Abstract
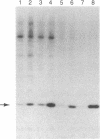
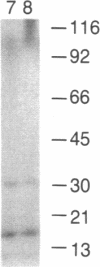
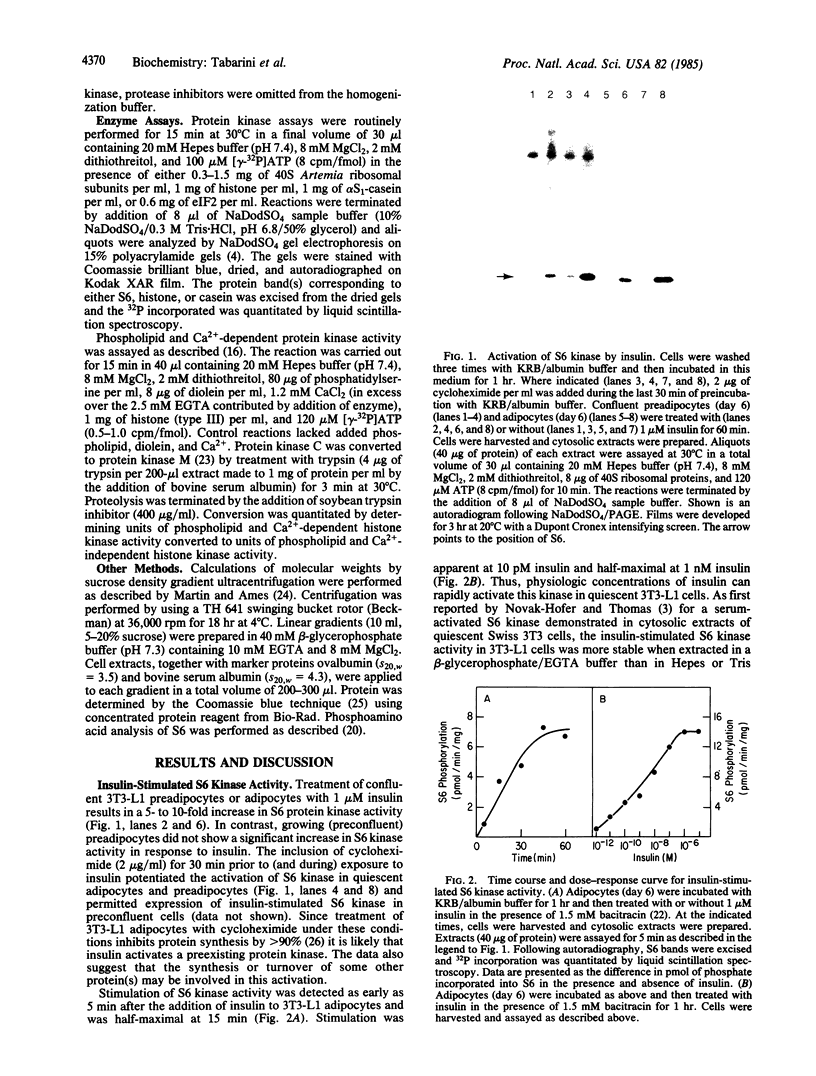
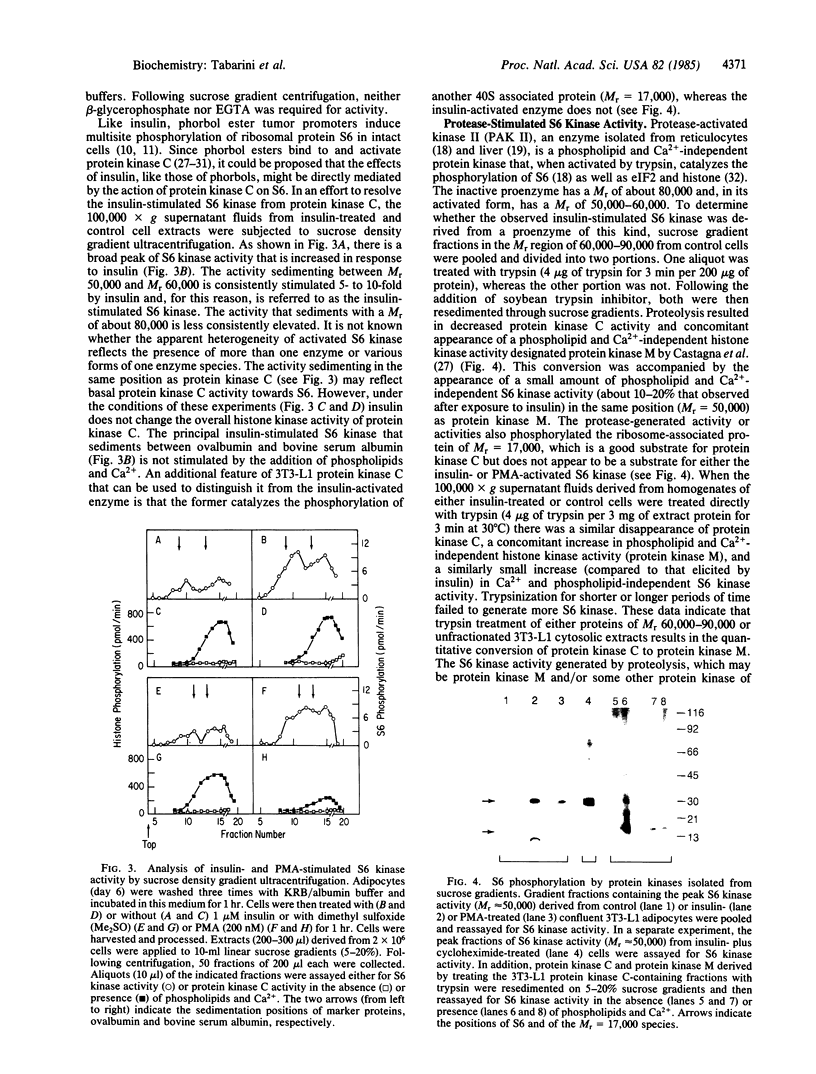
Treatment of 3T3-L1 cells with 0.1-1.0 nM insulin results in rapid (5-15 min) activation of a soluble protein kinase that phosphorylates serine residues in ribosomal protein S6. The insulin-stimulated kinase activity is detectable in confluent, nongrowing preadipocytes and adipocytes. In the presence of 2 micrograms of cycloheximide per ml, preconfluent 3T3-L1 cells also respond to insulin by acquiring an S6 kinase activity whose properties are the same as those of the enzyme activity elicited by insulin alone in growth-inhibited cells. The principal insulin-stimulated S6 kinase has a Mr of approximately equal to 50,000-60,000; there is a variable amount of activity that sediments with a Mr of about 80,000. The soluble enzyme exhibits optimal activity between pH 8 and pH 9, requires Mg2+ (10-20 mM), and is inhibited by Ca2+ (0.5 mM), Mn2+ (0.05 mM), and NaF (30 mM). GTP cannot substitute for ATP in the phosphotransferase reaction; cAMP, cGMP, phosphatidylserine plus diolein, the cAMP-dependent protein kinase inhibitor, and heparin (0.7 micrograms/ml) are without effect. Although treatment of 3T3-L1 cells with insulin does not influence the activity or the subcellular distribution of the phospholipid and Ca2+-dependent protein kinase C, exposure to the phorbol tumor promoter phorbol 12-myristate 13-acetate (PMA) results in translocation of protein kinase C to the membrane and activation of a soluble phospholipid and Ca2+-independent S6 protein kinase that has the same magnitude of activity and sedimentation behavior as the insulin-induced activity. Trypsin treatment of either 3T3-L1 cytosolic extracts or partially purified 3T3-L1 protein kinase C generates a small amount of S6 kinase activity of Mr 50,000. This activity, resolved by sucrose gradient centrifugation, is less active than that elicited by either insulin or PMA and, unlike the activities generated by insulin and PMA, is associated with histone kinase activity. The data suggest that the S6 kinase elicited by either insulin or PMA is neither protein kinase C, its phospholipid, and Ca2+-independent proteolytic derivative nor the result of proteolytic activation of an inactive proenzyme that can be reproduced by trypsin treatment of cell extracts in vitro.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashendel C. L., Staller J. M., Boutwell R. K. Identification of a calcium- and phospholipid- dependent phorbol ester binding activity in the soluble fraction of mouse tissues. Biochem Biophys Res Commun. 1983 Feb 28;111(1):340–345. doi: 10.1016/s0006-291x(83)80157-0. [DOI] [PubMed] [Google Scholar]
- Blenis J., Spivack J. G., Erikson R. L. Phorbol ester, serum, and rous sarcoma virus transforming gene product induce similar phosphorylations of ribosomal protein S6. Proc Natl Acad Sci U S A. 1984 Oct;81(20):6408–6412. doi: 10.1073/pnas.81.20.6408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castagna M., Takai Y., Kaibuchi K., Sano K., Kikkawa U., Nishizuka Y. Direct activation of calcium-activated, phospholipid-dependent protein kinase by tumor-promoting phorbol esters. J Biol Chem. 1982 Jul 10;257(13):7847–7851. [PubMed] [Google Scholar]
- Donahue M. J., Masaracchia R. A. Phosphorylation of ribosomal protein S6 at multiple sites by a cyclic AMP-independent protein kinase from lymphoid cells. J Biol Chem. 1984 Jan 10;259(1):435–440. [PubMed] [Google Scholar]
- Gabrielli B., Wettenhall R. E., Kemp B. E., Quinn M., Bizonova L. Phosphorylation of ribosomal protein S6 and a peptide analogue of S6 by a protease-activated kinase isolated from rat liver. FEBS Lett. 1984 Oct 1;175(2):219–226. doi: 10.1016/0014-5793(84)80740-1. [DOI] [PubMed] [Google Scholar]
- Issinger O. G., Beier H., Speichermann N., Flokerzi V., Hofmann F. Comparison of phosphorylation of ribosomal proteins from HeLa and Krebs II ascites-tumour cells by cyclic AMP-dependent and cyclic GMP-dependent protein kinases. Biochem J. 1980 Jan 1;185(1):89–99. doi: 10.1042/bj1850089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraft A. S., Anderson W. B., Cooper H. L., Sando J. J. Decrease in cytosolic calcium/phospholipid-dependent protein kinase activity following phorbol ester treatment of EL4 thymoma cells. J Biol Chem. 1982 Nov 25;257(22):13193–13196. [PubMed] [Google Scholar]
- Kraft A. S., Anderson W. B. Phorbol esters increase the amount of Ca2+, phospholipid-dependent protein kinase associated with plasma membrane. Nature. 1983 Feb 17;301(5901):621–623. doi: 10.1038/301621a0. [DOI] [PubMed] [Google Scholar]
- Lastick S. M., McConkey E. H. HeLa ribosomal protein S6. Insulin and dibutyryl cyclic AMP affect different phosphopeptides. J Biol Chem. 1981 Jan 25;256(2):583–585. [PubMed] [Google Scholar]
- Lastick S. M., Nielsen P. J., McConkey E. H. Phosphorylation of ribosomal protein S6 in suspension cultured HeLa cells. Mol Gen Genet. 1977 Apr 29;152(3):223–230. doi: 10.1007/BF00693074. [DOI] [PubMed] [Google Scholar]
- Le Peuch C. J., Ballester R., Rosen O. M. Purified rat brain calcium- and phospholipid-dependent protein kinase phosphorylates ribosomal protein S6. Proc Natl Acad Sci U S A. 1983 Nov;80(22):6858–6862. doi: 10.1073/pnas.80.22.6858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lubben T. H., Traugh J. A. Cyclic nucleotide-independent protein kinases from rabbit reticulocytes. Purification and characterization of protease-activated kinase II. J Biol Chem. 1983 Nov 25;258(22):13992–13997. [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- Niedel J. E., Kuhn L. J., Vandenbark G. R. Phorbol diester receptor copurifies with protein kinase C. Proc Natl Acad Sci U S A. 1983 Jan;80(1):36–40. doi: 10.1073/pnas.80.1.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsen-Hamilton M., Hamilton R. T., Allen W. R., Potter-Perigo S. Synergistic stimulation of S6 ribosomal protein phosphorylation and DNA synthesis by epidermal growth factor and insulin in quiescent 3T3 cells. Cell. 1982 Nov;31(1):237–242. doi: 10.1016/0092-8674(82)90423-8. [DOI] [PubMed] [Google Scholar]
- Novak-Hofer I., Thomas G. An activated S6 kinase in extracts from serum- and epidermal growth factor-stimulated Swiss 3T3 cells. J Biol Chem. 1984 May 10;259(9):5995–6000. [PubMed] [Google Scholar]
- Perisic O., Traugh J. A. Protease-activated kinase II as the potential mediator of insulin-stimulated phosphorylation of ribosomal protein S6. J Biol Chem. 1983 Aug 25;258(16):9589–9592. [PubMed] [Google Scholar]
- Rodriguez-Pena A., Rozengurt E. Disappearance of Ca2+-sensitive, phospholipid-dependent protein kinase activity in phorbol ester-treated 3T3 cells. Biochem Biophys Res Commun. 1984 May 16;120(3):1053–1059. doi: 10.1016/s0006-291x(84)80213-2. [DOI] [PubMed] [Google Scholar]
- Rosen O. M., Smith C. J., Fung C., Rubin C. S. Development of hormone receptors and hormone responsiveness in vitro. Effect of prolonged insulin treatment on hexose uptake in 3T3-L1 adipocytes. J Biol Chem. 1978 Oct 25;253(20):7579–7583. [PubMed] [Google Scholar]
- Rubin C. S., Hirsch A., Fung C., Rosen O. M. Development of hormone receptors and hormonal responsiveness in vitro. Insulin receptors and insulin sensitivity in the preadipocyte and adipocyte forms of 3T3-L1 cells. J Biol Chem. 1978 Oct 25;253(20):7570–7578. [PubMed] [Google Scholar]
- Smith C. J., Wejksnora P. J., Warner J. R., Rubin C. S., Rosen O. M. Insulin-stimulated protein phosphorylation in 3T3-L1 preadipocytes. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2725–2729. doi: 10.1073/pnas.76.6.2725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spector T. Refinement of the coomassie blue method of protein quantitation. A simple and linear spectrophotometric assay for less than or equal to 0.5 to 50 microgram of protein. Anal Biochem. 1978 May;86(1):142–146. doi: 10.1016/0003-2697(78)90327-5. [DOI] [PubMed] [Google Scholar]
- Takai Y., Kishimoto A., Inoue M., Nishizuka Y. Studies on a cyclic nucleotide-independent protein kinase and its proenzyme in mammalian tissues. I. Purification and characterization of an active enzyme from bovine cerebellum. J Biol Chem. 1977 Nov 10;252(21):7603–7609. [PubMed] [Google Scholar]
- Thomas G., Martin-Pérez J., Siegmann M., Otto A. M. The effect of serum, EGF, PGF2 alpha and insulin on S6 phosphorylation and the initiation of protein and DNA synthesis. Cell. 1982 Aug;30(1):235–242. doi: 10.1016/0092-8674(82)90029-0. [DOI] [PubMed] [Google Scholar]
- Thomas G., Siegmann M., Gordon J. Multiple phosphorylation of ribosomal protein S6 during transition of quiescent 3T3 cells into early G1, and cellular compartmentalization of the phosphate donor. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3952–3956. doi: 10.1073/pnas.76.8.3952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trevillyan J. M., Kulkarni R. K., Byus C. V. Tumor-promoting phorbol esters stimulate the phosphorylation of ribosomal protein S6 in quiescent Reuber H35 hepatoma cells. J Biol Chem. 1984 Jan 25;259(2):897–902. [PubMed] [Google Scholar]
- Walsh D. A., Ashby C. D., Gonzalez C., Calkins D., Fischer E. H. Krebs EG: Purification and characterization of a protein inhibitor of adenosine 3',5'-monophosphate-dependent protein kinases. J Biol Chem. 1971 Apr 10;246(7):1977–1985. [PubMed] [Google Scholar]
- Wettenhall R. E., Morgan F. J. Phosphorylation of hepatic ribosomal protein S6 on 80 and 40 S ribosomes. Primary structure of S6 in the region of the major phosphorylation sites for cAMP-dependent protein kinases. J Biol Chem. 1984 Feb 25;259(4):2084–2091. [PubMed] [Google Scholar]
- Zasloff M., Ochoa S. Purification of eukaryotic initiation factor 1 (EIF1) from Artemia salina embryos. Methods Enzymol. 1974;30:197–206. doi: 10.1016/0076-6879(74)30022-5. [DOI] [PubMed] [Google Scholar]
- del Grande R. W., Traugh J. A. Phosphorylation of 40-S ribosomal subunits by cAMP-dependent, cGMP-dependent and protease-activated protein kinases. Eur J Biochem. 1982 Apr 1;123(2):421–428. doi: 10.1111/j.1432-1033.1982.tb19785.x. [DOI] [PubMed] [Google Scholar]