Abstract
Introduction
In the previous study, we found that the inhibition of phosphatidylinositol 3-kinase (PI3K) by LY294002 induced SGC7901 cell death in vitro. We did not know whether SN50, which is a specific inhibitor of nuclear factor κB (NF-κB), could increase the cell death induction of gastric cancer of LY294002 in vitro, and we also wanted to know the mechanism of it, which might be applied to clinical tumor therapy.
Material and methods
The 3-(4,5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide (MTT) assay was used to determine the cytotoxic effects of the drugs. Mitochondrial membrane potential was measured using the fluorescent probe JC-1. Hoechst 33258 staining was used to detect apoptosis and necrosis morphological changes after LY294002 and/or SN50 treatment. Expression of p53, PUMA and Beclin1 were determined with real-time polymerase chain reaction (RT-PCR) analysis. We used transmission electron microscopy to identify ultrastructural changes in SGC7901 cells after LY294002 and/or SN50 treatment.
Results
In this study, we found that treating the human gastric cancer cells SGC7901 with SN50 could significantly enhance the effects of LY294002 on inducing cell death after 24 h, compared to the control group (p < 0.05). Detection of mitochondrial potential and transmission electron microscopic examination indicated that the rate of cell death increased progressively. The expression of p53, PUMA and Beclin1 was up-regulated.
Conclusions
The NF-κB inhibitor SN50 could enhance the role of LY294002 on inducing cell death of human gastric cancer cells SGC7901, which might be a promising new approach to gastric cancer therapy.
Keywords: phosphatidylinositol 3-kinase, nuclear factor κB, gastric cancer, cell death
Introduction
Cancers of the gastrointestinal tract account for 22% of all cancers and represent a major health threat worldwide [1]. Gastric cancer is the fourth most frequently diagnosed malignancy worldwide, accounting for 12% of all cancer-related deaths [2, 3]. In Asia and parts of South America in particular, gastric cancer is the most common epithelial malignancy and a leading cause of cancer-related death.
The phosphatidylinositol 3-kinase (PI3K) pathway plays a central role in the regulation of cell proliferation, growth, differentiation, and survival [4, 5]. Dysregulation of this pathway is frequently observed in a variety of tumors, including colon carcinoma and brain, breast, ovarian and other tumors [6–9]. Therefore, inhibition of PI3K signaling is under investigation as a potentially useful approach for cancer treatment.
LY294002 is a specific inhibitor of PI3K and could induce cell death by activating p53 and up- regulating PUMA, suggesting that the LY294002-induced apoptosis of SGC7901 cells may be mediated by the p53 signaling pathway [10].
Nuclear factor κB (NF-κB) has been linked to control of cell growth and oncogenesis. The roles of NF-κB in cancer appear to be complex, but are likely to involve the ability of this transcription factor to control programmed cell death (PCD) and cell-cycle progression, and possibly cell differentiation, angiogenesis and cell migration.
SN50, a cell-permeable peptide, can specifically inhibit the NF-κB, the mechanism of action of which may include its suppression of growth factors, cytokines, chemokines, and metalloproteinases [11], but it is not completely understood whether the inhibitory action of SN50 is specific to NF-κB signaling or results from suppression of other signaling cascades [12]. Importantly, it has been reported that NF-κB is activated in cancer cells by several chemotherapies and by radiation, and that in many cases this response inhibits the capacity of the cancer cells for radio- and chemotherapy induced cell death [13]. Therefore, inhibition of NF-κB is under investigation as a potentially useful approach for cancer treatment. However, the detailed mechanisms are also poorly understood.
Apoptosis and autophagy are evolutionarily conserved processes that regulate cell fate, both of which are important in tumorigenesis and oncotherapy. P53 and its target gene PUMA play important roles in apoptosis regulation. Beclin1, which is a marker of the start of the autophagy process, also takes part in regulation of different steps of autophagosome formation and maturation. Deletion or mutation of Beclin1 will cause cell death reduction which leads to tumorigenesis [14–16].
In the present study, we investigated whether the inhibition of PI3K by LY294002 induces SGC7901 cell death in vitro. We did not know whether SN50, which is an inhibitor of NF-κB, could increase the cell death induction of gastric cancer of LY294002 in vitro, and we also wanted to know the mechanism of it, which might be applied to clinical tumor therapy.
Material and methods
Material
SGC7901 gastric cancer cells were obtained from the Shanghai Institute of Cell Biology, Chinese Academy of Sciences (Shanghai, China). RPMI1640 medium was obtained from Gibco (Rockville, MD, USA). Fetal bovine serum was provided by Hangzhou Sijiqing Biological Engineering Material Co., Ltd. (Hangzhou, China). L-glutamine and MTT were provided by Sigma (St Louis, MO, USA). Hoechst 33258 was purchased from Beyotime (Nantong, China). Primers, RNAiso Reagent kit, PrimescriptTM RT Reagent Kit, and SYBR Premix EX TaqTM kit were purchased from TAKARA (Dalian, China), and iCycler 5 from BioRad (USA).
Drug preparation
LY294002 (Cell Signaling Technology, Beverly, MA, USA) was diluted in DMSO to create a stock solution that was stored according to the manufacturer's suggestions. After diluting 10–4 with distilled sterilization water, the final solution used in the experiments was 50 µmol/l. This concentration of LY294002 was selected on the basis of our experiments on SGC7901 cells. SN50 (BIOMOL, Plymouth Meeting, PA, USA) was diluted in distilled sterilization water to create a stock solution stored according to the manufacturer's instructions. The final concentration of the SN50 solution used was 18 µmol/l. This concentration of SN50 was selected on the basis of our previous experiments in vivo and in vivo [17, 18].
Cell culture
SGC7901 gastric cancer cells were maintained in RPMI1640 medium containing 10% heat-inactivated fetal bovine serum, 0.03% L-glutamine and incubated in a 5% CO2 atmosphere at 37°C. Cells in a mid-log phase were used in experiments.
Cell viability assay
Cell viability was assessed with MTT assay. To determine the effects of SN50 on enhancing the role of LY294002 on SGC7901 cells, cells were plated into 96-well microplates (7 × 103 cells/well) and cultured for 24 h. Then LY294002 (50 µmol/l), SN50 (18 µmol/l) and LY294002+SN50 were added to the culture medium and cell viability was assessed with MTT 24 h after drug treatment. Later MTT (Sigma, St Louis, MO, USA) solution was added to the culture medium (5 mg/ml final concentration) 4 h before the end of treatment and the reaction was stopped by addition of 10% acidified SDS 100 µl. The absorbance value (A) at 570 nm was read using an automatic multiwell spectrophotometer (Bio-Rad, Richmond, CA, USA). The percentage of cell death was calculated as follows: cell death (%) = (1 – A of experiment well/A of positive control well) × 100%.
Hoechst 33258 staining
After treatment, cell cultures were washed twice with PBS and incubated with 2 µM Hoechst 33258 (Beyotime, Nantong, China) for 1 h in the dark at 37°C. After washing thrice with phosphate buffered saline (PBS), the cells were viewed with a fluorescence microscope (Nikon, Tokyo, Japan) equipped with a UV filter. The images were recorded on a computer with a digital camera (DXM 1200; Nikon) attached to the microscope, and the images were analyzed with Image-Pro Plus software (Media Cybernetics company, USA). The Hoechst reagent was taken up by the nuclei of the cells, and apoptotic cells exhibited bright blue fluorescence.
Detection of mitochondrial potential (ΔΨ)
Mitochondrial ΔΨ was determined using the KeyGEN Mitochondrial Membrane Sensor Kit (KeyGEN, Nanjing, China). The Mitosensor dye aggregates in the mitochondria of healthy cells and emits red fluorescence against a green monomeric cytoplasmic background staining. However, in cells with a collapsed mitochondrial ΔΨ, the dye cannot accumulate in the mitochondria and remains in monomeric form throughout the cells with green fluorescence [19]. Briefly, SGC7901 cells were incubated with LY294002, SN50 and LY294002 + SN50 in 6-well plates for the indicated times and then pelleted, washed with PBS, and resuspended in 0.5 ml of diluted Mitosensor reagent (1 µmol/ml in incubation buffer). After the cells were incubated with the Mitosensor reagent for 20 min, 0.2 ml of incubation buffer was added and cells were centrifuged then resuspended in 40 µl of incubation buffer. Finally, the cells were washed and resuspended in 1 ml PBS for flow cytometry analysis.
Real-time quantitative RT-PCR analysis of p53, Beclin1 and PUMA
Total RNA was extracted using the RNAiso kit (TaKaRa). For extracting total mRNA, SGC7901 cells were treated with LY294002 (50 µmol/l), SN50 (18 µmol/l) and LY294002+SN50 for 6 h before being harvested. First-strand cDNA was generated via reverse transcription of 2 µg of total RNA using random primers and the Primescript RT Reagent Kit (TaKaRa) in a total reaction volume of 20 µl according to the manufacturer's instructions. The sequences of the forward and reverse oligonucleotide primers, specific to the chosen candidate and housekeeping genes, were designed with Primer5 software (available from frodo.wi.mit.edu/cgi-bin/primer5/primer5_www.cgi). For p53 the primers were: forward, 5’-ACTAAGCG-AGCACTGCCCAAC-3’; reverse, 5’-CCTCATTCA- GCTCTCGGAACATC-3’ (GenBank NM_000546; nucleotides 1161–1290). For PUMA the primers were: forward, 5’-CGACCTCAACGCACAGTACGA-3’; reverse, 5’-GGCACCTAATTGGGCTCCATC-3’ (GenBank NM_014417; nucleotides 719–868). For Beclin1 the primers were: forward, 5’- CCAGATGC-GTTATGCCCAGAC-3’; reverse, 5’-CATTCCATTC-CACGGGAACAC-3’ (GenBank NM_003766; nucleotides 889-1037). For β-actin the primers were: forward, 5’-ATTGCCGACAGGATGCAGA-3’; reverse, 5’-GAGTACTTGCGCTCAGGAGGA-3’ (GenBank NM_ 001101; nucleotides 998–1086). Real-time quantitative RT-PCR was performed using the iCycler 5 thermal cycler (BioRad, Hercules, CA, USA). An 80-fold dilution of each cDNA was amplified in a 20 µl volume, using the SYBR Premix EX Taq kit (TaKaRa), with a 500 nM final concentration of each primer. The amplification specificity was checked using melting curve analysis. Threshold cycle Ct, which correlates inversely with the target mRNA level, was calculated using the second derivative maximum algorithm provided by the Light-Cycler software. For each cDNA, all target gene mRNA levels were normalized to β-actin mRNA levels. Results are expressed as the ratio of normalized target gene mRNA levels in treated cells relative to those in untreated cells.
Transmission electron microscopic examination
Pursuant to treatment with LY294002, SN50 or LY294002 + SN50, cells were fixed in ice-cold 2.5% glutaraldehyde in 0.1 M PBS and preserved at 4°C for further processing. When processing resumed, cells were post-fixed in 1% osmium tetroxide in the same buffer, dehydrated in graded alcohols, embedded in Epon 812, sectioned with an ultra-microtome, stained with uranyl acetate and lead citrate followed by examination with a transmission electron microscope (Philips CM120, Dutch).
Statistical analysis
All data are presented as mean ± SD. Statistical analysis was carried out by ANOVA followed by Dunnett's t-test, considering p < 0.05 as significant.
Results
SN50 enhanced tumor cell growth inhibition of LY294002
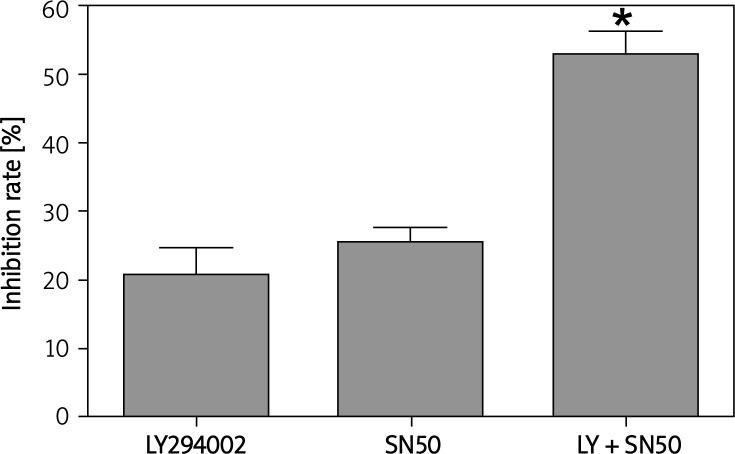
MTT assays revealed that after 24 h of LY294002 (50 µmol/l) and SN50 (18 µmol/l) treatment, the rate of inhibition reached 20.71 ±4.13% and 25.69 ±3.87%. When SGC7901 cells were incubated with LY294002 and SN50 together, the inhibition rate rose to 52.93±2.35% (Figure 1).
Figure 1.
SN50 enhanced tumor cell growth inhibition of LY294002. SGC7901 cells (7 × 104 cells/ml) were cultured with various doses of LY294002, SN50 and LY294002 + SN50 for 24 h and cell viability was analyzed by MTT assay. Values were given as mean ± SD of three independent experiments
*p < 0.05 compared to control group
SN50 enhanced the effects of inducing cell apoptosis of LY294002
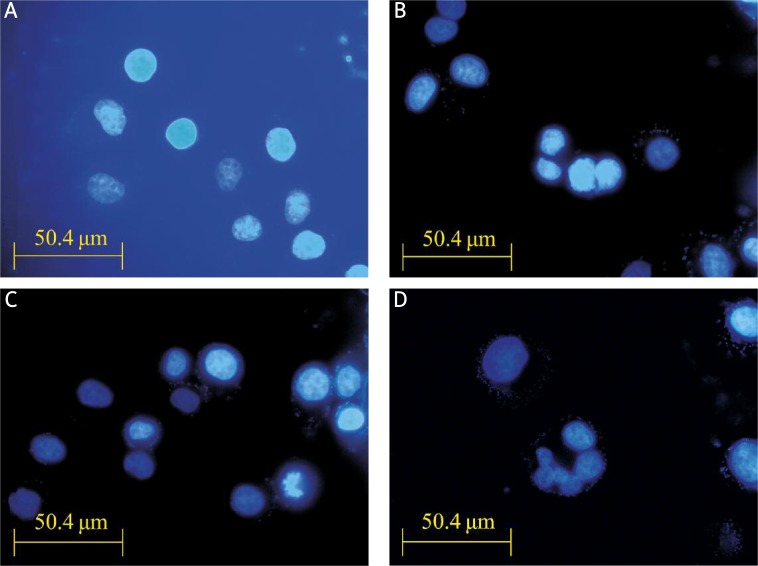
Treatment with 50 µmol/l LY294002 for 12 h in SGC7901 cells produced intense Hoechst-positive staining for condensed nuclei indicative of apoptosis. A significant increase in Hoechst staining was observed along with apoptosis when cells were treated with 50 µmol/l LY294002 + 18 µmol/l SN50. The results indicate that SN50 enhanced the effects of inducing cell apoptosis of LY294002 (Figure 2).
Figure 2.
Hoechst 33258 staining showed that SN50 enhanced the effects of inducing cell apoptosis of LY294002. SGC7901 cells were incubated with LY294002 (50 μmol/l) or SN50 (18 μmol/l) for the indicated time and stained with Hoechst 33258 (10 mmol/l). Fluorescence particles showed apoptosis. A – Control, B – LY294002, C – SN50, D – LY294002 + SN50 12 h after treatment (400×) (n = 3)
SN50 enhanced the effects of inducing mitochondrial dysfunction of LY294002
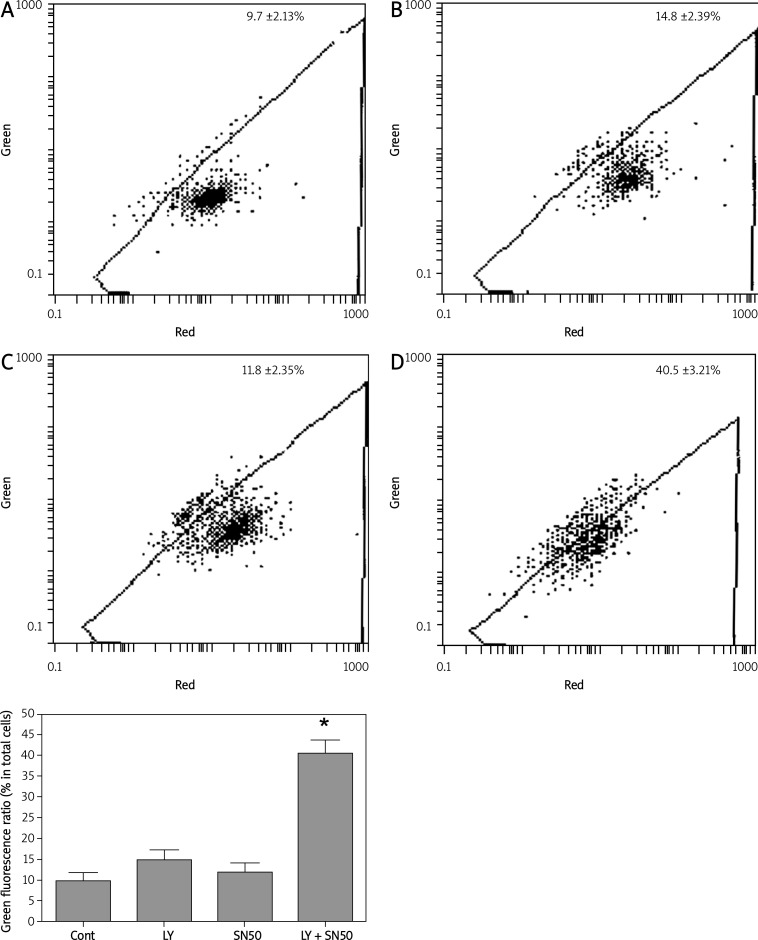
In the present study, mitochondrial membrane potential was examined using the fluorescent dye JC-1. We detected a collapse in mitochondrial membrane potential (Ψ) at 6 h after LY294002 or SN50 treatment, as indicated by increased emission of green fluorescence. This change reached a maximum after LY294002 + SN50 treatment (Figure 3). A collapse in mitochondrial membrane potential always indicates cell death, LY294002 or SN50 induced mitochondrial dysfunction and activated cell death in SGC7901 cells, and the results described here prove that SN50 enhanced the effects of LY294002 in inducing cell death in SGC7901 cells.
Figure 3.
Flow cytometry analysis of mitochondrial membrane potential in the control, LY294002, SN50 and LY294002 + SN50-treated SGC7901 cells. The percentage of cells emitting green fluorescence light increased to 14.8 ±2.39%, 11.8 ±2.35% and 40.5 ±3.21% of total cells after 6 h respectively
SN50 enhanced the effects of LY294002 in up-regulating the expression of Beclin1
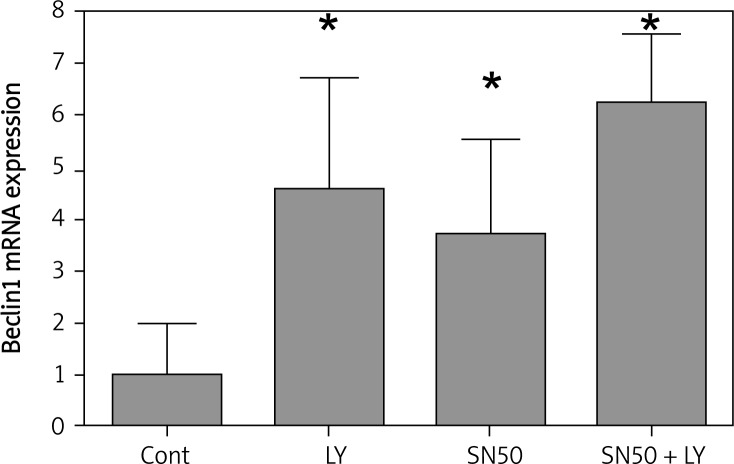
To determine whether SN50 increases the role of LY294002 in up-regulating the expression of autophagic genes, real-time quantitative reverse transcription-PCR analysis was used to detect the expression of Beclin1. The results showed that the basal level of Beclin1 mRNA in SGC7901 cells was low. After incubating with LY294002 or SN50, the Beclin1 mRNA expression was increased after 6 h, and when treated with LY294002 + SN50, the Beclin1 mRNA expression was increased significantly (Figure 4).
Figure 4.
Real-time quantitative RT-PCR analysis of the autophagic gene Beclin1 of the control and drug treated SGC7901 cells. Cells were treated with LY294002 (50 μmol/l), SN50 (18 μmol/l) and LY294002 + SN50 for 6 h. Values were given as mean ± SD of three independent experiments. Statistical analysis was carried out with ANOVA followed by Student t-test
*p < 0.05 compared to control group
SN50 increased the role of LY294002 in up-regulating the expression of p53 and PUMA
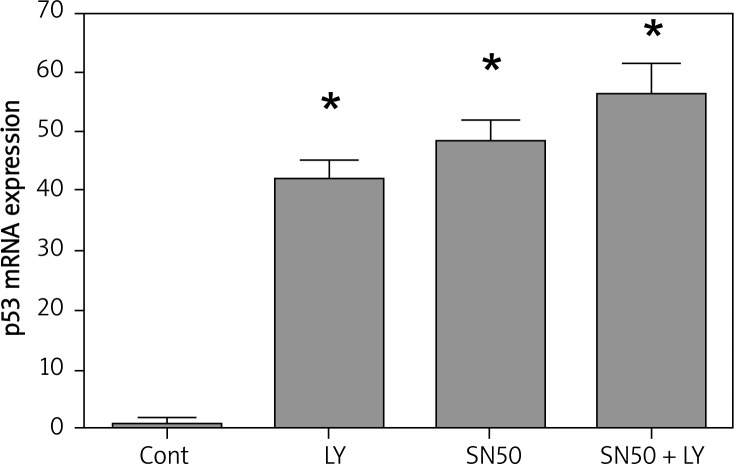
The tumor suppressor and nuclear transcription factor P53 is a tetramer phosphoprotein that can regulate several major cellular functions including gene transcription, DNA synthesis, DNA repair, cell cycle regulation, senescence, and cell death [20, 21]. p53 is activated by cell stress and DNA damage and, depending upon the severity of the stress and the particular cell type, may aid in adaptive responses to the stress or may trigger cell cycle arrest or apoptosis [22]. When LY294002 or SN50 was applied, the expression of p53 was activated. When treated with LY294002 + SN50, the p53 mRNA expression was increased significantly (Figure 5).
Figure 5.
SN50 increased the role of LY294002 in upregulating the expression of p53. Real-time quantitative RT-PCR analysis of the p53 of the control and drug treated SGC7901 cells. Cells were treated with LY294002 (50 μmol/l), SN50 (18 μmol/l) and LY294002 + SN50 for 6 h. Values were given as mean ± SD of three independent experiments. Statistical analysis was carried out with ANOVA followed by Student t-test
*p < 0.05 compared to control group
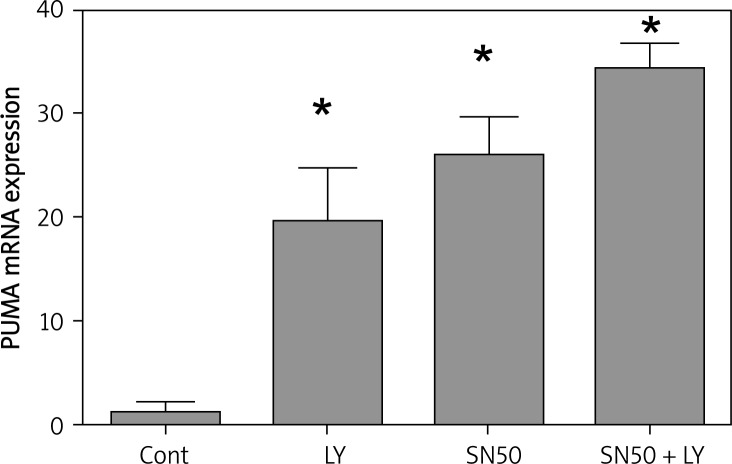
PUMA is a downstream target of the p53 tumor suppressor gene and a member of the BH3-only group of Bcl-2 family proteins [23, 24]. The activation of PUMA by DNA damage is dependent on p53 and is mediated by the direct binding of p53 to the PUMA promoter region [25]. PUMA plays an essential role in p53-dependent and -independent apoptosis induced by a variety of stimuli [23]. In this study, when LY294002 or SN50 was applied, the expression of PUMA was activated, and when treated with LY294002 + SN50, the PUMA mRNA expression were increased significantly (Figure 6).
Figure 6.
SN50 increased the role of LY294002 in upregulating the expression of PUMA. Real-time quantitative RT-PCR analysis of the PUMA of the control and drug treated SGC7901 cells. Cells were treated with LY294002 (50 μmol/l), SN50 (18 μmol/l) and LY294002 + SN50 for 6 h. Values were given as mean ± SD of three independent experiments. Statistical analysis was carried out with ANOVA followed by Student t-test
*p < 0.05 compared to control group
SN50 enhanced the effects of LY294002 in activating impairment of mitochondria
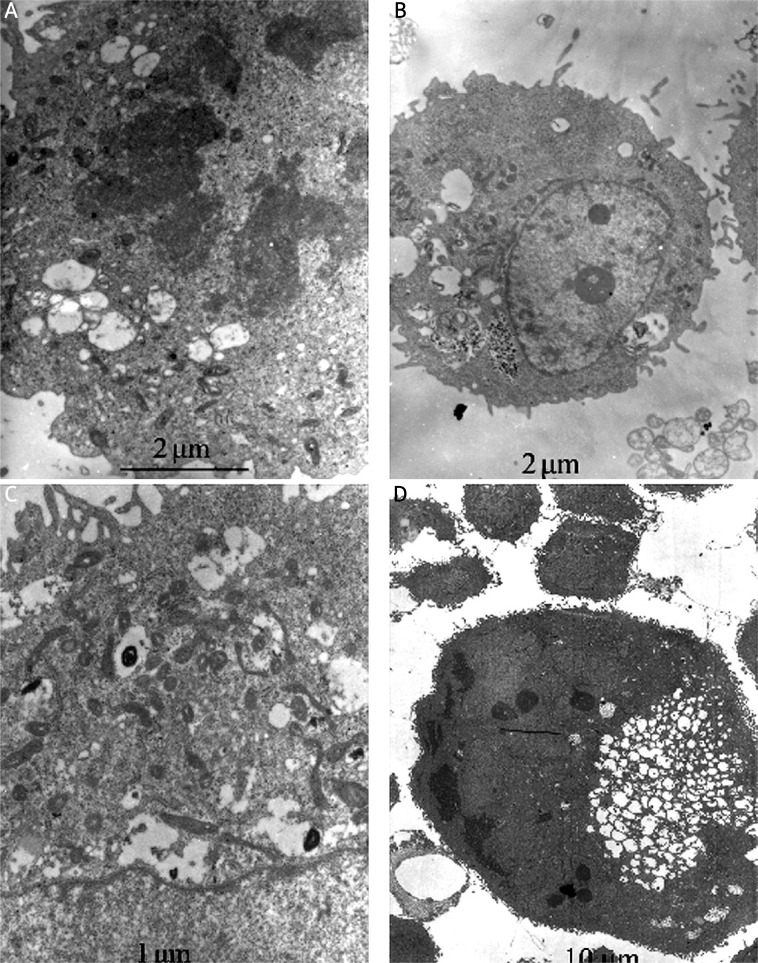
We used transmission electron microscopy to identify ultrastructural changes in SGC7901 cells after LY294002 or SN50 treatment. Control cells showed a round shape and contained normal looking organelles, nucleus and chromatin (Figure 7A), while LY294002-treated cells exhibited signs of organelle morphology as shown in Figure 7B. Mitochondria displayed vacuolization and swelling with a complete loss of cristae (Figure 7C). With the treatment of SN50, a number of isolated membranes, derived possibly from ribosome-free endoplasmic reticulum, were seen. These isolated membranes were elongated and curved to engulf cytoplasmic fraction and organelles (Figure 7C). These membrane structures formed autophagosome traits with double or multi-membranes and then fused with lysosomes in the formation of autolysosomes. The lysosome staining darkened, indicating the activation of lysosomal enzymes (Figure 7C). And with the treatment of LY294002 and SN50, cell apoptosis was observed. Meanwhile, chromatin margination was found in addition to nuclear condensation and pyknosis. The loss of organelles and cytoplasm vacuolization were also observed when SN50 was incubation with LY294002 (Figure 7D).
Figure 7.
Ultrastructure of SGC7901 cells underwent autophagy and apoptosis after drug treatment. A – Non-treated SGC7901 control cells, B – LY294002 (12 h), C – SN50 (12 h), D – LY294002+SN50 (12 h)
Discussion
We found that the PI3K inhibitor LY294002 reduced the viability of and induced apoptosis in SGC7901 gastric cancer cells, demonstrating the cytotoxic effects of LY294002. We also found that LY294002 increased the expression of p53 and PUMA. Our results showed that inhibition of the PI3K signaling pathway is a potential strategy for managing gastric cancers [10].
Nuclear factor κB plays important roles in proliferation and survival of tumor cells. Nuclear factor κB binding sites have been identified in the promoter region of cyclin D1. Nuclear factor κB promotes cyclin D1 expression and cell cycle progression [26, 27]. Nuclear factor κB activity has been shown to inhibit activation of caspase-8, thus inhibiting apoptosis initiation [28]. Thus activation of NF-κB confers resistance of tumor cells to radio-chemotherapy induced cytotoxicity [29, 30].
P53 is known to play important roles in cell death by regulating expression of pro-apoptotic proteins. PUMA is one of the p53 target genes involved in apoptosis. The activation of PUMA by DNA damage is dependent on p53 and is mediated by the direct binding of p53 to the PUMA promoter region [31]. PUMA plays an essential role in p53-dependent and -independent apoptosis induced by a variety of stimuli [25, 32]. Here, we demonstrated that the inhibitor of NF-κB p65 nuclear import, SN50, significantly enhanced the role of LY294002 in up-regulating the levels of PUMA. In supporting this notion, we found that mitochondria membrane potential was collapsed after SN50 treatment. Mitochondria play a central role in regulating cell death and survival. Diverse pro-apoptotic stimuli act on mitochondria, triggering mitochondrial membrane potential collapse, cytochrome c release and caspase activation. The mitochondrial permeability transition (MPT) represents an important event initiating apoptotic cell death.
Beclin1 is the mammalian orthologue of the yeast ATG6-Vps30 gene. It can complement the defect in autophagy present in ATG6–/– yeast strains and stimulate autophagy when overexpressed in mammalian cells [33]. Beclin1 is monoallelically deleted in human breast and ovarian cancers and is expressed at reduced levels in those tumors [33, 34]. The present results suggest that autophagy is induced by SN50 and its activation may contribute to anti-tumor effects of LY294002.
In summary, the present study revealed a new mechanism associated with NF-κB and PI3K inhibition triggered impairment of cell proliferation and induction of cell death of cancer cells. Blocking NF-κB increases the role of LY294002 in up-regulating the expression of p53, PUMA and Beclin1 related to apoptosis and autophagy. These findings suggest that inhibition of NF-κB p65 can significantly enhance the anti-tumor effects of LY294002 on the gastric cancer cell line SGC7901 in vitro, and further indicated that development of SN50 and LY294002 based therapeutics might be a potential approach for gastric cancer management.
Acknowledgments
The project was supported by The Natural Science Foundation of China (No. 81172348), the Health Foundation of Jiangsu Province (H200719), and the Natural Science Foundation of Jiangsu Province (BK2008168), a Suzhou high-level talents project (2008-11) and the Science, Education and Health Foundation of Suzhou City (SWKQ0914).
References
- 1.Parkin DM, Bray FI, Devesa SS. Cancer burden in the year 2000. The global picture. Eur J Cancer. 2001;37(Suppl. 8):54–66. doi: 10.1016/s0959-8049(01)00267-2. [DOI] [PubMed] [Google Scholar]
- 2.Jemal A, Thomas A, Murray T, Thun M. Cancer statistics. CA Cancer J Clin. 2002;52:23–47. doi: 10.3322/canjclin.52.1.23. [DOI] [PubMed] [Google Scholar]
- 3.Zheng L, Wang L, Ajani J, Xie K. Molecular basis of gastric cancer development and progression. Gastric Cancer. 2004;7:61–77. doi: 10.1007/s10120-004-0277-4. [DOI] [PubMed] [Google Scholar]
- 4.Vogt PK. PI 3-kinase, mTOR, protein synthesis and cancer. Trends Mol Med. 2001;7:482–4. doi: 10.1016/s1471-4914(01)02161-x. [DOI] [PubMed] [Google Scholar]
- 5.Oldham S, Hafen E. Insulin/IGF and target of rapamycin signaling: a TOR de force in growth control. Trends Cell Biol. 2003;13:79–85. doi: 10.1016/s0962-8924(02)00042-9. [DOI] [PubMed] [Google Scholar]
- 6.Ma J, Sawai H, Matsuo Y, et al. IGF-1 and PTEN regulate the proliferation and invasiveness of colon cancer cells through opposite effects on PI3K/Akt signaling. Arch Med Sci. 2009;5:195–206. [Google Scholar]
- 7.Choe G, Horvath S, Cloughesy TF, et al. Analysis of the phosphatidylinositol 3’-kinase signaling pathway in glioblastoma patients in vivo. Cancer Res. 2003;63:2742–6. [PubMed] [Google Scholar]
- 8.Neve RM, Holbro T, Hynes NE. Distinct roles for phosphoinositide 3-kinase, mitogen-activated protein kinase and p38 MAPK in mediating cell cycle progression of breast cancer cells. Oncogene. 2002;21:4567–76. doi: 10.1038/sj.onc.1205555. [DOI] [PubMed] [Google Scholar]
- 9.Philp AJ, Campbell IG, Leet C, et al. The phosphatidylinositol 3’-kinase p85alpha gene is an oncogene in human ovarian and colon tumors. Cancer Res. 2001;61:7426–79. [PubMed] [Google Scholar]
- 10.Xing CG, Zhu BS, Liu HH, et al. LY294002 induces p53-dependent apoptosis of SGC7901 gastric cancer cells. Acta Pharmacologica Sinica. 2008;29:489–98. doi: 10.1111/j.1745-7254.2008.00770.x. [DOI] [PubMed] [Google Scholar]
- 11.Saika S, Miyamoto T, Yamanaka O, et al. Therapeutic effect of topical administration of SN50, an inhibitor of nuclear factor-kappaB, in treatment of corneal alkali burns in mice. Am J Pathol. 2005;166:1393–403. doi: 10.1016/s0002-9440(10)62357-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Boothby M. Specificity of sn50 for NF-kappaB? Nat Immunol. 2001;2:471–2. doi: 10.1038/88652. [DOI] [PubMed] [Google Scholar]
- 13.Baldwin A. Control of oncogenesis and cancer therapy resistance by the transcription factor NF-kappaB. J Clin Invest. 2001;107:241–6. doi: 10.1172/JCI11991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Edinger AL, Thompson CB. Defective autophagy leads to cancer. Cancer Cell. 2003;4:422–4. doi: 10.1016/s1535-6108(03)00306-4. [DOI] [PubMed] [Google Scholar]
- 15.Ahn CH, Jeong EG, Lee JW, et al. Expression of Beclin1, an autophagy-related protein, in gastric and colorectal cancers. APMIS. 2007;115:1344–9. doi: 10.1111/j.1600-0463.2007.00858.x. [DOI] [PubMed] [Google Scholar]
- 16.Sun Y, Zhang J, Peng ZL. Beclin1 induces autophagy and its potential contributions to sensitizes SiHa cells to carboplatin therapy. Int J Gynecol Cancer. 2009;19:772–6. doi: 10.1111/IGC.0b013e31819d7d10. [DOI] [PubMed] [Google Scholar]
- 17.Zhu BS, Xing CG, Lin F, Fan XQ, Zhao K, Qin ZH. Blocking NF-kappaB nuclear translocation leads to p53-related autophagy activation and cell apoptosis. World J Gastroenterol. 2011;17:478–87. doi: 10.3748/wjg.v17.i4.478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sun JL, Zhu BS, Gong W, et al. Effects of combined therapy of LY294002 and SN50 on nude mice model with gastric cancer [Chinese] Zhonghua Wei Chang Wai Ke Za Zhi. 2011;14:364–7. [PubMed] [Google Scholar]
- 19.Paglin S, Hollister T, Delohery T, et al. A novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles. Cancer Res. 2001;61:439–44. [PubMed] [Google Scholar]
- 20.Sherr CJ, McCormick F. The RB and p53 pathways in cancer. Cancer Cell. 2002;2:103–12. doi: 10.1016/s1535-6108(02)00102-2. [DOI] [PubMed] [Google Scholar]
- 21.Hofseth LJ, Hussain SP, Harris CC. p53: 25 years after its discovery. Trends Pharmacol Sci. 2004;25:177–81. doi: 10.1016/j.tips.2004.02.009. [DOI] [PubMed] [Google Scholar]
- 22.Joers A, Jaks V, Kase J, Toivo M. p53-dependent transcription can exhibit both on/off and graded response after genotoxic stress. Oncogene. 2004;23:6175–85. doi: 10.1038/sj.onc.1207864. [DOI] [PubMed] [Google Scholar]
- 23.Yu J, Zhang L, Hwang PM, Kinzler KW, Vogelstein B. PUMA induces the rapid apoptosis of colorectal cancer cells. Mol Cell. 2001;7:673–82. doi: 10.1016/s1097-2765(01)00213-1. [DOI] [PubMed] [Google Scholar]
- 24.Nakano K, Vousden KH. PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell. 2001;7:683–94. doi: 10.1016/s1097-2765(01)00214-3. [DOI] [PubMed] [Google Scholar]
- 25.Yu J, Zhang L. No PUMA, no death: implications for p53-dependent apoptosis. Cancer Cell. 2003;4:248–9. doi: 10.1016/s1535-6108(03)00249-6. [DOI] [PubMed] [Google Scholar]
- 26.Westwick JK, Lee RJ, Lambert QT, et al. Transforming potential of Db1 family proteins correlates with transcription from the cyclin D1 promoter but not with activation of Jun NH2-terminal kinase, p38:Mpk2, serum response factor, or c-Jun. J Biol Chem. 1998;273:16739–47. doi: 10.1074/jbc.273.27.16739. [DOI] [PubMed] [Google Scholar]
- 27.Westwick JK, Lambert QT, Clark GJ, et al. Rac regulation of transformation, gene expression and actin organisation by multiple, PAK-independent pathways. Mol Cell Biol. 1997;17:1324–35. doi: 10.1128/mcb.17.3.1324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mitsiades N, Mitsiades CS, Poulaki V. Biologic sequelae of nuclear factor-kappaB blockade in multiple myeloma: therapeutic applications. Blood. 2002;9:4079–86. doi: 10.1182/blood.v99.11.4079. [DOI] [PubMed] [Google Scholar]
- 29.Wang CY, Guttridge DC, Mayo MW, Baldwin AS., Jr NF-kappaB induces expression of the Bcl-2 homologue A1/Bfl-1 to preferentially suppress chemotherapy-induced apoptosis. Mol Cell Biol. 1999;19:5923–9. doi: 10.1128/mcb.19.9.5923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang CY, Mayo MW, Korneluk RG, Goeddel DV, Baldwin AS., Jr NF-kappaB antiapoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 activation. Science. 1998;281:1680–3. doi: 10.1126/science.281.5383.1680. [DOI] [PubMed] [Google Scholar]
- 31.Yu J, Zhang L, Hwang PM, Kinzler KW, Vogelstein B. PUMA induces the rapid apoptosis of colorectal cancer cells. Mol Cell. 2001;7:673–82. doi: 10.1016/s1097-2765(01)00213-1. [DOI] [PubMed] [Google Scholar]
- 32.Nakano K, Vousden KH. PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell. 2001;7:683–94. doi: 10.1016/s1097-2765(01)00214-3. [DOI] [PubMed] [Google Scholar]
- 33.Lum JJ, DeBerardinis RJ, Thompson CB. Autophagy in metazoans: cell survival in the land of plenty. Nat Rev Mol Cell Biol. 2005;6:439–48. doi: 10.1038/nrm1660. [DOI] [PubMed] [Google Scholar]
- 34.Aita VM, Liang XH, Murty VV, et al. Cloning and genomic organization of Beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics. 1999;59:59–65. doi: 10.1006/geno.1999.5851. [DOI] [PubMed] [Google Scholar]