Abstract
Cancer is the second leading cause of death in the world. Despite advances in the diagnosis and treatment, overall survival of patients still remains poor. Hence, there is an urgent need for development of new anticancer agents. Considering promising biological activity of 1,3,4-thiadiazole derivatives, in the present study, synthesis and cytotoxicity assessment of new derivatives of this ring was done. All synthesized compounds were characterized by NMR, IR and MS spectroscopic methods. Obtained data from MTT assay showed that all compounds 3a- 3l had better anticancer activity against MDA(breast cancer) compared to PC3(prostate cancer) and U87(Glioblastoma). Compound 3 g with m-OCH3 moiety on the phenyl ring was the most potent one in this series with IC50 = 9 μM against MDA breast cell line in comparison with imatinib (IC50 = 20 μM) as reference drug.
Key Words: Synthesis; 1,3,4-Thiadiazole; MTT assay; Breast cancer
Introduction
Cancer is a disease in which cells can be aggressive, invasive and/or metastatic. These are three malignant properties of cancer cells that differentiate them from benign tumors which are self-limited in their growth and do not invade or metastasize (1-4). Cancer is the second leading cause of death in the world. Despite advances in the diagnosis and treatment, overall survival of patients still remains poor. Until recently, surgery, chemotherapy, radiotherapy and endocrine therapyhave been the standard treatment options available for cancer patients. This has improved survival in several types of solid tumors; however, drug toxicity and emergence of drug resistance have been the major causes of failure in treatment. Hence, there is an urgent need for discovery of new anticancer agents to overcome the disadvantages of the currently available anticancer drugs (3, 5-7).
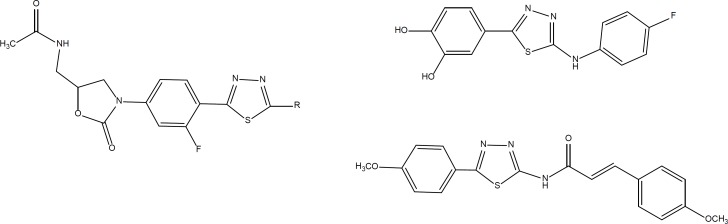
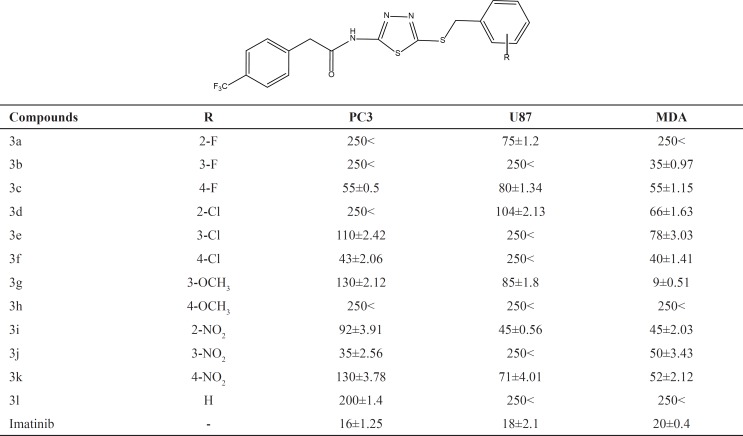
Diverse chemical structure containing 1,3,4-Thiadiazole have been reported with potential anticancer activity (Figure 1) (8-13). Recently Maurizio Botta and coworkers reported the discovery of new derivatives of N-(5- (benzylthio)-1,3,4-thiadiazol-2-yl) benzamide as potent dual inhibitors of abl and src tyrosine kinases (14, 15) (Figure 2). In the present study, we synthesized a new series of these derivatives and evaluated their preliminary anticancer activity in-vitro against three cancer cell lines.
Figure 1.
Structures of some 1,3,4-thiadiazole-based compounds with anticancer activity
Figure 2.
Total structure of 5-amino-1,3,4-thiadiazole-2-thiol derivatives as anticancer agents
Results and Discussion
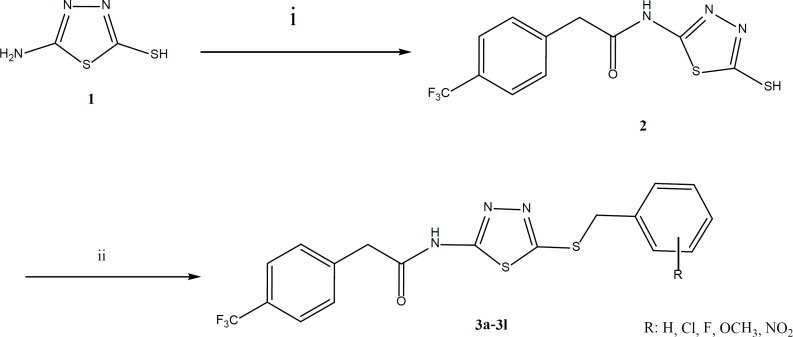
According to Table 1, all synthesized compounds 3a-3l were evaluated against three cancer cell lines. Overall, the best anticancer activity were observed against MDA breast cancer cell line compared to other cell lines (PC3 and U87). Substitution of various moieties such as F, Cl, nitro and methoxy were done at different positions of the phenyl ring to study the electronic effects of the related substituents.
Table 1.
Cytotoxicity results (IC50, μM) of compounds 3a-3l against cancerous cell lines, PC3 (Prostate cancer), U87(Glioblastoma) and MDA(Breast cancer).
|
Fluorine substitution showed the best anticancer properties against MDA cell line at position 3(meta) of the phenyl ring (compound 3b) with IC50 = 35 μM. Comparison of cytotoxic effects of fluorinated derivatives in all cell lines exhibited that para position is the best position for rendering of the optimal activity(compound 3c). Chlorinated derivatives demonstrated better cytotoxic activity at position para (compound 3f) compared to other positions of the phenyl ring. Compound 3f rendered more cytotoxic activity toward PC3 and MDA cell lines. Investigation of the role of methoxy group at positions meta and para caused a high cytotoxic effects for compound 3g with m-methoxy substituent in all cell lines. This compound showed higher activity with IC50 = 9 μM compared to imatinib (IC50 = 20 μM) in MDA breast cancer cell line. Substitution of nitro group at position 2 of the phenyl ring led to an acceptable cytotoxicity against U87 and MDA cell lines rather than PC3 cell line. Nitro moiety rendered the best anticancer activity at position 3(meta) against PC3 cell line. Insertion of the phenyl ring without any moiety resulted in implausible effects in all cell lines.
Experimental
Chemistry
All chemical compounds were purchased from commercial suppliers of Merck and Aldrich companies. The purity of the prepared compounds was proved by thin layer chromatography (TLC) using various solvents of different polarities. Merck silica gel 60 F254 plates were applied for analytical TLC. Column chromatography was performed on Merck silica gel (70-230 mesh) for purification obtained compounds. 1H-NMR spectra were recorded using a Brucker 200 MHz spectrometer, and chemical shifts were expressed as δ (ppm) with tetramethylsilane (TMS) as internal standard. The IR spectra were obtained on a Shimadzu 470 spectrophotometer (potassium bromide disks). Melting points were determined using electrothermal melting point analyzer apparatus and were uncorrected. The mass spectra were run on a Finigan TSQ-70 spectrometer (Finigan, USA) at 70 eV. All cell lines were purchased from the Pasteur Institute of Iran.
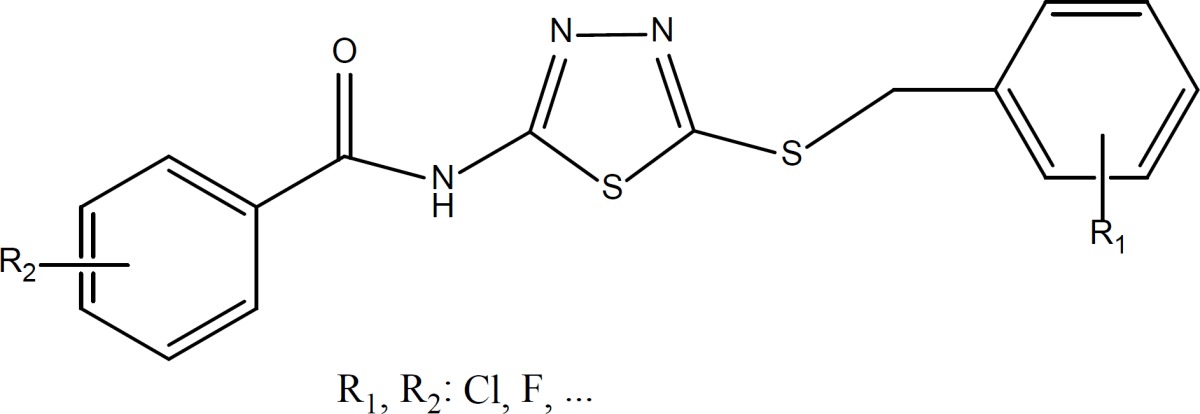
According to the Figure 3, 5-amino- 1,3,4-thiadiazole-2-thiol (1) was treated with 4-trifluoromethylphenylacetic acid for direct coupling of acid with amine. The reaction was carried out in the presence of EDC and hydroxybenzotriazole (HOBt) in acetonitrile as solvent. The termination of reaction was proved by thin layer chromatography (TLC). After completion, the solvent was evaporated using rotary evaporator apparatus and ethyl acetate and water were added. The aqueous phase was removed and the organic phase was washed two times by sodium bicarbonate 5%, diluted sulfuric acid and brine (16-19). Anhydrous sodium sulfate was added for drying and filtration was done. Ethyl acetate was removed under reduced pressure and a yellow powder was obtained. The obtained product was used after crystallization from ethanol for the next step. Various benzyl chloride derivatives were reacted with compound 2 for obtaining the final appropriate products (3a-3l). The 1H NMR, IR and MS spectra were used to confirm the synthesized compounds.
Figure 3.
Synthetic procedure of compounds 3a-3l, Reagents and conditions i) 4-Trifluoromethylphenylacetic acid, EDC, HOBt, CH3CN, rt, 24 h, ii) Benzyl chloride derivatives, KOH, EtOH, reflux, 24 h
Synthesis of N-(5-Mercapto-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (2)
In a flask, equimolar amounts of 4-trifluoromethylphenylacetic acid, EDC and HOBtin acetonitrile solvent were stirred for 30 min and then equimolar quantity of 5-amino-1,3,4-thiadiazole-2-thiol was added. The stirring condition was continued for 24 h. The end point of the reaction was determined by thin layer chromatography(TLC). Acetonitrile was removed under reduced pressure and ethyl acetate/water was added. The aqueous layer was removed and organic layer was washed two times by sodium bicarbonate 5%, diluted sulfuric acid and brine. Anhydrous sodium sulfate was added for drying and then filtered. The ethyl acetate was evaporated using rotary evaporator apparatus. The obtained yellowish solid was washed by dry ether and used for the next step.
mp. 171°C, Yield: 65%,C11H8F3N3OS2, MW: 319 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 3.76 (s, 2H, -CH2CO-), 3.95 (s, 1H, -SH), 7.58 (m, 2H, J = 8Hz), 8.19 (m, 2H, J = 8Hz, 4-trifluoromethylphenyl), 12.76 (brs, 1H, NH).IR (KBr, cm-1) ῡ: 3265, 1697, 1580, 1519, 1321, 1155, 1103, 1064, 821, 705.MS(m/z, %): M++2: 322(10), M+: 320(10), 279(45), 276(35), 167(80), 159(95), 149(100), 133(15), 109(15), 71(15), 57(20).
General procedure for synthesis of compounds 3a-3l
Equimolar quantities of appropriate benzyl chloride derivative was treated with 2-(4-fluorophenyl)-N-(5-mercapto-1,3,4-thiadiazol-2-yl)acetamide (2). Equimolar amount of potassium hydroxide in absolute ethanol was added to convert the thiol moiety to the thiolate anion. Then, the related benzyl chloride derivative was added to the reaction medium and reflux condition was performed for 24 h. Crushed ice was added and the precipitate filtered, washed by cool water and purified by appropriate procedures such as crystallization or column chromatography (EtOAC/Petroleum ether: 3/2).
N-(5-(2-Fluorobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3a)
mp. 201 °C, Yield: 42%,C18H13F4N3OS2, MW: 427 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 4.00 (s, 2H, -CH2CO-), 4.53 (s, 2H, -CH2S-), 7.16-7.54 (m, 2-fluorobenzyl), 7.59 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.76 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 13.00 (s, NH). IR(KBr, cm-1) ῡ: 3158, 3050, 2898, 1690, 1560, 1493, 1456, 1421, 1402, 1337, 1301, 1236, 1168, 1117, 1071, 1019, 972, 844, 757, 693, 655.MS(m/z, %): M+: 427(95), 377(60), 325(35), 297(30), 236(25), 236(15), 159(75), 109(100).
N-(5-(3-Fluorobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3b)
mp. 190 °C, Yield: 58%,C18H13F4N3OS2, MW: 427 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 3.93 (s, 2H, -CH2CO-), 4.53 (s, 2H, -CH2S-), 7.01-7.47 (m, 3-fluorobenzyl), 7.58 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.75 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 12.98 (s, NH).IR(KBr, cm-1) ῡ: 3448, 3156, 2916, 1699, 1562, 1488, 1358, 1326, 1172, 1110, 1068, 837.MS(m/z, %): M+: 427(85), 377(80), 325(40), 297(25), 236(20), 159(70), 109(100).
N-(5-(4-Fluorobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3c)
mp. 166 °C, Yield: 40%,C18H13F4N3OS2, MW: 427 g/mol,1H NMR (DMSO-d6 , 200 MHz) δ: 3.99 (s, 2H, -CH2CO-), 4.51 (s, 2H, -S-CH2-), 7.24 (t, 2H, 4-fluorobenzyl), 7.48 (t, 2H, 4-fluorobenzyl), 7.58 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.75 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 12.97 (s, NH).IR(KBr, cm-1) ῡ: 3370, 3030, 2920, 2829, 1701, 1558, 1508, 1323, 1219, 1160, 1109, 1060, 835.MS(m/z, %): M+: 427(90), 377(75), 325(40), 297(30), 236(25), 159(75), 109(100).
N-(5-(2-Chlorobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3d)
mp. 208 °C, Yield: 54%,C18H13ClF3N3OS2, MW: 443 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 4.00 (s, 2H, -CH2CO-), 4.59 (s, 2H, -CH2S-), 7.35 (m, 2-chlorophenyl), 7.48 (d, 2H, J = 12 Hz, 4-trifluoromethylphenyl), 7.55 (m, 2-chlorophenyl), 7.97(d, 2H, J = 12 Hz, 4-trifluoromethylphenyl), 13.00 (s, NH). IR(KBr, cm-1) ῡ: 3447, 3165, 2922, 1700, 1570, 1445, 1326, 1171, 1109, 1067, 868, 759.MS(m/z, %): M+:443(10), 410(55), 409(85), 159(40), 127(50), 125(100), 109(10), 89(10).
N-(5-(3-Chlorobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3e)
mp. 198 °C, Yield: 57%,C18H13ClF3N3OS2, MW: 443 g/mol,1H NMR (DMSO-d6 , 200 MHz) δ: 4.00 (s, 2H, -CH2CO-), 4.59 (s, 2H, -S-CH2-), 7.33-7.40 (m, 3-chlorobenzyl), 7.43 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.56 (m, 3-chlorobenzyl), 7.75 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 13.00 (s, NH).IR(KBr, cm-1) ῡ: 3427, 3165, 2914, 2730, 1700, 1569, 1446, 1356, 1326, 1301, 1170, 1112, 1066, 868, 839, 759.MS(m/z, %): M+:443(15), 410(60), 409(65), 282(10), 159(55), 127(35), 125(100), 89(10).
N-(5-(4-Chlorobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3f)
mp. 198 °C, Yield: 34%,C18H13ClF3N3OS2, MW: 443 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 3.99 (s, 2H, -CH2CO-), 4.52 (s, 2H, -S-CH2-), 7.38-7.48 (m, 4-chlorobenzyl), 7.58 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.78 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 12.97 (s, NH).IR(KBr, cm-1) ῡ: 3440, 3130, 3040, 2877, 1690, 1556, 1334, 1163, 1118, 1068, 1020, 846, 745, 702.MS(m/z, %): M++1: 444(10), M+:443(10), 410(60), 409(80), 282(12), 178(12), 159(40), 127(50), 125(100), 109(10), 89(10).
N-(5-(3-Methoxybenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3g)
mp. 160 °C, Yield: 32%,C19H16F3N3O2S2, MW: 439 g/mol,1H NMR (DMSO-d6 , 200 MHz) δ:1H NMR (DMSO-d6 , 200 MHz) δ: 3.80 (s, 3H, -OCH3), 3.99 (s, 2H, -CH2CO-), 4.48 (s, 2H, -S-CH2-),7.06(m, 3H, 3-methoxybenzyl), 7.32(m, 1H, 3-methoxybenzyl), 7.58(d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.75 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 12.97 (s, NH).IR(KBr, cm-1) ῡ:3167, 3038,2946, 1735, 1702, 1607, 1577, 1488, 1438, 1358, 1325, 1302, 1271, 1158, 1113, 1068, 839, 777, 736.MS(m/z, %): M+: 439(40), 159(75), 122(45), 121(100), 109(35).
N-(5-(4-Methoxybenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3h)
mp. 219 °C, Yield: 36%,C19H16F3N3O2S2, MW: 439 g/mol,1H NMR (DMSO-d6 , 200 MHz) δ: 3.76 (s, 3H, -OCH3), 3.99 (s, 2H, -CH2CO-), 4.46 (s, 2H, -S-CH2-), 6.92 (d, 2H, J = 8 Hz, 4-methoxybenzyl), 7.35 (d, 2H, J = 8 Hz, 4-methoxybenzyl), 7.58 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.75 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 12.96 (s, NH).IR(KBr, cm-1) ῡ: 3265, 3045, 2870, 1691, 1554, 1510, 1400, 1332, 1298, 1170, 1122, 1107, 1066, 827. MS(m/z, %): M++2: 441(15), M++1: 440(20), M+: 439(25), 159(60), 122(45), 121(100), 109(30).
N-(5-(2-Nitrobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3i)
mp. 146 °C, Yield: 35%,C18H13F3N4O3S2, MW: 454 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 4.00 (s, 2H, -CH2CO-), 4.79 (s, 2H, -CH2S-), 7.49 (m, aromatic), 8.11 (m, aromatic), 8.21 (m, aromatic), 13.00 (s, NH). IR(KBr, cm-1) ῡ: 3447, 3165, 2923, 1698, 1612, 1573, 1527, 1443, 1335, 1168, 1106, 1066, 830, 827, 702.MS(m/z,%): M+: 454(15), 270(35), 242(65), 225(15), 195(100), 179(65), 165(85), 136(70), 106(45), 90(50), 78(35).
N-(5-(3-Nitrobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3j)
mp. 132 °C, Yield: 45%,C18H13F3N4O3S2, MW: 454 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 3.84 (s, 2H, -CH2CO-), 4.51 (s, 2H, -CH2S-), 7.71(m, 5H, aromatic), 8.16(m, 3H, aromatic), 13(brs, NH).IR(KBr, cm-1) ῡ: 3318, 3154, 2850, 1692, 1629, 1562, 1527, 1508, 1487, 1348, 1329, 1163, 1117, 1073, 810, 747.MS(m/z,%): M++1: 455(7), M+: 454(15), 270(30), 242(75), 195(100), 179(70), 165(90), 136(60), 106(35), 90(45), 78(60).
N-(5-(4-Nitrobenzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3k)
mp. 198 °C, Yield: 54%,C18H13F3N4O3S2, MW: 454 g/mol,1H NMR (DMSO-d6, 200 MHz) δ: 3.99 (s, 2H, -CH2CO-), 4.66 (s, 2H, -S-CH2-),7.55(m, 2H, aromatic), 7.77(m, 4H, aromatic), 8.25(m, 2H, aromatic), 12.99(brs, NH).IR(KBr, cm-1) ῡ:3265, 3153, 3040, 2912, 2852, 1697, 1554, 1517, 1342, 1323, 1155, 1103, 1064, 1020, 960, 830.MS(m/z,%): M++1: 455(10), M+: 454(10), 270(40), 242(75), 225(40), 195(100), 179(95), 165(85), 136(60), 106(50), 90(50), 78(60).
N-(5-(Benzylthio)-1,3,4-thiadiazol-2-yl)-2-(4-(trifluoromethyl)phenyl)acetamide (3l)
mp. 203 °C, Yield: 47%,C18H14F3N3OS2, MW: 409 g/mol,1H NMR (DMSO-d6 , 200 MHz) δ: 3.99 (s, 2H, -CH2CO-), 4.52 (s, 2H, -S-CH2-), 7.30-7.49 (m, 5H, benzyl), 7.58 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 7.75 (d, 2H, J = 8 Hz, 4-trifluoromethylphenyl), 12.97 (s, NH).IR(KBr, cm-1) ῡ: 3375, 3040, 1701, 1556, 1508, 1323, 1294, 1220, 1159, 1107, 1066, 1020, 827, 700.MS(m/z, %): M+: 410(100), 409(95), 408(95), 159(75), 148(60), 91(75).
MTT assay
Diverse derivatives of 1,3,4-thiadiazole (compounds 3a-3l) were tested for cytotoxic activity at 0.1-250 μg/mL concentration in three human cancer cell lines of PC3 cell (prostate cancer), U87 (gliobalstoma) and MDA (breast cancer). Cells from different cell lines were seeded in 96-well plates at the density of 8000–10,000 viable cells per well and incubated for 48 h to allow cell attachment.The cells were then incubated for another 48-96 h (depends to cell cycle of each cell line) with various concentrations of compounds 3a-3l. Cells were then washed inPBS, and 20 μL of MTT (3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide solution (5 mg/mL) were added to each well. An additional 4 h of incubation at 37°C were done, and then the medium was discarded. Dimethyl sulfoxide (60 μL) was added to each well, and the solution was vigorously mixed to dissolve the purple tetrazolium crystals. The absorbance of each well was measured by plate reader (Anthous 2020; Austria) at a test wavelength of 550 nm against a standard reference solution at 690 nm. The amount of produced purple formazan is proportional to the number of viable cells (16).
Acknowledgment
Authors are grateful from research council of Kermanshah University of Medical Sciences for financial support. This article has been deduced from the PharmD thesis of Mr Zaman Hasanvand.
References
- 1.Bhuva HA, Kini SJ. Synthesis, anticancer activity and docking of some substituted benzothiazoles as tyrosine kinase inhibitors. J. Mol. Graph. Model. 2010;29:32–37. doi: 10.1016/j.jmgm.2010.04.003. [DOI] [PubMed] [Google Scholar]
- 2.Albreht T, Mckee M, Alexe DM, Coleman MP, Martin-Moreno JM. Making progress against cancer in Europe in 2008. Eur. J. cancer . 2008;44:1451–56. doi: 10.1016/j.ejca.2008.02.015. [DOI] [PubMed] [Google Scholar]
- 3.Yang XH, Wen Q, Zhao TT, Sun J, Li X, Xing M, Lu X, Zhu HL. Synthesis, biological evaluation, and molecular docking studies of cinnamic acyl 1,3,4-thiadiazole amide derivatives as novel antitubulin agents. Bioorg. Med. Chem. 2012;20:1181–1187. doi: 10.1016/j.bmc.2011.12.057. [DOI] [PubMed] [Google Scholar]
- 4.Sun J, Yang YS, Li W, Zhang YB, Wang XL, Tang JF, Zhu XL. Synthesis, biological evaluation and molecular docking studies of 1,3,4-thiadiazole derivatives containing 1,4-benzodioxan as potential antitumor agents. Bioorg. Med. Chem. Lett. 2011;21:6116–6121. doi: 10.1016/j.bmcl.2011.08.039. [DOI] [PubMed] [Google Scholar]
- 5.Levitzki A. Protein tyrosine kinase inhibitors as novel therapeutic agents. Pharmacol. Ther. 1999;82:231–39. doi: 10.1016/s0163-7258(98)00066-7. [DOI] [PubMed] [Google Scholar]
- 6.Ferlay J, Autier P, Boniol M, Heanue M, Colombet M, Boyle P. Estimates of the cancer incidence and mortality in Europe in 2006. Ann. Oncol. 2007;18:581–592. doi: 10.1093/annonc/mdl498. [DOI] [PubMed] [Google Scholar]
- 7.Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, Thun MJ. Cancer Statistics, 2006. Cancer J. Clin. 2006;56:106–130. doi: 10.3322/canjclin.56.2.106. [DOI] [PubMed] [Google Scholar]
- 8.Dalip Kumar, Reddy Vaddula B, Kuei-Hua Chang KH, Shah K. One-pot synthesis and anticancer studies of 2-arylamino-5-aryl-1,3,4-thiadiazoles. Bioorg. Med. Chem. Lett. 2011;21:2320–2323. doi: 10.1016/j.bmcl.2011.02.083. [DOI] [PubMed] [Google Scholar]
- 9.Farshori NN, Banday RM, Ahmad A, Khan AU, Rauf A. Synthesis, characterization, and in vitro antimicrobial activities of 5-alkenyl/hydroxyalkenyl-2-phenylamine-1,3,4-oxadiazoles and thiadiazoles. Bioorg. Med. Chem. Lett. 2010;20:1933–1938. doi: 10.1016/j.bmcl.2010.01.126. [DOI] [PubMed] [Google Scholar]
- 10.Matysiak J, Opolski A. Synthesis and antiproliferative activity of N-substituted 2-amino-5-(2,4-dihydroxyphenyl)-1,3,4-thiadiazoles. Bioorg. Med. Chem. 2006;14:4483–4489. doi: 10.1016/j.bmc.2006.02.027. [DOI] [PubMed] [Google Scholar]
- 11.Yusuf M, Payal Jain P. Synthesis and biological significances of 1,3,4-thiadiazolines and related heterocyclic compounds. Arab. J. Chem. 2011 in press. [Google Scholar]
- 12.Rzeski W, Matysiakb J, Kandefer-Szerszen M. Anticancer, neuroprotective activities and computational studies of 2-amino-1,3,4-thiadiazole based compound. Bioorg. Med. Chem. 2007;15:3201–3207. doi: 10.1016/j.bmc.2007.02.041. [DOI] [PubMed] [Google Scholar]
- 13.Chou JY, Lai SY, Pan SL, Jow GM, Chern JW, Guh JH. Investigation of anticancer mechanism of thiadiazole-based compound in human non-small cell lung cancer A549 cells. Biochem. Pharmacol. 2003;66:115–124. doi: 10.1016/s0006-2952(03)00254-5. [DOI] [PubMed] [Google Scholar]
- 14.Radi M, Crespan E, Botta G, Falchi F, Maga G, Manetti F. Discovery and SAR of 1,3,4-thiadiazole derivatives as potentAbl tyrosine kinase inhibitors and cytodifferentiating agents. Bioorg. Med. Chem. Lett. 2008;18:1207–1211. doi: 10.1016/j.bmcl.2007.11.112. [DOI] [PubMed] [Google Scholar]
- 15.Crespan E, Radi M, Zanoli S, Schenone S, Botta M, Maga G. Dual Src and Abl inhibitors target wild type Abl and the AblT315I Imatinib-resistant mutant with different mechanisms. Bioorg. Med. Chem. 2010;18:3999–4008. doi: 10.1016/j.bmc.2010.04.024. [DOI] [PubMed] [Google Scholar]
- 16.Aliabadi A, Shamsa F, Ostad SN, Emami S, Shafiee A, Davoodi J, Foroumadi A. Synthesis and biological evaluation of 2-Phenylthiazole-4-carboxamide derivatives as anticancer agents. Eur. J. Med. Chem. 2010;11:5384–5389. doi: 10.1016/j.ejmech.2010.08.063. [DOI] [PubMed] [Google Scholar]
- 17.Valentina O, Maria TC, Valentina L, Cenzo C. Synthesis and evaluation of antitumoral activity of ester and amide derivatives of 2-arylamino-6-trifluoromethyl-3-pyridine carboxylic acids. Bioorg. Med. Chem. 2008;16:2367–2378. doi: 10.1016/j.bmc.2007.11.069. [DOI] [PubMed] [Google Scholar]
- 18.Eliseu O, De Oliveira, Carlos AB, Maria A, B DS, Richard A G. Synthesis of pyrrolidine-substituted benzamides via iodocyclization of β-enaminoesters. Tetrahedron Lett. 2007;48:6393–6396. [Google Scholar]
- 19.Lutz FT, Tobias H, Tim F, Ingrid S. Synthesis and Biological Evaluation of Novel Analogues and Prodrugs of the Cytotoxic Antibiotic CC-1065 for Selective Cancer Therapy. Eur. J. Org. Chem. 2002;10:1634–1645. [Google Scholar]