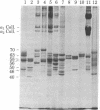
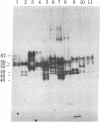
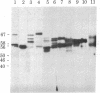
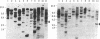Abstract
We have traced the evolutionary origins of keratin-like sequences to the genomes of lower eukaryotes. The proteins encoded by these genes have evolved to form the intermediate filaments that comprise the backbone of vertebrate skin cells. Two related but distinct types of keratins encoded by two separate multigene subfamilies are expressed in the epidermal keratinocytes of vertebrate species from fish to human. Both at the level of protein and at the level of DNA, these two classes of keratins are coordinately conserved throughout vertebrate evolution, indicating the central role that both types of keratins must play in the assembly and structure of the 8-nm filament.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baden H. P., Lee L. D. Fibrous protein of human epidermis. J Invest Dermatol. 1978 Aug;71(2):148–151. doi: 10.1111/1523-1747.ep12546905. [DOI] [PubMed] [Google Scholar]
- Baden H. P., Maderson P. F. Morphological and biophysical identification of fibrous proteins in the amniote epidermis. J Exp Zool. 1970 Jun;174(2):225–232. doi: 10.1002/jez.1401740211. [DOI] [PubMed] [Google Scholar]
- Baden H., Sviokla S., Roth I. The structural protein of reptilian scales. J Exp Zool. 1974 Feb;187(2):287–294. doi: 10.1002/jez.1401870212. [DOI] [PubMed] [Google Scholar]
- Bereiter-Hahn J., Osborn M., Weber K., Vöth M. Filament organization and formation of microridges at the surface of fish epidermis. J Ultrastruct Res. 1979 Dec;69(3):316–330. doi: 10.1016/s0022-5320(79)80050-7. [DOI] [PubMed] [Google Scholar]
- Brysk M. M., Gray R. H., Bernstein I. A. Tonofilament protein from newborn rat epidermis. Isolation, localization, and biosynthesis of marker of epidermal differentiation. J Biol Chem. 1977 Mar 25;252(6):2127–2133. [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Cleveland D. W., Lopata M. A., MacDonald R. J., Cowan N. J., Rutter W. J., Kirschner M. W. Number and evolutionary conservation of alpha- and beta-tubulin and cytoplasmic beta- and gamma-actin genes using specific cloned cDNA probes. Cell. 1980 May;20(1):95–105. doi: 10.1016/0092-8674(80)90238-x. [DOI] [PubMed] [Google Scholar]
- Culbertson V. B., Freedberg I. M. Mammalian epidermal keratin: isolation and characterization of the alpha-helical proteins from newborn rat. Biochim Biophys Acta. 1977 Jan 25;490(1):178–191. doi: 10.1016/0005-2795(77)90118-0. [DOI] [PubMed] [Google Scholar]
- Dale B. A., Stern I. B., Rabin M., Huang L. The identification of fibrous proteins in fetal rat epidermis by electrophoretic and immunologic techniques. J Invest Dermatol. 1976 Apr;66(4):230–235. doi: 10.1111/1523-1747.ep12482148. [DOI] [PubMed] [Google Scholar]
- Dellagi K., Brouet J. C., Perreau J., Paulin D. Human monoclonal IgM with autoantibody activity against intermediate filaments. Proc Natl Acad Sci U S A. 1982 Jan;79(2):446–450. doi: 10.1073/pnas.79.2.446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falkner F. G., Saumweber H., Biessmann H. Two Drosophila melanogaster proteins related to intermediate filament proteins of vertebrate cells. J Cell Biol. 1981 Oct;91(1):175–183. doi: 10.1083/jcb.91.1.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchs E. V., Coppock S. M., Green H., Cleveland D. W. Two distinct classes of keratin genes and their evolutionary significance. Cell. 1981 Nov;27(1 Pt 2):75–84. doi: 10.1016/0092-8674(81)90362-7. [DOI] [PubMed] [Google Scholar]
- Fuchs E., Green H. Changes in keratin gene expression during terminal differentiation of the keratinocyte. Cell. 1980 Apr;19(4):1033–1042. doi: 10.1016/0092-8674(80)90094-x. [DOI] [PubMed] [Google Scholar]
- Fuchs E., Green H. Multiple keratins of cultured human epidermal cells are translated from different mRNA molecules. Cell. 1979 Jul;17(3):573–582. doi: 10.1016/0092-8674(79)90265-4. [DOI] [PubMed] [Google Scholar]
- Fuchs E., Green H. The expression of keratin genes in epidermis and cultured epidermal cells. Cell. 1978 Nov;15(3):887–897. doi: 10.1016/0092-8674(78)90273-8. [DOI] [PubMed] [Google Scholar]
- Geisler N., Plessmann U., Weber K. Related amino acid sequences in neurofilaments and non-neural intermediate filaments. Nature. 1982 Apr 1;296(5856):448–450. doi: 10.1038/296448a0. [DOI] [PubMed] [Google Scholar]
- Geisler N., Weber K. Comparison of the proteins of two immunologically distinct intermediate-sized filaments by amino acid sequence analysis: desmin and vimentin. Proc Natl Acad Sci U S A. 1981 Jul;78(7):4120–4123. doi: 10.1073/pnas.78.7.4120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gown A. M., Vogel A. M. Monoclonal antibodies to intermediate filament proteins of human cells: unique and cross-reacting antibodies. J Cell Biol. 1982 Nov;95(2 Pt 1):414–424. doi: 10.1083/jcb.95.2.414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greer C., Schekman R. Actin from Saccharomyces cerevisiae. Mol Cell Biol. 1982 Oct;2(10):1270–1278. doi: 10.1128/mcb.2.10.1270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haber J. E., Peloquin J. G., Halvorson H. O., Borisy G. G. Colcemid inhibition of cell growth and the characterization of a colcemid-binding activity in Saccharomyces cerevisiae. J Cell Biol. 1972 Nov;55(2):355–367. doi: 10.1083/jcb.55.2.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanukoglu I., Fuchs E. The cDNA sequence of a Type II cytoskeletal keratin reveals constant and variable structural domains among keratins. Cell. 1983 Jul;33(3):915–924. doi: 10.1016/0092-8674(83)90034-x. [DOI] [PubMed] [Google Scholar]
- Hanukoglu I., Fuchs E. The cDNA sequence of a human epidermal keratin: divergence of sequence but conservation of structure among intermediate filament proteins. Cell. 1982 Nov;31(1):243–252. doi: 10.1016/0092-8674(82)90424-x. [DOI] [PubMed] [Google Scholar]
- Hanukoglu I., Tanese N., Fuchs E. Complementary DNA sequence of a human cytoplasmic actin. Interspecies divergence of 3' non-coding regions. J Mol Biol. 1983 Feb 5;163(4):673–678. doi: 10.1016/0022-2836(83)90117-1. [DOI] [PubMed] [Google Scholar]
- Henrikson R. C., Matoltsy A. G. The fine structure of teleost epidermis. 1. Introduction and filament-containing cells. J Ultrastruct Res. 1967 Dec 12;21(3):194–212. doi: 10.1016/s0022-5320(67)80091-1. [DOI] [PubMed] [Google Scholar]
- Inoue N., Fukuyama K., Epstein W. L. Immunochemical studies of proteins in epidermal cornified cells of human and newborn rat. Biochim Biophys Acta. 1976 Jul 19;439(1):95–106. doi: 10.1016/0005-2795(76)90165-3. [DOI] [PubMed] [Google Scholar]
- Kalfayan L., Wensink P. C. Developmental regulation of Drosophila alpha-tubulin genes. Cell. 1982 May;29(1):91–98. doi: 10.1016/0092-8674(82)90093-9. [DOI] [PubMed] [Google Scholar]
- Kim K. H., Rheinwald J. G., Fuchs E. V. Tissue specificity of epithelial keratins: differential expression of mRNAs from two multigene families. Mol Cell Biol. 1983 Apr;3(4):495–502. doi: 10.1128/mcb.3.4.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai E. Y., Walsh C., Wardell D., Fulton C. Programmed appearance of translatable flagellar tubulin mRNA during cell differentiation in Naegleria. Cell. 1979 Aug;17(4):867–878. doi: 10.1016/0092-8674(79)90327-1. [DOI] [PubMed] [Google Scholar]
- Lazarides E. Intermediate filaments: a chemically heterogeneous, developmentally regulated class of proteins. Annu Rev Biochem. 1982;51:219–250. doi: 10.1146/annurev.bi.51.070182.001251. [DOI] [PubMed] [Google Scholar]
- Matoltsy A. G., Huszar T. Keratinization of the reptilian epidermis: an ultrastructural study of the turtle skin. J Ultrastruct Res. 1972 Jan;38(1):87–101. doi: 10.1016/s0022-5320(72)90085-8. [DOI] [PubMed] [Google Scholar]
- Matoltsy A. G. Keratinization of the avian epidermis: an ultrastructural study of the newborn chick skin. J Ultrastruct Res. 1969 Dec;29(5):438–458. doi: 10.1016/s0022-5320(69)90065-3. [DOI] [PubMed] [Google Scholar]
- Moll R., Franke W. W., Schiller D. L., Geiger B., Krepler R. The catalog of human cytokeratins: patterns of expression in normal epithelia, tumors and cultured cells. Cell. 1982 Nov;31(1):11–24. doi: 10.1016/0092-8674(82)90400-7. [DOI] [PubMed] [Google Scholar]
- Neff N. F., Thomas J. H., Grisafi P., Botstein D. Isolation of the beta-tubulin gene from yeast and demonstration of its essential function in vivo. Cell. 1983 May;33(1):211–219. doi: 10.1016/0092-8674(83)90350-1. [DOI] [PubMed] [Google Scholar]
- Ng R., Abelson J. Isolation and sequence of the gene for actin in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1980 Jul;77(7):3912–3916. doi: 10.1073/pnas.77.7.3912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruss R. M., Mirsky R., Raff M. C., Thorpe R., Dowding A. J., Anderton B. H. All classes of intermediate filaments share a common antigenic determinant defined by a monoclonal antibody. Cell. 1981 Dec;27(3 Pt 2):419–428. doi: 10.1016/0092-8674(81)90383-4. [DOI] [PubMed] [Google Scholar]
- Reeves O. R. Adult amphibian epidermal proteins: biochemical characterization and developmental appearance. J Embryol Exp Morphol. 1975 Aug;34(1):55–73. [PubMed] [Google Scholar]
- Rice R. H., Means G. E. Radioactive labeling of proteins in vitro. J Biol Chem. 1971 Feb 10;246(3):831–832. [PubMed] [Google Scholar]
- Roop D. R., Hawley-Nelson P., Cheng C. K., Yuspa S. H. Keratin gene expression in mouse epidermis and cultured epidermal cells. Proc Natl Acad Sci U S A. 1983 Feb;80(3):716–720. doi: 10.1073/pnas.80.3.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schliwa M. Cytoarchitecture of surface layer cells of the teleost epidermis. J Ultrastruct Res. 1975 Sep;52(3):377–386. doi: 10.1016/s0022-5320(75)80076-1. [DOI] [PubMed] [Google Scholar]
- Schweizer J., Goerttler K. Synthesis in vitro of keratin polypeptides directed by mRNA isolated from newborn and adult mouse epidermis. Eur J Biochem. 1980 Nov;112(2):243–249. doi: 10.1111/j.1432-1033.1980.tb07200.x. [DOI] [PubMed] [Google Scholar]
- Skerrow D., Hunter I. Protein modifications during the keratinization of normal and psoriatic human epidermis. Biochim Biophys Acta. 1978 Dec 20;537(2):474–484. doi: 10.1016/0005-2795(78)90532-9. [DOI] [PubMed] [Google Scholar]
- Skerrow D., Matoltsy A. G., Matoltsy M. N. Isolation and characterization of the helical regions of epidermal prekeratin. J Biol Chem. 1973 Jul 10;248(13):4820–4826. [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Steinert P. M., Idler W. W. The polypeptide composition of bovine epidermal alpha-keratin. Biochem J. 1975 Dec;151(3):603–614. doi: 10.1042/bj1510603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinert P. M., Rice R. H., Roop D. R., Trus B. L., Steven A. C. Complete amino acid sequence of a mouse epidermal keratin subunit and implications for the structure of intermediate filaments. Nature. 1983 Apr 28;302(5911):794–800. doi: 10.1038/302794a0. [DOI] [PubMed] [Google Scholar]
- Sun T. T., Green H. Keratin filaments of cultured human epidermal cells. Formation of intermolecular disulfide bonds during terminal differentiation. J Biol Chem. 1978 Mar 25;253(6):2053–2060. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valenzuela P., Quiroga M., Zaldivar J., Rutter W. J., Kirschner M. W., Cleveland D. W. Nucleotide and corresponding amino acid sequences encoded by alpha and beta tubulin mRNAs. Nature. 1981 Feb 19;289(5799):650–655. doi: 10.1038/289650a0. [DOI] [PubMed] [Google Scholar]
- Vandekerckhove J., Weber K. Mammalian cytoplasmic actins are the products of at least two genes and differ in primary structure in at least 25 identified positions from skeletal muscle actins. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1106–1110. doi: 10.1073/pnas.75.3.1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyld J. A., Brush A. H. The molecular heterogeneity and diversity of reptilian keratins. J Mol Evol. 1979 Apr 12;12(4):331–347. doi: 10.1007/BF01732028. [DOI] [PubMed] [Google Scholar]
- Yuspa S. H., Poirier M. C., Harness J. R., Olsom D. R., Steinert P. M. Specific quantification of mouse and human keratin proteins by radioimmunoassay. Biochem J. 1980 Apr 1;187(1):281–284. doi: 10.1042/bj1870281. [DOI] [PMC free article] [PubMed] [Google Scholar]