beta 1 decrease ovariectomy-stimulated osteoclastic resorption in vivo in rats. J Bone Miner Res. 1995;10:971–7. doi: 10.1002/jbmr.5650100619. http://dx.doi.org/10.1002/jbmr.5650100619. [DOI] [PubMed] [Google Scholar]
- 154.Finkelman RD, Bell NH, Strong DD, Demers LM, Baylink DJ. Ovariectomy selectively reduces the concentration of transforming growth factor beta in rat bone: implications for estrogen deficiency-associated bone loss. Proc Natl Acad Sci U S A. 1992;89:12190–3. doi: 10.1073/pnas.89.24.12190. http://dx.doi.org/10.1073/pnas.89.24.12190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Delvig AA, Lee JJ, Chrzanowska-Lightowlers ZM, Robinson JH. TGF-beta1 and IFN-gamma cross-regulate antigen presentation to CD4 T cells by macrophages. J Leukoc Biol. 2002;72:163–6. [PubMed] [Google Scholar]
- 156.Lee YJ, Han Y, Lu HT, Nguyen V, Qin H, Howe PH, et al. TGF-beta suppresses IFN-gamma induction of class II MHC gene expression by inhibiting class II transactivator messenger RNA expression. J Immunol. 1997;158:2065–75. [PubMed] [Google Scholar]
- 157.Bord S, Beavan S, Ireland D, Horner A, Compston JE. Mechanisms by which high-dose estrogen therapy produces anabolic skeletal effects in postmenopausal women: role of locally produced growth factors. Bone. 2001;29:216–22. doi: 10.1016/s8756-3282(01)00501-4. http://dx.doi.org/10.1016/S8756-3282(01)00501-4. [DOI] [PubMed] [Google Scholar]
- 158.Goldring SR, Gravallese EM. Mechanisms of bone loss in inflammatory arthritis: diagnosis and therapeutic implications. Arthritis Res. 2000;2:33–7. doi: 10.1186/ar67. http://dx.doi.org/10.1186/ar67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Pelletier JP, Martel-Pelletier J, Abramson SB. Osteoarthritis, an inflammatory disease: potential implication for the selection of new therapeutic targets. Arthritis Rheum. 2001;44:1237–47. doi: 10.1002/1529-0131(200106)44:6<1237::AID-ART214>3.0.CO;2-F. http://dx.doi.org/10.1002/1529-0131-(200106)44:6<1237::AID-ART214>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- 160.Bronner F, Farach-Carson MC. Bone and osteoarthritis. London: Springer; 2007. http://dx.doi.org/10.1007/978-1-84628-701-5. [Google Scholar]
- 161.Fernandes JC, Martel-Pelletier J, Pelletier JP. The role of cytokines in osteoarthritis pathophysiology. Biorheology. 2002;39:237–46. [PubMed] [Google Scholar]
- 162.Blom AB, van der Kraan PM, van den Berg WB. Cytokine targeting in osteoarthritis. Curr Drug Targets. 2007;8:283–92. doi: 10.2174/138945007779940179. http://dx.doi.org/10.2174/138945007779940179. [DOI] [PubMed] [Google Scholar]
- 163.Zhang X, Mao Z, Yu C. Suppression of early experimental osteoarthritis by gene transfer of interleukin-1 receptor antagonist and interleukin-10. J Orthop Res. 2004;22:742–50. doi: 10.1016/j.orthres.2003.12.007. http://dx.doi.org/10.1016/j.orthres.2003.12.007. [DOI] [PubMed] [Google Scholar]
- 164.Attur MG, Dave M, Cipolletta C, Kang P, Goldring MB, Patel IR, et al. Reversal of autocrine and paracrine effects of interleukin 1 (IL-1) in human arthritis by type II IL-1 decoy receptor. Potential for pharmacological intervention. J Biol Chem. 2000;275:40307–15. doi: 10.1074/jbc.M002721200. http://dx.doi.org/10.1074/jbc.M002721200. [DOI] [PubMed] [Google Scholar]
- 165.Glasson SS. In vivo osteoarthritis target validation utilizing genetically-modified mice. Curr Drug Targets. 2007;8:367–76. doi: 10.2174/138945007779940061. http://dx.doi.org/10.2174/138945007779940061. [DOI] [PubMed] [Google Scholar]
- 166.Clements KM, Price JS, Chambers MG, Visco DM, Poole AR, Mason RM. Gene deletion of either interleukin-1beta, interleukin-1beta-converting enzyme, inducible nitric oxide synthase, or stromelysin 1 accelerates the development of knee osteoarthritis in mice after surgical transection of the medial collateral ligament and partial medial meniscectomy. Arthritis Rheum. 2003;48:3452–63. doi: 10.1002/art.11355. http://dx.doi.org/10.1002/art.11355. [DOI] [PubMed] [Google Scholar]
- 167.Blom AB, van Lent PL, Holthuysen AE, van der Kraan PM, Roth J, van Rooijen N, et al. Synovial lining macrophages mediate osteophyte formation during experimental osteoarthritis. Osteoarthritis Cartilage. 2004;12:627–35. doi: 10.1016/j.joca.2004.03.003. http://dx.doi.org/10.1016/j.joca.2004.03.003. [DOI] [PubMed] [Google Scholar]
- 168.Blaney Davidson EN, van der Kraan PM, van den Berg WB. TGF-beta and osteoarthritis. Osteoarthritis Cartilage. 2007;15:597–604. doi: 10.1016/j.joca.2007.02.005. http://dx.doi.org/10.1016/j.joca.2007.02.005. [DOI] [PubMed] [Google Scholar]
- 169.Scharstuhl A, Vitters EL, van der Kraan PM, van den Berg WB. Reduction of osteophyte formation and synovial thickening by adenoviral overexpression of transforming growth factor beta/bone morphogenetic protein inhibitors during experimental osteoarthritis. Arthritis Rheum. 2003;48:3442–51. doi: 10.1002/art.11328. http://dx.doi.org/10.1002/art.11328. [DOI] [PubMed] [Google Scholar]
- 170.Lis K, Sypniewska G, Nowacki W. IGF-1, cytokines and biochemical bone turnover markers in synovial fluid and serum of patients with primary and secondary osteoarthritis of the hip. BiochemMed. 2006;16:127–36. [Google Scholar]
- 171.Veys EM, Menkes CJ, Emery P. A randomized, double-blind study comparing twenty-four-week treatment with recombinant interferon-gamma versus placebo in the treatment of rheumatoid arthritis. Arthritis Rheum. 1997;40:62–8. doi: 10.1002/art.1780400110. http://dx.doi.org/10.1002/art.1780400110. [DOI] [PubMed] [Google Scholar]
- 172.Hofbauer LC, Lacey DL, Dunstan CR, Spelsberg TC, Riggs BL, Khosla S. Interleukin-1beta and tumor necrosis factor-alpha, but not interleukin-6, stimulate osteoprotegerin ligand gene expression in human osteoblastic cells. Bone. 1999;25:255–9. doi: 10.1016/s8756-3282(99)00162-3. http://dx.doi.org/10.1016/S8756-3282-(99)00162-3. [DOI] [PubMed] [Google Scholar]
- 173.Quinn JM, Horwood NJ, Elliott J, Gillespie MT, Martin TJ. Fibroblastic stromal cells express receptor activator of NF-kappa B ligand and support osteoclast differentiation. J Bone Miner Res. 2000;15:1459–66. doi: 10.1359/jbmr.2000.15.8.1459. http://dx.doi.org/10.1359/jbmr.2000.15.8.1459. [DOI] [PubMed] [Google Scholar]
- 174.Jimi E, Nakamura I, Duong LT, Ikebe T, Takahashi N, Rodan GA, et al. Interleukin 1 induces multinucleation and bone-resorbing activity of osteoclasts in the absence of osteoblasts/stromal cells. Exp Cell Res. 1999;247:84–93. doi: 10.1006/excr.1998.4320. http://dx.doi.org/10.1006/excr.1998.4320. [DOI] [PubMed] [Google Scholar]
- 175.Lader CS, Flanagan AM. Prostaglandin E2, interleukin 1alpha, and tumor necrosis factor-alpha increase human osteoclast formation and bone resorption in vitro. Endocrinology. 1998;139:3157–64. doi: 10.1210/endo.139.7.6085. http://dx.doi.org/10.1210/en.139.7.3157. [DOI] [PubMed] [Google Scholar]
- 176.Lam J, Takeshita S, Barker JE, Kanagawa O, Ross FP, Teitelbaum SL. TNF-alpha induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J Clin Invest. 2000;106:1481–8. doi: 10.1172/JCI11176. http://dx.doi.org/10.1172/JCI11176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Kobayashi K, Takahashi N, Jimi E, Udagawa N, Takami M, Kotake S, et al. Tumor necrosis factor alpha stimulates osteoclast differentiation by a mechanism independent of the ODF/RANKL-RANK interaction. J Exp Med. 2000;191:275–86. doi: 10.1084/jem.191.2.275. http://dx.doi.org/10.1084/jem.191.2.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Kim N, Kadono Y, Takami M, Lee J, Lee SH, Okada F, et al. Osteoclast differentiation independent of the TRANCE-RANK-TRAF6 axis. J Exp Med. 2005;202:589–95. doi: 10.1084/jem.20050978. http://dx.doi.org/10.1084/jem.20050978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Kudo O, Fujikawa Y, Itonaga I, Sabokbar A, Torisu T, Athanasou NA. Proinflammatory cytokine (TNFalpha/IL-1alpha) induction of human osteoclast formation. J Pathol. 2002;198:220–7. doi: 10.1002/path.1190. http://dx.doi.org/10.1002/path.1190. [DOI] [PubMed] [Google Scholar]
- 180.Palmqvist P, Persson E, Conaway HH, Lerner UH. IL-6, leukemia inhibitory factor, and oncostatin M stimulate bone resorption and regulate the expression of receptor activator of NF-kappa B ligand, osteoprotegerin, and receptor activator of NF-kappa B in mouse calvariae. J Immunol. 2002;169:3353–62. doi: 10.4049/jimmunol.169.6.3353. [DOI] [PubMed] [Google Scholar]
- 181.Kudo O, Sabokbar A, Pocock A, Itonaga I, Fujikawa Y, Athanasou NA. Interleukin-6 and interleukin-11 support human osteoclast formation by a RANKL-independent mechanism. Bone. 2003;32:1–7. doi: 10.1016/s8756-3282(02)00915-8. http://dx.doi.org/10.1016/S8756-3282(02)00915-8. [DOI] [PubMed] [Google Scholar]
- 182.Devlin RD, Reddy SV, Savino R, Ciliberto G, Roodman GD. IL-6 mediates the effects of IL-1 or TNF, but not PTHrP or 1,25(OH)2D3, on osteoclast-like cell formation in normal human bone marrow cultures. J Bone Miner Res. 1998;13:393–9. doi: 10.1359/jbmr.1998.13.3.393. http://dx.doi.org/10.1359/jbmr.1998.13.3.393. [DOI] [PubMed] [Google Scholar]
- 183.Gorny G, Shaw A, Oursler MJ. IL-6, LIF, and TNF-alpha regulation of GM-CSF inhibition of osteoclastogenesis in vitro. Exp Cell Res. 2004;294:149–58. doi: 10.1016/j.yexcr.2003.11.009. http://dx.doi.org/10.1016/j.yexcr.2003.11.009. [DOI] [PubMed] [Google Scholar]
- 184.Liu XH, Kirschenbaum A, Yao S, Levine AC. Interactive effect of interleukin-6 and prostaglandin E2 on osteoclastogenesis via the OPG/RANKL/RANK system. Ann N Y Acad Sci. 2006;1068:225–33. doi: 10.1196/annals.1346.047. http://dx.doi.org/10.1196/annals.1346.047. [DOI] [PubMed] [Google Scholar]
- 185.Bendre MS, Margulies AG, Walser B, Akel NS, Bhattacharrya S, Skinner RA, et al. Tumor-derived interleukin-8 stimulates osteolysis independent of the receptor activator of nuclear factor-kappaB ligand pathway. Cancer Res. 2005;65:11001–9. doi: 10.1158/0008-5472.CAN-05-2630. http://dx.doi.org/10.1158/0008-5472.CAN-05-2630. [DOI] [PubMed] [Google Scholar]
- 186.Horwood NJ, Elliott J, Martin TJ, Gillespie MT. Osteotropic agents regulate the expression of osteoclast differentiation factor and osteoprotegerin in osteoblastic stromal cells. Endocrinology. 1998;139:4743–6. doi: 10.1210/endo.139.11.6433. http://dx.doi.org/10.1210/en.139.11.4743. [DOI] [PubMed] [Google Scholar]
- 187.Nakchbandi IA, Mitnick MA, Masiukiewicz US, Sun BH, Insogna KL. IL-6 negatively regulates IL-11 production in vitro and in vivo. Endocrinology. 2001;142:3850–6. doi: 10.1210/endo.142.9.8368. http://dx.doi.org/10.1210/en.142.9.3850. [DOI] [PubMed] [Google Scholar]
- 188.Ogata Y, Kukita A, Kukita T, Komine M, Miyahara A, Miyazaki S, et al. A novel role of IL-15 in the development of osteoclasts: inability to replace its activity with IL-2. J Immunol. 1999;162:2754–60. [PubMed] [Google Scholar]
- 189.Kotake S, Udagawa N, Takahashi N, Matsuzaki K, Itoh K, Ishiyama S, et al. IL-17 in synovial fluids from patients with rheumatoid arthritis is a potent stimulator of osteoclastogenesis. J Clin Invest. 1999;103:1345–52. doi: 10.1172/JCI5703. http://dx.doi.org/10.1172/JCI5703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Yago T, Nanke Y, Ichikawa N, Kobashigawa T, Mogi M, Kamatani N, et al. IL-17 induces osteoclastogenesis from human monocytes alone in the absence of osteoblasts, which is potently inhibited by anti-TNF-alpha antibody: a novel mechanism of osteoclastogenesis by IL-17. J Cell Biochem. 2009;108:947–55. doi: 10.1002/jcb.22326. http://dx.doi.org/10.1002/jcb.22326. [DOI] [PubMed] [Google Scholar]
- 191.Mabilleau G, Sabokbar A. Interleukin-32 promotes osteoclast differentiation but not osteoclast activation. PLoS One. 2009;4:e4173. doi: 10.1371/journal.pone.0004173. http://dx.doi.org/10.1371/journal.pone.0004173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Kamolmatyakul S, Chen W, Li YP. Interferon-gamma down-regulates gene expression of cathepsin K in osteoclasts and inhibits osteoclast formation. J Dent Res. 2001;80:351–5. doi: 10.1177/00220345010800011001. http://dx.doi.org/10.1177/00220345010800011001. [DOI] [PubMed] [Google Scholar]
- 193.Takahashi N, Mundy GR, Roodman GD. Recombinant human interferon-gamma inhibits formation of human osteoclast-like cells. J Immunol. 1986;137:3544–9. [PubMed] [Google Scholar]
- 194.Kohara H, Kitaura H, Fujimura Y, Yoshimatsu M, Morita Y, Eguchi T, et al. IFN-gamma directly inhibits TNF-alpha-induced osteoclastogenesis in vitro and in vivo and induces apoptosis mediated by Fas/Fas ligand interactions. Immunol Lett. 2011;137:53–61. doi: 10.1016/j.imlet.2011.02.017. http://dx.doi.org/10.1016/j.imlet.2011.02.017. [DOI] [PubMed] [Google Scholar]
- 195.Fox SW, Chambers TJ. Interferon-gamma directly inhibits TRANCE-induced osteoclastogenesis. Biochem Biophys Res Commun. 2000;276:868–72. doi: 10.1006/bbrc.2000.3577. http://dx.doi.org/10.1006/bbrc.2000.3577. [DOI] [PubMed] [Google Scholar]
- 196.Zheng H, Yu X, Collin-Osdoby P, Osdoby P. RANKL stimulates inducible nitric-oxide synthase expression and nitric oxide production in developing osteoclasts. An autocrine negative feedback mechanism triggered by RANKL-induced interferon-beta via NF-kappaB that restrains osteoclastogenesis and bone resorption. J Biol Chem. 2006;281:15809–20. doi: 10.1074/jbc.M513225200. http://dx.doi.org/10.1074/jbc.M513225200. [DOI] [PubMed] [Google Scholar]
- 197.Cheng J, Liu J, Shi Z, Xu D, Luo S, Siegal GP, et al. Interleukin-4 inhibits RANKL-induced NFATc1 expression via STAT6: a novel mechanism mediating its blockade of osteoclastogenesis. J Cell Biochem. 2011;112:3385–92. doi: 10.1002/jcb.23269. http://dx.doi.org/10.1002/jcb.23269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Kamel Mohamed SG, Sugiyama E, Shinoda K, Hounoki H, Taki H, Maruyama M, et al. Interleukin-4 inhibits RANKL-induced expression of NFATc1 and c-Fos: a possible mechanism for downregulation of osteoclastogenesis. Biochem Biophys Res Commun. 2005;329:839–45. doi: 10.1016/j.bbrc.2005.02.049. http://dx.doi.org/10.1016/j.bbrc.2005.02.049. [DOI] [PubMed] [Google Scholar]
- 199.Palmqvist P, Lundberg P, Persson E, Johansson A, Lundgren I, Lie A, et al. Inhibition of hormone and cytokine-stimulated osteoclastogenesis and bone resorption by interleukin-4 and interleukin-13 is associated with increased osteoprotegerin and decreased RANKL and RANK in a STAT6-dependent pathway. J Biol Chem. 2006;281:2414–29. doi: 10.1074/jbc.M510160200. http://dx.doi.org/10.1074/jbc.M510160200. [DOI] [PubMed] [Google Scholar]
- 200.Yamada A, Takami M, Kawawa T, Yasuhara R, Zhao B, Mochizuki A, et al. Interleukin-4 inhibition of osteoclast differentiation is stronger than that of interleukin-13 and they are equivalent for induction of osteoprotegerin production from osteoblasts. Immunology. 2007;120:573–9. doi: 10.1111/j.1365-2567.2006.02538.x. http://dx.doi.org/10.1111/j.1365-2567.2006.02538.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Stein NC, Kreutzmann C, Zimmermann SP, Niebergall U, Hellmeyer L, Goettsch C, et al. Interleukin-4 and interleukin-13 stimulate the osteoclast inhibitor osteoprotegerin by human endothelial cells through the STAT6 pathway. J Bone Miner Res. 2008;23:750–8. doi: 10.1359/jbmr.080203. http://dx.doi.org/10.1359/jbmr.080203. [DOI] [PubMed] [Google Scholar]
- 202.Mirosavljevic D, Quinn JM, Elliott J, Horwood NJ, Martin TJ, Gillespie MT. T-cells mediate an inhibitory effect of interleukin-4 on osteoclastogenesis. J Bone Miner Res. 2003;18:984–93. doi: 10.1359/jbmr.2003.18.6.984. http://dx.doi.org/10.1359/jbmr.2003.18.6.984. [DOI] [PubMed] [Google Scholar]
- 203.Wei S, Wang MW, Teitelbaum SL, Ross FP. Interleukin-4 reversibly inhibits osteoclastogenesis via inhibition of NF-kappa B and mitogen-activated protein kinase signaling. J Biol Chem. 2002;277:6622–30. doi: 10.1074/jbc.M104957200. http://dx.doi.org/10.1074/jbc.M104957200. [DOI] [PubMed] [Google Scholar]
- 204.Dai SM, Nishioka K, Yudoh K. Interleukin (IL) 18 stimulates osteoclast formation through synovial T cells in rheumatoid arthritis: comparison with IL1 beta and tumour necrosis factor alpha. Ann Rheum Dis. 2004;63:1379–86. doi: 10.1136/ard.2003.018481. http://dx.doi.org/10.1136/ard.2003.018481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Hartgring SA, Bijlsma JW, Lafeber FP, van Roon JA. Interleukin-7 induced immunopathology in arthritis. Ann Rheum Dis. 2006;65(Suppl 3):iii69–74. doi: 10.1136/ard.2006.058479. http://dx.doi.org/10.1136/ard.2006.058479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Weitzmann MN, Cenci S, Rifas L, Brown C, Pacifici R. Interleukin-7 stimulates osteoclast formation by up-regulating the T-cell production of soluble osteoclastogenic cytokines. Blood. 2000;96:1873–8. [PubMed] [Google Scholar]
- 207.Toraldo G, Roggia C, Qian WP, Pacifici R, Weitzmann MN. IL-7 induces bone loss in vivo by induction of receptor activator of nuclear factor kappa B ligand and tumor necrosis factor alpha from T cells. Proc Natl Acad Sci U S A. 2003;100:125–30. doi: 10.1073/pnas.0136772100. http://dx.doi.org/10.1073/pnas.0136772100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Amcheslavsky A, Bar-Shavit Z. Interleukin (IL)-12 mediates the anti-osteoclastogenic activity of CpG-oligodeoxynucleotides. J Cell Physiol. 2006;207:244–50. doi: 10.1002/jcp.20563. http://dx.doi.org/10.1002/jcp.20563. [DOI] [PubMed] [Google Scholar]
- 209.Yoshimatsu M, Kitaura H, Fujimura Y, Eguchi T, Kohara H, Morita Y, et al. IL-12 inhibits TNF-alpha induced osteoclastogenesis via a T cell-independent mechanism in vivo. Bone. 2009;45:1010–6. doi: 10.1016/j.bone.2009.07.079. http://dx.doi.org/10.1016/j.bone.2009.07.079. [DOI] [PubMed] [Google Scholar]
- 210.Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–8. doi: 10.1038/nature04753. http://dx.doi.org/10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- 211.Ju JH, Cho ML, Moon YM, Oh HJ, Park JS, Jhun JY, et al. IL-23 induces receptor activator of NF-kappaB ligand expression on CD4+ T cells and promotes osteoclastogenesis in an autoimmune arthritis model. J Immunol. 2008;181:1507–18. doi: 10.4049/jimmunol.181.2.1507. [DOI] [PubMed] [Google Scholar]
- 212.Duvallet E, Semerano L, Assier E, Falgarone G, Boissier MC. Interleukin-23: a key cytokine in inflammatory diseases. Ann Med. 2011;43:503–11. doi: 10.3109/07853890.2011.577093. http://dx.doi.org/10.3109/07853890.2011.577093. [DOI] [PubMed] [Google Scholar]
- 213.Kamiya S, Nakamura C, Fukawa T, Ono K, Ohwaki T, Yoshimoto T, et al. Effects of IL-23 and IL-27 on osteoblasts and osteoclasts: inhibitory effects on osteoclast differentiation. J Bone Miner Metab. 2007;25:277–85. doi: 10.1007/s00774-007-0766-8. http://dx.doi.org/10.1007/s00774-007-0766-8. [DOI] [PubMed] [Google Scholar]