Abstract
E2F factors are implicated in various cellular processes including specific gene induction at the G1/S transition of the cell cycle. We present in this study a novel regulatory aspect for the tobacco large subunit of ribonucleotide reductase (R1a) and its encoding gene (RNR1a) in the UV-C response. By structural analyses, two E2F sites were identified on the promoter of this gene. Functional analysis showed that, in addition to their role in the specific G1/S induction of the RNR1a gene, both E2F sites were important for regulating specific RNR1a gene expression in response to UV-C irradiation in non-synchronized and synchronized cells. Concomitantly, western blot and cellular analyses showed an increase of a 60 kDa E2F factor and a transient translocation of a GFP-R1a protein fusion from cytoplasm to nucleus in response to UV irradiation.
INTRODUCTION
In mammals, the E2F transcription factor plays a crucial role in various cellular processes, such as proliferation, apoptosis and differentiation (1). In this context, extensive studies have demonstrated the role of E2F transcription factors in the control of cell cycle progression and their requirement for G1/S induction of genes linked to DNA replication (2). The mammalian E2F family consists of seven members, where E2F1 to E2F5 factors possess a trans-activation domain and a Rb binding domain while the E2F6 and E2F7 factors contain neither (3,4). E2F-mediated response is achieved by heterodimerization of E2F with its dimerization partner (DP) and binding to their target DNA motif, TTTSSCGC. In quiescent cells, E2F4 and E2F5 were shown to silence expression of various genes, which are in turn derepressed upon cell cycle activation. Transcriptional repression is mediated through binding of E2F4/p130 or E2F5/p107 repressor complexes to the cell cycle-dependent element (CDE) and cell cycle gene homology region (CHR) on their target gene promoters. E2F1, E2F2 and E2F3 factors are activators of genes that are highly transcribed during S phase (5). Recent data obtained from microarray analyses extend the role of E2F to other pathways, such as DNA repair and mitosis (6).
In plants, six E2F factors have been cloned in Arabidopsis (7,8). Similar to their mammalian E2F1-3 counterparts, AtE2Fa-c factors can transactivate E2F targets and present conserved residues for DNA binding, dimerization, transactivation and Rb binding (7,9). The other E2F factors named DP-E2F-like factors (DEL1, 2, 3) lack dimerization, trans-activation and Rb binding domains but contain a duplicated DNA binding domain (DBD) (7), as recently observed for the newly identified human E2F7 factor (4). These DEL factors act as transcriptional repressors (7,10). In plants, only a few genes have been investigated for E2F-controlled transcriptional regulation. The tobacco RNR2 promoter was the first to be described to have an in vivo E2F-mediated regulation. According to the promoter context, E2F elements act only as activators at the entry of S phase (11) or switch from a repressor to an activator element during cell cycle progression (12). E2F elements in the tobacco PCNA promoter drive transcriptional activation in proliferating cells (13) but behave as repressors in differentiated tissues (14). An extensive analysis of potential E2F targets in the genome of Arabidopsis revealed that various genes belonging to different categories (e.g. stress or signal transduction) are regulated by the E2F pathway (15). Nevertheless, only limited functional data on the role of E2F elements in regulating plant promoters are available to date (16).
Ribonucleotide reductase plays an important role in providing the cell with the dNTP pool needed for DNA replication and urgent DNA repair (17). A malfunction of RNR in providing the cell with an unbalanced dNTP pool can lead to misincorporation of dNTPs into DNA and therefore to genetic abnormalities (18). Consequently, RNR is submitted to a strict regulation that maintains the four dNTPs at optimal levels during DNA replication and repair. In yeast, RNR is controlled at different levels during DNA replication as well as upon DNA damage: transcription, post-transcription (protein stability) and RNR activity (allosteric control and activity inhibitor) (19–22). In the DNA damage response, one gene encoding the large subunit, namely RNR3, is exclusively induced upon DNA damage. In addition to transciptional regulation, the Mec1/Rad53 pathway, a central player in the DNA damage response, regulates RNR activity through phosphorylation of Sml1 which binds to yeast RNR and inhibits its activity (23). In mammals, activation of the small subunit RNR gene p53R2 depends on p53 after DNA damage (24). This emphasizes the importance of dNTP regulation after DNA damage in yeast and mammals and therefore the need to investigate further RNR regulation in plants.
This paper focuses on the involvement of E2F in the regulation of the tobacco RNR1a gene expression in both cell cycle and UV-C response. Structural analyses show that two E2F elements present on the RNR1a promoter interact with a purified tobacco E2F factor as well as with specific nuclear complexes. Functional analyses reveal that both E2F elements are important for driving RNR1a promoter induction at the G1/S transition in synchronized BY-2 tobacco cells, as well as in response to UV irradiation in dividing non-synchronized cells and S phase cells. UV-C irradiation of mid-log phase cells led to an increase of a 60 kDa E2F factor as well as to a transient translocation of an EGFP-R1a fusion protein from cytoplasm to nucleus. Compared with mammals, the present study reports a new aspect of RNR regulation in UV-C response involving both E2F-mediated transcriptional control as well as subcellular localization of the R1a protein.
MATERIALS AND METHODS
Plant cell culture, synchronization and UV-C irradiation
The BY-2 tobacco cell suspension was maintained by weekly subculture as described (25) at 27°C. For cell synchronization, freshly sub-cultured stationary phase cells were treated with aphidicolin (3 µg/ml, Sigma) for 24 h and extensively washed with sucrose (30 g/l). DNA synthesis and mitotic index were monitored as described previously (26).
Cell irradiation was performed inside a Stratalinker 2400 (Stratagene) irradiation chamber equipped with 254 nm UV-C light bulbs. Fifty millilitres of cells were irradiated in a large Petri dish without a lid. After irradiation, cells were cultured in liquid medium at 27°C.
RNA isolation and northern blot analysis
Total RNA was isolated from BY-2 cells, analysed by formaldehyde-agarose gel electrophoresis and transferred onto a Hybond N+ membrane (Amersham Biosciences) as described (11). Hybridization was performed overnight at 42°C with 32P-labelled probes in the presence of 50% formamide.
Nuclear extracts and gel shift assays
Nuclear extracts were prepared as described previously (12). Gel shift assays were performed in the presence of 6 µg of nuclear extract or 300 ng of purified tobacco protein obtained as described (12). Protein samples were incubated with 20 000 c.p.m. of radio-labelled probes in 20 µl of binding buffer (25 mM HEPES, pH 7.5, 50 mM KCl, 5 mM MgCl2, 0.1 mM EDTA, 10% glycerol, 5 mM β-mercaptoethanol, 0.5 mM PMSF, 250 µg/l pepstatin, 500 µg/l leupeptin) in the presence of 0.05% Nonidet P-40 and 1 µg of poly(dI–dC)–poly(dI–dC) (Pharmacia Biotech). The unrelated oligonucleotide used in the gel shift assay was TGCCATCACGAA-GCTTAC TAATATGAAC. For competition assays, a molar excess of unlabelled double-stranded oligonucleotides or 2 µl of antibody (anti E2F5; Santa Cruz) was included in the reaction. The samples were electrophoresed on 4.5% polyacrylamide gels in 0.25× TBE at 4°C (8 V/cm). After electrophoresis, the gels were dried and autoradiographed.
Western blot analysis
Nuclear proteins were analysed by SDS–PAGE and transferred to Immobilon-P membranes (Millipore) using a mini transblot transfer cell apparatus (Bio-Rad). The blots were incubated with a polyclonal serum raised against the DBD of the human E2F5 factor (Santa Cruz), and immunodetection was performed using ECL chemiluminescence detection reagents (Pierce).
Cloning of the RNR1a promoter
The RNR1a promoter sequence was amplified by inverse PCR (IPCR) on tobacco genomic DNA. A fragment from –1151 bp to the ATG start codon was obtained. The DNA region from –423 to –20 bp was amplified by PCR with XbaI–BamHI linkers and was subcloned into the XbaI–BamHI sites of the pBluescript (pKS) vector to give the reference construct used in our experiments. This construct was mutated in both E2F sites (dE2F/CDE and pE2F) by PCR-based site-directed mutagenesis, using the same nucleotide changes as in gel shift assays (indicated in Fig. 2), and gave the dpE2FMU construct.
Figure 2.
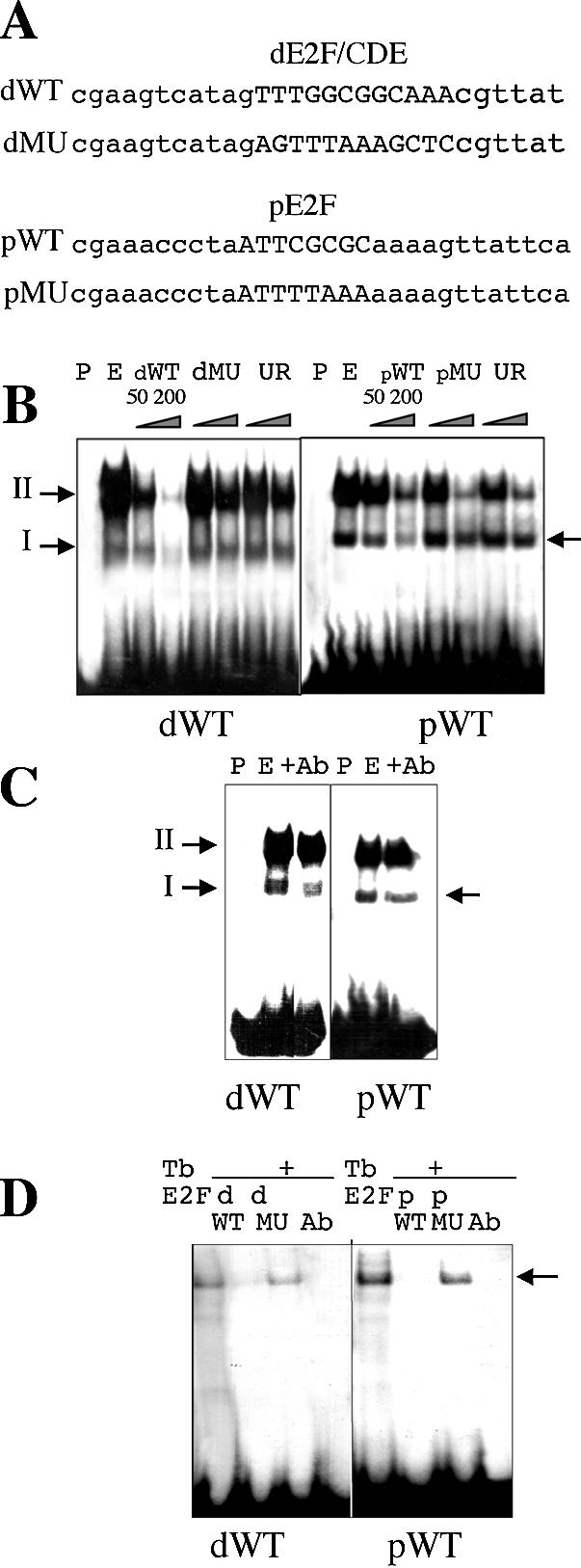
Binding properties of the E2F elements of the RNR1a promoter. (A) dWT and pWT oligonucleotides carrying either dE2F/CDE or pE2F motifs are indicated as well as their mutated versions in their E2F sites, dMU and pMU, respectively. (B) The WT oligonucleotides were used as probes (P) in gel shift experiments performed with nuclear extracts (E) prepared from mid-log phase BY-2 cells. The dWT probe revealed two specific complexes, I and II, whose binding was competed by a 50–200-fold molar excess of unlabelled oligonucleotides (dWT) but not by the E2F-mutated (dMU) and unrelated (UR) oligonucleotides. In contrast, a single specific complex was revealed with the pWT probe (see arrow): binding is competed by an excess of the pWT oligonucleotides but not by the pMU oligonucleotides. (C) E2F factor is part of nuclear complexes bound to E2F elements of the RNR1a promoter. Binding of specific complexes is competed by 2 µl of antibody directed against the DBD of human E2F5 (Ab). (D) Specific interaction between E2F sites of the RNR1a promoter and a purified tobacco E2F factor (TbE2F). A complex is detected (see arrow) with both WT probes and the complex is competed by a 200-fold molar excess of the wild-type unlabelled probe (pWT or dWT) and 2 µl of antibody raised against the DBD of human E2F5 (Ab) but not by a 200-fold excess of E2F-mutated oligonucleotides (dMU or pMU).
Promoter constructs and generation of transgenic cells
The WT promoter construct and its E2F-mutated version were subcloned by PCR into the KpnI–NcoI sites of the luciferase (LUC)-intron reporter gene plasmid pLuk07 (27), to replace the original CaMV 35S promoter. In addition, a minimal KpnI–NcoI promoter restricted to the TATA box (–108 to –20 bp) was similarly subcloned into the pLuk07 vector.
Then, each KpnI–XbaI fragment carrying the promoter-LUC fusion was cloned into the KpnI–XbaI sites of the binary vector pCGN1549 (Calgene). Constructs were introduced into Agrobacterium tumefaciens LBA4404 and used to transform tobacco BY2 cells, as described (28). Approximately 1000 kanamycin-resistant calli were pooled and grown as suspension cultures. The transgenic cell suspensions were maintained by subculturing 2 ml of stationary phase cells in 80 ml of fresh medium supplemented with carbenicillin (500 µg/ml) and kanamycin (100 µg/ml). After four rounds of subculture, carbenicillin was omitted from the medium.
LUC assay
Two millilitres of cells were washed twice in PBS buffer (140 mM NaCl, 2.7 mM KCl, 1.5 mM KH2PO4, 4.3 mM Na2HPO4 pH 7.4) and lysed by a 10 min incubation at room temperature in 200 µl of lysis buffer (100 mM potassium phosphate buffer pH 7.8, 1 mM DTT and 0.2% Triton X-100). After centrifugation at 3500 g for 3 min, the supernatant was frozen in liquid nitrogen and stored at –80°C. Luciferase activity was measured using the Luciferase assay kit (Tropix) in a microplate luminometer (TR 717 Tropix Perkin Elmer Applied Biosystems) according to the manufacturer’s instructions.
EGFP construct, observation of the GFP fluorescence and cell treatment with leptomycin B
The open-reading frame of the RNR1a cDNA (accession no. Y10861) was amplified with the flanking attB sites and subcloned into the pDONR201 vector (Invitrogen, Gaithersburg, MD, USA) by recombination with the attP sites of the vector mediated by the GATEWAY™ BP Clonase™ enzyme mix. Then, this open-reading frame was transferred downstream from the EGFP into the pK7WGF2 binary destination vector (29) by using the GATEWAY™ LR Clonase™ enzyme mix. Upon subcloning into Agrobacterium LBA4404, the EGFP-R1a construct was used to stably transform BY-2 cells, as described previously (28). Calli were maintained as suspension cultures in selective medium supplemented with kanamycin (100 µg/ml). GFP fluorescence was analysed using a Zeiss LSM 510 confocal laser scanning microscope with the 488 nm ray of the argon laser and a barrier filter of 505–550 nm.
As specific inhibitor of nuclear export, we used leptomycin B (LMB; Sigma). Experiments were performed on BY2 mid-log phase cells in the presence of LMB (0.1 µM). After overnight incubation at 24°C, GFP fluorescence was observed.
RESULTS
Tobacco RNR1a promoter analysis
Our previous results showed that the small multigenic RNR1 gene family was cell cycle regulated in synchronized BY2 cells (30). To investigate the specific transcriptional regulation of the RNR1a gene, a 1151 bp promoter sequence was amplified by IPCR from tobacco genomic DNA. Various potential cis-elements were identified upstream from the ATG codon (Fig. 1), notably a TATA box (at –108 bp), two E2F elements related to the consensus TTTSSCGC: a proximal E2F element, called pE2F (ATTCGCGC, at –143 bp) and a distal reverse E2F element, named dE2F (GCGGCAAA) combined with a direct CDE-like element (TTTGGCGG), at –413 bp (31). A CHR-like element (CGTTAT) was identified downstream from the CDE element. Compared with the tobacco RNR2 promoter (12), the organization of E2F elements was different in the RNR1a promoter: the composite element dE2F/CDE was distal, whereas the other element was proximal and located next to a telo box (AAACCCTAA). Telo boxes were described to act in synergy with regulatory cis-elements in Arabidopsis (32,33). Potential Myb elements (CAACAG) were located next to the E2F motifs at –385 and –100 bp from the ATG, in a direct and reverse orientation, respectively (34). Interestingly, the Myb site at –385 bp was found downstream from the E2F/CDE element as for the RNR2 promoter. In the distal part of the promoter from –1115 to –1074 bp, putative scaffold attachment regions (SARs) were identified, which correspond to A/T boxes or T boxes and display intrinsic curved DNA as for animal SARs (35).
Figure 1.
Nucleotide sequence of the tobacco RNR1a promoter. A –1151pb DNA fragment upstream from the ATG was amplified by IPCR. The subcloned sequence tested for promoter activity in our experiments is delimited by stars. Putative E2F elements are boxed, the distal dE2F/CDE composite element as well as the proximal pE2F element are indicated. A putative CHR box is in italic, potential Myb and telo elements are in bold characters and a potential TATA box is framed. Orientation of the various cis-elements is indicated by horizontal arrows. Putative SAR elements are underlined.
Binding properties of the E2F motifs in the RNR1a promoter
To characterize the binding capacity of the E2F sites in the RNR1a promoter, we assayed the specific in vitro binding of nuclear complex(es) to these sites. To this end, gel shift assays were performed with either the dWT or pWT probes, carrying, respectively, the dE2F/CDE and pE2F motifs (Fig. 2A), in the presence of nuclear extracts from mid-log phase BY-2 cells. The dWT probe that contained both dE2F and CDE (repressor-like element) elements detected two specific complexes (I and II) while the pWT probe revealed a single specific complex (Fig. 2B). Binding of these specific complexes to their respective target was titrated out by an excess of their unlabelled WT probes but not by their E2F-mutated oligonucleotides (dMU and pMU oligonucleotides presented in Fig. 2A) nor by unrelated oligonucleotides (Fig. 2B). Binding of specific complexes to the pWT and dWT probes was partly prevented by a specific antibody raised against the DNA binding domain (DBD) of human E2F5 [which is well conserved between plants and animals (12)], when added to nuclear extracts prior to probe incubation (Fig. 2C). This suggests that E2F factors may belong to these complexes.
In addition, we checked the capacity of the E2F sites to bind a purified tobacco E2F protein (12). As shown in Figure 2D, binding of the tobacco E2F factor to the dWT or pWT probes was detected and could be competed, respectively, with the dWT or pWT wild-type sequences but not with the pMU or dMU mutated E2F sequences. In our experimental conditions, E2F binding might not be optimal since no DP was added, nevertheless the observed interaction is specific. A similar observation was reported when a wheat E2F factor was tested alone or in the presence of DP for interaction with specific E2F elements (36). Moreover, the antibody directed against the DNA binding domain (DBD) of human E2F5 prevented binding of the tobacco E2F factor to its target elements. Therefore, both E2F elements present in the RNR1a promoter showed binding capacity for E2F factors.
Specific RNR1a gene expression in tobacco BY-2 cells parallels RNR1a promoter activity
BY-2 tobacco cells were synchronized by aphidicolin and specific RNR1a mRNA levels were evaluated for 16 h following the release from the inhibitory block. Two parameters were measured during cell synchronization: the mitotic index and DNA synthesis, which allowed one to delimit the progression of mitosis and S phases, respectively (Fig. 3A). S1 represents the first S phase after release from the aphidicolin block and S2 the second S phase. Northern blot analysis was performed with extracts prepared from cells taken at different phases of the cell cycle. The blots were hybridized with a 3′ UTR region of the RNR1a cDNA. The RNR1a mRNA level paralleled DNA synthesis in both S1 and S2 phases (Fig. 3B) and also increased slightly at the G2/M transition, which could be related to RNR1a gene induction for DNA repair before entry into mitosis.
Figure 3.
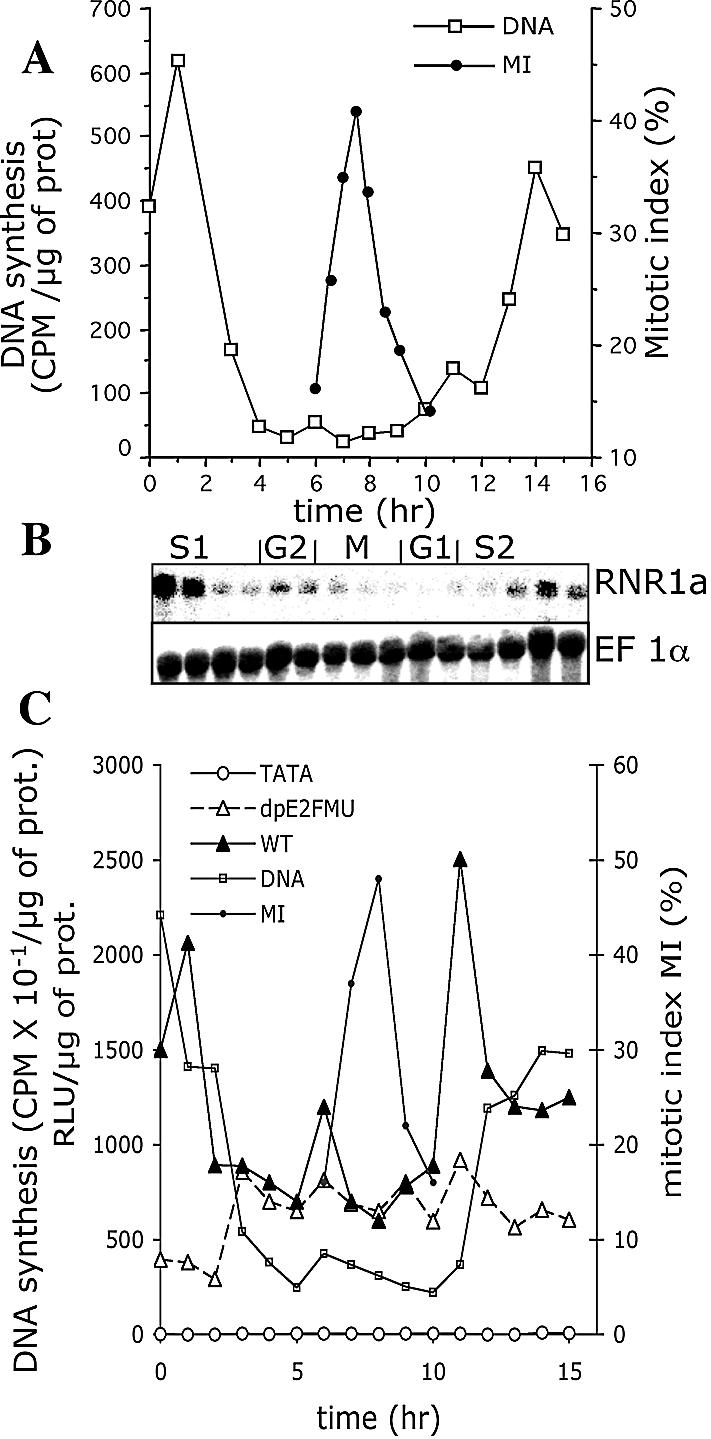
Specific expression of the RNR1a gene parallels its promoter activity during the cell cycle. (A) Analysis of parameters in cell cycle progression of synchronized tobacco BY-2 cells. Cells were synchronized by aphidicolin and after removal of the inhibitor, DNA synthesis was monitored by [3H]thymidine pulse labelling experiments (open squares) and mitotic index was determined by UV light microscopic analysis of cells stained with DAPI (filled circles). (B) Specific RNR1a gene expression in synchronized BY-2 cells. RNA samples (20 µg) prepared from cells taken every hour were blotted and hybridized to the 3′ specific region of the RNR1a cDNA. The constitutively expressed EF-1α gene (57) was blot-hybridized for loading control. (C) RNR1a promoter activity in synchronized BY-2 cells and role of E2F elements. RNR1a promoter constructs were fused to the luciferase reporter gene: the wild-type promoter (WT), the WT promoter mutated in both dE2F/CDE and pE2F sites (dpE2FMU) and the minimal promoter reduced to the TATA box (TATA). Mutations of the composite dE2F/CDE element and pE2F site were identical to those described in bandshift experiments (Fig. 2A). Transgenic BY-2 cells harbouring the various promoter constructs were synchronized by aphidicolin. After removal of aphidicholin, cell cycle progression was monitored by measurements of DNA synthesis (open squares) and mitotic index (filled circles). Similar synchronization profiles were obtained with the different constructs. At different time-points of the cell cycle, LUC activities were measured in the transgenic cells harbouring the following promoter constructs: WT (filled triangles), dpE2FMU (open triangles, broken line), TATA (open circles). RLU, relative light unit. Results were reproducible in three independent experiments with a pool of 1000 individual clones.
In order to determine if the RNR1a mRNA level was mainly due to transcriptional regulation, we investigated the RNR1a promoter activity through the cell cycle as well as the role of E2F elements in the control of its activity. Different constructs were generated: a 403 bp promoter (WT, delimited by stars in Fig. 1) containing E2F elements and most of the regulatory elements described above, a 403 bp mutated promoter (dpE2FMU) carrying both mutated E2F elements (dE2F/CDE and pE2F sites) and an 88 bp minimal promoter (TATA) reduced to the TATA box. These different constructs were fused to the luciferase reporter gene (LUC) and were stably transferred into BY-2 cells. Pools of 1000 individual clones were cultured as cell suspensions and analysed. After synchronization of the transgenic lines by aphidicolin, DNA synthesis and the mitotic index were monitored during cell cycle progression. Cell synchronization parameters were similar for the WT, dpE2FMU and TATA promoter constructs (Fig. 3C), with a maximal mitotic index (46–48%) at 8 h and a maximal second S phase at 12 h. LUC activity of the various constructs was measured concomitantly. WT promoter activity paralleled the RNR1a mRNA levels observed in Figure 3B. In the first S phase, after aphidicolin block release, the promoter was already induced, as we previously observed (30), then promoter activity increased slightly in the G2/M transition (6 h), decreased in mitosis and increased concurrently with DNA synthesis in the second S phase (10–12 h) to reach a 2.5-fold maximal level of induction. Interestingly, the activity of the promoter extended to its distal part (up to –1113 bp in Fig. 1) presented an overall higher activity (5-fold higher) compared with the –403 bp promoter (WT) but had a similar induction level in S phase (data not shown). The minimal promoter presented a very low basal activity. Finally, mutation of both E2F elements prevented any induction of the promoter at the G1/S transition. Therefore, we can conclude that E2F elements present in the 403 bp sequence are important for driving S phase induction of the RNR1a promoter during the cell cycle.
Involvement of E2F elements in the RNR1a promoter induction in non-synchronized cells upon UV-C irradiation
Since RNR1a mRNA levels increased concomitantly with RNR1a promoter activity at the G2/M transition, we correlate such RNR induction to DNA repair activity occurring before entry into mitosis. Therefore, we investigated the transcriptional regulation of the RNR1a gene in the DNA damage response as well as the involvement of E2F factors.
We analysed the DNA damage response of the WT or dpE2FMU promoter fused to the LUC reporter gene in transgenic BY-2 cells. Mid-log phase cells were submitted to a 450 J/m2 UV-C irradiation and LUC activity was measured at 0 and 4 h following cell irradiation. Such UV-C energy was previously shown to induce efficient DNA repair in Arabidopsis cells (37) and was used to test gene induction into BY-2 cells (38). As seen in Figure 4A, the activity of the WT promoter increased to 2.5-fold at 4 h following irradiation but not for the dpE2FMU promoter. In conclusion, the RNR1a promoter is induced upon UV-C irradiation in BY-2 cells and E2F motifs are important in mediating such a response.
Figure 4.
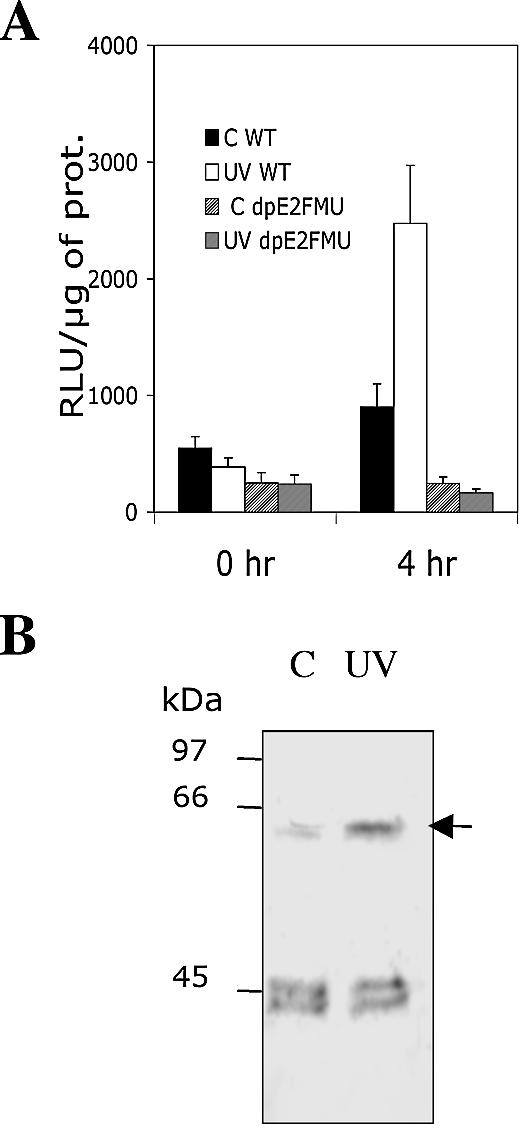
E2F-mediated regulation of the RNR1a gene in the UV-C response. (A) LUC activity was measured in mid-log phase BY-2 transgenic cells harbouring the WT (non-hatched bars) or dpE2FMU (hatched bars) promoter constructs. Transgenic cells with the WT (empty bars) or dpE2FMU constructs (fine hatched bars) were taken at 0 and 4 h following UV-C irradiation at 450 J/m2. In parallel, control non-irradiated cells harbouring the WT (black bars) or dpE2FMU constructs (large hatched bars) were analysed. Results were reproducible in three individual experiments, error bars are indicated. (B) E2F factor analysis in 6 µg of nuclear extracts from mid-log phase cells which were non-irradiated (C) or taken at 4 h after irradiation (UV-C, 450 J/m2). The protein gel blot was incubated with the antibody directed against the well conserved DBD of human E2F5 factor. Various E2F factors present in the BY-2 cells were detected.
Subsequently, we analysed the level of E2F factors upon irradiation of mid-log phase BY-2 cells. Nuclear extracts were prepared from control non-irradiated cells (C) or cells taken at 4 h after UV-C irradiation (UV) at 450 J/m2 (Fig. 4B). An antibody directed against the human E2F5 DBD, which is well conserved among eukaryotes, revealed the E2F factors present in the tobacco nuclear extracts with apparent molecular masses of 45 and 60 kDa (12). The levels of a 60 kDa E2F factor were considerably increased upon UV irradiation, suggesting a role for this E2F factor in the DNA damage response.
RNR1a gene response to a replication block and UV-C irradiation in synchronized S phase cells
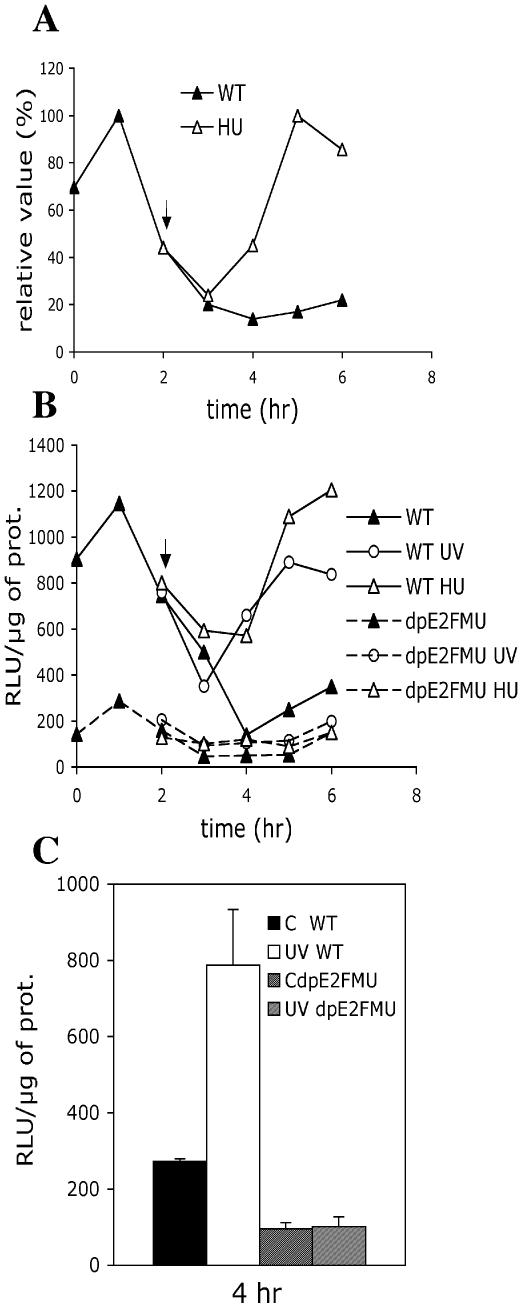
We further investigated the DNA damage response of the RNR1a gene in synchronized cells. Specific RNR1a mRNA levels were evaluated in mid-S phase cells upon DNA replication block mediated by hydroxyurea (HU). Two hours after aphidicolin removal, cells were treated with HU (60 mM) (Fig. 5A). The RNR1a mRNA level paralleled the control 1 h after HU treatment, then increased to its maximum (five times higher than the control) at 4 h after drug application. Promoter activity was also evaluated in mid-S phase transgenic cells upon DNA replication block via HU or UV-C irradiation (450 J/m2). LUC expression driven by the WT promoter construct paralleled RNR1a mRNRA accumulation after adding HU, suggesting that the specific mRNA increase was mainly due to transcriptional activation of the RNR1a promoter (Fig. 5B). In response to UV-C, the WT promoter construct increased to a maximal level (4-fold compared with the non-irradiated control), 4 h after cell irradiation. As RNR1a promoter was similarly induced upon HU or UV-C treatment, such induction might be related to a feedback RNR regulation due to a DNA replication block that probably involves a common signalling pathway. In addition, mutation of E2F elements prevented any promoter induction in HU- or UV-treated cells, which reinforces the important role of these cis-elements in the RNR1a HU–UV response. Moreover, a 3-fold WT promoter induction was observed 4 h after cell irradiation (Fig. 5C), even in stationary phase cells shown to be arrested in G1 (39). No induction was detected when both E2F sites were mutated in the promoter. Thus, these data suggest that induction of the RNR1a promoter in G1-arrested cells may be a direct response to UV-C irradiation.
Figure 5.
RNR1a transcript levels and promoter activity in response to a replication block or UV-C irradiation during S phase. (A) Kinetics of RNR1a transcript accumulation in synchronized tobacco cells following a replication block in mid-S phase. Synchronized cells were treated with HU (60 mM) 2 h after removal of aphidicolin (arrow). RNA samples from control (filled triangles) or treated cells (open triangles) were hybridized to the 3′ specific probe of the RNR1a cDNA. Signals were standardized using the constitutively expressed EF1α gene. The plotted values are relative to the maximal level obtained in the control. (B) The activity of the WT (solid line) or dpE2FMU (broken line) promoter constructs was evaluated in the synchronized BY-2 transgenic cells following a replication block in mid-S phase. Two hours after removal of aphidicolin (see arrow), cells were either treated with HU (60 mM) or UV-C (450 J/m2). LUC activity was measured in the control cells (filled triangles) and in HU (open triangles) or UV (open circles) treated cells. Results were reproducible in three individual experiments. (C) LUC activity was measured in G1-arrested BY-2 cells (stationary phase cells). Transgenic lines carried the WT (non-hatched bars) or dpE2FMU (hatched bars) promoter constructs. Transgenic cells with the WT (empty bars) or dpE2FMU constructs (fine hatched bars) were taken at 4 h following UV-C irradiation at 450 J/m2. In parallel, control non-irradiated cells harbouring the WT (black bars) or dpE2FMU constructs (large hatched bars) were analysed. Results were reproducible in three individual experiments; error bars are indicated.
Transient subcellular relocalization of the large subunit R1a upon UV-C irradiation
To extend our study to the cellular level, we analysed the subcellular localization of the R1a large subunit encoded by the RNR1a gene. An EGFP:R1a protein fusion was constitutively expressed in transgenic BY-2 cells. While the GFP:R1a fusion was only located in the cytoplasm of control mid-log phase cells (Fig. 6), it transiently accumulated in the nuclei of irradiated cells, 3 h following UV-C irradiation at 450 J/m2, then returned to the cytoplasm at 4 and 5 h after irradiation. Interestingly, when cells were treated before irradiation by leptomycine B (L), an inhibitor of nuclear export, no cytoplasmic relocalization of the GFP:R1a was observed in irradiated cells at 4 and 5 h after irradiation. In cells expressing GFP alone, fluorescence was both detected in cytoplasm and nucleus at all times, in conditions of cell growth or after cell irradiation. Consequently, regulation of the R1a large subunit in the UV-C response involved both an active cytoplasmic–nuclear and nuclear–cytoplasmic shuttling of the R1a subunit as well as a transcriptional induction of the encoded RNR1a gene mediated by E2F.
Figure 6.
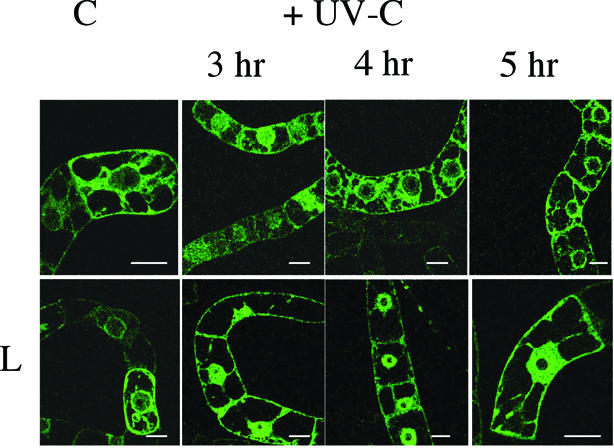
Transient subcellular localization of a constitutively expressed EGFP-R1a fusion upon UV-C irradiation. The EGFP:R1a fusion protein was stably expressed in transgenic BY-2 cells under the control of a 35S promoter. Fluorescence was observed in non-irradiated cells (C) or in cells at 3, 4 and 5 h following UV-C irradiation (450 J/m2). LMB (L) was added overnight (0.1 µM) before cell irradiation. These photographs are representatives of four individual experiments. Bar = 10 µm.
DISCUSSION
Complex regulated expression of the RNR1a gene
Our detailed analysis during cell cycle progression revealed a complex expression pattern of the RNR1a gene that belongs to the small multigenic RNR1 family in tobacco. While expression of the RNR1b gene was restricted to S phase (11), expression of the RNR1a gene was detected in both G2 and S phases, with a higher level in S phase (Fig. 3B). This result is in agreement with the complex expression pattern of the RNR genes deduced from the transcriptome analysis of synchronized BY-2 cells (40). Thus, RNR1a gene induction before mitosis may either correspond to dNTPs synthesis for urgent DNA repair or to another RNR function, such as an activator of microtubule nucleation, as described in Xenopus (41).
RNR1a promoter sequence analysis (Fig. 1) revealed the presence of a composite E2F/CDE element similar to those previously described in the tobacco RNR2 and PCNA promoter sequences (12,42). The potential SAR sequences present in the distal part of the promoter might correspond to an enhancer element that increased overall activity of the promoter during the cell cycle without affecting the induction level at the G1/S transition of the cell cycle. MAR sequences were found to act as enhancers in various tobacco genes, such as the chitinase or phaseolin genes (43,44).
Two E2F sites control cell cycle-regulated transcription of the RNR1a gene
Our results emphasize the important role played by the E2F elements in regulating RNR1a gene expression during the cell cycle. In the gel shift assays (Fig. 2), both dE2F/CDE and pE2F elements interact with specific nuclear complexes in mid-log phase cells and can bind with the purified tobacco E2F factor. Therefore, both E2F elements of the RNR1a promoter may be regulated through binding of E2F factors since cross-reacting antibody directed against the human E2F5 DBD partly prevents formation of the specific complexes. When mutated in its E2F elements, RNR1a promoter activity was considerably reduced at the entry of S phase compared with the wild-type promoter activity (Fig. 3C). That suggests that both E2F elements are important for driving S phase induction of the RNR1a promoter, as was observed for the RNR2, RNR1b promoters (11,12). Nevertheless, we cannot exclude that other cis-elements present on the RNR1a promoter might act as positive regulators in E2F-mediated regulation. Indeed, a telo box is found next to E2F sites in promoters of other tobacco and Arabidopsis genes, such as PCNA, DHFR and RNR (45), that are similarly induced at the G1/S transition; this raises the possibility that the telo box might be involved in a common regulation pathway in synergy with E2F for genes involved in DNA synthesis.
The diversity of the E2F family in plants (8,16) could reflect the complexity of expression regulation of various target genes. For instance, the six E2F proteins found in Arabidopsis can be classified as activators or repressors of gene expression, with repression being achieved by the DEL members without the need of DPs (7,13). Analysis of the specific E2F complexes associated with the E2F elements of the RNR1a promoter should give further insight in the mode of action of the various plant E2F factors in concert with specific co-regulators.
E2F-mediated transcriptional induction of the RNR1a gene and subcellular relocalization of the R1a protein upon UV-C irradiation
The more interesting feature is the E2F-mediated transcriptional induction of the RNR1a gene in the UV-C response.
Four hours after cell irradiation, RNR1a promoter activity was shown to be induced in mid-log phase cells (Fig. 4A) as well as in non-growing cells. Similar data had already been reported for yeast and mouse RNR induction in response to DNA damage by UV light (46,47). In BY-2 cells, only the auxin-regulated arcA3 gene has been reported to be induced by UV-C, at 4 h after irradiation of the cells (38).
Mutation of both E2F elements resulted in a decreased RNR1a promoter activity in growing cells, but increased activity was no more observed in response to UV-C irradiation, thus demonstrating the importance of E2F elements in inducing the RNR1a gene upon UV-C irradiation. In addition, RNR genes were recently shown to be induced upon genotoxic stress in Arabidopsis (48), underlining that RNR induction is mediated through different DNA damaging agents.
In synchronized cells, RNR1a promoter activity was considerably induced in S phase in the presence of DNA replication blocking agents like HU or by UV-C irradiation. Enhanced promoter activity might overcome the S phase block and/or provide the dNTP pool for efficient DNA repair, as it was previously described for RNR genes in yeast (46). Such induction may be related to the ATR pathway, described in mammals to be involved in the S phase checkpoint (49). As E2F factors are important in mediating RNR induction in the DNA damage response, they could act as downstream signals of the ATR-DNA damage regulatory network. Specific complexes are involved in RNR induction DNA damage in yeast and mammals. Indeed, DNA damage responsive elements present in yeast RNR promoters are activated through derepression of CRT1 complexes (50,51), whereas the RNR2p53 gene expression is induced via the p53 pathway in mammals (24). It will be interesting to characterize the specific E2F complexes involved in the DNA repair response in plants. In addition, the RNR1a induction upon UV-C irradiation observed in G1-arrested cells (i.e. stationary cells) 4 h after irradiation, demonstrates that the RNR1a gene can be induced outside S phase by UV-induced DNA damage.
Upon UV-C treatment of the cells, we observed accumulation of an E2F factor with an apparent molecular weight of 60 kDa (Fig. 4B). This E2F factor increase could result from transcriptional induction and/or stabilization of an activator E2F factor, as described for the Arabidopsis E2Fa factor upon DNA damage (48). E2F factor increase was also described in mammals where E2F1 factor was stabilized by phosphorylation in the DNA damage response (52). Upon UV irradiation, the R1a subunit is transiently translocated from cytoplasm to nucleus and may act as a signal for transcriptional induction of the RNR1a gene. Active shuttling of the R1a subunit between the two cellular compartments might involve potential nuclear localization and nuclear export signals identified on the protein, however, functional experiments are needed to show their implication in the R1a shuttling. Upon irradiation, R2 subunit relocalized to the cytoplasm where R1 resided in yeast (53), whereas both R1 and R2p53 were shown to translocate from cytoplasm to nucleus in mammals (54). Therefore, the RNR response to UV-C in plants appears to be intermediate between that of mammals and yeast. The recent discovery of the equivalent of a human ATM pathway in plants (55) suggests that similar signalling exists between human and plants, though no p53 or Chk2 factors have been identified through the complete genome sequencing of Arabidopsis (56). In conclusion, these are the first results demonstrating the implication of E2F elements in specific RNR1a gene induction upon UV-C irradiation in plants as well as transient R1a protein shuttling from cytoplasm to nucleus and nucleus to cytoplasm.
Further investigations are required to determine whether the R1a subunit could directly associate with damaged DNA or be part of transcriptional complexes for RNR1a gene induction upon DNA damage. Since RNR1a and RNR1b genes showed different responses when DNA replication was blocked in S phase (11), they probably are controlled by different regulatory pathways (ATM or ATR) involving specific downstream effectors. Connection of E2F factors to these pathways will need further analyses.
Acknowledgments
ACKNOWLEDGEMENTS
We thank Dr L. Mankin for supplying the LUC-int vector, Calgene for providing the PCGN vector and Dr M. Sekine and Dr D. Inze for their kind gift of purified tobacco E2F factor and pK7WGF2 binary vector, respectively. We also thank G. Vetter for helpful discussion. We thank B. Clément, M. Messmer and J. Betou for technical help. We are grateful to Dr P. Pfeiffer for critical reading of the manuscript. This work was supported by the Centre National de la Recherche Scientifique (CNRS) and Université Louis Pasteur (Strasbourg). The Inter-Institute Zeiss LSM510 confocal microscopy platform was cofinanced by the CNRS, Université Louis Pasteur, the Région Alsace, the Association de la Recherche sur le Cancer, and the Ligue Nationale contre le Cancer.
REFERENCES
- 1.Muller H., Bracken,A.P., Vernell,R., Moroni,M.C., Christians,F., Grassilli,E., Prosperini,E., Vigo,E., Oliner,J.D. and Helin,K. (2001) E2Fs regulate the expression of genes involved in differentiation, development, proliferation and apoptosis. Genes Dev., 15, 267–285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dyson N. (1998) The regulation of E2F by pRB-family proteins. Genes Dev., 12, 2245–2262. [DOI] [PubMed] [Google Scholar]
- 3.Trimarchi J.M., Fairchild,B., Wen,J. and Lees,J.A. (2001) The E2F6 transcription factor is a component of the mammalian Bmi1-containing polycomb complex. Proc. Natl Acad. Sci. USA, 98, 1519–1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.de Bruin A., Maiti,B., Jakoi,L., Timmers,C., Buerki,R. and Leone,G. (2003) Identification and characterization of E2F7, a novel mammalian E2F family member capable of blocking cellular proliferation. J. Biol. Chem., 278, 42041–42049. [DOI] [PubMed] [Google Scholar]
- 5.Takahashi Y., Rayman,J.B. and Dynlacht,B.D. (2000) Analysis of promoter binding by the E2F and pRB families in vivo: distinct E2F proteins mediate activation and repression. Genes Dev., 14, 804–816. [PMC free article] [PubMed] [Google Scholar]
- 6.Ren B., Cam,H., Takahashi,Y., Volkert,T., Terragni,J., Young,R.A. and Dynlacht,B.D. (2002) E2F integrates cell cycle progression with DNA repair, replication and G(2)/M checkpoints. Genes Dev., 16, 245–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mariconti L., Pellegrini,B., Cantoni,R., Stevens,R., Bergounioux,C., Cella,R. and Albani,D. (2002) The E2F family of transcription factors from Arabidopsis thaliana. Novel and conserved components of the retinoblastoma/E2F pathway in plants. J. Biol. Chem., 277, 9911–9919. [DOI] [PubMed] [Google Scholar]
- 8.Vandepoele K., Raes,J., De Veylder,L., Rouze,P., Rombauts,S. and Inze,D. (2002) Genome-wide analysis of core cell cycle genes in Arabidopsis. Plant Cell, 14, 903–916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kosugi S. and Ohashi,Y. (2002) Interaction of the Arabidopsis E2F and DP proteins confers their concomitant nuclear translocation and transactivation. Plant Physiol., 128, 833–843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kosugi S. and Ohashi,Y. (2002) E2Ls, E2F-like repressors of Arabidopsis that bind to E2F sites in a monomeric form. J. Biol. Chem., 277, 16553–16558. [DOI] [PubMed] [Google Scholar]
- 11.Chabouté M.E., Clement,B. and Philipps,G. (2002) S phase and meristem-specific expression of the tobacco RNR1b gene is mediated by an E2F element located in the 5′ leader sequence. J. Biol. Chem., 277, 17845–17851. [DOI] [PubMed] [Google Scholar]
- 12.Chabouté M.E., Clément,B., Sekine,M., Philipps,G. and Chaubet-Gigot,N. (2000) Cell cycle regulation of the tobacco ribonucleotide reductase small subunit gene is mediated by E2F-like elements. Plant Cell, 12, 1987–2000. [PMC free article] [PubMed] [Google Scholar]
- 13.Kosugi S. and Ohashi,Y. (2002) E2F sites that can interact with E2F proteins cloned from rice are required for meristematic tissue-specific expression of rice and tobacco proliferating cell nuclear antigen promoters. Plant J., 29, 45–59. [DOI] [PubMed] [Google Scholar]
- 14.Egelkrout E.M., Robertson,D. and Hanley-Bowdoin,L. (2001) Proliferating cell nuclear antigen transcription is repressed through an E2F consensus element and activated by geminivirus infection in mature leaves. Plant Cell, 13, 1437–1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ramirez-Parra E., Frundt,C. and Gutierrez,C. (2003) A genome-wide identification of E2F-regulated genes in Arabidopsis. Plant J., 33, 801–811. [DOI] [PubMed] [Google Scholar]
- 16.Gutierrez C., Ramirez-Parra,E., Castellano,M.M. and del Pozo,J.C. (2002) G(1) to S transition: more than a cell cycle engine switch. Curr. Opin. Plant Biol., 5, 480–486. [DOI] [PubMed] [Google Scholar]
- 17.Elledge S.J., Zhou,Z., Allen,J.B. and Navas,T.A. (1993) DNA damage and cell cycle regulation of ribonucleotide reductase. Bioessays, 15, 333–339. [DOI] [PubMed] [Google Scholar]
- 18.Lengauer C., Kinzler,K.W. and Vogelstein,B. (1998) Genetic instabilities in human cancers. Nature, 396, 643–649. [DOI] [PubMed] [Google Scholar]
- 19.Elledge S.J., Zhou,Z. and Allen,J.B. (1992) Ribonucleotide reductase: regulation, regulation, regulation. Trends Biochem. Sci., 17, 119–123. [DOI] [PubMed] [Google Scholar]
- 20.Chabes A. and Thelander,L. (2000) Controlled protein degradation regulates ribonucleotide reductase activity in proliferating mammalian cells during the normal cell cycle and in response to DNA damage and replication blocks. J. Biol. Chem., 275, 17747–17753. [DOI] [PubMed] [Google Scholar]
- 21.Thelander L. and Reichard,P. (1979) Reduction of ribonucleotides. Annu. Rev. Biochem., 48, 133–158. [DOI] [PubMed] [Google Scholar]
- 22.Zhao X., Muller,E.G. and Rothstein,R. (1998) A suppressor of two essential checkpoint genes identifies a novel protein that negatively affects dNTP pools. Mol. Cell, 2, 329–340. [DOI] [PubMed] [Google Scholar]
- 23.Zhao X., Chabes,A., Domkin,V., Thelander,L. and Rothstein,R. (2001) The ribonucleotide reductase inhibitor Sml1 is a new target of the Mec1/Rad53 kinase cascade during growth and in response to DNA damage. EMBO J., 20, 3544–3553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tanaka H., Arakawa,H., Yamaguchi,T., Shiraishi,K., Fukuda,S., Matsui,K., Takei,Y. and Nakamura,Y. (2000) A ribonucleotide reductase gene involved in a p53-dependent cell-cycle checkpoint for DNA damage. Nature, 404, 42–49. [DOI] [PubMed] [Google Scholar]
- 25.Nagata T., Nemoto,Y. and Hazezawa,S. (1992) Tobacco BY2 cell line as the ‘Hela’ cell in the cell biology of higher plants. Int. Rev. Cytol., 132, 1–30. [Google Scholar]
- 26.Reichheld J.P., Sonobe,S., Clément,B., Chaubet,N. and Gigot,C. (1995) Cell cycle-regulated histone gene expression in synchronized plant cells. Plant J., 7, 245–252. [Google Scholar]
- 27.Mankin S.L., Allen,G.C. and Thompson,W.F. (1997) Introduction of a plant intron into the luciferase gene of Photinus pyralis. Plant Mol. Biol. Rep., 15, 186–196. [Google Scholar]
- 28.An G. (1987) Binary Ti vectors for plant transformation and promoter analysis. Methods Enzymol., 153, 292–305. [Google Scholar]
- 29.Karimi M., Inze,D. and Depicker,A. (2002) GATEWAY vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci., 7, 193–195. [DOI] [PubMed] [Google Scholar]
- 30.Chabouté M.E., Combettes,B., Clément,B., Gigot,C. and Philipps,G. (1998) Molecular characterization of tobacco ribonucleotide reductase RNR1 and RNR2 cDNAs and cell cycle-regulated expression in synchronized plant cells. Plant Mol. Biol., 38, 797–806. [DOI] [PubMed] [Google Scholar]
- 31.Zwicker J., Lucibello,F.C., Wolfraim,L.A., Gross,C., Truss,M., Engeland,K. and Muller,R. (1995) Cell cycle regulation of the cyclin A, cdc25C and cdc2 genes is based on a common mechanism of transcriptional repression. EMBO J., 14, 4514–4522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Regad F., Lebas,M. and Lescure,B. (1994) Interstitial telomeric repeats within the Arabidopsis thaliana genome. J. Mol. Biol., 239, 163–169. [DOI] [PubMed] [Google Scholar]
- 33.Manevski A., Bertoni,G., Bardet,C., Tremousaygue,D. and Lescure,B. (2000) In synergy with various cis-acting elements, plant insterstitial telomere motifs regulate gene expression in Arabidopsis root meristems. FEBS Lett., 483, 43–46. [DOI] [PubMed] [Google Scholar]
- 34.Stracke R., Werber,M. and Weisshaar,B. (2001) The R2R3-MYB gene family in Arabidopsis thaliana. Curr. Opin. Plant Biol., 4, 447–456. [DOI] [PubMed] [Google Scholar]
- 35.Gasser S.M. and Laemmli,U.K. (1986) Cohabitation of scaffold binding regions with upstream/enhancer elements of three developmentally regulated genes of D. melanogaster. Cell, 46, 521–530. [DOI] [PubMed] [Google Scholar]
- 36.Ramirez-Parra E. and Gutierrez,C. (2000) Characterization of wheat DP, a heterodimerization partner of the plant E2F transcription factor which stimulates E2F-DNA binding. FEBS Lett., 486, 73–78. [DOI] [PubMed] [Google Scholar]
- 37.Li A., Schuermann,D., Gallego,F., Kovalchuk,I. and Tinland,B. (2002) Repair of damaged DNA by Arabidopsis cell extract. Plant Cell, 14, 263–273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Perennes C., Glab,N., Guglieni,B., Doutriaux,M.P., Phan,T.H., Planchais,S. and Bergounioux,C. (1999) Is arcA3 a possible mediator in the signal transduction pathway during agonist cell cycle arrest by salicylic acid and UV irradiation? J. Cell Sci., 112, 1181–1190. [DOI] [PubMed] [Google Scholar]
- 39.Planchais S., Glab,N., Trehin,C., Perennes,C., Bureau,J.M., Meijer,L. and Bergounioux,C. (1997) Roscovitine, a novel cyclin-dependent kinase inhibitor, characterizes restriction point and G2/M transition in tobacco BY-2 cell suspension. Plant J., 12, 191–202. [DOI] [PubMed] [Google Scholar]
- 40.Breyne P., Dreesen,R., Vandepoele,K., De Veylder,L., Van Breusegem,F., Callewaert,L., Rombauts,S., Raes,J., Cannoot,B., Engler,G. et al. (2002) Transcriptome analysis during cell division in plants. Proc. Natl Acad. Sci. USA, 99, 14825–14830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Takada S., Shibata,T., Hiraoka,Y. and Masuda,H. (2000) Identification of ribonucleotide reductase protein R1 as an activator of microtubule nucleation in Xenopus egg mitotic extracts. Mol. Biol. Cell, 11, 4173–4187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Egelkrout E.M., Mariconti,L., Settlage,S.B., Cella,R., Robertson,D. and Hanley-Bowdoin,L. (2002) Two E2F elements regulate the proliferating cell nuclear antigen promoter differently during leaf development. Plant Cell, 14, 3225–3236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fukuda Y. and Nishikawa,S. (2003) Matrix attachment regions enhance transcription of a downstream transgene and the accessibility of its promoter region to micrococcal nuclease. Plant Mol. Biol., 51, 665–675. [DOI] [PubMed] [Google Scholar]
- 44.van der Geest A.H. and Hall,T.C. (1997) The beta-phaseolin 5′ matrix attachment region acts as an enhancer facilitator. Plant Mol. Biol., 33, 553–557. [DOI] [PubMed] [Google Scholar]
- 45.Tremousaygue D., Garnier,L., Bardet,C., Dabos,P., Herve,C. and Lescure,B. (2003) Internal telomeric repeats and ‘TCP domain’ protein-binding sites co-operate to regulate gene expression in Arabidopsis thaliana cycling cells. Plant J., 33, 957–966. [DOI] [PubMed] [Google Scholar]
- 46.Elledge S.J. and Davis,R.W. (1989) DNA damage induction of ribonucleotide reductase. Mol. Cell Biol., 9, 4932–4940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Filatov D., Bjorklund,S., Johansson,E. and Thelander,L. (1996) Induction of the mouse ribonucleotide reductase R1 and R2 genes in response to DNA damage by UV light. J. Biol. Chem., 271, 23698–23704. [DOI] [PubMed] [Google Scholar]
- 48.Chen I.P., Haehnel,U., Altschmied,L., Schubert,I. and Puchta,H. (2003) The transcriptional response of Arabidopsis to genotoxic stress—a high density colony array study (HDCA). Plant J., 35, 771–786. [DOI] [PubMed] [Google Scholar]
- 49.Shiloh Y. (2001) ATM and ATR: networking cellular responses to DNA damage. Curr. Opin. Genet. Dev., 11, 71–77. [DOI] [PubMed] [Google Scholar]
- 50.Huang M., Zhou,Z. and Elledge,S.J. (1998) The DNA replication and damage checkpoint pathways induce transcription by inhibition of the Crt1 repressor. Cell, 94, 595–605. [DOI] [PubMed] [Google Scholar]
- 51.Li B. and Reese,J.C. (2000) Derepression of DNA damage-regulated genes requires yeast TAF(II)s. EMBO J., 19, 4091–4100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hofferer M., Wirbelauer,C., Humar,B. and Krek,W. (1999) Increased levels of E2F-1-dependent DNA binding activity after UV- or gamma-irradiation. Nucleic Acids Res., 27, 491–495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yao R., Zhang,Z., An,X., Bucci,B., Perlstein,D.L., Stubbe,J. and Huang,M. (2003) Subcellular localization of yeast ribonucleotide reductase regulated by the DNA replication and damage checkpoint pathways. Proc. Natl Acad. Sci. USA, 100, 6628–6633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Xue L., Zhou,B., Liu,X., Qiu,W., Jin,Z. and Yen,Y. (2003) Wild-type p53 regulates human ribonucleotide reductase by protein–protein interaction with p53R2 as well as hRRM2 subunits. Cancer Res., 63, 980–986. [PubMed] [Google Scholar]
- 55.Garcia V., Bruchet,H., Camescasse,D., Granier,F., Bouchez,D. and Tissier,A. (2003) AtATM is essential for meiosis and the somatic response to DNA damage in plants. Plant Cell, 15, 119–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.The Arabidopsis Genome Initiative (2000) Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature, 408, 796–815. [DOI] [PubMed] [Google Scholar]
- 57.Regad F., Herve,C., Marinx,O., Bergounioux,C., Tremousaygue,D. and Lescure,B. (1995) The tef1 box, a ubiquitous cis-acting element involved in the activation of plant genes that are highly expressed in cycling cells. Mol. Gen. Genet., 248, 703–711. [DOI] [PubMed] [Google Scholar]