Significance
Synapses between neurons are the primary means by which information flows through the nervous system. The molecular and functional complexity of synapses has been appreciated for some time, but the mechanisms by which synapses assemble remain unclear. We previously identified a conserved pair of signaling proteins, SAD kinases, required for two early events in the formation of axons by developing neurons. Here, we demonstrate that this same pair of signaling molecules acts in a later step to promote the maturation of axonal segments into presynaptic nerve terminals. Thus, SAD kinases may serve to coordinate multiple aspects of neuronal development.
Keywords: neuromuscular junction, sympathetic ganglion, spinal cord, Ia synapse, Calyx of Held
Abstract
Axons develop in a series of steps, beginning with specification, outgrowth, and arborization, and terminating with formation and maturation of presynaptic specializations. We found previously that the SAD-A and SAD-B kinases are required for axon specification and arborization in subsets of mouse neurons. Here, we show that following these steps, SAD kinases become localized to synaptic sites and are required within presynaptic cells for structural and functional maturation of synapses in both peripheral and central nervous systems. Deleting SADs from sensory neurons can perturb either axonal arborization or nerve terminal maturation, depending on the stage of deletion. Thus, a single pair of kinases plays multiple, sequential roles in axonal differentiation.
Axons differentiate in a prolonged series of steps. Initially, one of several equivalent neurites is specified as axonal (1). The axon then grows, often over long distances, to reach target regions. Within the targets, axons branch to form complex arbors that contact postsynaptic cells (2). Some contacts then differentiate into nerve terminals that contain active zones, calcium channels, and concentrations of synaptic vesicles and mitochondria (3). These components are organized in a precise manner to facilitate rapid release of neurotransmitter in response to entry of calcium through voltage-gated channels (4).
Formation of the presynaptic apparatus itself occurs in several stages. Some components are transported together in packets from cell bodies to synaptic sites (5, 6). These packets are then combined with other components to form transmitter release sites (7). Vesicles and mitochondria accumulate in juxtaposition to these sites, perhaps trapped by cytoskeletal elements anchored to active zones (8, 9). Synapses then mature functionally, adding components, exchanging embryonic for adult isoforms, and acquiring complex topologies (10, 11). During this period, many synapses grow, the number of release sites increases, and neurotransmission becomes more synchronized and reliable.
Signals from postsynaptic and neighboring glial cells organize formation and maturation of nerve terminals, ensuring precise matching of presynaptic and postsynaptic structures. Numerous synaptic organizers that initiate this process have been identified, including soluble, membrane-anchored, and extracellular matrix molecules (12–15). In contrast, little is known about the signal transduction components through which the organizers act. Promising candidates include the Ser/Thr kinases SAD-A and SAD-B, mammalian orthologes of Caenorhabditis elegans SAD-1 (16–18). Sad-1 was identified in a screen for mutations that affected vesicle clustering at active zones (16, 19). We found previously that mouse SAD-A and SAD-B play essential roles in two earlier phases of axonal differentiation, axonal specification and formation of terminal arbors (17, 18, 20). We show here that they are also required for maturation of nerve terminals at motor and autonomic synapses in the mouse peripheral nervous system and at central synapses in the brain and spinal cord. By playing discrete, sequential roles in a single cell type, SAD kinases can coordinate multiple phases of axonal development.
Results
SAD Kinases Regulate Synapse Maturation at the Neuromuscular Junction.
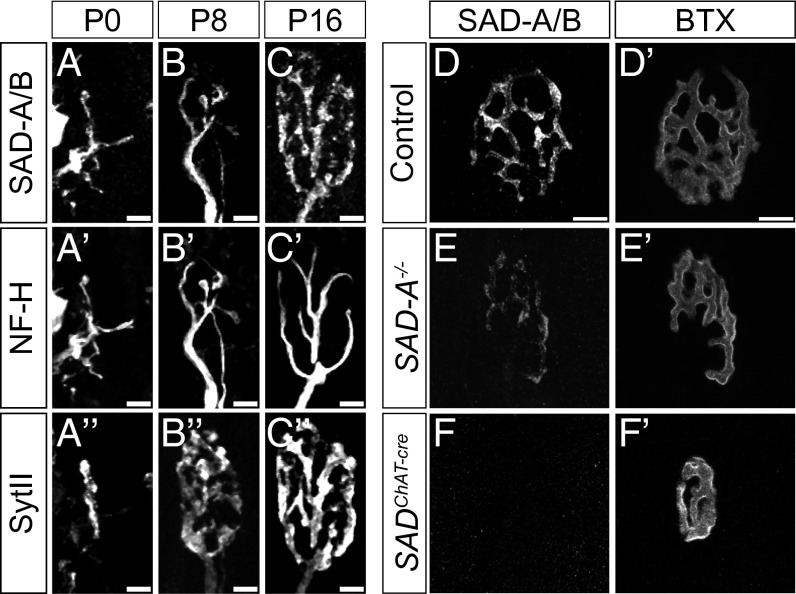
To begin this study, we analyzed the skeletal neuromuscular junction (NMJ), a large synapse that undergoes a stereotyped series of morphological changes during the first two postnatal weeks (21, 22). Immunostaining with an antibody that recognizes both SAD-A and SAD-B revealed that SADs are present throughout the axons of peripheral nerves at postnatal day (P)0 and P8 (Fig. 1 A–B′′). During the second postnatal week, the localization of SAD-A/B shifted, leading to a concentration in motor nerve terminals (labeled with antibodies to Synaptotagmin II [SytII], a component of synaptic vesicles; Fig. 1C and ref. 23). In adults, SAD-A/B protein was largely localized to nerve terminals (Fig. 1D). SAD-A/B protein also underwent a shift in distribution from axonal tracts to synaptic neuropil in spinal cord and retina (Fig. S1).
Fig. 1.
SAD kinases localize to synaptic sites during development. Sections of splenius rectus muscle were stained with antibodies to SAD-A/B (A–F) plus anti-neurofilament heavy subunit (NF-H, A′–C′) and anti-synaptotagmin II (SytII, A′′–C′′) and/or BTX (D′–F′). SADs are redistributed from axons (labeled with anti–NF-H) to endplates (labeled with anti-SytII) postnatally. SAD immunoreactivity at NMJs is decreased in SAD-A−/− animals and is completely lost in SADChAT-cre animals at P35 (D–F), showing that motor nerve terminals contain both SAD-A and SAD-B. (Scale bars: A–C′′, 5 µm; D and D′, 10 µm.)
Because antibodies specific for either SAD-A or SAD-B stain tissue poorly, we used SAD-A and SAD-B mutants to determine which SAD proteins are present in motor nerve terminals. Levels of immunoreactivity were reduced in SAD-A mutant mice and abolished in mice lacking both SAD-A and SAD-B (Fig. 1 D–F), indicating that both proteins are present at NMJs. Consistent with this observation, we detected no synaptic defects in mice lacking either SAD-A or SAD-B.
Because SAD-A;SAD-B double mutants (denoted, SAD-A/B−/−) die perinatally (17), we used a conditional SAD-A floxed allele (18) in conjunction with the SAD-B null allele to delete all copies of SADs in motor neurons. We used two Cre drivers: Isl1-cre (24) and ChAT-Cre (25) (Fig. S2A; denoted SADIsl1-cre and SADChAT-cre, respectively). SADs are deleted from motor neurons by embryonic day (E)13.5 and E15.5 in SADIsl1-cre and SADChAT-cre mice, respectively (18).
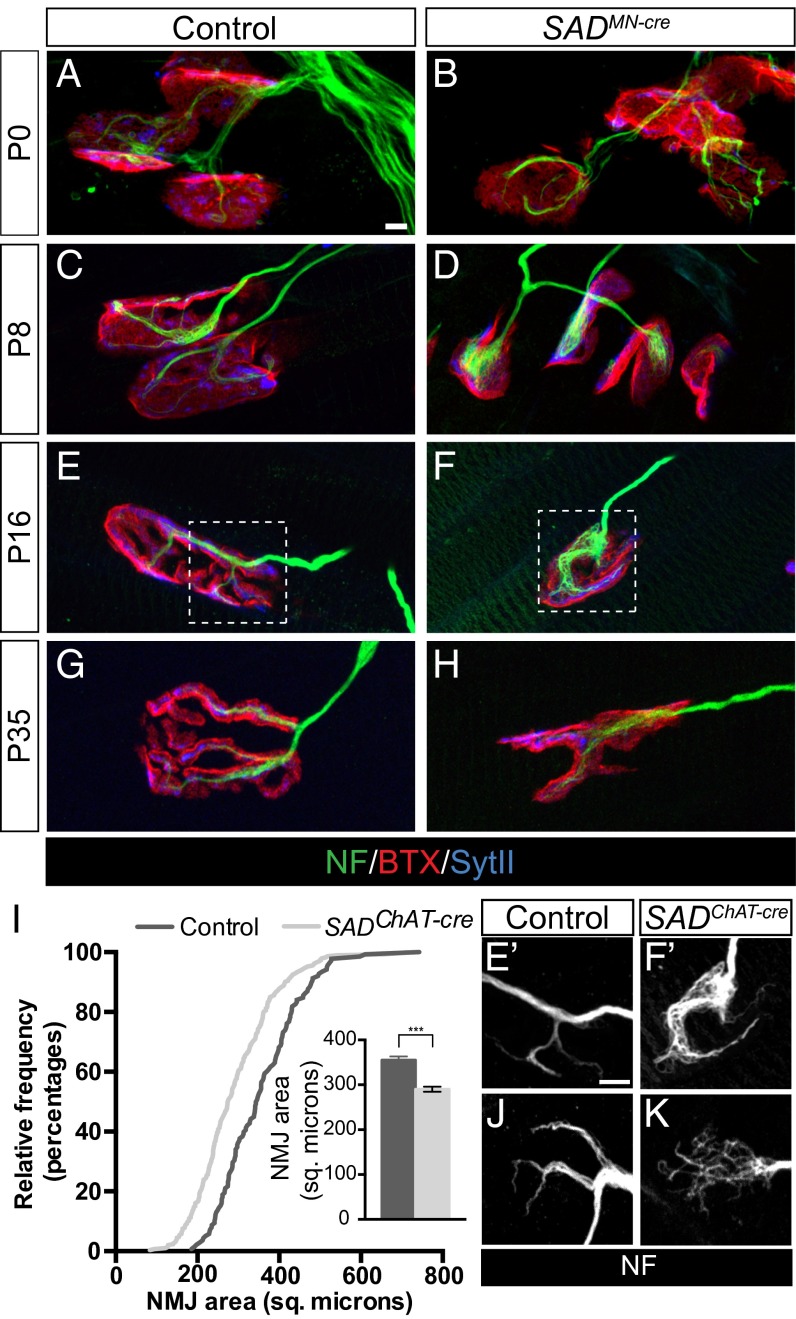
NMJs in the neonatal diaphragm muscle of control animals are plaque-shaped structures in which two or more neurofilament-positive motor axons contact an acetylcholine receptor (AChR)-rich aggregate on the postsynaptic membrane (21). Subsequently, the AChR clusters enlarge and acquire a complex, perforated topology that eventually resolves into a set of finger-like branches; in parallel, the overlying axons branch to maintain occupancy of the postsynaptic site. In both SAD-A/B−/− and SADIsl1-cre mutants, initial stages of synapse formation were normal as shown by apposition of structures stained for SytII and α-bungarotoxin (BTX), which binds to AChRs (Fig. 2 A–D). Because most SADIsl1-cre mutants died within 24 h after birth, we examined NMJ maturation in SADChAT-cre mutant animals, which survived until adulthood but exhibited mild postural tremors. In SADChAT-cre mutants, NMJ area was significantly reduced at P16, as was the overall topological complexity of the synapse (Fig. 2 E–I). We observed similar defects in the splenius rectus muscle of the upper back (Fig. S3 A and B). SADs are therefore not required for formation of NMJs, but rather are required presynaptically for synaptic maturation.
Fig. 2.
SAD kinases are required for the maturation of the neuromuscular junction. (A–H) Immunohistochemical analysis of diaphragm muscle of control and SADMN-cre (SADIsl1-cre in B and D; SADChAT-cre in F and H) animals reveals impaired growth and topological maturation of SAD-deficient NMJs. (I) Cumulative frequency histogram and graph (Inset) of endplate size in control and SADChAT-cre animals at P16 showing reduced NMJ size when SADs are deleted presynaptically (graph shows mean ± SEM of endplate size, n > 100, ***unpaired t test: P < 0.001). (E′, F′, J, and K) Endplates stained with anti-neurofilaments to show disorganization of neurofilament structures at SADChAT-cre NMJs. E′ and F′ show boxed areas from E and F at higher magnification. (Scale bars: 5 µm in A for A–H and E′ for E’, F′, J, and K.)
One prominent feature of SADChAT-cre mutant NMJs was disorganization of neurofilaments. Rather than containing tapered, finely branched neurofilament bundles, SADChAT-cre terminals showed disorganized bundles that covered a large portion of the endplate (Fig. 2 E–K). Ultrastructural analysis of NMJs from SADChAT-cre animals showed the presence of filamentous material adjacent to the presynaptic membrane, consistent with light microscopic analysis (Fig. S3 D and E). In many cases, the abnormally positioned filaments appeared to displace synaptic vesicles from normal-appearing active zones adjacent to postsynaptic folds (Fig. S3 C–I). No defects were observed in the postsynaptic apparatus by either light or electron microscopy, except the simplified topology noted above.
We also asked whether SADs are required for later steps in synaptic maturation or maintenance. A well-characterized aspect of synaptic maturation is elimination of supernumerary nerve terminals accompanied by growth of those that remain (26). We did not observe an increase in the number of multiply innervated NMJs at P16 in SADChAT-cre animals relative to controls. Further, we did not observe an increased severity of defects or an increased number of denervated NMJs in 3- to 4-mo-old SADChAT-cre animals (Fig. S3 J and K), indicating that SADs are not required for synaptic maintenance.
SADs Regulate Synaptic Maturation of Neuron-Neuron Synapses in an Autonomic Ganglion.
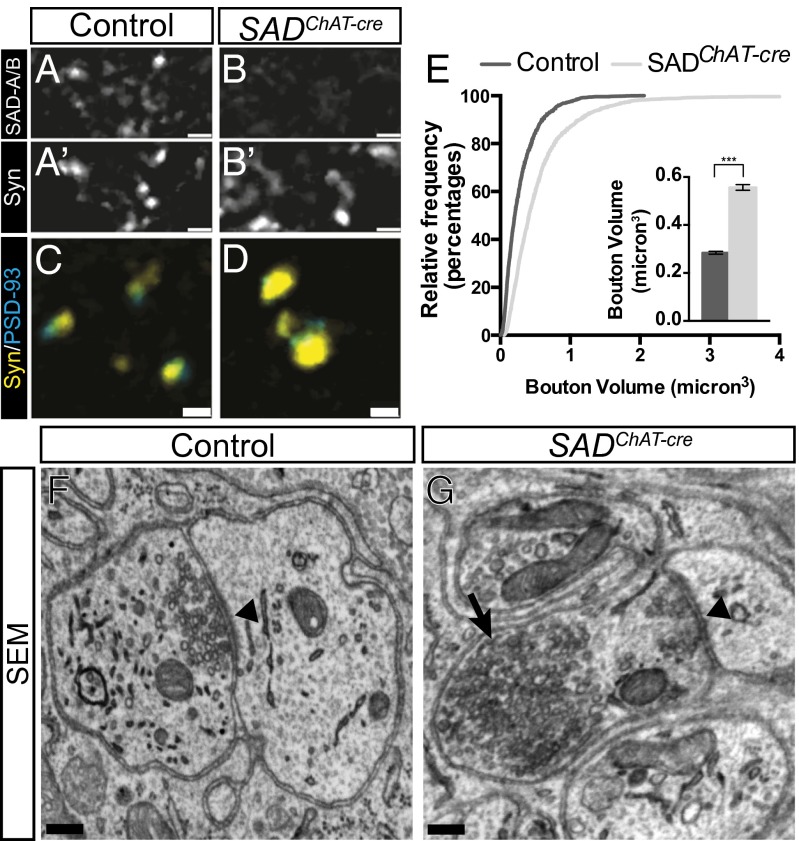
To ask whether neuron-neuron synapses require SADs for their development, we analyzed the cholinergic synapses made by autonomic preganglionic axons on adrenergic sympathetic neurons in the superior cervical ganglion (SCG). We used the ChAT-cre line to examine the role of SADs in the development of this neuron-neuron synapse as it acts in preganglionic motor neurons, but not their postsynaptic targets in the SCG (Fig. S2B). We used antibodies to synaptophysin (a synaptic vesicle protein) to mark nerve terminals and antibodies to the scaffolding protein PSD-93 to mark postsynaptic sites on ganglion cells (27). SAD-A/B colocalized with synaptophysin in wild-type ganglia, showing that these kinases are concentrated in nerve terminals in the SCG (Fig. 3 A and B).
Fig. 3.
Maturation of autonomic ganglionic synapses require SADs. (A–B’) SADs colocalize with presynaptic synaptophysin of control P16 SCG, but reactivity is lost in SADChAT-cre animals. (C and D) Enlargement of synaptophysin boutons in SADChAT-cre SCG relative to control. (E) Cumulative frequency histogram and graph (Inset) of bouton size in control and SADChAT-cre animals, measured from images such as those in C and D (graph shows mean ± SEM of bouton volume, n > 2,000, ***unpaired t test: P < 0.001). (F and G) Electron microscopy shows vesicles clustered at an active zone (arrowhead) in a control synapse (F), but large vesicle aggregates (arrow) localized away from the active zone in a mutant synapse. (Scale bars: A–B′, 4 µm; C and D, 2 µm; F and G, 200 nm.)
The number of synaptophysin- and PSD-93–rich punctae did not differ detectably between control and SADChAT-cre animals at any age examined (P8–adult), and the apposition of presynaptic to postsynaptic specializations was maintained in mutants (Fig. 3 C and D). However, at P8 and later, presynaptic punctae in SADChAT-cre mutants exhibited a marked increase in size and intensity of staining (Fig. 3 C–E). Similar results were obtained by using two other markers of nerve terminals, synaptotagmin II (SytII) and vesiular acetylcholine transporter (VaChT) (Fig. S4 A–D). Thus, preganglionic nerve terminals lacking SAD kinases mature aberrantly.
We used electron microscopy to analyze synaptic ultrastructure in SADChAT-cre ganglia. In controls, synapses were characterized by electron-dense presynaptic and postsynaptic membranes separated by a synaptic cleft, and by clusters of synaptic vesicles in close proximity to the presynaptic membrane (Fig. 3F and Fig. S3 E–G; ref. 28). In synapses of SADChAT-cre mice, in contrast, we observed synaptic vesicle aggregates that were several hundred nanometers in diameter and were displaced by >200 nm from a visible active zone (Fig. 3G and Fig. S4 H–J). Abnormalities were apparent in 24/41 terminals from three mutants, but in no control synapses (n = 18). This defect in the organization of vesicles at the nerve terminal is consistent with results from light microscopic analysis, and with the displacement of vesicles from release sites described above for NMJs.
SADs Regulate Maturation of the Calyx of Held.
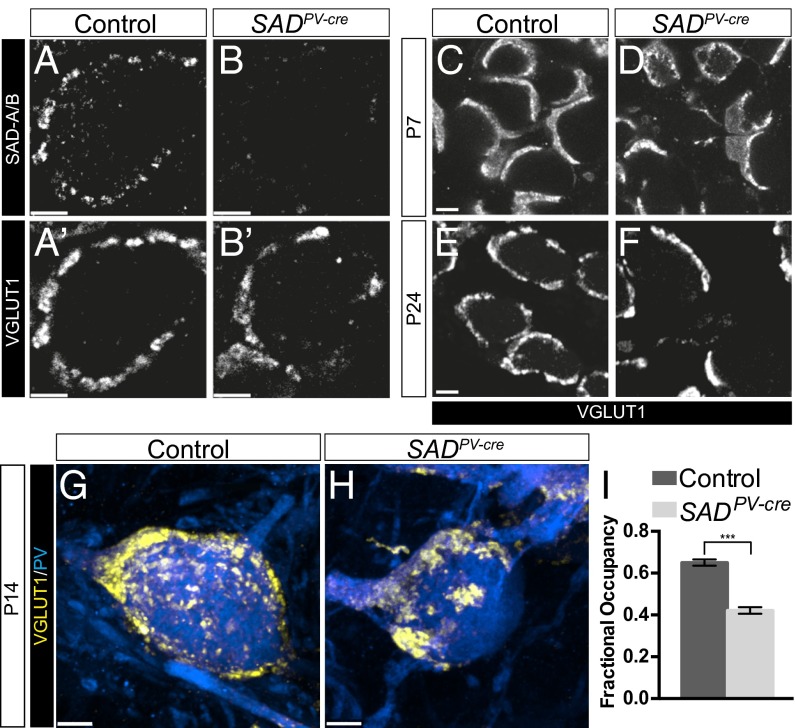
Does the role of SAD kinases in synapse development extend to the central nervous system? The small size and high density of most types of brain synapses hamper light microscopic analysis. An exception is the Calyx of Held, a large glutamatergic synapse in the median nucleus of the trapezoid body (MNTB) of the auditory brainstem. These synapses undergo dramatic growth during the first two postnatal weeks, during which nerve terminals envelope the MNTB principal cells to form calyces that contain many transmitter release sites (29, 30). SAD-A/B colocalized with the vesicular glutamate transporter VGLUT1 and SytII, both of which selectively mark calyces in the MNTB (ref. 31; Fig. 4 A and B). The calcium binding protein parvalbumin (PV) is expressed by the globular bushy cells of the ventral cochlear nucleus, which form calyces in the MNTB (32), so we used a PV-cre line to delete SADs from these cells. Cre activity was evident by P1 in presynaptic axons and was also expressed by some MNTB neurons by P6 (Fig. S2C).
Fig. 4.
SADs regulate the maturation of the Calyx of Held. (A–B′) SAD-A/B immunohistochemistry showing colocalization with the presynaptic marker VGLUT1, which is lost in calyces of SADPV-cre animals. (C–F) VGLUT1 immunohistochemistry of calyces at different ages showing defects in nerve terminal occupancy around MNTB cells. (G and H) En face view of P14 calyces showing defects in presynaptic growth and vesicle organization in SAD-deficient calyces. (I) Quantification of calyx occupancy in control and SADPV-cre animals at P24. Graph shows mean ± SEM, n = 40 calyces, ***unpaired t test: P < 0.001. (Scale bars: A–F, 5 µm; G and H, 4 µm.)
By P24, fully mature calyces in controls had undergone substantial growth as demonstrated by the presence of VGLUT1- and SytII-rich structures covering large portions of the surface of MNTB soma (Fig. 4 C, E, and G). In contrast, a substantial fraction of calyces in SADPV-cre animals showed reduced occupancy of the postsynaptic cell (Fig. 4 D, F, H, and I). This phenotype persisted at later stages, suggesting that calyx maturation was defective rather than merely delayed in SADPV-cre animals.
SADs Play Sequential Roles in Formation of Sensory Synapses on Motor Neurons.
We showed recently that SAD kinases are essential for terminal axon arborization by type Ia proprioceptive sensory neurons (IaPSNs) in the ventral spinal cord (ref. 18; Fig. S5A). These axons fail to contact and form synapses on motor neurons in SADIsl1-cre mice, in which SADs are deleted at ∼E13.5 (18). To ask whether SADs play a distinct, later role in formation of synapses by IaPSNs, we used the PV-cre line that acts selectively in proprioceptive sensory neurons in DRG (33). Using reporter lines, we found that PV-cre activity is only sporadically detectable at E15.5–E16 in DRG neurons, after IaPSNs have grown to the ventral horn (Figs. S2D and S5B). By P0, however, SAD protein is undetectable in the somata of PV+ DRG neurons of SADPV-cre mutants (Fig. S5 C and D). Consistent with these results, IaPSN projections were truncated in SADIsl1-cre mutants but IaPSN axons formed arbors in the ventral horn of SADPV-cre mutants (Fig. S6).
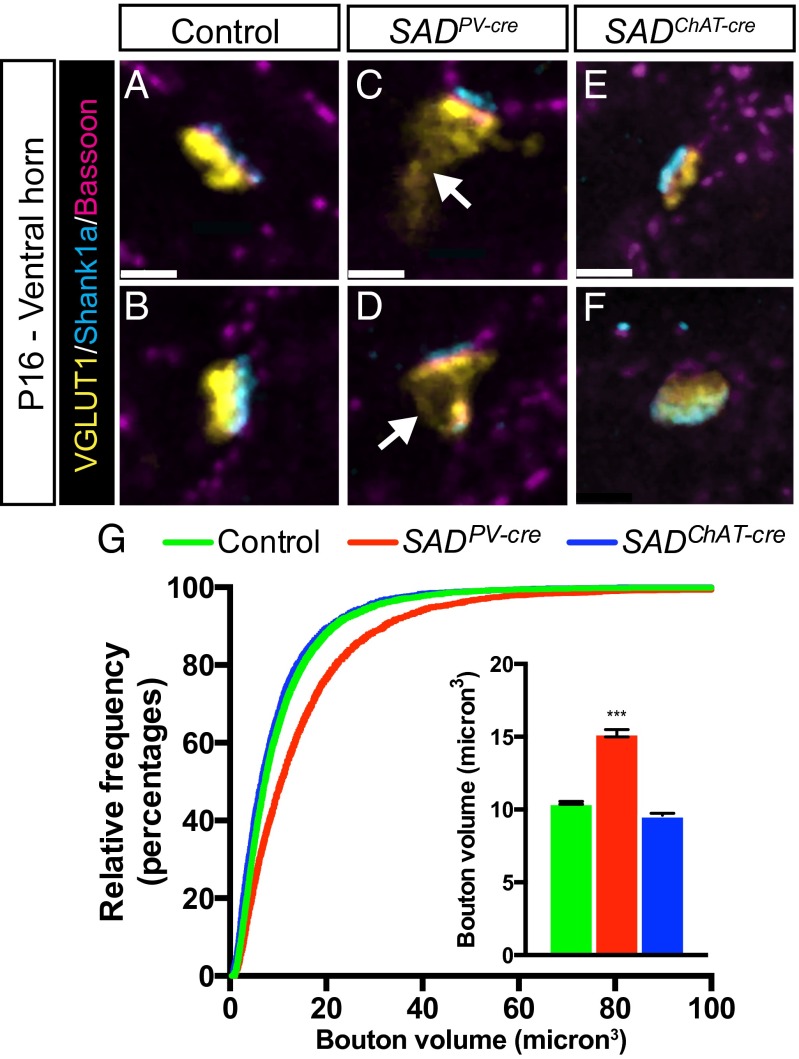
We analyzed Ia-motor neuron synapses by using VGLUT1 and Shank1a as selective presynaptic and postsynaptic markers, respectively (34). At P14–P16, VGLUT1-labeled Ia boutons in SADPV-cre spinal cord were significantly larger than those in controls (Fig. 5 A–D and G). As was seen in the cholinergic synapses in the SCG, single VGLUT1 boutons were often apposed to multiple Shank1a clusters. Expansion of VGLUT1 immunoreactivity in preterminal axon segments was observed at many synapses, and the average VGLUT1 bouton volume was increased by nearly 50% in SADPV-cre animals (Fig. 5G). Thus, SADs play multiple, sequential roles in the assembly of Ia-MN synapses.
Fig. 5.
Presynaptic SADs regulate IaPSN-MN synaptic maturation. (A–F) In control Ia boutons (A and B), the vesicle marker VGLUT1 is closely apposed to the active zone marker Bassoon and the postsynaptic marker Shank1a. VLUT1 immunoreactivity extends into preterminal regions when SADs are deleted presynaptically from sensory neurons (arrows in C and D) but not postsynaptically from motor neurons (E and F). (G) Cumulative frequency histogram and bar graph showing enlargement of VGLUT1 bouton volume with presynaptic but not postsynaptic SAD deletion (graph shows mean ± SEM, n = 4 animals, n > 5,000 boutons, ***unpaired t test: P < 0.001 compared with control and SADChatCre). (Scale bars: 2 µm.)
SAD Kinases Are Required Presynaptically but Not Postsynaptically for Nerve Terminal Differentiation.
In studies described so far, we deleted SADs from neurons that form nerve terminals in muscle, autonomic ganglia, and spinal cord and from both presynaptic and postsynaptic elements at the Calyx of Held. We next asked whether loss of SAD kinases from postsynaptic cells alone affects the maturation of nerve terminals that form on them. To this end, we examined Ia-motor neuron synapses in SADChAT-cre mice, in which SADs were deleted from motor neurons but not IaPSNs (Fig. S2E). Postsynaptic deletion of SADs in motor neurons had no detectable effect on the size and distribution of IaPSN (VGLUT1+) boutons or on the clustering of Shank1a at postsynaptic sites (Fig. 5 E–G). We also examined inhibitory terminals on motor neurons in SADChAT-cre animals. Inhibitory synapses labeled by antibodies to the vesicular amino acid transporter and glycine receptors were unaffected by the loss of SADs in postsynaptic partners (Fig. S7). Thus, the requirement for SADs in nerve terminal development is restricted to the presynaptic compartment at least in some types of synapses.
SAD Kinases Are Required for Functional Maturation of Synapses.
Loss of SAD from motor nerve terminals and the Calyx of Held led to decreased size of these large synapses, which is likely to result in decreased capacity for neurotransmitter release. In contrast, autonomic and sensory nerve terminals were enlarged in SAD mutants, raising the question of whether SADs could enhance their function. We used electrophysiological methods to address this issue.
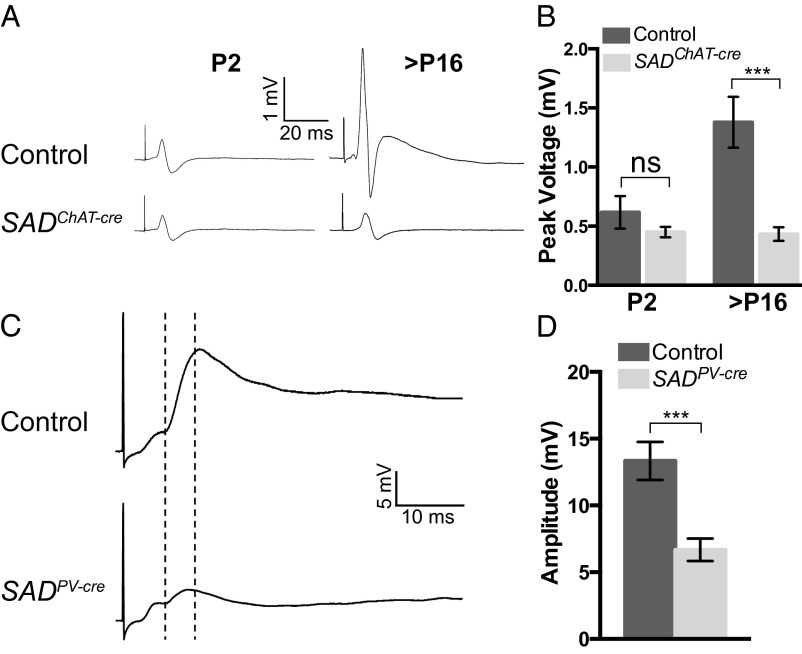
For SCG, we recorded compound action potentials from the postganglionic nerve upon evoked stimulation of presynaptic fibers. This method is widely used as an indirect measure of ganglionic transmission (35). At P2, the size of compound action potentials from control and SADChAT-cre animals were indistinguishable (Fig. 6 A and B). Over the following 2 wk, the size of the compound action potentials increased ∼threefold in controls. In SADChAT-cre mutant ganglia, in contrast, the responses in older ganglia were similar to those seen at P2, demonstrating that ganglionic transmission did not mature, consistent with a role for SAD kinases in promoting synaptic maturation (Fig. 6 A and B).
Fig. 6.
SADs mediate functional maturation of peripheral and central synapses. (A) Compound action potentials (CAP) from SADChAT-cre and age-matched control SCG. (B) Quantification of peak CAP voltage showing that postnatal enhancement in ganglionic transmission fails to occur in mutants. Graph shows mean ± SEM of CAP peak height. The difference between control (n = 6 ganglia) and SADChAT-cre (n = 5 ganglia) at P2 is not significant (unpaired t test, P = 0.32), but the difference at P16 (control n = 13; SADChAT-cre n = 11) is significant (unpaired t test P = 0.0007). ns, not significant. (C) Sample traces of homonymous EPSPs from quadriceps motor neurons in SADPV-cre mice at P5. The dashed lines indicate the latency to the onset and the amplitude of the monosynaptic responses. (D) Homonymous EPSP amplitude is reduced in SADPV-cre animals relative to controls (graph shows EPSP mean ± SEM, Control, n = 14; SADPV-cre, n = 13, unpaired t test: P = 0.0006).
To analyze IaPSN-motor neuron synapses, we first recorded from motor axons in L4 ventral roots following stimulation of L4 dorsal roots. Suprathreshold dorsal root stimulation induced a strong monosynaptic (short latency) response in motor axons of control animals at P0 (Fig. S8A). In SADIsl1-cre mutants, L4 dorsal root stimulation elicited only a weak monosynaptic response and failed to induce action potential firing in motor neurons in 4/4 animals (Fig. S8 A and B). In contrast, stimulation of L4 dorsal roots in SADPV-cre animals elicited a robust monosynaptic response with normal synaptic latency (Fig. S8 C–F). Thus, consistent with anatomical studies (Fig. 5), later deletion of SADs in IaPSNs permits formation of functional connections with motor neurons.
We then recorded intracellularly from motor neurons of P5–P7 SADPV-cre animals while stimulating Ia sensory axons in individual muscle nerves to ask whether loss of SADs from sensory neurons perturbs maturation of the Ia synapse. The latency of monosynaptic responses did not differ detectably between control and SADPV-cre mutants (Fig. S8G). However, we observed an ∼50% reduction in the average excitatory postsynaptic potential (EPSP) amplitude in SADPV-cre animals relative to controls (Fig. 6 C and D). Thus, in both autonomic preganglionic and Ia nerve terminals, loss of SADs leads to enlarged size but decreased function.
Finally, we asked whether impaired maturation of synapses in SAD mutants resulted from a defect in synaptic specificity, which could lead to formation of inappropriate synapses. In wild-type mice, IaPSNs arising from a particular muscle selectively synapse on “homonymous” motor neurons, which innervate the same muscle (36). Accordingly, stimulation of quadriceps or obturator muscle nerves elicited monosynaptic EPSPs in identified motor neurons innervating the same muscle, but not that of the nonhomonymous muscle in control mice. Specificity was maintained in SADPV-cre mutants (Fig. S8H), indicating that SADs are not required for establishing appropriate synaptic connections in this circuit.
Discussion
Our main result is that SAD kinases are required for the structural and functional maturation of synapses. We analyzed four synaptic types in detail, including ones in the peripheral motor system (the NMJ), the peripheral autonomic nervous system (synapses in the SCG), the spinal cord (synapses of Ia sensory afferents on motor neurons) and the brain (the Calyx of Held). In all cases, synapses formed in the absence of SADs, but the maturation of nerve terminals was compromised. We used immunohistochemical methods to document defects in all four synaptic types and complemented this work with ultrastructural analysis of the NMJ and SCG synapses. We also used electrophysiological methods to demonstrate functional defects in Ia-motor neuron and SCG synapses. Although our survey is incomplete, we conclude that SADs promote maturation of many, and perhaps all, types of synapses in mice. Favoring this idea, mutations of SAD orthologs in C. elegans and Drosophila also lead to presynaptic defects (16, 37).
Although presynaptic maturation was impaired in all four types of synapses examined, the defects varied among synaptic types. SADs are required for synaptic growth in motor nerve terminals and the Calyx of Held, two very large synapses with multiple release sites. However, at SCG and Ia-MN synapses, the defects involved distension of nerve terminals with, at least for SCG, aberrant vesicle aggregation. What processes might SADs control to promote growth of nerve terminals and localization of synaptic vesicles? SAD-deficient NMJs display disorganized neurofilament structures that do not branch appreciably, and SAD-deficient DRG neurons fail to form terminal axon branches in the central nervous system (18). SADs may therefore influence aspects of cytoskeletal dynamics as axons form branches either in the terminal field or at presynaptic sites where large areas of contact are made onto postsynaptic targets (NMJ and Calyx). Interestingly, SADs phosphorylate both γ-tubulin and the microtubule-associated protein tau, which regulates microtubule dynamics (17, 20, 38).
In Ia-MN and SCG nerve terminals, SADs appear to influence localization of synaptic vesicles at active zones. This effect may reflect the phosphorylation of another set of substrates by SADs. Plausible candidates include proteins involved in positioning vesicles at nerve terminals such as myosins or synapsins (39, 40). SAD-B also phosphorylates RIM proteins, which are components of a cytomatrix that is closely associated with active zones (41). Our ultrastructural analysis did not reveal obvious defects in the assembly of active zones at the NMJ or in SCG in SAD mutants. We therefore propose that SADs act as links between synaptic vesicles and presynaptic cytomatrix coordinating the assembly of nerve terminal structure as synapses mature.
In previous studies, we demonstrated roles of SAD kinases in the initial specification of axons and in the formation of their terminal arbors (17, 18, 20). Taken together with results presented here, this work establishes SADs as critical organizers of multiple steps in axonal development. However, roles of SADs are strikingly selective in at least two respects. First, roles of SADs appear to differ among neuronal types. SADs are required for axon specification and polarization of projection neurons in the cerebral cortex and hippocampus. In contrast, SADs are dispensable for polarization of multiple other neuronal types, including motor neurons, sensory neurons, retinal ganglion cells, photoreceptors, and retinal bipolar interneurons, all of which are strikingly polarized. Likewise, formation of terminal arbors is SAD-dependent in NT-3–responsive sensory neurons, whereas axons of motor neurons and NGF-responsive sensory neurons show no obvious arborization defects in the absence of SADs (18). Second, at least in the neurons examined to date, SADs are not involved in all aspects of axonal development. As noted in the Introduction, axons develop in a series of steps, including specification, elongation, formation of terminal arbors, formation of presynaptic specializations, and synaptic maturation (42). SADs are required in at least some neurons for axonal specification, arborization, and synaptic maintenance but not for axonal elongation or the initial steps in synapse formation.
These results raise two questions: First, how can SADs promote diverse steps in axonal morphogenesis, at least sometimes (for example, in Ia-PSN sensory neurons) in a single neuronal type? Second, why are effects of SADs confined to some neuronal types and some developmental steps? The likely answer to the first question is that SADs lie at the hub of a complex signaling pathway, with distinct inputs and outputs depending on the cell type and developmental stage. For example, LKB1 acts upstream of SADs in axonal specification, but not in axonal arborization (18, 20); in the latter case, and likely in synaptic maturation, other kinases are likely to activate SADs in responses to appropriate signals. Likewise, C. elegans SAD-1 acts through neurabins to regulate polarity, whereas its effects on synaptic differentiation are neurabin-independent (43). For the second question, one likely answer is that SADs act redundantly with other kinases. SADs are 2 members of a 12-member kinase subfamily, most of which are expressed in neurons. The MARK and NUAK members of the AMPK subfamily of kinases also influence neuronal polarity and axon branching, respectively (44, 45), and thus may act in parallel with SADs in some circumstances. NUAKs also influence presynaptic mitochondrial dynamics during synapse development (45). Although our analysis did not reveal changes in the distribution of mitochondria at SAD-deficient nerve terminals, we speculate that SADs and NUAKs may interact to regulate presynaptic development.
Finally, it will be interesting to ask whether SADs play even more roles in axons. One attractive possibility is that SADs are involved in synaptic function or plasticity (46). We have observed that, in response to depolarization, SADs undergo dramatic dephosphorylation of inhibitory sites, a regulatory control mechanism that facilitates SAD kinase activation (18). Thus, SADs may also alter synaptic structure or function in mature nerve terminals in response to activity-dependent signals.
Materials and Methods
Animals were used in accordance with protocols approved by Institutional Animal Use and Care Committee at Harvard University in accordance with National Institutes of Health guidelines. Mice carrying the SAD-A and SAD-B null alleles and the SAD-A floxed allele have been described (17, 18). SAD-Afl/+; SAD-B−/−; x-cre and SAD-Afl/fl; SAD-B−/− animals showed phenotypes similar to wild-type animals and were used as littermate controls. The Isl1-cre line (Isl1tm1(cre)Tmj; ref. 24) was provided by Tom Jessell, Columbia University, New York. The Chat-cre [Chattm1(cre)Lowl; ref. 25] and PV-cre (Pvalbtm1(cre)Arbr; ref. 33) lines were obtained from Jackson Laboratories. The RC::Epe, [Gt(ROSA)26Sortm6Dym provided by Susan Dymecki, Harvard Medical School, Boston; ref. 47] and Ai14 (Jackson Labs; ref. 48) cre reporter lines were used to determine patterns of cre expression. Animals were maintained on a mixed C57B6J/CD-1 genetic background.
Histological, ultrastructural, and electrophysiological methods are described in SI Materials and Methods.
Supplementary Material
Acknowledgments
We thank Susan Dymecki and Tom Jessell for mouse strains used in this study; Richard Schalek and Deborah Pelusi for technical assistance; and members of the J.R.S. laboratory for helpful suggestions. This work was supported by National Institutes of Health Grants R01 NS029169 and R21 NS076880. B.N.L. was supported by Damon Runyon Cancer Research Foundation Fellowship DRG-1914-06.
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1321990111/-/DCSupplemental.
References
- 1.Barnes AP, Polleux F. Establishment of axon-dendrite polarity in developing neurons. Annu Rev Neurosci. 2009;32:347–381. doi: 10.1146/annurev.neuro.31.060407.125536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gibson DA, Ma L. Developmental regulation of axon branching in the vertebrate nervous system. Development. 2011;138(2):183–195. doi: 10.1242/dev.046441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jin Y, Garner CC. Molecular mechanisms of presynaptic differentiation. Annu Rev Cell Dev Biol. 2008;24:237–262. doi: 10.1146/annurev.cellbio.23.090506.123417. [DOI] [PubMed] [Google Scholar]
- 4.Südhof TC. The presynaptic active zone. Neuron. 2012;75(1):11–25. doi: 10.1016/j.neuron.2012.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ahmari SE, Buchanan J, Smith SJ. Assembly of presynaptic active zones from cytoplasmic transport packets. Nat Neurosci. 2000;3(5):445–451. doi: 10.1038/74814. [DOI] [PubMed] [Google Scholar]
- 6.Wu YE, Huo L, Maeder CI, Feng W, Shen K. The balance between capture and dissociation of presynaptic proteins controls the spatial distribution of synapses. Neuron. 2013;78(6):994–1011. doi: 10.1016/j.neuron.2013.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Patel MR, et al. Hierarchical assembly of presynaptic components in defined C. elegans synapses. Nat Neurosci. 2006;9(12):1488–1498. doi: 10.1038/nn1806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Goldstein AYN, Wang X, Schwarz TL. Axonal transport and the delivery of pre-synaptic components. Curr Opin Neurobiol. 2008;18(5):495–503. doi: 10.1016/j.conb.2008.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gundelfinger ED, Fejtova A. Molecular organization and plasticity of the cytomatrix at the active zone. Curr Opin Neurobiol. 2012;22(3):423–430. doi: 10.1016/j.conb.2011.10.005. [DOI] [PubMed] [Google Scholar]
- 10.McMahon SA, Díaz E. Mechanisms of excitatory synapse maturation by trans-synaptic organizing complexes. Curr Opin Neurobiol. 2011;21(2):221–227. doi: 10.1016/j.conb.2010.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shi L, Fu AKY, Ip NY. Molecular mechanisms underlying maturation and maintenance of the vertebrate neuromuscular junction. Trends Neurosci. 2012;35(7):441–453. doi: 10.1016/j.tins.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 12.Hall AC, Lucas FR, Salinas PC. Axonal remodeling and synaptic differentiation in the cerebellum is regulated by WNT-7a signaling. Cell. 2000;100(5):525–535. doi: 10.1016/s0092-8674(00)80689-3. [DOI] [PubMed] [Google Scholar]
- 13.Umemori H, Linhoff MW, Ornitz DM, Sanes JR. FGF22 and its close relatives are presynaptic organizing molecules in the mammalian brain. Cell. 2004;118(2):257–270. doi: 10.1016/j.cell.2004.06.025. [DOI] [PubMed] [Google Scholar]
- 14.Linhoff MW, et al. An unbiased expression screen for synaptogenic proteins identifies the LRRTM protein family as synaptic organizers. Neuron. 2009;61(5):734–749. doi: 10.1016/j.neuron.2009.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Siddiqui TJ, Craig AM. Synaptic organizing complexes. Curr Opin Neurobiol. 2011;21(1):132–143. doi: 10.1016/j.conb.2010.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Crump JG, Zhen M, Jin Y, Bargmann CI. The SAD-1 kinase regulates presynaptic vesicle clustering and axon termination. Neuron. 2001;29(1):115–129. doi: 10.1016/s0896-6273(01)00184-2. [DOI] [PubMed] [Google Scholar]
- 17.Kishi M, Pan YA, Crump JG, Sanes JR. Mammalian SAD kinases are required for neuronal polarization. Science. 2005;307(5711):929–932. doi: 10.1126/science.1107403. [DOI] [PubMed] [Google Scholar]
- 18.Lilley BN, Pan YA, Sanes JR. SAD kinases sculpt axonal arbors of sensory neurons through long- and short-term responses to neurotrophin signals. Neuron. 2013;79(1):39–53. doi: 10.1016/j.neuron.2013.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim JSM, et al. A chemical-genetic strategy reveals distinct temporal requirements for SAD-1 kinase in neuronal polarization and synapse formation. Neural Dev. 2008;3:23. doi: 10.1186/1749-8104-3-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Barnes AP, et al. LKB1 and SAD kinases define a pathway required for the polarization of cortical neurons. Cell. 2007;129(3):549–563. doi: 10.1016/j.cell.2007.03.025. [DOI] [PubMed] [Google Scholar]
- 21.Sanes JR, Lichtman JW. Development of the vertebrate neuromuscular junction. Annu Rev Neurosci. 1999;22:389–442. doi: 10.1146/annurev.neuro.22.1.389. [DOI] [PubMed] [Google Scholar]
- 22.Wu H, Xiong WC, Mei L. To build a synapse: Signaling pathways in neuromuscular junction assembly. Development. 2010;137(7):1017–1033. doi: 10.1242/dev.038711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hagiwara A, Harada K, Hida Y, Kitajima I, Ohtsuka T. Distribution of serine/threonine kinase SAD-B in mouse peripheral nerve synapse. Neuroreport. 2011;22(7):319–325. doi: 10.1097/WNR.0b013e328346013c. [DOI] [PubMed] [Google Scholar]
- 24.Srinivas S, et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev Biol. 2001;1:4. doi: 10.1186/1471-213X-1-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rossi J, et al. Melanocortin-4 receptors expressed by cholinergic neurons regulate energy balance and glucose homeostasis. Cell Metab. 2011;13(2):195–204. doi: 10.1016/j.cmet.2011.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tapia JC, et al. Pervasive synaptic branch removal in the mammalian neuromuscular system at birth. Neuron. 2012;74(5):816–829. doi: 10.1016/j.neuron.2012.04.017. [DOI] [PubMed] [Google Scholar]
- 27.Parker MJ, Zhao S, Bredt DS, Sanes JR, Feng G. PSD93 regulates synaptic stability at neuronal cholinergic synapses. J Neurosci. 2004;24(2):378–388. doi: 10.1523/JNEUROSCI.3865-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Krishnaswamy A, Cooper E. An activity-dependent retrograde signal induces the expression of the high-affinity choline transporter in cholinergic neurons. Neuron. 2009;61(2):272–286. doi: 10.1016/j.neuron.2008.11.025. [DOI] [PubMed] [Google Scholar]
- 29.Schneggenburger R, Forsythe ID. The calyx of Held. Cell Tissue Res. 2006;326(2):311–337. doi: 10.1007/s00441-006-0272-7. [DOI] [PubMed] [Google Scholar]
- 30.Borst JGG, Soria van Hoeve J. The calyx of held synapse: From model synapse to auditory relay. Annu Rev Physiol. 2012;74:199–224. doi: 10.1146/annurev-physiol-020911-153236. [DOI] [PubMed] [Google Scholar]
- 31.Fox MA, Sanes JR. Synaptotagmin I and II are present in distinct subsets of central synapses. J Comp Neurol. 2007;503(2):280–296. doi: 10.1002/cne.21381. [DOI] [PubMed] [Google Scholar]
- 32.Solbach S, Celio MR. Ontogeny of the calcium binding protein parvalbumin in the rat nervous system. Anat Embryol (Berl) 1991;184(2):103–124. doi: 10.1007/BF00942742. [DOI] [PubMed] [Google Scholar]
- 33.Hippenmeyer S, et al. A developmental switch in the response of DRG neurons to ETS transcription factor signaling. PLoS Biol. 2005;3(5):e159. doi: 10.1371/journal.pbio.0030159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Betley JN, et al. Stringent specificity in the construction of a GABAergic presynaptic inhibitory circuit. Cell. 2009;139(1):161–174. doi: 10.1016/j.cell.2009.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kuba K, Koketsu K. Synaptic events in sympathetic ganglia. Prog Neurobiol. 1978;11(2):77–169. doi: 10.1016/0301-0082(78)90010-2. [DOI] [PubMed] [Google Scholar]
- 36.Mears SC, Frank E. Formation of specific monosynaptic connections between muscle spindle afferents and motoneurons in the mouse. J Neurosci. 1997;17(9):3128–3135. doi: 10.1523/JNEUROSCI.17-09-03128.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Baas S, et al. Sugar-free frosting, a homolog of SAD kinase, drives neural-specific glycan expression in the Drosophila embryo. Development. 2011;138(3):553–563. doi: 10.1242/dev.055376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Alvarado-Kristensson M, Rodríguez MJ, Silió V, Valpuesta JM, Carrera AC. SADB phosphorylation of gamma-tubulin regulates centrosome duplication. Nat Cell Biol. 2009;11(9):1081–1092. doi: 10.1038/ncb1921. [DOI] [PubMed] [Google Scholar]
- 39.Kneussel M, Wagner W. Myosin motors at neuronal synapses: Drivers of membrane transport and actin dynamics. Nat Rev Neurosci. 2013;14(4):233–247. doi: 10.1038/nrn3445. [DOI] [PubMed] [Google Scholar]
- 40.Chi P, Greengard P, Ryan TA. Synaptic vesicle mobilization is regulated by distinct synapsin I phosphorylation pathways at different frequencies. Neuron. 2003;38(1):69–78. doi: 10.1016/s0896-6273(03)00151-x. [DOI] [PubMed] [Google Scholar]
- 41.Inoue E, et al. SAD: A presynaptic kinase associated with synaptic vesicles and the active zone cytomatrix that regulates neurotransmitter release. Neuron. 2006;50(2):261–275. doi: 10.1016/j.neuron.2006.03.018. [DOI] [PubMed] [Google Scholar]
- 42.Lewis TL, Jr, Courchet J, Polleux F. Cell biology in neuroscience: Cellular and molecular mechanisms underlying axon formation, growth, and branching. J Cell Biol. 2013;202(6):837–848. doi: 10.1083/jcb.201305098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hung W, Hwang C, Po MD, Zhen M. Neuronal polarity is regulated by a direct interaction between a scaffolding protein, Neurabin, and a presynaptic SAD-1 kinase in Caenorhabditis elegans. Development. 2007;134(2):237–249. doi: 10.1242/dev.02725. [DOI] [PubMed] [Google Scholar]
- 44.Sapir T, et al. Antagonistic effects of doublecortin and MARK2/Par-1 in the developing cerebral cortex. J Neurosci. 2008;28(48):13008–13013. doi: 10.1523/JNEUROSCI.2363-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Courchet J, et al. Terminal axon branching is regulated by the LKB1-NUAK1 kinase pathway via presynaptic mitochondrial capture. Cell. 2013;153(7):1510–1525. doi: 10.1016/j.cell.2013.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fioravante D, Regehr WG. Short-term forms of presynaptic plasticity. Curr Opin Neurobiol. 2011;21(2):269–274. doi: 10.1016/j.conb.2011.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Purcell P, et al. Temporomandibular joint formation requires two distinct hedgehog-dependent steps. Proc Natl Acad Sci USA. 2009;106(43):18297–18302. doi: 10.1073/pnas.0908836106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Madisen L, et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci. 2010;13(1):133–140. doi: 10.1038/nn.2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.