Abstract
Background
Methicillin-resistant Staphylococcus aureus (MRSA) has become one of the most prevalent pathogens responsible for nosocomial infections throughout the world. As clinical MRSA diagnosis is concerned, current diagnostic methodologies are restricted by significant drawbacks and novel methods are required for MRSA detection. This study aimed at developing a simple loop-mediated isothermal amplification (LAMP) assay targeting on orfX for the rapid detection of methicillin-resistance Staphylococcus aureus (MRSA).
Results
The protocol was designed by targeting orfX, a highly conserved open reading frame in S. aureus. One hundred and sixteen reference strains, including 52 Gram-positive and 64 Gram-negative isolates, were included for evaluation and optimization of the orfX-LAMP assay. This assay had been further performed on 667 Staphylococcus (566 MRSA, 25 MSSA, 53 MRCNS and 23 MSCNS) strains and were comparatively validated by PCR assay using primers F3 and B3, with rapid template DNA processing, simple equipments (water bath) and direct result determination (both naked eye and under UV light) applied. The indispensability of each primer had been confirmed, and the optimal amplification was obtained under 65°C for 45 min. The 25 μl reactant was found to be the most cost-efficient volume, and the detection limit was determined to be 10 DNA copies and 10 CFU/reaction. High specificity was observed when orfX-LAMP assay was subjected to 116 reference strains. For application, 557 (98.4%, 557/566) and 519 (91.7%, 519/566) tested strains had been detected positive by LAMP and PCR assays. The detection rate, positive predictive value (PPV) and negative predictive value (NPV) of orfX-LAMP were 98.4%, 100% and 92.7% respectively.
Conclusions
The established orfX-LAMP assay had been demonstrated to be a valid and rapid detection method on MRSA.
Keywords: Loop-mediated isothermal amplification (LAMP), MRSA, OrfX
Background
As a group of Gram-positive bacteria, staphylococci strains are responsible for various tissues infection and a wide range of diseases, including skin infections, pneumonia, endocarditis, osteomyelitis, gastroenteritis, scalded skin syndrome and toxic shock syndrome [1-4]. Within species of staphylococci, Staphylococcus aureus has been considered to be a major concern in both medicine and food safety [1-3,5,6]. For other clinically significant Staphylococcus strains, coagulase-negative staphylococci (CoNS) strains have been reported as the most frequently isolated pathogens in intravascular catheter related infections (CRI) (accounting for approximately 28% of nosocomial bloodstream infection), thus become a frequent cause of nosocomial infection and bacteremia, especially in patients with indwelling medical devices [7-9]. Since the first report in 1961, methicillin-resistant Staphylococcus aureus (MRSA) has become one of the most prevalent pathogens responsible for nosocomial infections throughout the world, which has raised a global challenge for clinicians, hospital epidemiologists and administrators [10-16]. The mecA gene, encoding the PBP2a protein and causing methicillin resistance in staphylococci, is located on staphylococcal cassette chromosome mec (SCCmec). SCCmec contains the mec gene complex (the mecA gene and its regulators) and the ccr gene complex (encoding site-specific recombinases responsible for the mobility of SCCmec) [17-23]. In recent years, MRSA strains have been considered to be a major example of the leading “Super Bugs”, with broad resistance to practically all β-lactam antibiotics and usually other multiple drugs due to the associated resistance genes carried by SCCmec[1-3,17-20]. As consequence, the increasing awareness for the risk and hazard of MRSA strains raised the demands of an early, cost-effective, timely, specific and sensitive detection assay [5,6,10,20].
As clinical MRSA diagnosis is concerned, Staphylococcus strains have commonly been identified via routine standard procedures including colony morphology, Gram staining, testing of catalase, hyaluronidase and coagulase, as well as the Vitek 2 automated system and the API-Staph commercial kit, which makes the detection of 16SrRNA somehow irrelevant. However, despite the advantages of this assay, the further development and broad application of multiplex-PCR has been restricted by many factors, such as cross-reaction of different sets of primers, self-inhibition due to formation of dimmers, reduced amplification efficiency caused by the simultaneous and parallel amplification, undetectable influence of different targets, as well as the requirement for standard full use of external and internal quality control (both positive and negative control for each targets) for each assay. As a novel analytical assay since the past decade, mass spectrometry has been applied for detection and diagnostics of various clinical microorganisms, with advantages on high-throughput process, sensitivity and specificity [24]. However, this methodology requires trained personnel, operating space, complicated sample preparation procedure, as well as expensive equipment.
In the last decade, loop-mediated isothermal amplification (LAMP) had been reported as a novel nucleic acid amplification method [25-27] (Figure 1) and applied to the detection of various pathogenic organisms, including Escherichia coli O157 with the associated toxins stx1 and stx2[28], Yersinia pseudotuberculosis[29], Salmonella[30-33], Vibrio parahaemolyticus with the associated toxins tdh and trh[34-39], Psuedomonas aeruginosa[40] and Listeria monocytogenes[41]. The LAMP methodology relies on an auto-cycling strand displacement DNA synthesis performed by the Bst DNA polymerase large fragment under the isothermal conditions between 60-65°C, and the amplicons are mixtures of many different sizes of stem-loop DNAs with several inverted repeats of the target sequence and cauliflower-like structures with multiple loops, which significantly simplify the reaction itself and the result determination. The LAMP assay includes 4 or 6 primers (targeting on 6 or 8 specific regions) to perform the amplification, and requires 45-60 min and 30-45 min without and with loop primers, respectively. In comparision with PCR assay, the positive points of LAMP method were simplicity, rapidity, specificity and sensitivity (10-1000 times higher than PCR).
Figure 1.
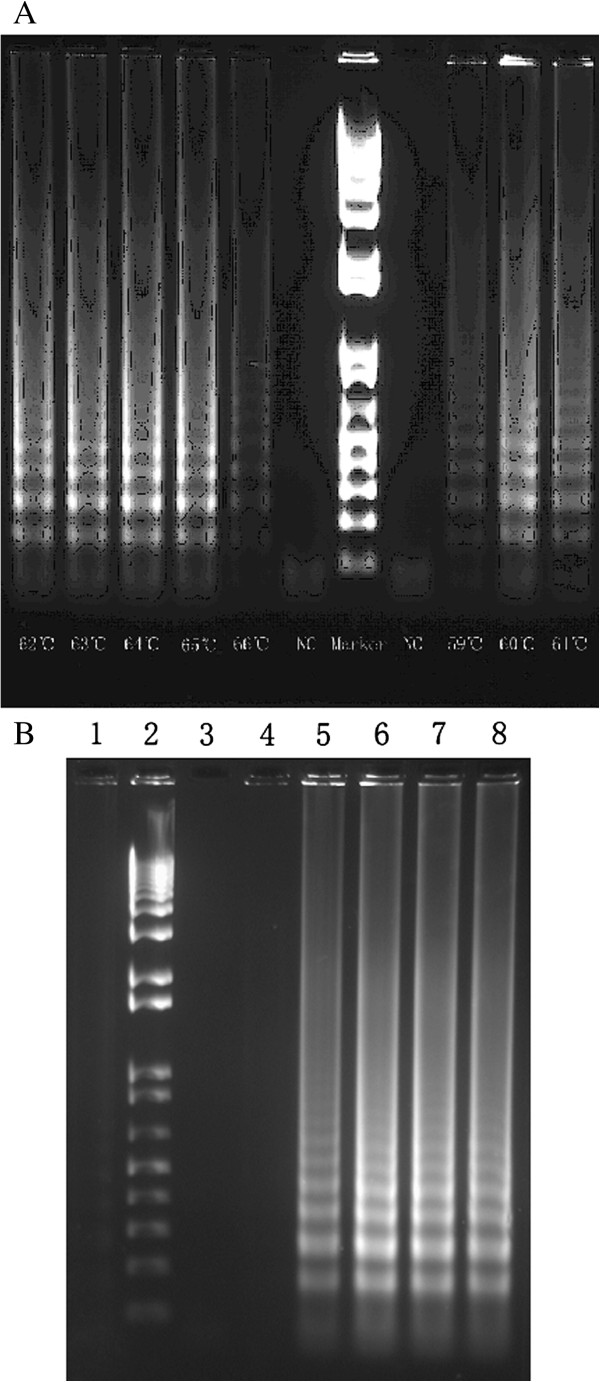
Electrophoresis of orfX-LAMP. A: Under different temperatures. B: Detection limit, lane 1-8: negative control (distilled water), DNA Marker, negative contro1 (template DNA sample of other bacteria), 1 copies, 10 copies, 102 copies, 103 copies and 104 copies.
In the current study, a simple orfX-LAMP assay had been developed, evaluated, optimized and further applied to detect a large scale of clinical MRSA strains, requiring approximately 60 mins for the whole process.
Results
Optimization of orfX-LAMP assay
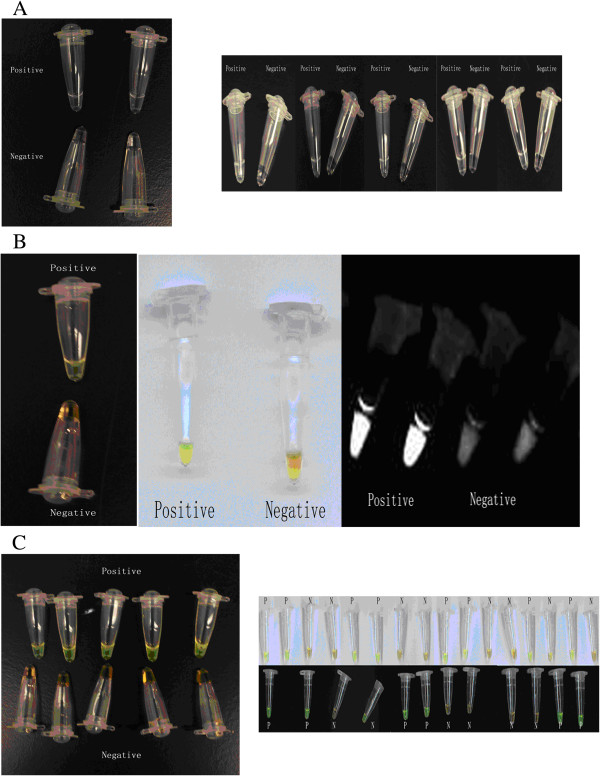
The orfX-LAMP amplification had generated a large number of ladder-like pattern bands on agarose gel due to the characteristic structure up to the loading wells, with a bottom band of 212-bp size amplicon obtained by sequencing. OrfX-LAMP assay had been performed under isothermal condition between 59°C and 66°C. Despite none of significant difference observed, the electrophoresis of LAMP products which were amplified under 64°C exhibited slightly larger amount of DNA amplicons when compared to other temperatures (Figure 1), which was consistent with studies previously. As reaction length was concerned, various time points had been studied under 65°C and with 10 ng template DNA, including 15 min, 30 min, 45 min, 60 min, 75 min, 90 min, 105 min and 120 min. For the LAMP assay with loop primers, amplification had been initially detected at 30 min, and reached the maximal at 45 min (Figure 2). Nevertheless, without loop primers, amplification products could not be observed until 90 min. All amplicons of orfX-LAMP assay had been determined by electrophoresis, with observation directly by naked eye, as well as under UV light with Sybr Green dyed (Figure 3).
Figure 2.

Evaluation of orfX-LAMP assay under different time points. A: Electrophoresis of orfX-LAMP reaction under different time points: 1-10: DNA Marker, 15, 30, 45, 60, 75, 90, 105 and 120 min, negative control. B: Sybr Green of orfX-LAMP reaction under different time points.
Figure 3.
Result determination of orfX-LAMP assays by color change. A: Results determination of orfX-LAMP by naked eyes. B: Results determination of orfX-LAMP by Sybr Green with dark background, light backgrounds and under UV light. C: Results determination of orfX-LAMP in application by Sybr Green.
Sensitivity and specificity of orfX-LAMP assays
The detection limit of orfX-LAMP assay had been studied by both minimal CFU of bacterial and template DNA amount, which was found to be 10 copies of DNA/tube and 10 CFU/reaction (LAMP was positive for sample containing 1X104 CFU/ml, with 1 μl was included in the reaction system) (Figure 1), in comparision with the detection limit of PCR which was 102 copies of DNA/tube and 103 CFU/reaction, indicating that the sensitive of LAMP was 10-100 times sensitive than PCR. As specificity was concerned, for primers, amplification could not be obtained in the absence of FIP, BIP, F3 or B3, due to the indispensable role each of the primers plays in auto-cycling strand placement. The LAMP was only performed in the existence of both inner primers and outer primers. In addition, for the orfX-LAMP assay, high specificity was acquired when this assay was subjected to 116 reference strains, without any false positive amplification for non-MRSA strains (Table 1).
Table 1.
Reference strains included in the evaluation of orfX -LAMP
| Reference strains | No. | orfX |
|---|---|---|
| Gram-positive microorganisms |
|
|
| Methicillin-resistant Staphylococcus aureus 10442, COL, N315, 85/2082, CA05, JCSC 1978, JCSC 4469, MR108, M03-68, WIS |
10 |
+ |
| Methicillin-sensitive Staphylococcus aureus ATCC6358, ATCC12598, ATCC12600, ATCC13150, ATCC13565, ATCC14458, ATCC19095, ATCC23235, ATCC27664, ATCC25923, ATCC29213 |
11 |
- |
| Methicillin-resistant coagulase-negative staphylococci: S. epidermidis ATCC29887, ATCC700586 |
2 |
- |
| Methicillin-sensitive coagulase-negative staphylococci: S. epidermidis ATCC12228, 6508, 9142, 9142-M10; S. haemolyticus 1922; S. hominis ATCC27844; S. capitis ATCC27840; S. saprophiticus ATCC35552; S. sciuri ATCC29062; S. schleiferi ATCC43808; S. intermedius ATCC49052 |
11 |
- |
|
Listeria monocytogenes ATCC15313ATCC19111, ATCC19114, ATCC19115, ATCC19116, ATCC19118, WF 03213, WF 06410, WF 08211 |
8 |
- |
|
Listeria invanovii ATCC19119, WF 06319 |
2 |
- |
|
Listeria welshimeri WF 05086 |
1 |
- |
|
Listeria seeligeri WF 04426 |
1 |
- |
|
Bacillus cereus ATCC11778, ATCC12480, ATCC13061, ATCC14579, ATCC25621, ATCC53522 |
6 |
- |
| Gram-negative organisms |
|
|
|
Escherichia coli O157: H7 WF01201 ATCC43889, NCTC12900 |
3 |
- |
|
Escherichia coli O157: H7 WF07803, WF06544, WF05395 |
3 |
- |
|
Escherichia coli O157: H7 WF04402, WF05311, WF06837 |
3 |
- |
|
Escherichia coli O157: H7 WF04587, WF08385, WF06349 |
3 |
- |
|
Escherichia coli O26: H11 WF054489 |
1 |
- |
|
Escherichia coli O127: H6 WF073522 |
1 |
- |
|
Escherichia coli O148: H28 WF063224 |
1 |
- |
|
Escherichia coli C600 ATCC25922, ATCC43984, ATCC43985, ATCC8739, C600, DH5α |
6 |
- |
|
Vibrio parahaemolyticus O1: K25 WF 04213 |
1 |
- |
|
Vibrio parahaemolyticus O1: K56 WF 02314 |
1 |
- |
|
Vibrio parahaemolyticus O3: K6 WF 04232 |
1 |
- |
|
Vibrio parahaemolyticus O3: K6 WF 01031, WF 04506, WF 06215 |
3 |
- |
|
Vibrio parahaemolyticus O3: K6 WF 02321, WF 03218, WF 05612 |
3 |
- |
|
Vibrio parahaemolyticus O3: K12 WF 02108 |
1 |
- |
|
Vibrio parahaemolyticus O3: K72 WF 02471 |
1 |
- |
|
Vibrio parahaemolyticus O4: K37 WF 01309, WF 04238 |
2 |
- |
|
Vibrio parahaemolyticus O4: K55 WF 03256, WF 07521 |
2 |
- |
|
Vibrio parahaemolyticus O4: K68 WF 07926, WF 03422, WF 02613 |
3 |
- |
|
Vibrio parahaemolyticus O10: K28 WF 06438 |
1 |
- |
|
Vibrio parahaemolyticus ATCC17802, ATCC27969 |
2 |
- |
|
Vibrio vulnificus ATCC27562 |
1 |
- |
|
Vibrio mimicus ATCC33653 |
1 |
- |
|
Psuedomonas aeruginosa ATCC 27853 |
1 |
- |
|
Salmonella enterica ATCC13076, ATCC14028, ATCC19585, ATCC29629 |
4 |
- |
|
Salmonella typhimurium ATCC 14028, WF 04313 |
2 |
- |
|
Salmonella choleraesuis ATCC 13312 |
1 |
- |
|
Salmonella enteritidis WF 05148, WF 07086 |
2 |
- |
|
Salmonella typhi WF 03201, WF 05026, WF 08138 |
3 |
- |
|
Salmonella paratyphi WF 06426 |
1 |
- |
|
Salmonella aberdeen WF 04542 |
1 |
- |
|
Salmonella gallinarum WF 05938 |
1 |
- |
|
Klebsiella pneumoniae ATCC 13883 |
1 |
- |
|
Enterobacter cloacae ATCC 23355 |
1 |
- |
|
Yersinia enterocolitica ATCC9610, ATCC27729 |
2 |
- |
| Total | 116 |
Application of LAMP assays on clinical MRSA strains
After establishment and optimization, orfX-LAMP assay were applied to the detection of 667 clinical Staphylococcus strains, including 566 MRSA, 25 MSSA, 53 MRCNS and 23 MSCNS strains (Table 2), with comparative validation by standard PCR assay. For application, rapid DNA preparation process, simple heating equipments and results determination by observation directly by naked eye and under UV light had been applied. As shown by the results, for total of 566 MRSA strains, 557 (98.4%, 557/566) and 519 (91.7%, 519/566) had been detected positive by LAMP and PCR assays, respectively. However, for other 101 non MRSA isolates, none and 3 yielded positive amplification. The detection rate, positive predictive value (PPV) and negative predictive value (NPV) of orfX-LAMP were 98.4%, 100% and 92.7%, respectively. All reaction had been performed in triplicate to ensure reproducibility.
Table 2.
Application of the orfX -LAMP on clinical Staphylococcus strains
| Samples | LAMP | PCR |
|---|---|---|
| MRSA |
|
|
| Bloodstream |
100% (36/36) |
94.4% (34/36) |
| Respiratory tract |
98.6% (145147) |
91.8% (135/147) |
| Skin and soft tissue |
98.0% (238/243) |
92.6% (225/243) |
| Urinary tract |
98.6% (73/74) |
91.9% (68/74) |
| Other |
98.5% (65/66) |
86.4% (57/66) |
| Subtotal |
98.4% (557/566) |
91.7% (519/566) |
| MSSA |
0% (0/25) |
0% (0/25) |
| MRCNS |
0% (0/53) |
0.0% (2/53) |
| MSCNS |
0% (0/23) |
0.0% (1/23) |
| Subtotal | 0% (0/101) | 0.0% (3/101) |
Samples, Blood culture specimens included blood inoculated into aerobic and/or anaerobic blood culture medium. Tracheal secretion specimens included secretions from the trachea and bronchia and bronchoalveolar lavage fluid specimens. Wound specimens included surgical wound specimens but also specimens from ulcers, fistulae, abscesses, drainage fluids, catheters sites, percutaneous endoscopic gastrostomy insertion sites, and tracheostomas. Urine contained native urine from urinary catheters or bladder puncture and inoculated culture systems. Other specimens consisted of skin swabs specimens (from the nares, axilla, groin, or perianal area), as well as swab specimens of the throat, tonsils, eye, ear, and vagina; body liquids; puncture exudates.
Discussion
As one of the globally widespread clinical pathogens, MRSA strains have attained a heightened concern for its rapid and accurate detection, which has become a common issue in the diagnostics of MRSA. Current MRSA diagnosis methodologies include routine standard procedures (including colony morphology, Gram staining, testing of catalase, hyaluronidase and coagulase), Vitek 2 automated system, API-Staph kit, immunological assays, mass spectrometry, PCR (regular PCR and quantitative PCR), etc. MRSA strains exhibit resistance to practically all β-lactam antibiotics and commonly other drugs due to the mecA and other resistance genes carried by SCCmec. However, for type I (1B) and IV (2B) SCCmec (with class B mec complex), the mecI-encoded repressor function may lead to the low-level methicillin resistance. In the case of type II (2A) and III (3A) SCCmec (with class A mec complex), the IS431-mediated deletion of mecI and further derepression of mecA transcription may cause the expression of methicillin resistance. As consequence, differing from biochemical methodologies, the accurate diagnostic identification of MRSA relies on the detection of highly specific and conserved targets within SCCmec. As orfX was considered to be highly specific target for both S. aureus and SCCmec, a distinctive MRSA detection methodology based on orfX-LAMP was developed in the present study. In addition, a number of attributes, including rapidity, simplicity in operation, specificity, sensitivity and expense, were also evaluated to verify the application of the orfX-LAMP assay for MRSA detection. Targeting on six distinct regions, inner and outer primers of the LAMP assay were highly specific in comparison with conventional PCR techniques. In this study, the specificity of the orfX-LAMP assay was verified by indispensibility of each primer, without any false positive test obtained from reference strains and 100% PPV for application. As sensitivity was concerned, in comparison with regular PCR (ranging from 103-105 CFU/reaction) [6] and previous LAMP detection methodoloy (ranging from 102-103 copies of DNA) [42], the detection limit of orfX-LAMP was found to be 10 copies DNA/tube and 10 CFU/reaction. For rapidity, approximate 60 min was required in the application of orfX-LAMP, including DNA process, isothermal reaction and result determination.
Conclusions
The first study on detection of MRSA by LAMP assay was reported lately [6], however, 3 individual LAMP assays with targets including 16SrRNA, femA and mecA were laborious and time and expense demanding. Also, in another preliminary Staphylococcus LAMP identification assay, 120 min and 2 targets (spaA and mecA) were required for the procedure [42]. In this current study, a simple LAMP assay targeting on orfX for rapid MRSA detection had been developed, evaluated and optimized, which required approximately 60 min for each test and used the highly conserved target orfX. This orfX-LAMP had been further applied to the detection of 667 clinical Staphylococcus strains, and the sensitivity, specificity, PPV and NPV were found to be 98.4%, 100%, 100% and 92.7%, respectively. In comparison with conventional PCR, this established LAMP assay exhibited advantages on detection limit, sensitivity, simplicity and rapidity. In conclusion, this orfX-LAMP assay had been demonstrated to be a valid and rapid detection method on MRSA, which will undoubtedly aid in the broad application of bacteriological MRSA detection.
Methods
Bacterial strains
For development and evaluation of the orfX-LAMP assay, 116 reference strains were included, with various species of gram-negative and -positive isolates (Table 1). As application was concerned, the optimized LAMP and PCR assays was performed on a total of 667 Staphylococcus strains, including 566 MRSA, 25 MSSA, 53 MRCNS and 23 MSCNS strains (Table 2), which had been previously isolated from various clinical samples during 2001-2006 and preliminarily identified [6,9,15]. All strains were identified as S. aureus using standard procedures: colony morphology, Gram staining, testing of catalase, hyaluronidase and coagulase, the Vitek 2 automated system and the API-Staph commercial kit. Methicillin resistance was determined by susceptibility testing on oxacillin-screening agar, confirmed by latex agglutination for PBP2a and mecA detection by PCR [43].
Primer design
The protocol of this LAMP assay for rapid MRSA detection was designed to target on the specific orfX, which located on the site of SCCmec and had been considered to be a highly conserved open reading frame in S. aureus. The sequences of orfX had been acquired on GenBank, including SCCmec type I (NCTC 10442, AB033763), SCCmec type II (N315, D86934), SCCmec type III (Mu50, gi:57634611), CA05, MR108, 86-3P, M03-6, 43000, SCCmec type V (WIS). A set of LAMP primers (Table 3) targeting 8 distinct regions on orfX was designed via PrimerExplorer (V4), including forward inner primer (FIP) with the complementary sequence of F1 (F1c), a T–T–T–T linker and F2, backward inner primer (BIP) with the complementary sequence of B1 (B1c), a T–T–T–T linker and B2, the outer primers F3 and B3 located outside of the F2 and B2 regions, loop primers LF and LB located between F2 and F1 or B1 and B2, respectively.
Table 3.
The sequences and information of LAMP primers
| Label | 5′pos | 3′pos | len | Tm | 5′dG | 3′dG | GCrate | Sequence |
|---|---|---|---|---|---|---|---|---|
| F3 |
204 |
222 |
19 |
56.03 |
-5.56 |
-4.06 |
0.42 |
ACCACAATCMACAGTCATT |
| B3 |
398 |
415 |
18 |
55.58 |
-7.53 |
-4.56 |
0.50 |
CCCGCATCATTTGATGTG |
| FIP |
|
|
39 |
|
|
|
|
CAAAGTCGCTTTGCCCTTGG-GATGCTATCTTCCGAAGGA |
| BIP |
|
|
41 |
|
|
|
|
GATCAAACGGCCTGCACAAG-GRAATGTCATTTTGCT RAATG |
| F2 |
243 |
261 |
19 |
55.33 |
-5.14 |
-4.71 |
0.47 |
GATGCTATCTTCCGAAGGA |
| F1c |
291 |
310 |
20 |
61.82 |
-4.16 |
-5.45 |
0.55 |
CAAAGTCGCTTTGCCCTTGG |
| B2 |
377 |
397 |
21 |
55.47 |
-4.51 |
-4.07 |
0.38 |
GR(G or A)AATGTCATTTTGCTRAATG |
| B1c |
326 |
345 |
20 |
61.55 |
-3.92 |
-4.66 |
0.55 |
GATCAAACGGCCTGCACAAG |
| LF |
266 |
286 |
21 |
61.30 |
-6.24 |
-5.45 |
0.48 |
TGCGTTGGTTCAATTCTTGGG |
| LB | 346 | 367 | 22 | 60.11 | -5.26 | -3.73 | 0.45 | GACGTCTTACAACGCAGTAACT |
Establishment and optimization of orfX-Lamp assay
To evaluate and optimize the orfX-LAMP assay, 52 Gram-positive and 64 -negative isolates were employed as reference strains. Cultural conditions and DNA extraction of gram-negative and gram-positive strains were performed as described previously [41,44-47]. In brief, template DNA from S. aureus strains were prepared from overnight Tryptic Soy Broth (TSB) cultures at 37°C with shaking, and the collected culture was then diluted 10-fold in 10 mM Tris-HCl (pH 8.0) containing 1 mM EDTA. The suspension was boiled for 10 min and further kept on ice. After centrifugation at 12,000 g for 3 min, the resulting supernatant was used as templates for LAMP and PCR assays. Evaluation and optimization of this orfX-LAMP assay included the study of mixture volumn (4 volumns, with 12.5 μl, 25 μl and 50 μl), reaction temperature (8 temperatures, with 59°C, 60°C, 61°C, 62°C, 63°C, 64°C, 65°C and 66°C), reaction time (8 time points, with15 min, 30 min, 45 min, 60 min, 75 min, 90 min, 105 min and 120 min), specificity (including the primers and strains) and detection limit. LAMP assays was carried out in 3 different reaction mixture volumns, containing 1.6 μM (each) of the primers FIP and BIP, 0.2 μM (each) of the primers F3 and B3, 0.8 μM (each) of primers LF and LB, 1.6 mM of deoxynucleoside triphosphates, 6 mM MgSO4, 1 M betain (Sigma, St. Louis, MO, USA), 1 X thermopol buffer (New England Biolabs, Ipswich, MA, USA), and specified amounts of target genomic DNA. The reaction was initiated by heating at 95°C for 3 min, then chilled on ice for 30 sec, with 1 μl (8 U) of Bst DNA polymerase (New England Biolabs, Ipswich, MA, USA) further added. After incubation at various temperatures ranging from 59°C to 66°C for 15 min-120 min, the reaction was terminated by heating at 80°C for 2 min. As PCR assay was concerned, the amplification was carried out in 50 μl reactant, using the outer primers F3 and B3. The thermal profile was 94°C for 5 min, followed by 30 cycles of 94°C for 30 sec, 50°C for 30 sec, and 72°C for 30 sec and a final extension cycle at 72°C for 7 min. The amplified products (5 μl/well) were analyzed by gel electrophoresis in 2% agarose gels and stained with ethidium bromide for 10 min. Template DNA from MRSA 85/2082 was diluted for serial 10-fold and the detection limit of LAMP and PCR assays were ascertained by both minimal CFU of bacterial and plasmid DNA (with orfX recombinated in a T vector). For LAMP assays, the lowest bands from amplicons were sequenced by an ABI PRISM 310 genetic analyzer (PE Biosystems, Foster City, CA, USA).
Application of LAMP assays on clinical MRSA strains
After optimization, the orfX-LAMP assay had been further performed on 667 Staphylococcus (566 MRSA, 25 MSSA, 53 MRCNS and 23 MSCNS) strains, and was comparatively validated by standard PCR assay. Template DNA was prepared through a rapid procedure as aforementioned, with the required time as less than 30 min. Heating and isothermal amplification were separately performed on simple equipments including water bath and heating block. Amplification products of LAMP assay were dyed with Sybr Green, positive or negative were determined through both visually observation of the color change by naked eye and a fluorescence assay under UV [6,28,33]. This experiment was performed in triplicate to ensure reproducibility.
Abbreviations
LAMP: Loop-mediated isothermal amplification; MRSA: Methicillin-resistant Staphylococcus aureus; MSSA: Methicillin-sensitive Staphylococcus aureus; MRCNS: Methicillin-resistant coagulase-negative staphylococci; MSCNS: Methicillin-sensitive coagulase-negative staphylococci.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
JS carried out the LAMP detection assays and drafted the manuscript. XL carried out the DNA preparation and participated in the bacterial processing. HC performed the statistical analysis. YL carried out the sample collection and bacterial processing. DC participated in the design of the study and revised the manuscript. YL conceived of the study and participated in its design and coordination. GY participate in the design of the study and manuscript revision. All authors read and approved the final manuscript.
Contributor Information
Jianyu Su, Email: jysu@scut.edu.cn.
Xiaochen Liu, Email: lxcjs123@qq.com.
Hemiao Cui, Email: 1072951969@qq.com.
Yanyan Li, Email: yanyanli1123@hotmail.com.
Dingqiang Chen, Email: jyksys@126.com.
Yanmei Li, Email: yanmeili@live.cn.
Guangchao Yu, Email: guangchaoyu@126.com.
Acknowledgements
We sincerely thank Dr. Zhenbo Xu from University of Maryland for his excellent support for this study. We are grateful for Dr. Xihong Zhao from Taiwan National University for his review for this manuscript. This work was supported by the National 973-Plan (2012CB720800), the National Natural Science Foundation of China (31201362 and 31101278), the national science and technology support program (2012BAD37B01), the Fundamental Research Funds for the Central Universities (2014ZM0060) and the Funding of Guangdong Outstanding Dissertation (Dr. Zhenbo Xu).
References
- Schito GC. The importance of the development of antibiotic resistance in Staphylococcus aureus. Clin Microbiol Infect. 2006;12(Suppl. 1):3–8. doi: 10.1111/j.1469-0691.2006.01343.x. [DOI] [PubMed] [Google Scholar]
- Tacconelli E. Methicillin-resistant Staphylococcus aureus: risk assessment and infection control policies. Clin Microbiol Infect. 2008;14:407–410. doi: 10.1111/j.1469-0691.2007.01936.x. [DOI] [PubMed] [Google Scholar]
- Harbarth S. Control of endemic methicillin-resistant Staphylococcus aureus –recent advances and future challenges. Clin Microbiol Infect. 2006;12:1154–1162. doi: 10.1111/j.1469-0691.2006.01572.x. [DOI] [PubMed] [Google Scholar]
- Xu Z, Li L, Shi L, Shirtliff M. Class 1 integron in staphylococci. Mol Biol Rep. 2011;38:5261–5279. doi: 10.1007/s11033-011-0676-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chambers HF. The changing epidemiology of Staphylococcus aureus? Emerg Infect Dis. 2001;7:178–182. doi: 10.3201/eid0702.010204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z, Li L, Chu J, Peters B, Harris M, Li B, Shi B, Shirtliff M. Development and application of loop-mediated isothermal amplification assays on rapid detection of various types of staphylococci strains. Food Res Int. 2012;47:166–173. doi: 10.1016/j.foodres.2011.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-Ami R, Navon-Venezia S, Chwartz D, Carmeli Y. Infection of a ventriculoatrial shunt with phenotypically variable Staphylococcus epidermidis masquerading as polymicrobial bacteremia due to various Coagulase-Negative Staphylococci and kocuria varians. J Clin Microbiol. 2003;41:2444–2447. doi: 10.1128/JCM.41.6.2444-2447.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nouwen JL, Van Belkum A, De Marie S, Sluijs J, Wielenga JJ, Kluytmans JW, Verbrugh HA. Clonal expansion of Staphylococcus epidermidis strains causing human catheter-related infections in a hemato-oncologic department. J Clin Microbiol. 1998;36:2696–2702. doi: 10.1128/jcm.36.9.2696-2702.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z, Shi L, Alam M, Li L, Yamasaki S. Integron-bearing methicillin-resistant coagulase-negative staphylococci in South China, 2001-2004. FEMS Microbiol Lett. 2008;278:223–230. doi: 10.1111/j.1574-6968.2007.00994.x. [DOI] [PubMed] [Google Scholar]
- Kluytmans-VandenBergh MFQ, Kluytmans JAJW. Community-acquired methicillin-resistant Staphylococcus aureus: current perspectives. Clin Microbiol Infect. 2006;12(Suppl. 1):9–15. doi: 10.1111/j.1469-0691.2006.01341.x. [DOI] [PubMed] [Google Scholar]
- Chang S, Sun C, Yang L, Luh K, Hsieh W. Increasing nosocomial infections of methicillin-resistant Staphylococcus aureus at a teaching hospital in Taiwan. Int J Antimicrob Agents. 1997;8:109–114. doi: 10.1016/S0924-8579(96)00360-3. [DOI] [PubMed] [Google Scholar]
- Zetola N, Francis JS, Nuermberger EL, Bishai WR. Community-acquired methicillin-resistant Staphylococcus aureus: an emerging threat. Lancet Infect Dis. 2008;5:275–286. doi: 10.1016/S1473-3099(05)70112-2. [DOI] [PubMed] [Google Scholar]
- Okuma K, Iwakawa K, Turnidge JD, Grubb WB, Bell JM, O’Brien FG, Coombs GW, Pearman JW, Tenover FC, Kapi M, Tiensasitorn C, Ito T, Hiramatsu K. Dissemination of new methicillin-resistant Staphylococcus aureus clones in the community. J Clin Microbiol. 2002;40:4289–4294. doi: 10.1128/JCM.40.11.4289-4294.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenesch F, Naimi T, Enright MC, Lina G, Nimmo GR, Heffernan H. Community-acquired methicillin-resistant Staphylococcus aureus carrying Panton-Valentine leukocidin genes: worldwide emergence. Emerg Infect Dis. 2003;9:978–984. doi: 10.3201/eid0908.030089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z, Shi L, Zhang C, Zhang L, Li X, Cao Y, Li L, Yamasaki S. Nosocomial infection caused by class 1 integron-carrying Staphylococcus aureus in a hospital in South China. Clin Microbiol Infect. 2007;13:980–984. doi: 10.1111/j.1469-0691.2007.01782.x. [DOI] [PubMed] [Google Scholar]
- Xu Z, Li L, Alam M, Yamasaki S, Shi L. First confirmation of integron-bearing methicillin-resistant Staphylococcus aureus. Curr Microbiol. 2008;57:264–268. doi: 10.1007/s00284-008-9187-8. [DOI] [PubMed] [Google Scholar]
- Ito T, Katayama Y, Asada K, Mori N, Tsutsumimoto K, Tiensasitorn C, Hiramatsu K. Structural comparison of three types of staphylococcal cassette chromosome mec integrated in the chromosome in methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 2001;45:1323–1336. doi: 10.1128/AAC.45.5.1323-1336.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katayama Y, Ito T, Hiramatsu K. A new class of genetic element, staphylococcus cassette chromosome mec, encodes methicillin resistance in Staphylococcus aureus. Antimicrob Agents Chemother. 2000;44:1549–1555. doi: 10.1128/AAC.44.6.1549-1555.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanssen A, Kjeldsen G, Sollid JUE. Local variants of staphylococcal cassette chromosome mec in sporadic methicillin-resistant Staphylococcus aureus and methicillin-resistant coagulase-negative Staphylococci: evidence of horizontal gene transfer? Antimicrob Agents Chemother. 2004;48:285–296. doi: 10.1128/AAC.48.1.285-296.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira DC, Lencastre H. Multiplex PCR strategy for rapid identification of structural types and variants of the mec element in methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 2002;46:2155–2161. doi: 10.1128/AAC.46.7.2155-2161.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qi W, Ender M, O’Brien F, Imhof A, Ruef C, McCallum N, Berger-Bächi B. Molecular epidemiology of methicillin-resistant staphylococcus aureus in Zürich, Switzerland (2003): prevalence of type IV SCCmec and a New SCCmec element associated with isolates from intravenous drug users. J Clin Microbiol. 2005;43:5164–5170. doi: 10.1128/JCM.43.10.5164-5170.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- You R, Gui Z, Xu Z, Shirtliff M, Yu G, Zhao X, Shi L, Li B, Su J, Li L. Methicillin-resistance staphylococcus aureus detection by an improved rapid PCR assay. Afr J Microbiol Res. 2012;6:7131–7133. [Google Scholar]
- Xu Z, Lin L, Zhao X, Chu J, Li B, Shi L, Su J, Shirtliff M. Development and application of a novel multiplex polymerase chain reaction (PCR) assay for rapid detection of various types of staphylococci strains. Afr J Microbiol Res. 2011;5:1869–1873. [Google Scholar]
- Sauer S, Kliem M. Mass spectrometry tools for the classification and identification of bacteria. Nat Rev Microbiol. 2010;8:74–82. doi: 10.1038/nrmicro2243. [DOI] [PubMed] [Google Scholar]
- Notomi T, Okayama H, Masubuchi H, Yonekawa T, Watanabe K, Amino N, Hase T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000;28:e63. doi: 10.1093/nar/28.12.e63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori Y, Nagamine K, Tomita N. Detection of loop-mediated isothermal amplification reaction by turbidity derived from magnesium pyrophosphate formation. Biochem Bioph Res Co. 2001;289:150–154. doi: 10.1006/bbrc.2001.5921. [DOI] [PubMed] [Google Scholar]
- Nagamine K, Watanabe K, Ohtsuka K. Loop-mediated isothermal amplification reaction using a nondenatured template. Clin Chem. 2001;47:1742–1743. [PubMed] [Google Scholar]
- Zhao X, Li Y, Wang L, You L, Xu Z, Li L, He X, Liu Y, Wang J, Yang L. Development and application of a loop-mediated isothermal amplification method on rapid detection Escherichia coli O157 strains from food samples. Mol Biol Rep. 2010;37:2183–2188. doi: 10.1007/s11033-009-9700-6. [DOI] [PubMed] [Google Scholar]
- Horisaka T, Fujita K, Iwata T, Nakadai A, Okatani AT, Horikita T, Taniguchi T, Honda E, Yokomizo Y, Hayashidani H. Sensitive and specific detection of Yersinia pseudotuberculosis by loop-mediated isothermal amplification. J Clin Microbiol. 2004;42:5349–5352. doi: 10.1128/JCM.42.11.5349-5352.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohtsuka M, Yanagawa K, Takatori K. Detection of Salmonella enterica in naturally contaminated liquid eggs by loop-mediated isothermal amplification, and characterization of Salmonella isolates. Appl Environ Microbiol. 2005;71:6730–6735. doi: 10.1128/AEM.71.11.6730-6735.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamura M, Ohba Y, Kikuchi S, Suzuki A, Tachizaki H, Takehara K, Ikedo M, Kojima T, Nakamura M. Loop-mediated isothermal amplification for the rapid, sensitive, and specific detection of the O9 group of Salmonella in chickens. Vet Microbiol. 2008;132:197–204. doi: 10.1016/j.vetmic.2008.04.029. [DOI] [PubMed] [Google Scholar]
- Ueda S, Kuwabara Y. The rapid detection of Salmonella from food samples by Loop-mediated isothermal amplification (LAMP) Biocontrol Sci. 2009;14:73–76. doi: 10.4265/bio.14.73. [DOI] [PubMed] [Google Scholar]
- Zhao X, Wang L, Chu J, Li Y, Li Y, Xu Z, Li L, Shirtliff M, He X, Liu Y, Wang J, Yang L. Development and application of a rapid and simple loop-mediated isothermal amplification method for food-borne Salmonella detection. Food Sci Biotechnol. 2010;19:1655–1659. doi: 10.1007/s10068-010-0234-4. [DOI] [Google Scholar]
- Chen S, Ge B. Development of a toxR-based loop-mediated isothermal amplification assay for detecting Vibrio parahaemolyticus. BMC Microbiol. 2010;10:41–45. doi: 10.1186/1471-2180-10-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nemoto J, Sugawara C, Akahane K, Hashimoto K, Kojima T, Ikedo M, Konuma H, Hara-Kudo Y. Rapid and specific detection of the thermostable direct hemolysin gene in Vibrio parahaemolyticus by loop-mediated isothermal amplification. J Food Prot. 2009;72:748–754. doi: 10.4315/0362-028x-72.4.748. [DOI] [PubMed] [Google Scholar]
- Yamazaki W, Ishibashi M, Kawahara R, Inoue K. Development of a loop-mediated isothermal amplification assay for sensitive and rapid detection of Vibrio parahaemolyticus. BMC Microbiol. 2008;8:163–167. doi: 10.1186/1471-2180-8-163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamazaki W, Kumeda Y, Misawa N, Nakaguchi Y, Nishibuchi M. Development of a loop-mediated isothermal amplification assay for sensitive and rapid detection of the tdh and trh genes of Vibrio parahaemolyticus and related Vibrio species. Appl Environ Microbiol. 2010;76:820–828. doi: 10.1128/AEM.02284-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X, Wang L, Chu J, Li Y, Li Y, Xu Z, Li L, Shirtliff M, He X, Liu Y, Wang J, Yang L. Rapid detection of Vibrio parahaemolyticus strains and virulent factors by loop-mediated isothermal amplification assays. Food Sci Biotechnol. 2010;19:1191–1197. doi: 10.1007/s10068-010-0170-3. [DOI] [Google Scholar]
- Wang L, Zhao X, Chu J, Li Y, Li Y, Li C, Xu Z, Zhong Q. Application of an improved loop-mediated isothermal amplification detection of Vibrio parahaemolyticus from various seafood samples. Afr J Microbiol Res. 2011;5:5765–5771. [Google Scholar]
- Zhao X, Wang L, Li Y, Xu Z, Li L, He X, Liu Y, Wang J, Yang L. Development and application of a loop-mediated isothermal amplification method on rapid detection of Pseudomonas aeruginosa strains. World J Microbiol Biotechnol. 2011;27:181–184. doi: 10.1007/s11274-010-0429-0. [DOI] [Google Scholar]
- Wang L, Li Y, Xu Z, Zhong Q. Development and application of a simple loop-mediated isothermal amplification method on rapid detection of Listeria monocytogenes strains. Mol Biol Rep. 2012;39:445–449. doi: 10.1007/s11033-011-0757-7. [DOI] [PubMed] [Google Scholar]
- Misawa Y, Yoshida A, Saito R, Yoshida H, Okuzumi K, Ito N, Okada M, Moriya K, Koike K. Application of loop-mediated isothermal amplification technique to rapid and direction detection of methicillin-resistant Staphylococcus aureus (MRSA) in blood cultures. J Infect Chemother. 2007;13:134–140. doi: 10.1007/s10156-007-0508-9. [DOI] [PubMed] [Google Scholar]
- Xu Z, Li L, Shirtliff M, Peters B, Li B, Peng Y, Alam M, Yamasaki S, Shi L. Resistance class 1 integron in clinical methicillin-resistant Staphylococcus aureus strains in Southern China, 2001-2006. Clin Microbiol Infect. 2011;17:714–717. doi: 10.1111/j.1469-0691.2010.03379.x. [DOI] [PubMed] [Google Scholar]
- Parida M, Horioke K, Ishida H. Rapid detection and differentiation of dengue virus serotypes by a real-time reverse transcription-loop-mediated isothermal amplification assay. J Clin Microbiol. 2005;43:2895–2903. doi: 10.1128/JCM.43.6.2895-2903.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaneko H, Kawana T, Fukushima E, Suzutani T. Tolerance of loop-mediated isothermal amplification to a culture medium and biological substances. J Biochem Bioph Meth. 2007;70:499–501. doi: 10.1016/j.jbbm.2006.08.008. [DOI] [PubMed] [Google Scholar]
- Xu Z, Li L, Shirtliff M, Alam M, Yamasaki S, Shi L. Occurrence and characteristics of class 1 and 2 integrons in Pseudomonas aeruginosa isolates from patients in Southern China. J Clin Microbiol. 2009;47:230–234. doi: 10.1128/JCM.02027-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z, Li L, Shirtliff M, Peters B, Peng Y, Alam M, Yamasaki S, Shi L. First report of class 2 integron in clinical Enterococcus faecalis and class 1 integron in Enterococcus faecium in South China. Diag Microbiol Infect Dis. 2010;68:315–317. doi: 10.1016/j.diagmicrobio.2010.05.014. [DOI] [PubMed] [Google Scholar]