Abstract
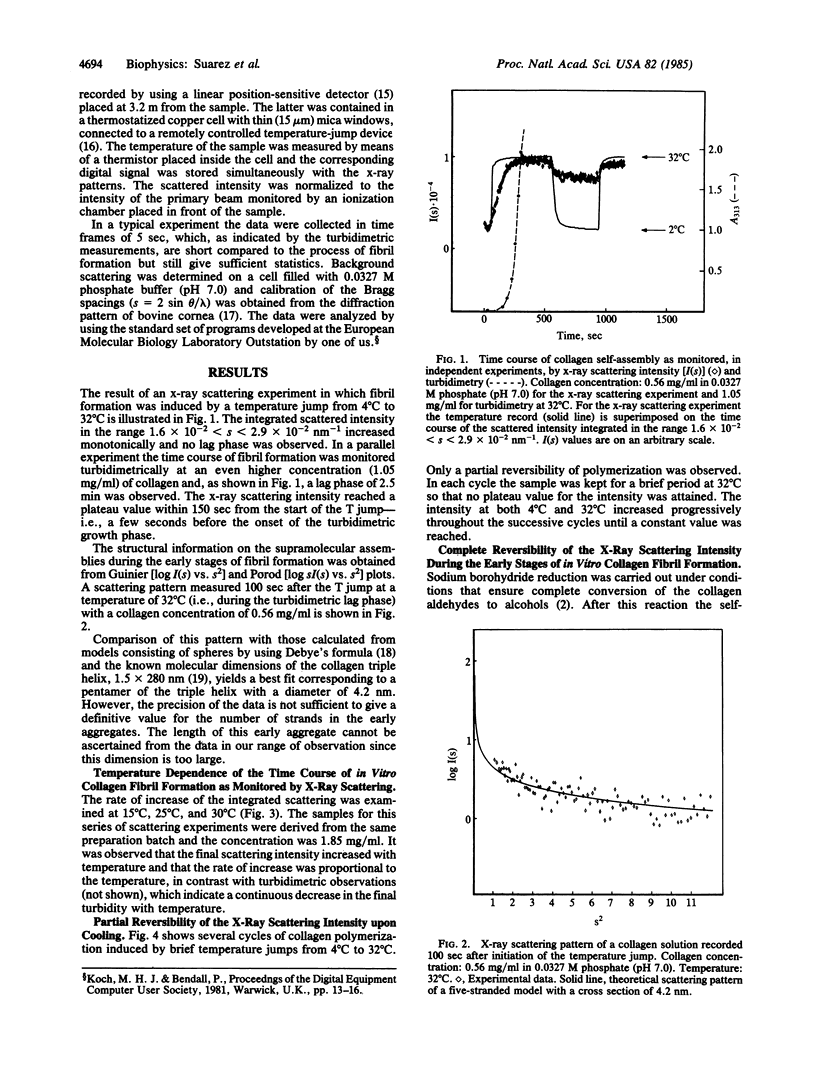
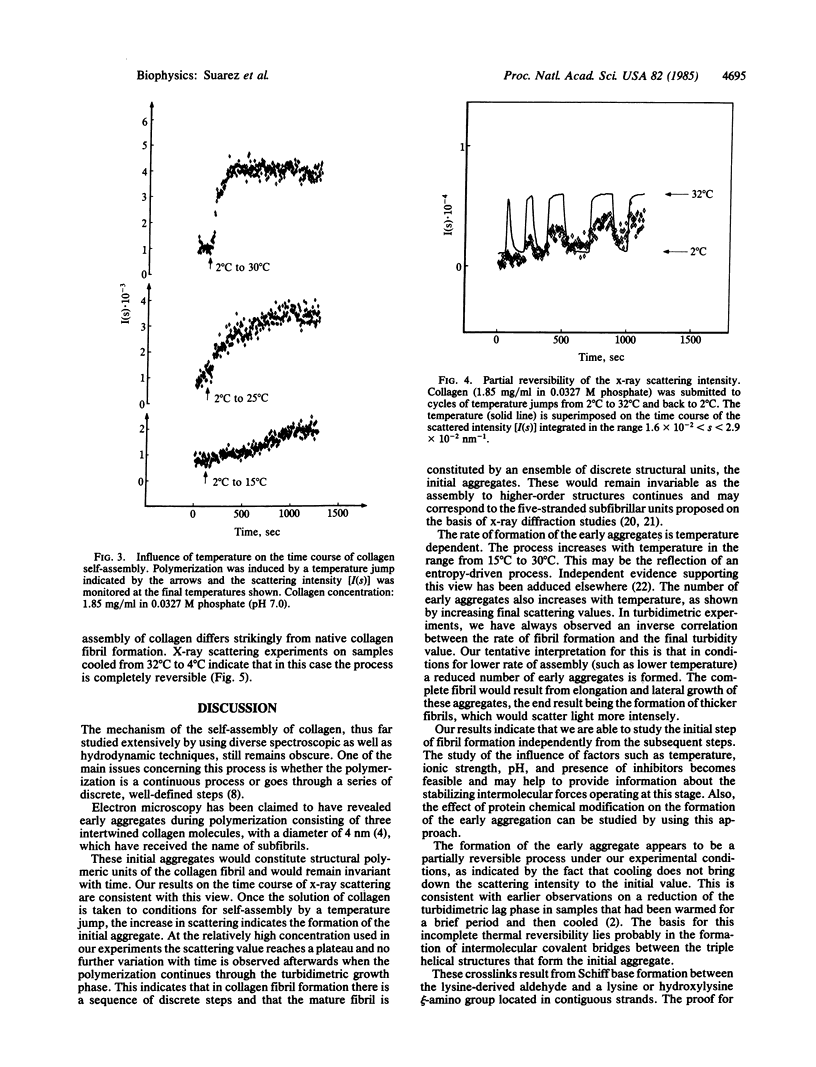
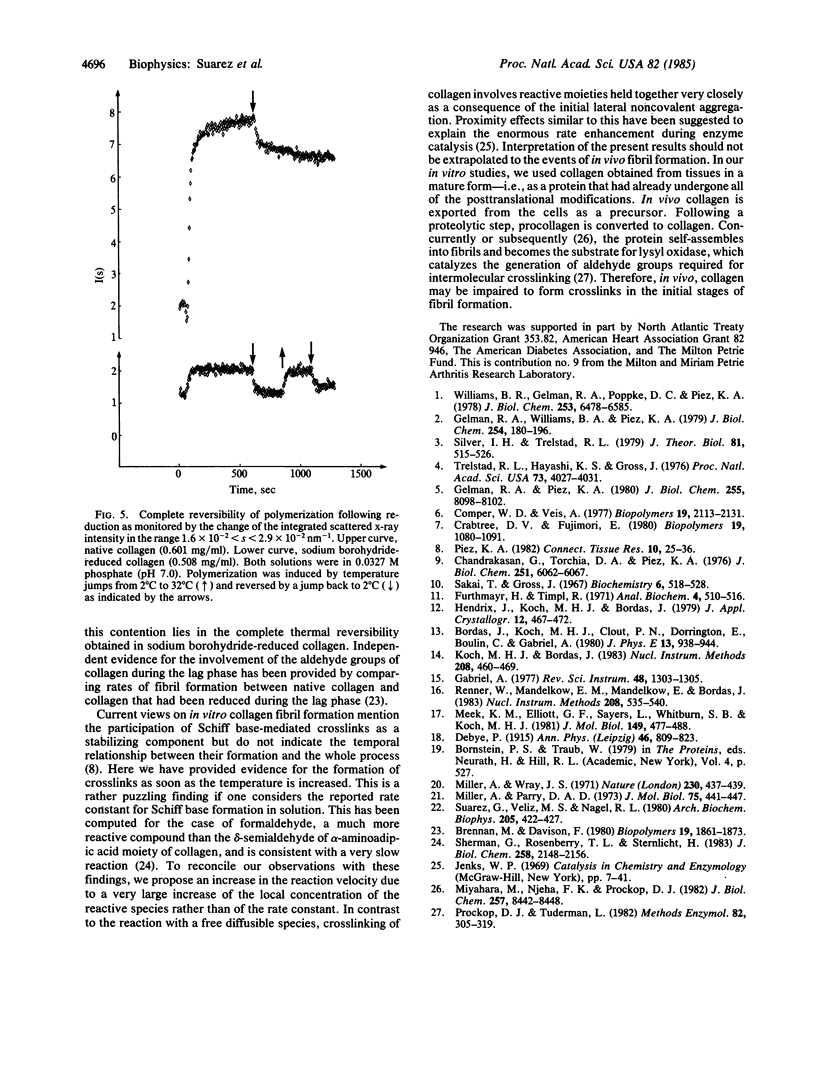
The time course of in vitro collagen fibril formation was monitored by synchrotron radiation x-ray scattering. Collagen polymerization was induced by a temperature jump from 4 degrees C to 32 degrees C and the solution scattering pattern was recorded continuously with a time resolution of a few seconds. The scattered intensity increased as soon as the final temperature was attained, without discernible lag phase, when the collagen concentration was about 0.56 mg/ml, whereas turbidimetric measurements revealed a lag phase of ca. 2.5 min at 1.05 mg/ml. A direct correlation was found between the temperature and both the rate of formation and the total amount of early aggregates. The formation of these aggregates was only partially reversed by lowering the temperature, except when the collagen had been prevented from forming Schiff base-mediated intermolecular crosslinks by sodium borohydride reduction. In this case, formation of aggregates was completely reversible. We conclude that the aggregates that appear to correspond to the subfibrils proposed on the basis of independent methods are formed and simultaneously crosslinked in the early phases of in vitro collagen self-assembly.
Full text
PDF
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brennan M., Davison P. F. Role of aldehydes in collagen fibrillogenesis in vitro. Biopolymers. 1980 Oct;19(10):1861–1873. doi: 10.1002/bip.1980.360191013. [DOI] [PubMed] [Google Scholar]
- Chandrakasan G., Torchia D. A., Piez K. A. Preparation of intact monomeric collagen from rat tail tendon and skin and the structure of the nonhelical ends in solution. J Biol Chem. 1976 Oct 10;251(19):6062–6067. [PubMed] [Google Scholar]
- Comper W. D., Veis A. The mechanism of nucleation for in vitro collagen fibril formation. Biopolymers. 1977 Oct;16(10):2113–2131. doi: 10.1002/bip.1977.360161004. [DOI] [PubMed] [Google Scholar]
- Crabtree D. V., Fujimori E. Fluorescent-labeled collagen: age-related differences and fluorescence changes during the lag phase of fibril formation. Biopolymers. 1980 May;19(5):1081–1091. doi: 10.1002/bip.1980.360190511. [DOI] [PubMed] [Google Scholar]
- Furthmayr H., Timpl R. Characterization of collagen peptides by sodium dodecylsulfate-polyacrylamide electrophoresis. Anal Biochem. 1971 Jun;41(2):510–516. doi: 10.1016/0003-2697(71)90173-4. [DOI] [PubMed] [Google Scholar]
- Gelman R. A., Piez K. A. Collagen fibril formation in vitro. A quasielastic light-scattering study of early stages. J Biol Chem. 1980 Sep 10;255(17):8098–8102. [PubMed] [Google Scholar]
- Gelman R. A., Williams B. R., Piez K. A. Collagen fibril formation. Evidence for a multistep process. J Biol Chem. 1979 Jan 10;254(1):180–186. [PubMed] [Google Scholar]
- Meek K. M., Elliott G. F., Sayers Z., Whitburn S. B., Koch M. H. Interpretation of the meridional x-ray diffraction pattern from collagen fibrils in corneal stroma. J Mol Biol. 1981 Jul 5;149(3):477–488. doi: 10.1016/0022-2836(81)90482-4. [DOI] [PubMed] [Google Scholar]
- Miller A., Parry D. A. Structure and packing of microfibrils in collagen. J Mol Biol. 1973 Apr 5;75(2):441–447. doi: 10.1016/0022-2836(73)90035-1. [DOI] [PubMed] [Google Scholar]
- Miyahara M., Njieha F. K., Prockop D. J. Formation of collagen fibrils in vitro by cleavage of procollagen with procollagen proteinases. J Biol Chem. 1982 Jul 25;257(14):8442–8448. [PubMed] [Google Scholar]
- Piez K. A. Structure and assembly of the native collagen fibril. Connect Tissue Res. 1982;10(1):25–36. doi: 10.3109/03008208209034403. [DOI] [PubMed] [Google Scholar]
- Prockop D. J., Tuderman L. Posttranslational enzymes in the biosynthesis of collagen: extracellular enzymes. Methods Enzymol. 1982;82(Pt A):305–319. doi: 10.1016/0076-6879(82)82068-5. [DOI] [PubMed] [Google Scholar]
- Sakai T., Gross J. Some properties of the products of reaction of tadpole collagenase with collagen. Biochemistry. 1967 Feb;6(2):518–528. doi: 10.1021/bi00854a021. [DOI] [PubMed] [Google Scholar]
- Sherman G., Rosenberry T. L., Sternlicht H. Identification of lysine residues essential for microtubule assembly. Demonstration of enhanced reactivity during reductive methylation. J Biol Chem. 1983 Feb 25;258(4):2148–2156. [PubMed] [Google Scholar]
- Silver F. H., Trelstad R. L. Linear aggregation and the turbidimetric lag phase: type I collagen fibrillogenesis in vitro. J Theor Biol. 1979 Dec 7;81(3):515–526. doi: 10.1016/0022-5193(79)90049-3. [DOI] [PubMed] [Google Scholar]
- Suarez G., Veliz M., Nagel R. L. Role of hydrophobic interactions in collagen fibril formation: effect of alkylureas in vitro. Arch Biochem Biophys. 1980 Dec;205(2):422–427. doi: 10.1016/0003-9861(80)90125-3. [DOI] [PubMed] [Google Scholar]
- Trelstad R. L., Hayashi K., Gross J. Collagen fibrillogenesis: intermediate aggregates and suprafibrillar order. Proc Natl Acad Sci U S A. 1976 Nov;73(11):4027–4031. doi: 10.1073/pnas.73.11.4027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams B. R., Gelman R. A., Poppke D. C., Piez K. A. Collagen fibril formation. Optimal in vitro conditions and preliminary kinetic results. J Biol Chem. 1978 Sep 25;253(18):6578–6585. [PubMed] [Google Scholar]


