Abstract
As a young medical resident, I encountered a patient suffering from spontaneous coronary vasospasm and was puzzled by these dramatic alterations in vasomotion. This encounter piqued my interest in understanding the drivers of vascular reactivity. In a paper published in the JCI, my colleagues and I revealed a role for superoxide production in the vascular dysfunction associated with hypercholesterolemia. Subsequent work by our group and others has unveiled complex associations between ROS generation and vascular disease.
While a second-year internal medicine resident on the Duke coronary care unit in the 1970s, I admitted a young lady who was having repeated episodes of chest pain and profound ST-segment elevation on her electrocardiogram. During several of these episodes, she developed ventricular fibrillation that required cardioversion. An emergency cardiac catheterization revealed that her coronary arteries were overtly normal, but her left anterior descending coronary artery spontaneously developed spasm to the point of closure. Thankfully, these episodes eventually resolved. She obviously had variant angina, and I was impressed with the dramatic nature of her illness and the idea that diseases could alter vasomotion in such a striking fashion.
An experimental model and search for mechanisms
After my residency and cardiology fellowship, I accepted a postdoctoral research fellowship under the tutelage of Melvin Marcus, Allyn Mark, and Frank Abboud at the University of Iowa. There, I met Donald Heistad and Mark Armstrong, who were studying the effects of cholesterol and atherosclerosis on vascular function. Don had made the exciting discovery that hypercholesterolemia altered vascular reactivity. Specifically, he demonstrated that administration of serotonin, normally a vasodilator, paradoxically evokes severe vascular spasm in cholesterol-fed monkeys (1). Other groups made similar observations in cholesterol-fed rabbits (2). These findings were reminiscent of my patient described above. Shortly before Don’s report, Robert Furchgott discovered that agonists, including acetylcholine, bradykinin, serotonin, and vasopressin, promote vasodilation by stimulating endothelium-derived relaxing factor (EDRF) release (3, 4); however, these same agonists evoked vasoconstriction following removal of the endothelium by mechanical abrasion. In discussions with Don and Mark, we hypothesized that the endothelium of cholesterol-fed monkeys was deficient in EDRF production. During a stint in Paul Vanhoutte’s laboratory at the Mayo Clinic, I learned to perform isometric tension studies of isolated vessels and established this method at the University of Iowa. Don and Mark provided iliac arteries from cholesterol-fed and control animals, and we demonstrated that atherosclerotic vessels had severely blunted endothelium-dependent vasodilation and sporadically demonstrated paradoxical vasoconstriction, much like the coronary artery of my patient at Duke (5). Others confirmed that hypercholesterolemia alters endothelium-dependent vasodilation (6), which was a fascinating result with clinical implications; however, the mechanisms responsible for hypercholesterolemia-associated changes in endothelium-dependent vasodilation were unclear.
Identifying a role of ROS
Our understanding of disease-dependent changes in EDRF production was advanced by the nearly concurrent observations that the EDRF was NO (7) and that the EDRF could be inactivated by superoxide (8). Because NO and superoxide react with one another at a near-diffusion-limited rate (9), we hypothesized that the hypercholesterolemia-associated defect in endothelium-dependent vasodilation was due to oxidative inactivation of NO by superoxide. At the time (late 1980s), apolipoprotein E–null mice, which develop severe hypercholesterolemia, were not available; therefore, we used cholesterol-fed rabbits as a model of hypercholesterolemia and early atherosclerosis. Fortunately, around this time I met Jim Bates, a faculty member in the Department of Anesthesia at the University of Iowa, who contributed substantially to these experiments (Figure 1). We used ozone-linked chemiluminescence to measure NO release from isolated perfused vascular segments and observed that vessels from cholesterol-fed rabbits produced NO, but it was largely released in the oxidized form of nitrite (NO2–) (10). Furthermore, cholesterol-fed rabbits treated with the superoxide scavenger polyethylene-glycolated superoxide dismutase exhibited improved endothelium-dependent vasodilation (11). Together, our results strongly suggested superoxide involvement in hypercholesterolemia-associated vascular dysfunction and in other molecular events in atherosclerosis.
Figure 1. Collaboration.

David Harrison (left), David Ku (center), and James Bates (right) at a Keystone Conference, circa 1990.
We next established methods to measure superoxide production by intact vessels. This was challenging, because many commonly used assays to study isolated enzymes or chemical reactions were either insensitive or subject to interference. After exploring several techniques, we settled on chemiluminescence-based methods to study intact vessels, and Dr. Yuichi Ohara, a postdoctoral fellow in our laboratory at the time, was instrumental in adapting this technique. We were surprised to discover that vessels from cholesterol-fed rabbits produced three times more superoxide than did normal vessels (12). Strikingly, removal of the vessel endothelium completely normalized superoxide production. Thus, hypercholesterolemia stimulated superoxide production in the very cells that were synthesizing NO, leading to NO oxidation prior to leaving the endothelium. A consequence of this reaction is formation of the strong oxidant peroxynitrite (OONO-), which oxidizes LDL, depletes antioxidants, and leads to the formation of isoprostanes. The isoprostanes are particularly interesting. These compounds, discovered in the early 1990s by my current colleague Jack Roberts (13), are potent vasoconstrictors and could therefore contribute to abnormal vasomotion in conditions of excessive oxidation. ROS are also implicated in many aspects of vascular disease, including lipid oxidation (14), adhesion molecule expression (15), platelet aggregation (16), thrombosis, matrix remodeling, and plaque rupture (17). Thus, superoxide production by the endothelium not only affects vasomotor tone, but also contributes to lesion formation.
Subsequent studies and complexity
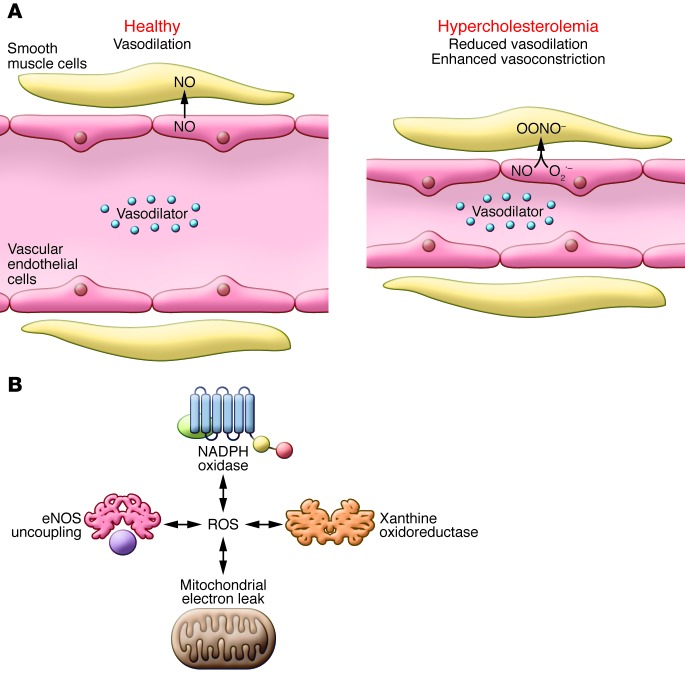
Our discoveries led to a simple concept (Figure 2A): hypercholesterolemia stimulates an increase in superoxide, which oxidizes NO, preventing endothelium-dependent vasodilation and in some cases unmasking vasoconstriction. The phrase, “reduced NO bioavailability” became commonly used in reference to conditions in which NO is produced but biologically unavailable through oxidation. Subsequent work from many groups expanded this concept, and it is now clear that as atherosclerosis progresses, plaque-associated and vascular smooth muscle cells produce ROS (18, 19). We initially found that oxypurinol reduced vascular superoxide production and endothelium-dependent vasodilation, suggesting a role for xanthine oxidase in early atherosclerosis (12). Subsequently, we learned that additional sources of ROS are important in vascular diseases, including the NADPH oxidases, uncoupled NOSs, and mitochondria. Our laboratory and others have shown that these various sources are interdependent (Figure 2B), and ROS from one source can activate ROS production by others in a feed-forward fashion (20–22). The NADPH oxidases appear to lead these interactions. For example, NADPH oxidase–produced ROS can oxidize tetrahydrobiopterin, an NOS cofactor, and this event uncouples these enzymes, resulting in the production of superoxide rather than NO (23). My colleague, Hua Cai, revealed that hydrogen peroxide inhibits the expression of dihydrofolate reductase, an enzyme involved in maintaining cellular levels of tetrahydrobiopterin, leading to eNOS uncoupling (24). Furthermore, NADPH oxidase–associated ROS disrupt mitochondrial electron transfer, leading to ROS production (25). The feed-forward nature of ROS production is recapitulated in several pathophysiological states (20).
Figure 2. Evolving concepts of vascular ROS production.
(A) Healthy vascular ECs respond to the presence of vasodilators by producing NO, which diffuses into smooth muscles and promotes vasodilation. Under hypercholesterolemia conditions, vascular ECs produce superoxide in addition to NO in response to vasodilators. Superoxide and NO interact within vascular ECs to produce OONO–, which is a strong oxidant that contributes to vascular dysfunction in individuals with hypercholesterolemia. (B) Interplay between various sources of ROS in vascular cells. NADPH oxidase–produced ROS can oxidize the eNOS cofactor tetrahydrobiopterin (purple), which uncouples these enzymes, resulting in ROS production. NADPH oxidase–associated ROS also disrupt mitochondrial electron transfer, leading to ROS production. Furthermore, ROS from other sources promote the formation of ROS by xanthine oxidoreductase. This feed-forward nature of ROS production promotes several pathophysiological states.
It should be noted that all methods for measuring superoxide and related ROS are imperfect. Dr. Sergey Dikalov joined our group and helped us adapt additional methods for ROS detection and quantification, including electron spin resonance, HPLC to monitor oxidation of dihydroethidium, and cytochrome c reduction, as well as hydrogen peroxide–detecting methods. Use of these methods confirmed our initial chemiluminescence-based measurements and proved extremely useful.
Clinical hurdles and therapeutic implications
Several large clinical trials analyzing the use of antioxidant vitamins in cardiovascular disease have failed to show benefit. In fact, high doses of vitamin E seem to be harmful (26). We now understand that ROS act as signaling molecules and are essential for cell growth and survival. Hydrogen peroxide released from the endothelial mitochondria acts as a hyperpolarizing factor to mediate vasodilation (27). Because ROS production can be highly localized and have different roles in various subcellular compartments, treatment with nonspecific antioxidants has proven problematic. An alternative approach has been to develop specific NADPH oxidase (NOX) inhibitors. Recently, Gray and colleagues demonstrated that hyperglycemia activates NOX1 (28), and deletion of Nox1 in mice prevented the diabetes-associated acceleration of atherosclerosis. Excitingly, Gray et al. determined that pharmacological inhibition of NOX1 attenuates lesion formation in hypercholesterolemic mice (28). Mitochondria-targeted antioxidants have proven beneficial in experimental hypertension and type 2 diabetes, perhaps by specifically targeting pathological ROS in mitochondria, without disrupting ROS signaling at other cellular sites (29).
In the years since our initial observations, it has become clear that the etiology of coronary spasm is complicated and involves perturbed endothelial NO production, oxidative injury, inflammation, and enhanced vascular smooth muscle constriction. Other causes of endothelial dysfunction in addition to NO oxidation have been described. Nevertheless, the experience of caring for a patient with coronary spasm left a lasting impression and attracted me to this area of research.
Acknowledgments
I would like to thank Donald Heistad and Allyn Mark for their editorial critique and suggestions.
Footnotes
Conflict of interest: The author has declared that no conflict of interest exists.
Citation for this article: J Clin Invest. 2014;124(2):473–475. doi:10.1172/JCI70336.
References
- 1.Heistad DD, Armstrong ML, Marcus ML, Piegors DJ, Mark AL. Augmented responses to vasoconstrictor stimuli in hypercholesterolemic and atherosclerotic monkeys. Circ Res. 1984;54(6):711–718. doi: 10.1161/01.RES.54.6.711. [DOI] [PubMed] [Google Scholar]
- 2.Henry PD, Yokoyama M. Supersensitivity of atherosclerotic rabbit aorta to ergonovine. Mediation by a serotonergic mechanism. J Clin Invest. 1980;66(2):306–313. doi: 10.1172/JCI109858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Furchgott RF, Zawadzki JV. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980;288(5789):373–376. doi: 10.1038/288373a0. [DOI] [PubMed] [Google Scholar]
- 4.Cherry PD, Furchgott RF, Zawadzki JV, Jothianandan D. Role of endothelial cells in relaxation of isolated arteries by bradykinin. Proc Natl Acad Sci U S A. 1982;79(6):2106–2110. doi: 10.1073/pnas.79.6.2106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Freiman PC, Mitchell GG, Heistad DD, Armstrong ML, Harrison DG. Atherosclerosis impairs endothelium-dependent vascular relaxation to acetylcholine and thrombin in primates. Circ Res. 1986;58(6):783–789. doi: 10.1161/01.RES.58.6.783. [DOI] [PubMed] [Google Scholar]
- 6.Bossaller C, Yamamoto H, Lichtlen PR, Henry PD. Impaired cholinergic vasodilation in the cholesterol-fed rabbit in vivo. Basic Res Cardiol. 1987;82(4):396–404. doi: 10.1007/BF01907027. [DOI] [PubMed] [Google Scholar]
- 7.Palmer RM, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987;327(6122):524–526. doi: 10.1038/327524a0. [DOI] [PubMed] [Google Scholar]
- 8.Rubanyi GM, Vanhoutte PM. Superoxide anions and hyperoxia inactivate endothelium-derived relaxing factor. Am J Physiol. 1986;250(5 pt 2):H822–H827. doi: 10.1152/ajpheart.1986.250.5.H822. [DOI] [PubMed] [Google Scholar]
- 9.Huie RE, Padmaja S. The reaction of NO with superoxide. Free Radic Res Commun. 1993;18(4):195–199. doi: 10.3109/10715769309145868. [DOI] [PubMed] [Google Scholar]
- 10.Minor RL, Jr, Myers PR, Guerra R, Jr, Bates JN, Harrison DG. iet-induced atherosclerosis increases the release of nitrogen oxides from rabbit aorta. J Clin Invest. 1990;86(6):2109–2116. doi: 10.1172/JCI114949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mugge A, Elwell JH, Peterson TE, Hofmeyer TG, Heistad DD, Harrison DG. Chronic treatment with polyethylene-glycolated superoxide dismutase partially restores endothelium-dependent vascular relaxations in cholesterol-fed rabbits. Circ Res. 1991;69(5):1293–1300. doi: 10.1161/01.RES.69.5.1293. [DOI] [PubMed] [Google Scholar]
- 12.Ohara Y, Peterson TE, Harrison DG. Hypercholesterolemia increases endothelial superoxide anion production. J Clin Invest. 1993;91(6):2546–2551. doi: 10.1172/JCI116491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Morrow JD, Awad JA, Boss HJ, Blair IA, Roberts LJ 2nd. Non-cyclooxygenase-derived prostanoids (F2-isoprostanes) are formed in situ on phospholipids. Proc Natl Acad Sci U S A. 1992;89(22):10721–10725. doi: 10.1073/pnas.89.22.10721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chisolm GM 3rd, Hazen SL, Fox PL, Cathcart MK. The oxidation of lipoproteins by monocytes-macrophages. Biochemical and biological mechanisms. J Biol Chem. 1999;274(37):25959–25962. doi: 10.1074/jbc.274.37.25959. [DOI] [PubMed] [Google Scholar]
- 15.Cayatte AJ, et al. S17834, a new inhibitor of cell adhesion and atherosclerosis that targets nadph oxidase. Arterioscler Thromb Vasc Biol. 2001;21(10):1577–1584. doi: 10.1161/hq1001.096723. [DOI] [PubMed] [Google Scholar]
- 16.Siess W. Platelet interaction with bioactive lipids formed by mild oxidation of low-density lipoprotein. Pathophysiol Haemost Thromb. 2006;35(3–4):292–304. doi: 10.1159/000093222. [DOI] [PubMed] [Google Scholar]
- 17.Rajagopalan S, Meng XP, Ramasamy S, Harrison DG, Galis ZS. Reactive oxygen species produced by macrophage-derived foam cells regulate the activity of vascular matrix metalloproteinases in vitro. Implications for atherosclerotic plaque stability. J Clin Invest. 1996;98(11):2572–2579. doi: 10.1172/JCI119076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miller FJ, Jr, Gutterman DD, Rios CD, Heistad DD, Davidson BL. Superoxide production in vascular smooth muscle contributes to oxidative stress and impaired relaxation in atherosclerosis. Circ Res. 1998;82(12):1298–1305. doi: 10.1161/01.RES.82.12.1298. [DOI] [PubMed] [Google Scholar]
- 19.Guzik TJ, et al. Calcium-dependent NOX5 nicotinamide adenine dinucleotide phosphate oxidase contributes to vascular oxidative stress in human coronary artery disease. J Am Coll Cardiol. 2008;52(22):1803–1809. doi: 10.1016/j.jacc.2008.07.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rathore R, et al. Hypoxia activates NADPH oxidase to increase [ROS]i and [Ca2+]i through the mitochondrial ROS-PKCepsilon signaling axis in pulmonary artery smooth muscle cells. Free Radic Biol Med. 2008;45(9):1223–1231. doi: 10.1016/j.freeradbiomed.2008.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee SB, Bae IH, Bae YS, Um HD. Link between mitochondria and NADPH oxidase 1 isozyme for the sustained production of reactive oxygen species and cell death. J Biol Chem. 2006;281(47):36228–36235. doi: 10.1074/jbc.M606702200. [DOI] [PubMed] [Google Scholar]
- 22.Landmesser U, et al. Angiotensin II induces endothelial xanthine oxidase activation: role for endothelial dysfunction in patients with coronary disease. Arterioscler Thromb Vasc Biol. 2007;27(4):943–948. doi: 10.1161/01.ATV.0000258415.32883.bf. [DOI] [PubMed] [Google Scholar]
- 23.Landmesser U, et al. Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J Clin Invest. 2003;111(8):1201–1209. doi: 10.1172/JCI14172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chalupsky K, Cai H. Endothelial dihydrofolate reductase: critical for nitric oxide bioavailability and role in angiotensin II uncoupling of endothelial nitric oxide synthase. Proc Natl Acad Sci U S A. 2005;102(25):9056–9061. doi: 10.1073/pnas.0409594102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Doughan AK, Harrison DG, Dikalov SI. Molecular mechanisms of angiotensin II-mediated mitochondrial dysfunction: linking mitochondrial oxidative damage and vascular endothelial dysfunction. Circ Res. 2008;102(4):488–496. doi: 10.1161/CIRCRESAHA.107.162800. [DOI] [PubMed] [Google Scholar]
- 26.Miller ER, et al. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005;142(1):37–46. doi: 10.7326/0003-4819-142-1-200501040-00110. [DOI] [PubMed] [Google Scholar]
- 27.Liu Y, Bubolz AH, Mendoza S, Zhang DX, Gutterman DD. H2O2 is the transferrable factor mediating flow-induced dilation in human coronary arterioles. Circ Res. 2011;108(5):566–573. doi: 10.1161/CIRCRESAHA.110.237636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gray SP, et al. NADPH oxidase 1 plays a key role in diabetes mellitus-accelerated atherosclerosis. Circulation. 2013;127(18):1888–1902. doi: 10.1161/CIRCULATIONAHA.112.132159. [DOI] [PubMed] [Google Scholar]
- 29.Dikalova AE, et al. Therapeutic targeting of mitochondrial superoxide in hypertension. Circ Res. 2010;107(1):106–116. doi: 10.1161/CIRCRESAHA.109.214601. [DOI] [PMC free article] [PubMed] [Google Scholar]