Abstract
The presence of dendritic cells, antigen-presenting cells that link innate and adaptive immunity, is necessary to generate and maintain the production of antiphospholipid antibodies in response to exposed intracellular phospholipids on the outer surface of apoptotic cells. In turn, antiphospholipid antibodies enhance dendritic cell-induced inflammatory and proatherogenic responses in a number of conditions that are associated with accelerated atherosclerosis, including diabetes, chronic kidney disease, periodontal infections, and aging. While altering dendritic cells by modifying the ubiquitin-proteasome system enhances antiphospholipid antibody production and leads to development of accelerated atherosclerosis and autoimmune features, inducing tolerance by dendritic cell manipulation leads to decreased atherosclerosis and thrombosis. Therefore, further translational studies are needed to understand the interplay between dendritic cells and antiphospholipid antibodies, and to develop potential new therapies for antiphospholipid syndrome and atherosclerosis. Here we review current experimental and translational studies that have examined the role of dendritic cells in antiphospholipid antibody formation and in antiphospholipid-associated atherosclerosis and thrombosis.
Keywords: dendritic cells, antiphospholipid antibodies, atherosclerosis, antiphospholipid syndrome
1.1 Introduction
Antiphospholipid antibodies (aPL Abs) are a heterogeneous group of Abs against phospholipids or phospholipid-binding proteins found not only in individuals with autoimmune diseases, but also in individuals without overt autoimmune diseases [1]. Antiphospholipid syndrome (APS) is characterized by the presence of persistent aPL Abs associated with arterial and/or venous thrombosis and increased pregnancy morbidity [2; 3]. While not all aPL Abs are pathogenic, it has been well established that Abs against cardiolipin (aCL Abs) and particularly Abs against beta2 glycoprotein I (anti-β2-GPI Abs), a plasma protein that binds to negatively charged phospholipids, play an instrumental role in the pathogenesis of APS [4; 5; 6; 7]. While an association between aPL Abs and accelerated atherosclerosis with and without APS has also been proposed, the exact mechanisms are not well understood [8; 9; 10; 11; 12; 13; 14].
Atherosclerosis is a complex process that is initiated when low density lipoprotein (LDL) molecules are deposited in the arterial walls, where they become oxidized by reactive oxygen species or enzymes, such as myeloperoxidase or lipoxygenase. Oxidized LDL (oxLDL) particles are phagocytosed by macrophages and become foam cells that are subsequently deposited within arterial walls, where they release pro-inflammatory cytokines such as TNF-α and IL-1, matrix metalloproteinases and oxygen-activated radicals. In addition to macrophages, monocytes, neutrophils and dendritic cells (DCs) also contribute to endothelial activation and increased expression of adhesion molecules including E-selectin and VCAM-1, which leads to further immune system activation and plaque formation. Acute plaque rupture and thrombus formation can cause stroke or acute myocardial infarction [15; 16; 17; 18; 19; 20].
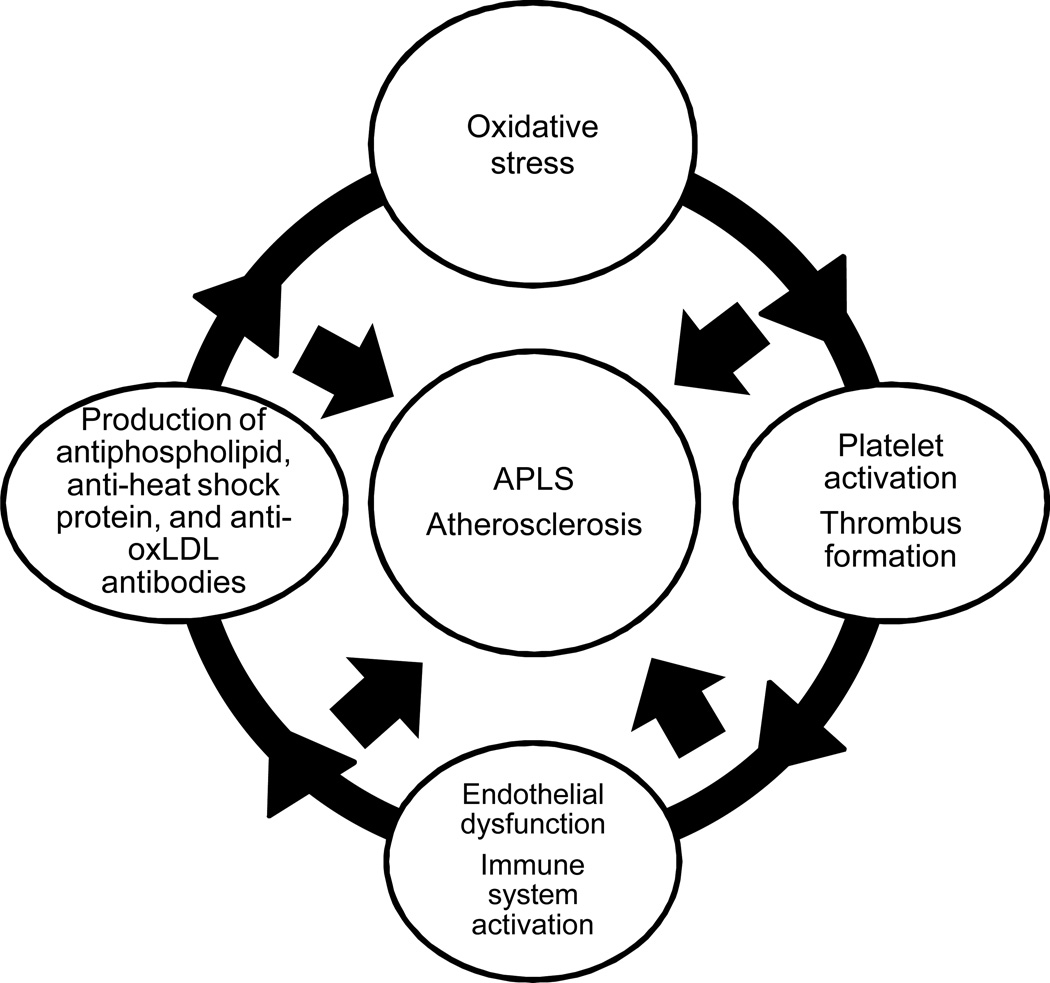
APS and atherosclerosis share several similar features: increased levels of Abs to oxLDL, phospholipids and heat shock proteins, endothelial dysfunction, platelet activation and thrombus formation, increased oxidative stress and increased immune cell activation (Figure 1). Several more potential contributions of aPL Abs in atherosclerosis have been proposed, including proatherogenic modification of LDL and high density lipoprotein (HDL), increased endothelial damage and immune system activation [8; 9; 10; 11; 13]. However, the epidemiologic evidence that the presence of aPL Abs may be a serologic marker or an independent risk factor for atherosclerosis is inconclusive [21; 22].
Figure 1. Common pathways in antiphospholipid syndrome and in atherosclerosis.
APLS and atherosclerosis share several common pathways including production of antibodies to phospholipids, heat shock proteins, and oxidized LDL; endothelial dysfunction; immune cell activation; oxidative stress; platelet activation and thrombus formation.
Here we review experimental and translational evidence suggesting that DCs may play an important role in the generation and maintenance of aPL Abs, and that illustrate a key connection between DCs, aPL Abs, innate and adaptive immunity and atherosclerosis.
1.2 The role of DCs in autoimmunity and atherosclerosis
DCs are antigen-presenting cells that link innate and adaptive immunity, and play an important role in the pathogenesis of atherosclerosis [20] and autoimmune diseases [23]. Conventional DCs search peripheral tissues for pathogens, secrete cytokines, present antigen, and activate naive T-cells. In addition, conventional DCs play a role in inducing and maintaining central and peripheral self-tolerance and minimizing autoimmune reactions. Plasmacytoid DCs (pDCs) migrate from the blood into lymph nodes and predominantly modulate immune responses to viruses by expressing toll-like receptors (TLRs), especially TLR-7 and TLR-9, and producing type I interferons [24; 25; 26; 27].
DCs play a complex role in early and advanced atherosclerosis. DCs promote early atherosclerosis and inflammation primarily through macrophage activation and a relative shift toward Th1 responses. While Th1 responses are proatherogenic, Th2 responses have both proatherogenic and protective effects. Furthermore, in advanced atherosclerotic plaques, accumulation of pDCs contributes to plaque instability and endothelial damage [28]. In addition, interactions between microbial pathogens and pDCs may link atherosclerosis to infections, explaining increased plaque vulnerability during acute infections [15].
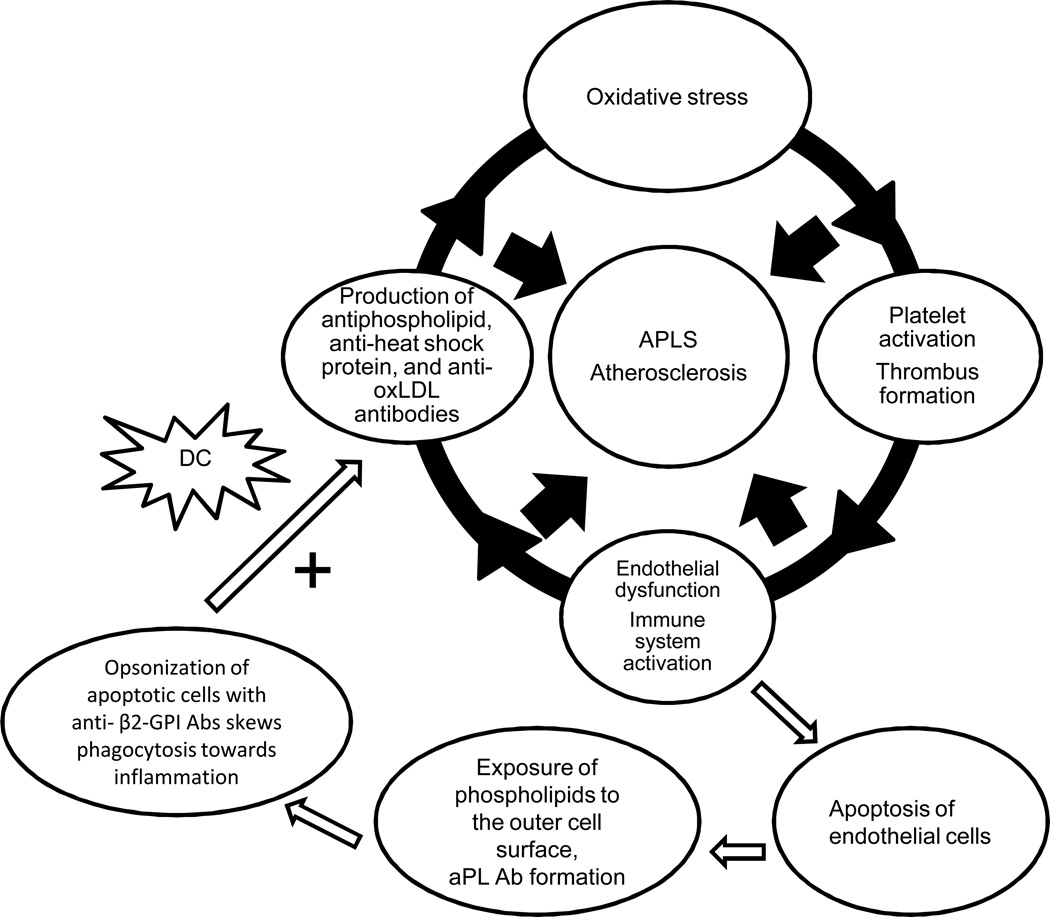
1.3 The presence of DCs is required to initiate production of aPL Abs (Figure 2)
Figure 2. DCs are needed to develop anti - β2-GPI Abs.
Early endothelial damage in the arterial vessels can lead to the exposure of phospholipids to the outer cell surface and aPL antibody formation. Opsonization of apoptotic cells with anti-β2-GPI Abs skews phagocytosis toward inflammation. DCs preferentially internalize apoptotic cells opsonized by anti-β2-GPI Abs, leading to sustained generation of anti-β2-GPI Abs.
The presence of apoptotic cells and DCs is needed to generate aPL Abs and to promote aPL-specific autoimmune responses. In intact cells, negatively charged phospholipids are not present on the outer cell surface and do not contribute to antibody formation. In inflammatory conditions such as autoimmune diseases and atherosclerosis, early endothelial damage in the arterial vessels can lead to the exposure of phospholipids to the outer cell surface and promote antibody formation [2; 29]. Indeed, several studies have demonstrated that apoptosis is an important initial step in formation of aPL Abs [29; 30; 31].
The initial evidence demonstrating the cooperative role of DCs and apoptotic cells in producing aPL Abs came from experiments showing that apoptotic cells were recognized by monoclonal Abs to β2-glycoprotein I (β2-GPI), and that DCs preferentially internalize apoptotic cells opsonized by anti-β2-GPI Abs. When apoptosis is induced in vitro in murine cells and analyzed with flow cytometry, apoptotic cells bound to anti-β2-GPI Abs are internalized more efficiently by DCs [32].
In contrast to necrotic cells, apoptotic cells usually produce only minimal immune responses and do not initiate inflammatory reactions. However, while exposure of anionic phospholipids serves as an anti-inflammatory and immunosuppressive signal, opsonization of apoptotic cells with aPL Abs skews phagocytosis toward inflammation. Presentation of cryptic epitopes from apoptotic cells in an inflammatory state such as atherosclerosis or an autoimmune disease may impair T cell tolerance, and increase survival of autoreactive T and B cells in the lymph nodes [33].
Similarly, the binding of β2-GPI to anionic phospholipids facilitates the processing and presentation of a cryptic epitope that activates pathogenic autoreactive T cells. When DCs and macrophages are pulsed with phospholipid-bound β2-GPI, an autoreactive T-cell response is induced, while β2-GPI alone or phospholipid alone do not. In addition, in vitro autoreactive T-cell responses were elicited when peripheral blood T cells from healthy individuals were stimulated with DCs pre-incubated with phospholipid-bound β2-GPI. Thus, activated β2-GPI-reactive T cells would subsequently stimulate B cells to produce pathogenic anti-β2-GPI Abs [34].
Additional evidence that DCs play an important role in developing Abs to β2-GPI came from a series of in vivo experiments. When NZB×NZWF mice were immunized with DCs that had macropinocytosed β2-GPI or phagocytosed apoptotic thymocytes that displayed β2-GPI, the mice developed anti-β2-GPI Abs and autoimmune features. However, apoptotic cells that were opsonized with β2-GPI did not induce anti-β2-GPI Abs or autoimmune features when DCs were not present [35].
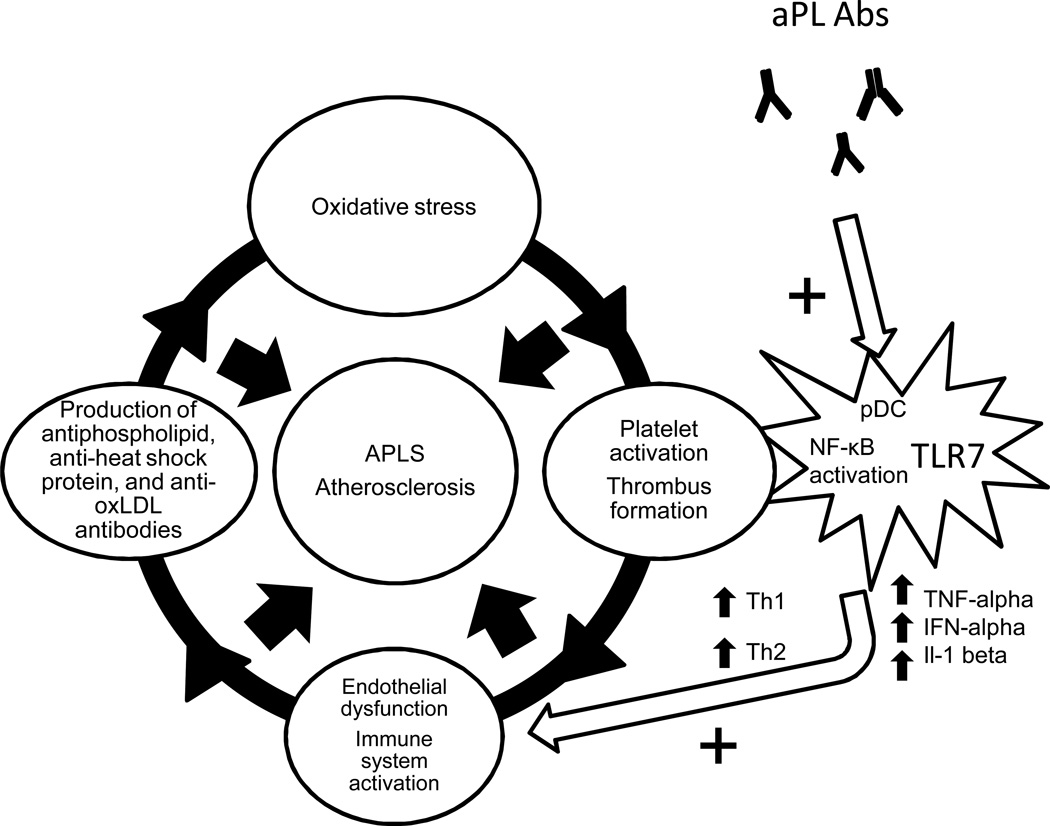
1.4 Synergistic effects of aPL and Toll-like receptors on pDCs (Figure 3)
Figure 3. Synergistic effects of aPL Abs and TLR7 on pDC maturation.
aPL Abs work synergistically with TLR7 to promote pDC maturation and pro-inflammatory function by activating the NF-κB signaling pathway.
Based on the data presented above, it is clear that the presence of DCs is important in aPL Ab formation. It would therefore follow that DCs are critical to the development of the clinical features of APS. Here we present evidence for an additional mechanism by which DCs may fulfill this role, demonstrating the synergistic association of aPL Abs with the Toll-like receptor (TLR) activation to promote pro-inflammatory functions of pDCs.
Stimulating human monocytes and pDCs in vitro with ligands of Toll-like receptor 7 (TLR7) and TLR8 leads to increased production of IL1-beta, an important mediator of inflammation associated with endothelial activation and damage. When human monoclonal IgG aPL Ab generated from a patient with APS is added, TLR7 mRNA expression is upregulated and production of IL1-beta is substantially increased, suggesting that the TLR7 signaling pathway synergistically increases IL-1beta production induced by aPL Abs in pDCs [36].
Further evidence of this synergistic effect is based on the observation that aPL Abs induce TLR7 mRNA expression in human pDCs in a dose-dependent manner. Prinz et al proposed a novel aPL Ab-dependent mechanism of TLR activation, whereby aPL Abs can accumulate in the endosomal compartment of pDCs and induce NADPH oxidation, most likely via interaction of these Abs with endosomal lysobisphosphatidic acid. NADPH oxidation stimulates activation of nuclear transcription factor NF-κB and a subsequent increase in TLR7 gene expression in mouse pDCs. Thus, aPL Abs sensitize pDCs to TLR7 ligands and promote increased production of IFN-alpha and TNF-alpha by pDCs in response to sub-threshold levels of aPL Abs [37]. Notably, both of these cytokines play an important role in atherosclerosis and in autoimmunity: TNF-alpha is pathogenic in atherosclerosis [38] and in endothelial cell activation by anti-β2-GPI Abs in APS [39]; IFN-alpha is implicated in premature atherosclerosis in systemic lupus erythematosus (SLE) [40].
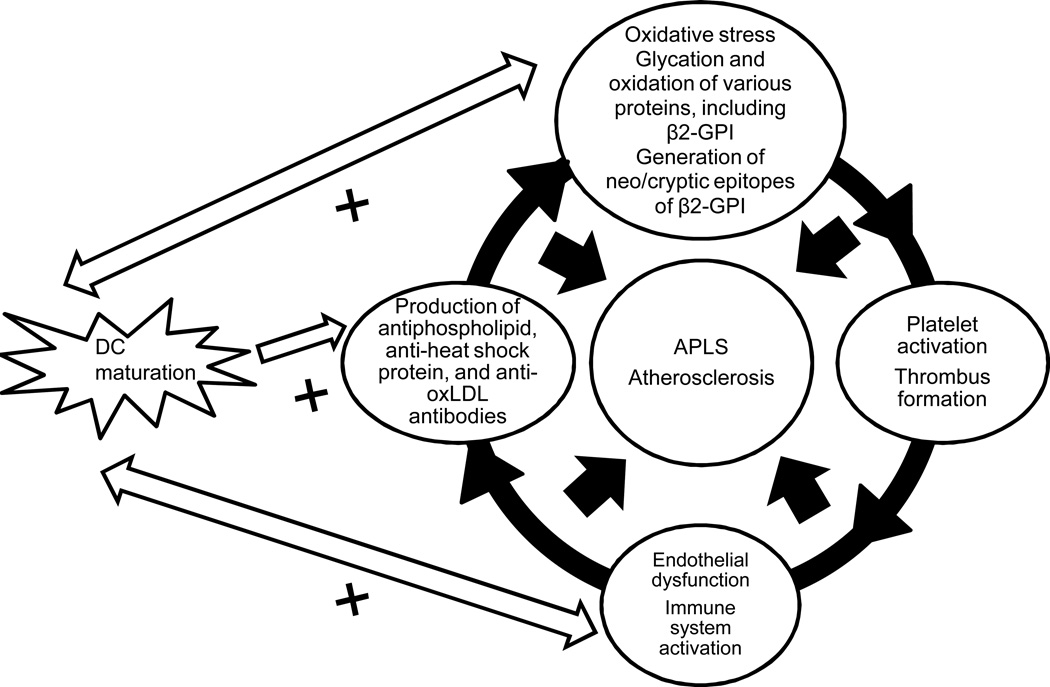
1.5 Interactions of oxidized/glycated beta2-GPI with DCs enhance their pro-atherogenic properties (Figure 4)
Figure 4. Interactions between DCs and aPL Abs may be enhanced by glycation and oxidation of aPL Abs.
In atherosclerosis, diabetes and aging, the microenvironment predisposes local β2GPI to glycation and/or oxidation, initiating a local autoimmune process leading to inflammation, endothelial dysfunction and accelerated atherosclerosis.
Although the evidence that β2-GPI generates strong proinflammatory and proatherogenic DC-mediated immune responses is convincing, the origin of these immunogenic properties remains elusive. One possible explanation of this phenomenon may involve oxidation of β2-GPI. In individuals with atherosclerosis, β2-GPI is found in carotid plaques [43], implying that oxidation of β2-GPI can take place in atherosclerotic vessels in a manner similar to LDL oxidation as described above. Incubation of oxidized β2-GPI with DCs from healthy human donors caused DCs to mature and initiate Th1 and Th2 responses, which are characterized by increased secretion of interleukin 12 (IL-12), IL-1beta, IL-6, IL-8, tumor necrosis factor alpha (TNF-alpha) and IL-10. However, DC maturation and NF-κB activation was prevented when DCs were incubated with β2-GPI that was first pretreated with the antioxidant alpha-tocopherol, suggesting that oxidation was an important step that potentially linked anti-β2-GPI Abs, DCs, and atherosclerosis. Oxidized β2-GPI has also been shown to generate neo/cryptic epitopes of β2-GPI, which caused maturation and activation of monocyte-derived DCs [41; 42].
The in vitro interaction of oxidized β2-GPI with DCs also results in interleukin-1 receptor associated kinase (IRAK) phosphorylation and NF-κB pathway activation [42]. The NF-κB signaling pathway plays a central role in all stages of atherosclerosis, from early plaque formation to acute coronary syndromes (recently reviewed by Pamukcu et al [44]). Similarly, activation of the NF-κB signaling pathway leading to upregulation of tissue factor expression in monocytes and endothelial cells is a major component of pathogenesis in APS [45]. Translational studies are needed to determine if physiologic oxidation of β2-GPI in individuals with atherosclerosis also leads to activation of the NF-κB signaling pathway in vivo.
Another proposed mechanism of DC activation involves nonenzymatic glycation of β2-GPI [46]. Glycation is a nonenzymatic posttranslational modification in which a sugar molecule binds to a protein or a lipid molecule. Subsequent oxidation of these compounds generates advanced glycation end products (AGE). Deposition of AGE in the vessel wall may contribute to endothelial activation, inflammation, and plaque formation in various conditions including aging, diabetes, and chronic kidney disease [47; 48].
Glycated β2GPI has been shown to activate monocyte-derived immature DCs from healthy human donors in vitro. This, in turn, induces upregulation of the receptor for AGE (RAGE) on monocyte-derived DCs. When DCs are stimulated with AGE-β2GPI complexes, there is a shift in naive T lymphocyte responses toward both Th1 and Th2 responses. In addition, glycated β2GPI causes phenotypic and functional maturation of immature DCs that involve activation of p38 MAPK, ERK and NF-κB. Both p38 MAPK and ERK are involved in cardiovascular remodeling [49], while activation of p38 MAPK plays an important role in diabetes-associated cardiovascular disease (reviewed by Liu and Cao [50]). Evidence from these studies strongly suggests that in diseases associated with accelerated atherosclerosis such as diabetes and chronic kidney disease, as well as in the natural aging process, the vascular microenvironment can predispose local β2GPI to glycation and/or oxidation. These structural changes in β2GPI act as inflammatory stimuli that activate immunogenic DCs and alter tolerogenic DC function, resulting in autoreactive T-cell responses and the development of autoimmune features and atherosclerosis [41].
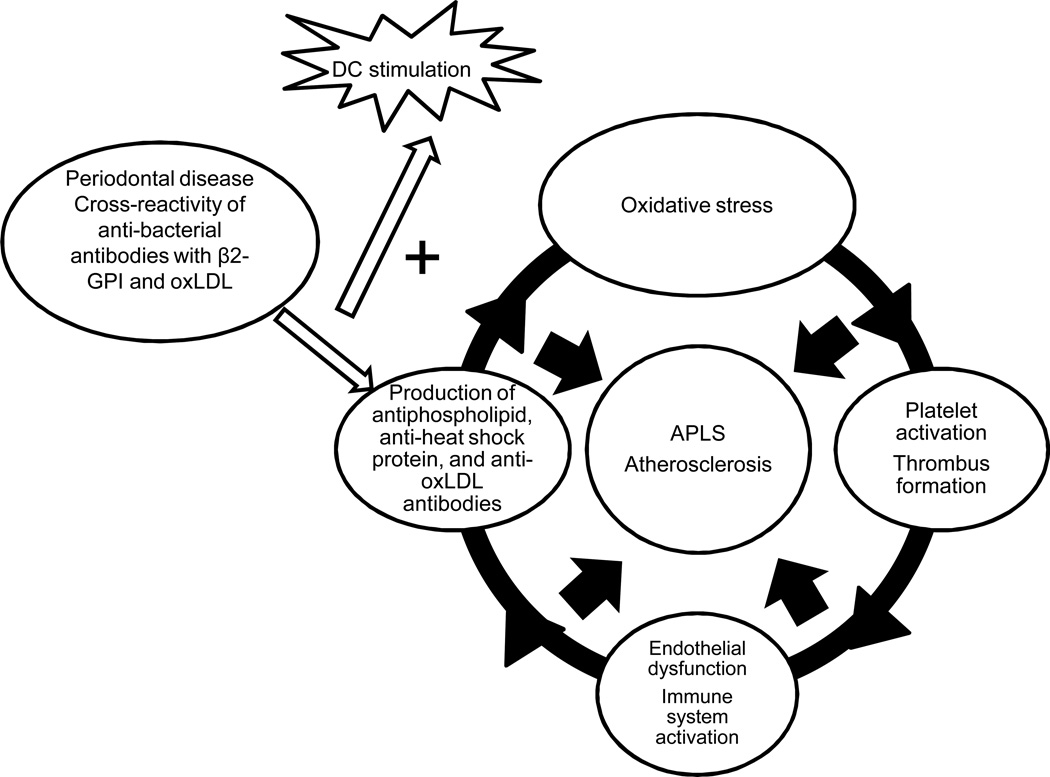
1.6 DCs, aPL Abs and periodontal infections (Figure 5)
Figure 5. A possible link between periodontal disease, aPL Abs, DCs, and atherosclerosis.
Bacteria associated with periodontal disease share homologous sequences with β2GPI and promote formation of pathogenic aPL Abs, which cross react with oxLDL and form immune complexes that stimulate DCs and macrophages more robustly than bacteria or oxLDL alone.
There is some evidence that DCs also serve as a link between aPL Abs and the development of atherosclerosis in individuals with chronic infections such as periodontal disease. Associations between various types of infections and increased production of aPL Abs with or without APS have been reported, suggesting that infectious agents may induce inflammatory responses and serve as the initial trigger of the production of Abs that cross-react with β2GPI [51]. An association between periodontal disease and atherosclerosis has been demonstrated in several studies, although the evidence is controversial (as recently reviewed by Lockhart et al [52]). In particular, P. gingivalis infection has been shown to accelerate the development and progression of atherosclerosis in apoE-deficient mice [53]. Patients with periodontal disease have been shown to have much higher levels of IgG and IgM aCL Abs in their serum samples compared with those from healthy controls. There was also a significant association between the presence of aPL Abs and increased levels of soluble adhesion molecules such as sICAM-1, sE-selectin and sVCAM-1 [54; 55], suggesting that these Abs are pathogenic and are associated with the endothelial activation and vascular inflammation seen in atherosclerosis. A similar process has been observed with aPL Abs from APS patients [5]. Furthermore, significantly higher rates of spontaneous transformation of monocytes into DCs have been observed in monocyte cultures from people with periodontal disease compared with healthy controls [56].
Some bacterial pathogens contain antigens with peptide sequences similar to the TLRVYK sequence in β2GPI. These pathogens can induce cross-reactive aCL Abs that cause endothelial activation and pregnancy complications in mice [57]. A search of the Swiss-Prot protein sequence database identified pathogens from several species of bacteria associated with periodontal disease with conserved TLRVYK sequences. Together, these studies suggest that cross-reactivity between bacterial pathogens and β2GPI leads to enhanced generation of anti-β2GPI Abs in individuals with periodontal infections [58]. Another study demonstrated that aPL Abs induced by P. gingivalis and A. actinomycetemcomitans also cross react with oxidized LDL and form immune complexes. These immune complexes stimulate DCs and macrophages significantly more than bacteria or oxidized LDL alone [56]. The association between various Abs, DCs, and periodontal disease was comprehensively examined in a recent review [59], which concluded that aPL Ab-mediated uptake of oxLDL or bacteria enhances DC maturation, NK-cell-IFN-γ responses and Th1 responses that sustain inflammation, thus providing an additional possible link between periodontal infections, aPL Abs and DCs.
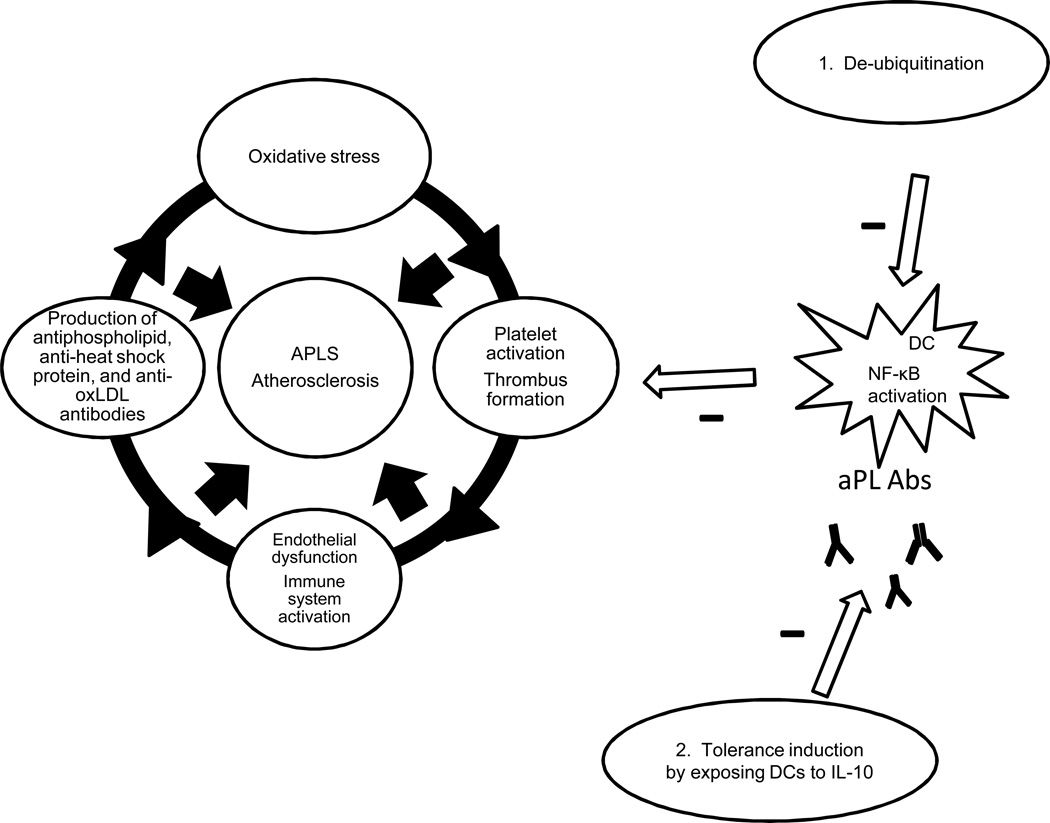
1.7 DCs may be altered to either promote or decrease DC-aPL interactions in APS and atherosclerosis (Figure 6)
Figure 6. Deubiquitination and tolerance induction may alter DCs to decrease DC-aPL interactions in APS and atherosclerosis.
1. Dysregulation of the ubiquitin pathway may partly explain why only some individuals develop persistent aPL Abs, autoimmune features and atherosclerosis. Therefore, de-ubiquitination may decrease DC-aPL interactions. 2. DCs exposed to IL-10 decrease atherosclerotic plaque formation in mice and T-cell responses to b2GPI in APS.
1.7.1 The role of the ubiquitin pathway
While glycation, oxidation and bacterial infections represent environmental exposures that modify β2GPI and/or potentiate interactions between aPL and DCs, alterations in the ubiquitin pathway in DCs may explain why not all individuals develop aPL Abs, autoimmunity and atherosclerosis.
The ubiquitin-proteasome system is responsible for the nonlysosomal degradation of intracellular proteins via conjugation of these proteins with ubiquitin and formation of ubiquitin chains [26], and is thought to play an important role in autoimmunity and atherosclerosis. If ubiquitination is not terminated appropriately, either because of genetic alterations or because of an imbalance between ubiquitin conjugated products and proteasomal degradation, there is a shift towards vascular damage and autoimmunity [26; 60; 61].
The ubiquitin editing enzyme A20 (also known as TNF-α-induced protein [TNFAIP]-3) is a cytoplasmic protein that inhibits inflammatory responses by deubiquitinating intermediate proteins in the NF-κB signaling pathway [60; 61]. Associations between several single-nucleotide polymorphisms in the A20 genomic locus and various autoimmune diseases including SLE, rheumatoid arthritis and systemic sclerosis have been described previously [62]. In a mouse model lacking A20 specifically in DCs (A20-deficient DCs), several abnormalities within A20-deficient DCs were observed, including activation of the classical NF-κB pathway and JNK phosphorylation and spontaneous maturation of these cells. Furthermore, A20-deficient DCs produce extremely high levels of pro-inflammatory cytokines that lead to increased B cell activation and differentiation into plasma cells. A20-deficient DCs are also associated with an increased T cell differentiation into Th1 and Th17 cells and a decrease in regulatory T cell (Treg) production compared with A20-sufficient DCs. Finally, mice lacking A20 in DCs developed high serum concentrations of IgG and IgA aCL Abs, along with auto Abs to double stranded DNA and RNP, and developed thrombocytopenia, recurrent abortions and other autoimmune features present in SLE and APS. These data strongly suggest that deregulation of the ubiquitin pathway leads to antibody formation and clinical features of APS [61].
While the above study did not evaluate the development of atherosclerosis in these mice, there have been several studies suggesting that the ubiquitin-proteasome system plays an important role in degradation of oxidized proteins, and that accelerated protein ubiquitination is associated with increased oxidative stress [63; 64]. However, high-levels of oxidative stress can also reduce proteasome activity. In unstable carotid plaques the presence of ubiquitin conjugates is significantly greater compared with stable plaques. The imbalance between an increase in ubiquitin conjugates and a decrease in proteasome activity results in cytotoxic protein aggregates in atherosclerotic plaques that become further oxidized and difficult to unfold or degrade [65].
1.7.2 Stimulation of tolerogenic DCs
In addition to promoting immune activation, DCs may also promote peripheral tolerance and decreased inflammation by inducing regulatory T-cell responses and T cell anergy following exposure to Il-10 [24; 25; 26; 27]. Therefore, DC manipulation to promote tolerance is a potential therapeutic target for treating APS and atherosclerosis. To test this theory in atherosclerosis-prone transgenic mice, DCs from these mice were pulsed with IL-10 and apolipoprotein B-100 (an LDL component) and then incubated with the murine spleen cells. As a result, the DCs induced decreased Th1 and Th2 responses and increased regulatory T cell responses. Furthermore, when hypercholesterolemic mice were injected with DCs that were pre-incubated with IL-10 and apolipoprotein B-100, there was a significant reduction in atherosclerotic plaques in the aorta, decreased CD4 T-cell deposition in the arterial wall and reduced signs of systemic inflammation [20; 66; 67].
Tolerogenic DCs may also induce tolerance to β2-GPI in effector/memory T cells from patients with APS. Human monocyte-derived DCs from 20 APS patients that were treated with IL-10 and transforming growth factor β-1 decreased proliferation of β2-GPI-specific effector/memory CD4 T-cells. Thus, tolerogenic DCs can induce β2-GPI -specific unresponsiveness in APS, perhaps by inducing apoptosis of autoreactive T cells or promoting generation of regulatory T cells [68].
1.8 Conclusions
We reviewed here the evidence that DCs play an important role in sustained production of aPL Abs triggered by endothelial damage in autoimmune diseases and in atherosclerosis. In turn, aPL Abs enhance DC-induced inflammatory responses by working synergistically with TLR and activating the NF-κB signaling pathway, which is important for the pathogenesis of both APS and atherosclerosis. Interactions between DCs and aPL Abs may be enhanced by various mechanisms including glycation, oxidation, and cross-reactivity with bacterial pathogens and oxLDL, providing a possible link between DCs, aPL Abs, APS and atherosclerosis. Genetic or pharmacologic alterations of DCs modulate interactions between DC and aPL Abs, and can either enhance or decrease the development of autoimmune features and accelerated atherosclerosis. However, additional translational studies are needed to understand the significance of these mechanisms in human disease. Further experimental studies should also evaluate the role of other targets of aPL Abs (i.e. phosphatidylserine, phosphatidylethanolamine, lysobisphosphatidic acid, and annexin A5) in atherosclerosis.
References
- 1.Petri M. Epidemiology of the antiphospholipid antibody syndrome. J Autoimmun. 2000;15:145–151. doi: 10.1006/jaut.2000.0409. [DOI] [PubMed] [Google Scholar]
- 2.Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med. 2002;346:752–763. doi: 10.1056/NEJMra002974. [DOI] [PubMed] [Google Scholar]
- 3.Miyakis S, Lockshin MD, Atsumi T, Branch DW, Brey RL, Cervera R, Derksen RH, PG DEG, Koike T, Meroni PL, Reber G, Shoenfeld Y, Tincani A, Vlachoyiannopoulos PG, Krilis SA. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS) J Thromb Haemost. 2006;4:295–306. doi: 10.1111/j.1538-7836.2006.01753.x. [DOI] [PubMed] [Google Scholar]
- 4.Meroni PL, Borghi MO, Raschi E, Tedesco F. Pathogenesis of antiphospholipid syndrome: understanding the antibodies. Nat Rev Rheumatol. 2011;7:330–339. doi: 10.1038/nrrheum.2011.52. [DOI] [PubMed] [Google Scholar]
- 5.Pierangeli SS, Chen PP, Raschi E, Scurati S, Grossi C, Borghi MO, Palomo I, Harris EN, Meroni PL. Antiphospholipid antibodies and the antiphospholipid syndrome: pathogenic mechanisms. Semin Thromb Hemost. 2008;34:236–250. doi: 10.1055/s-0028-1082267. [DOI] [PubMed] [Google Scholar]
- 6.Matsuura E, Shen L, Matsunami Y, Quan N, Makarova M, Geske FJ, Boisen M, Yasuda S, Kobayashi K, Lopez LR. Pathophysiology of beta2-glycoprotein I in antiphospholipid syndrome. Lupus. 2010;19:379–384. doi: 10.1177/0961203310361352. [DOI] [PubMed] [Google Scholar]
- 7.de Groot PG, Urbanus RT. The significance of autoantibodies against beta2-glycoprotein I. Blood. 2012;120:266–274. doi: 10.1182/blood-2012-03-378646. [DOI] [PubMed] [Google Scholar]
- 8.Ames PR, Margarita A, Alves JD. Antiphospholipid antibodies and atherosclerosis: insights from systemic lupus erythematosus and primary antiphospholipid syndrome. Clin Rev Allergy Immunol. 2009;37:29–35. doi: 10.1007/s12016-008-8099-5. [DOI] [PubMed] [Google Scholar]
- 9.Jara LJ, Medina G, Vera-Lastra O. Systemic antiphospholipid syndrome and atherosclerosis. Clin Rev Allergy Immunol. 2007;32:172–177. doi: 10.1007/s12016-007-0008-9. [DOI] [PubMed] [Google Scholar]
- 10.Jara LJ, Medina G, Vera-Lastra O, Shoenfeld Y. Atherosclerosis and antiphospholipid syndrome. Clin Rev Allergy Immunol. 2003;25:79–88. doi: 10.1385/CRIAI:25:1:79. [DOI] [PubMed] [Google Scholar]
- 11.Matsuura E, Lopez LR, Shoenfeld Y, Ames PR. beta2-glycoprotein I and oxidative inflammation in early atherogenesis: A progression from innate to adaptive immunity? Autoimmun Rev. 2012 doi: 10.1016/j.autrev.2012.04.003. [DOI] [PubMed] [Google Scholar]
- 12.McMahon M, Hahn BH. Atherosclerosis and systemic lupus erythematosus: mechanistic basis of the association. Curr Opin Immunol. 2007;19:633–639. doi: 10.1016/j.coi.2007.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Narshi CB, Giles IP, Rahman A. The endothelium: an interface between autoimmunity and atherosclerosis in systemic lupus erythematosus? Lupus. 2011;20:5–13. doi: 10.1177/0961203310382429. [DOI] [PubMed] [Google Scholar]
- 14.Profumo E, Buttari B, Alessandri C, Conti F, Capoano R, Valesini G, Salvati B, Rigano R. Beta2-glycoprotein I is a target of T cell reactivity in patients with advanced carotid atherosclerotic plaques. Int J Immunopathol Pharmacol. 2010;23:73–80. doi: 10.1177/039463201002300107. [DOI] [PubMed] [Google Scholar]
- 15.Weber C, Noels H. Atherosclerosis: current pathogenesis and therapeutic options. Nat Med. 2011;17:1410–1422. doi: 10.1038/nm.2538. [DOI] [PubMed] [Google Scholar]
- 16.Hansson GK. Regulation of immune mechanisms in atherosclerosis. Ann N Y Acad Sci. 2001;947:157–165. discussion 165-6. [PubMed] [Google Scholar]
- 17.Hansson GK. Immune mechanisms in atherosclerosis. Arterioscler Thromb Vasc Biol. 2001;21:1876–1890. doi: 10.1161/hq1201.100220. [DOI] [PubMed] [Google Scholar]
- 18.Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685–1695. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- 19.Virella G, Atchley D, Koskinen S, Zheng D, Lopes-Virella MF. Proatherogenic and proinflammatory properties of immune complexes prepared with purified human oxLDL antibodies and human oxLDL. Clin Immunol. 2002;105:81–92. doi: 10.1006/clim.2002.5269. [DOI] [PubMed] [Google Scholar]
- 20.Keaney JF., Jr Immune modulation of atherosclerosis. Circulation. 2011;124:e559–e560. doi: 10.1161/CIRCULATIONAHA.111.074096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Broder A, Tobin JN, Putterman C. High antiphospholipid antibody levels are associated with statin use and may reflect chronic endothelial damage in non-autoimmune thrombosis: cross-sectional study. J Clin Pathol. 2012;65:551–556. doi: 10.1136/jclinpath-2011-200613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Artenjak A, Lakota K, Frank M, Cucnik S, Rozman B, Bozic B, Shoenfeld Y, Sodin-Semrl S. Antiphospholipid antibodies as non-traditional risk factors in atherosclerosis based cardiovascular diseases without overt autoimmunity. A critical updated review. Autoimmun Rev. 2012 doi: 10.1016/j.autrev.2012.03.002. [DOI] [PubMed] [Google Scholar]
- 23.Rutella S, De Cristofaro R, Ferraccioli G. Function and dysfunction of dendritic cells in autoimmune rheumatic diseases. Hum Immunol. 2009;70:360–373. doi: 10.1016/j.humimm.2009.01.023. [DOI] [PubMed] [Google Scholar]
- 24.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 25.Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, Pulendran B, Palucka K. Immunobiology of dendritic cells. Annu Rev Immunol. 2000;18:767–811. doi: 10.1146/annurev.immunol.18.1.767. [DOI] [PubMed] [Google Scholar]
- 26.Murphy K, Travers P, Walport M, Janeway C. Janeway's immunobiology. New York: Garland Science; 2008. [Google Scholar]
- 27.Steinman RM, Idoyaga J. Features of the dendritic cell lineage. Immunol Rev. 2010;234:5–17. doi: 10.1111/j.0105-2896.2009.00888.x. [DOI] [PubMed] [Google Scholar]
- 28.Ketelhuth DF, Hansson GK. Cellular immunity, low-density lipoprotein and atherosclerosis: break of tolerance in the artery wall. Thromb Haemost. 2011;106:779–786. doi: 10.1160/TH11-05-0321. [DOI] [PubMed] [Google Scholar]
- 29.Levine JS, Subang R, Koh JS, Rauch J. Induction of anti-phospholipid autoantibodies by beta2-glycoprotein I bound to apoptotic thymocytes. J Autoimmun. 1998;11:413–424. doi: 10.1006/jaut.1998.0235. [DOI] [PubMed] [Google Scholar]
- 30.Price BE, Rauch J, Shia MA, Walsh MT, Lieberthal W, Gilligan HM, O'Laughlin T, Koh JS, Levine JS. Anti-phospholipid autoantibodies bind to apoptotic, but not viable, thymocytes in a beta 2-glycoprotein I-dependent manner. J Immunol. 1996;157:2201–2208. [PubMed] [Google Scholar]
- 31.Tolomeo T, Rico De Souza A, Roter E, Dieude M, Amireault P, Subang R, Levine JS, Rauch J. T cells demonstrate a Th1-biased response to native beta2-glycoprotein I in a murine model of anti-phospholipid antibody induction. Autoimmunity. 2009;42:292–295. doi: 10.1080/08916930902828254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rovere P, Manfredi AA, Vallinoto C, Zimmermann VS, Fascio U, Balestrieri G, Ricciardi-Castagnoli P, Rugarli C, Tincani A, Sabbadini MG. Dendritic cells preferentially internalize apoptotic cells opsonized by anti-beta2-glycoprotein I antibodies. J Autoimmun. 1998;11:403–411. doi: 10.1006/jaut.1998.0224. [DOI] [PubMed] [Google Scholar]
- 33.Gaipl US, Beyer TD, Baumann I, Voll RE, Stach CM, Heyder P, Kalden JR, Manfredi A, Herrmann M. Exposure of anionic phospholipids serves as anti-inflammatory and immunosuppressive signal--implications for antiphospholipid syndrome and systemic lupus erythematosus. Immunobiology. 2003;207:73–81. doi: 10.1078/0171-2985-00217. [DOI] [PubMed] [Google Scholar]
- 34.Kuwana M, Matsuura E, Kobayashi K, Okazaki Y, Kaburaki J, Ikeda Y, Kawakami Y. Binding of beta 2-glycoprotein I to anionic phospholipids facilitates processing and presentation of a cryptic epitope that activates pathogenic autoreactive T cells. Blood. 2005;105:1552–1557. doi: 10.1182/blood-2004-08-3145. [DOI] [PubMed] [Google Scholar]
- 35.Bondanza A, Rovere-Querini P, Zimmermann VS, Balestrieri G, Tincani A, Sabbadini MG, Manfredi AA. Requirement for dendritic cells in the establishment of anti-phospholipid antibodies. Autoimmunity. 2007;40:302–306. doi: 10.1080/08916930701356572. [DOI] [PubMed] [Google Scholar]
- 36.Hurst J, Prinz N, Lorenz M, Bauer S, Chapman J, Lackner KJ, von Landenberg P. TLR7 and TLR8 ligands and antiphospholipid antibodies show synergistic effects on the induction of IL-1beta and caspase-1 in monocytes and dendritic cells. Immunobiology. 2009;214:683–691. doi: 10.1016/j.imbio.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 37.Prinz N, Clemens N, Strand D, Putz I, Lorenz M, Daiber A, Stein P, Degreif A, Radsak M, Schild H, Bauer S, von Landenberg P, Lackner KJ. Antiphospholipid antibodies induce translocation of TLR7 and TLR8 to the endosome in human monocytes and plasmacytoid dendritic cells. Blood. 2011;118:2322–2332. doi: 10.1182/blood-2011-01-330639. [DOI] [PubMed] [Google Scholar]
- 38.Kleinbongard P, Heusch G, Schulz R. TNFalpha in atherosclerosis, myocardial ischemia/reperfusion and heart failure. Pharmacol Ther. 2010;127:295–314. doi: 10.1016/j.pharmthera.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 39.Dunoyer-Geindre S, de Moerloose P, Galve-de Rochemonteix B, Reber G, Kruithof EK. NFkappaB is an essential intermediate in the activation of endothelial cells by anti-beta(2)-glycoprotein 1 antibodies. Thromb Haemost. 2002;88:851–857. [PubMed] [Google Scholar]
- 40.Denny MF, Thacker S, Mehta H, Somers EC, Dodick T, Barrat FJ, McCune WJ, Kaplan MJ. Interferon-alpha promotes abnormal vasculogenesis in lupus: a potential pathway for premature atherosclerosis. Blood. 2007;110:2907–2915. doi: 10.1182/blood-2007-05-089086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Buttari B, Profumo E, Capozzi A, Sorice M, Rigano R. Oxidized human beta2-glycoprotein I: its impact on innate immune cells. Curr Mol Med. 2011;11:719–725. doi: 10.2174/156652411798062377. [DOI] [PubMed] [Google Scholar]
- 42.Buttari B, Profumo E, Mattei V, Siracusano A, Ortona E, Margutti P, Salvati B, Sorice M, Rigano R. Oxidized beta2-glycoprotein I induces human dendritic cell maturation and promotes a T helper type 1 response. Blood. 2005;106:3880–3887. doi: 10.1182/blood-2005-03-1201. [DOI] [PubMed] [Google Scholar]
- 43.George J, Harats D, Gilburd B, Afek A, Levy Y, Schneiderman J, Barshack I, Kopolovic J, Shoenfeld Y. Immunolocalization of beta2-glycoprotein I (apolipoprotein H) to human atherosclerotic plaques: potential implications for lesion progression. Circulation. 1999;99:2227–2230. doi: 10.1161/01.cir.99.17.2227. [DOI] [PubMed] [Google Scholar]
- 44.Pamukcu B, Lip GY, Shantsila E. The nuclear factor--kappa B pathway in atherosclerosis: a potential therapeutic target for atherothrombotic vascular disease. Thromb Res. 2011;128:117–123. doi: 10.1016/j.thromres.2011.03.025. [DOI] [PubMed] [Google Scholar]
- 45.Kinev AV, Roubey RA. Tissue factor in the antiphospholipid syndrome. Lupus. 2008;17:952–958. doi: 10.1177/0961203308096662. [DOI] [PubMed] [Google Scholar]
- 46.Buttari B, Profumo E, Capozzi A, Facchiano F, Saso L, Sorice M, Rigano R. Advanced glycation end products of human beta(2) glycoprotein I modulate the maturation and function of DCs. Blood. 2011;117:6152–6161. doi: 10.1182/blood-2010-12-325514. [DOI] [PubMed] [Google Scholar]
- 47.Mohar DS, Barseghian A, Haider N, Domanski M, Narula J. Atherosclerosis in chronic kidney disease: lessons learned from glycation in diabetes. Med Clin North Am. 2012;96:57–65. doi: 10.1016/j.mcna.2011.11.005. [DOI] [PubMed] [Google Scholar]
- 48.Cerami A, Vlassara H, Brownlee M. Protein glycosylation and the pathogenesis of atherosclerosis. Metabolism. 1985;34:37–42. doi: 10.1016/s0026-0495(85)80008-1. [DOI] [PubMed] [Google Scholar]
- 49.Muslin AJ. MAPK signalling in cardiovascular health and disease: molecular mechanisms and therapeutic targets. Clin Sci (Lond) 2008;115:203–218. doi: 10.1042/CS20070430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Liu Z, Cao W. p38 mitogen-activated protein kinase: a critical node linking insulin resistance and cardiovascular diseases in type 2 diabetes mellitus. Endocr Metab Immune Disord Drug Targets. 2009;9:38–46. doi: 10.2174/187153009787582397. [DOI] [PubMed] [Google Scholar]
- 51.Amital H, Govoni M, Maya R, Meroni PL, Ori B, Shoenfeld Y, Tincani A, Trotta F, Sarzi-Puttini P, Atzeni F. Role of infectious agents in systemic rheumatic diseases. Clin Exp Rheumatol. 2008;26:S27–S32. [PubMed] [Google Scholar]
- 52.Lockhart PB, Bolger AF, Papapanou PN, Osinbowale O, Trevisan M, Levison ME, Taubert KA, Newburger JW, Gornik HL, Gewitz MH, Wilson WR, Smith SC, Jr, Baddour LM. Periodontal disease and atherosclerotic vascular disease: does the evidence support an independent association?: a scientific statement from the American Heart Association. Circulation. 2012;125:2520–2544. doi: 10.1161/CIR.0b013e31825719f3. [DOI] [PubMed] [Google Scholar]
- 53.Seymour GJ, Ford PJ, Cullinan MP, Leishman S, Yamazaki K. Relationship between periodontal infections and systemic disease. Clin Microbiol Infect. 2007;13(Suppl 4):3–10. doi: 10.1111/j.1469-0691.2007.01798.x. [DOI] [PubMed] [Google Scholar]
- 54.Schenkein HA, Gunsolley JC, Best AM, Harrison MT, Hahn CL, Wu J, Tew JG. Antiphosphorylcholine antibody levels are elevated in humans with periodontal diseases. Infect Immun. 1999;67:4814–4818. doi: 10.1128/iai.67.9.4814-4818.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Schenkein HA, Best AM, Brooks CN, Burmeister JA, Arrowood JA, Kontos MC, Tew JG. Anti-cardiolipin and increased serum adhesion molecule levels in patients with aggressive periodontitis. J Periodontol. 2007;78:459–466. doi: 10.1902/jop.2007.060305. [DOI] [PubMed] [Google Scholar]
- 56.Barbour SE, Ishihara Y, Fakher M, Al-Darmaki S, Caven TH, Shelburne CP, Best AM, Schenkein HA, Tew JG. Monocyte differentiation in localized juvenile periodontitis is skewed toward the dendritic cell phenotype. Infect Immun. 2002;70:2780–2786. doi: 10.1128/IAI.70.6.2780-2786.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Blank M, Krause I, Fridkin M, Keller N, Kopolovic J, Goldberg I, Tobar A, Shoenfeld Y. Bacterial induction of autoantibodies to beta2-glycoprotein-I accounts for the infectious etiology of antiphospholipid syndrome. J Clin Invest. 2002;109:797–804. doi: 10.1172/JCI12337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vaarala O, Alfthan G, Jauhiainen M, Leirisalo-Repo M, Aho K, Palosuo T. Crossreaction between antibodies to oxidised low-density lipoprotein and to cardiolipin in systemic lupus erythematosus. Lancet. 1993;341:923–925. doi: 10.1016/0140-6736(93)91213-6. [DOI] [PubMed] [Google Scholar]
- 59.Tew JG, El Shikh ME, El Sayed RM, Schenkein HA. Dendritic cells, antibodies reactive with oxLDL, and inflammation. J Dent Res. 2012;91:8–16. doi: 10.1177/0022034511407338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Martin F, Dixit VM. A20 edits ubiquitin and autoimmune paradigms. Nat Genet. 2011;43:822–823. doi: 10.1038/ng.916. [DOI] [PubMed] [Google Scholar]
- 61.Kool M, van Loo G, Waelput W, De Prijck S, Muskens F, Sze M, van Praet J, Branco-Madeira F, Janssens S, Reizis B, Elewaut D, Beyaert R, Hammad H, Lambrecht BN. The ubiquitin-editing protein A20 prevents dendritic cell activation, recognition of apoptotic cells, and systemic autoimmunity. Immunity. 2011;35:82–96. doi: 10.1016/j.immuni.2011.05.013. [DOI] [PubMed] [Google Scholar]
- 62.Musone SL, Taylor KE, Nititham J, Chu C, Poon A, Liao W, Lam ET, Ma A, Kwok PY, Criswell LA. Sequencing of TNFAIP3 and association of variants with multiple autoimmune diseases. Genes Immun. 2011;12:176–182. doi: 10.1038/gene.2010.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shang F, Gong X, Taylor A. Activity of ubiquitin-dependent pathway in response to oxidative stress. Ubiquitin-activating enzyme is transiently up-regulated. J Biol Chem. 1997;272:23086–23093. doi: 10.1074/jbc.272.37.23086. [DOI] [PubMed] [Google Scholar]
- 64.Grune T, Reinheckel T, Davies KJ. Degradation of oxidized proteins in mammalian cells. FASEB J. 1997;11:526–534. [PubMed] [Google Scholar]
- 65.Versari D, Herrmann J, Gossl M, Mannheim D, Sattler K, Meyer FB, Lerman LO, Lerman A. Dysregulation of the ubiquitin-proteasome system in human carotid atherosclerosis. Arterioscler Thromb Vasc Biol. 2006;26:2132–2139. doi: 10.1161/01.ATV.0000232501.08576.73. [DOI] [PubMed] [Google Scholar]
- 66.Hermansson A, Johansson DK, Ketelhuth DF, Andersson J, Zhou X, Hansson GK. Immunotherapy with tolerogenic apolipoprotein B-100-loaded dendritic cells attenuates atherosclerosis in hypercholesterolemic mice. Circulation. 2011;123:1083–1091. doi: 10.1161/CIRCULATIONAHA.110.973222. [DOI] [PubMed] [Google Scholar]
- 67.Hjerpe C, Johansson D, Hermansson A, Hansson GK, Zhou X. Dendritic cells pulsed with malondialdehyde modified low density lipoprotein aggravate atherosclerosis in Apoe(−/−) mice. Atherosclerosis. 2010;209:436–441. doi: 10.1016/j.atherosclerosis.2009.10.003. [DOI] [PubMed] [Google Scholar]
- 68.Torres-Aguilar H, Blank M, Kivity S, Misgav M, Luboshitz J, Pierangeli SS, Shoenfeld Y. Tolerogenic dendritic cells inhibit antiphospholipid syndrome derived effector/memory CD4(+) T cell response to beta2GPI. Ann Rheum Dis. 2012;71:120–128. doi: 10.1136/annrheumdis-2011-200063. [DOI] [PubMed] [Google Scholar]