Abstract
Testosterone is the most abundant circulating androgen, and can be converted to dihydrotestosterone (DHT), a more potent androgen, by the 5α-reductase enzymes in target tissues. Current treatments for prostate cancer consist of reducing androgen levels by chemical or surgical castration or pure antiandrogen therapy that directly targets the androgen receptor (AR). Although these therapies reduce tumor burden and AR activity, the cancer inevitably recurs within 18–30 months. An approach targeting the androgen–AR axis at different levels could, therefore, improve the efficacy of prostate cancer therapy. Inhibition of 5α-reductase is one such approach; however, the two largest trials to investigate the use of the 5α-reductase inhibitors (5ARIs) finasteride and dutasteride in patients with prostate cancer have shown that, although the incidence of cancer was reduced by 5ARI treatment, those cancers that were detected were more aggressive than in patients treated with placebo. Thus, the best practice for using these drugs to prevent and treat prostate cancer remains unclear.
Introduction
Prostate cancer is the most frequently diagnosed cancer and the third most common cause of cancer-related deaths among men in developed countries.1 Prostate cancer-related deaths have declined over the past decade, owing to improved methods for early detection and diagnosis and more-effective therapeutic strategies.
Deregulation of the androgen–androgen receptor (AR) pathway is a hallmark of prostate cancer.2,3 Testosterone, the most abundant circulating androgen, is converted to dihydrotestosterone (DHT), which has a greater affinity for the AR than testosterone, by the 5α-reductase isoenzymes.4–7 During embryogenesis and throughout adulthood, androgens mediate the development, growth and maintenance of the male genitalia and secondary sexual characteristics.6 In addition to their importance in normal physiology, androgens also have a key role in the genesis and progression of diseases such as benign prostatic hyperplasia (BPH) and prostate cancer.8–10
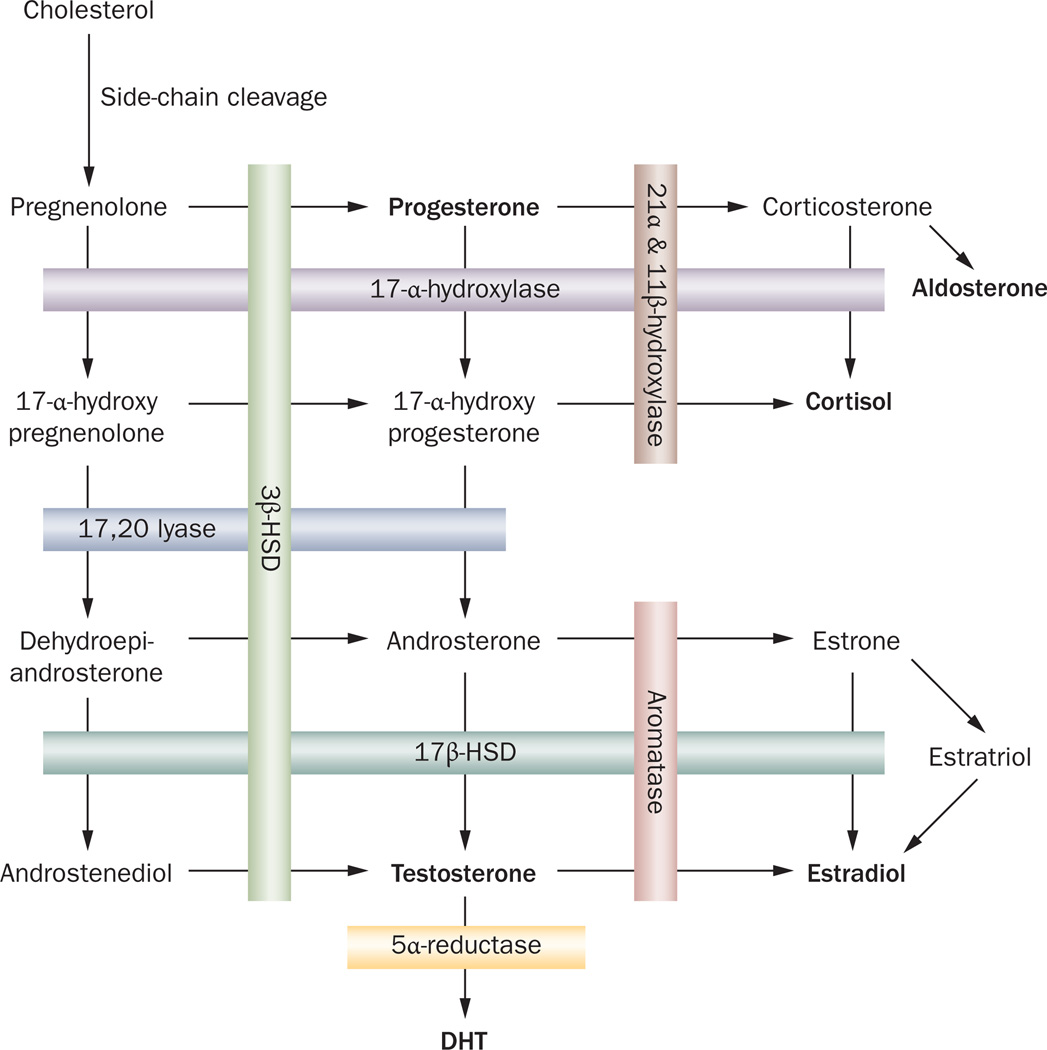
The steroid biosynthetic pathway involves the sequential enzymatic modification of the common precursor cholesterol to generate androgens, estrogens, progestogens and corticosteroids (Figure 1).11 Androgens—19-carbon compounds that form a subset within the steroid biosynthetic pathway—control development, growth and maintenance of male sexual characteristics.6,11 Testosterone is synthesized in the testis by the Leydig cells under the control of luteinizing hormone (LH) from the pituitary gland, internalized in prostate cells by passive diffusion, and converted to DHT by the 5α-reductase isoenzymes. The proposed mechanism of conversion of testosterone to DHT requires a reducing cofactor that will act as a hydride donor to the testosterone. For 5α-reductase, the cofactor is membrane-bound nicotinamide dinucleotide phosphate (NADPH). 5α-reductase forms a complex with NADPH that interacts with the substrate forming a ternary complex. The hydride from NADPH is transferred to carbon-5 of the aromatic ring, forming DHT. Once DHT is released, the 5α-reductase–NADP− binary complex dissociates, and the enzyme can catalyze a new reaction.12
Figure 1. The steroidogenesis pathway.
The common precursor cholesterol is sequentially modified by the enzymes to synthesize the steroid hormones. Testosterone is converted to its more potent form, DHT, by the 5α-reductase enzymes. Abbreviations: DHT, dihydrotestosterone; HSD, hydroxysteroid dehydrogenase.
There are three isoforms of the 5α-reductase enzymes, encoded by different genes and with differential expression patterns. The type 1 isoform is encoded by a gene on chromosome 5 and is expressed primarily in skin and liver.13,14 The gene encoding type 2 5α-reductase is on chromosome 2 and is expressed predominantly in stromal and basal epithelial cells of the prostate.13–15 Deficiency of type 2 5α-reductase, but not type 1, results in a lack of development of accessory sex organs.16,17 Interestingly, in prostate cancer, expression of both of these isoforms is increased, which could contribute to the enlargement of the organ.18,19 The type 3 5α-reductase isoenzyme is ubiquitously expressed in androgenic and nonandrogenic tissues, and elevated levels are found in prostate cancer cell lines.20–22 Type 3 5α-reductase reduces polyprenols to dolichols, which have a role in N-linked protein glycosylation, an essential post-translational modification to target proteins for secretion or membrane localization.23 No work has been published on the function of type 3 5α-reductase in prostate cancer.
Both testosterone and DHT are natural ligands of the AR, a member of the steroid nuclear receptor super-family. Although the association rates of androgens to the AR are similar, DHT dissociates from the AR at a much slower rate, resulting in a more stable and active DHT–AR complex. DHT is, therefore, a more potent activator of AR than its precursor, testosterone.24–26 Indeed, animal models lacking 5α-reductase enzymes and humans with a mutation in the 5α-reductase gene (resulting in an inactive enzyme) display impaired development of male sexual organs, highlighting the importance of DHT synthesis in male physiology.6,27,28
This Review will summarize the current knowledge of the role of testosterone and DHT in prostate disease and discuss the rationale for using treatments that target the synthesis of DHT in the treatment of prostate cancer. We will also bring together the data from the two largest trials of 5α-reductase inhibitors (5ARIs) in prostate cancer, and consider the future of these drugs in clinical practice.
The androgen receptor
Until it is activated by androgens, the AR is localized in the cytoplasm. After binding to testosterone or DHT, AR undergoes a conformational modification that enables it to translocate into the nucleus.29–31 In the nucleus, AR forms homodimers and interacts with nucleotide-specific sequences on the DNA of target genes, known as androgen response elements. The AR–DNA interaction recruits the transcriptional machinery that is necessary to modulate expression of genes involved in cell proliferation, survival and death, or genes that code for secretory proteins such as PSA. This makes PSA a useful biochemical tool to determine AR activity and assess the responsiveness of prostate cancer to treatment.32–34
The androgen receptor axis in prostate cancer
The relevance of androgens in the genesis and progression of prostate cancer was first described 70 years ago by Charles Huggins and Clarence Hodges,35 and because of their findings, clinical or surgical castration is the primary treatment for locally advanced prostate cancer.36 These therapies target the synthesis of the most abundant circulating androgens: testosterone and DHT. Chemical inhibition of androgen synthesis with precursor homologs, either alone or in combination with surgical castration by orchiectomy and radiation, is a primary treatment for locally advanced prostate cancer. These treatments result in a reduction in circulating androgens, a decrease in PSA levels (as an indicator of AR activity), and reduced tumor burden. However, after a period of 18–30 months, the tumor recurs and the disease progresses to become castration-resistant prostate cancer (CRPC).37–39
Although CRPC cells are resistant to the low levels of circulating androgens, basic and clinical evidence supports the paradigm that a functional AR is required for tumor growth and survival. AR functionality is achieved by several mechanisms.40–42 In CRPC, levels of AR are elevated compared to less-advanced tumors, with amplification of the AR gene and increased synthesis of the AR protein.43,44 Increased levels of the AR can, therefore, maximize the effect of the low androgen levels in the cell. In addition, gain-in-function point mutations in AR have been described. These mutations enable the AR to strongly bind to natural ligands or to interact with other steroids (such as adrenal androgens), leading to AR activation and promotion of cell growth, proliferation and survival.3,41,44
Intratumoral androgenesis
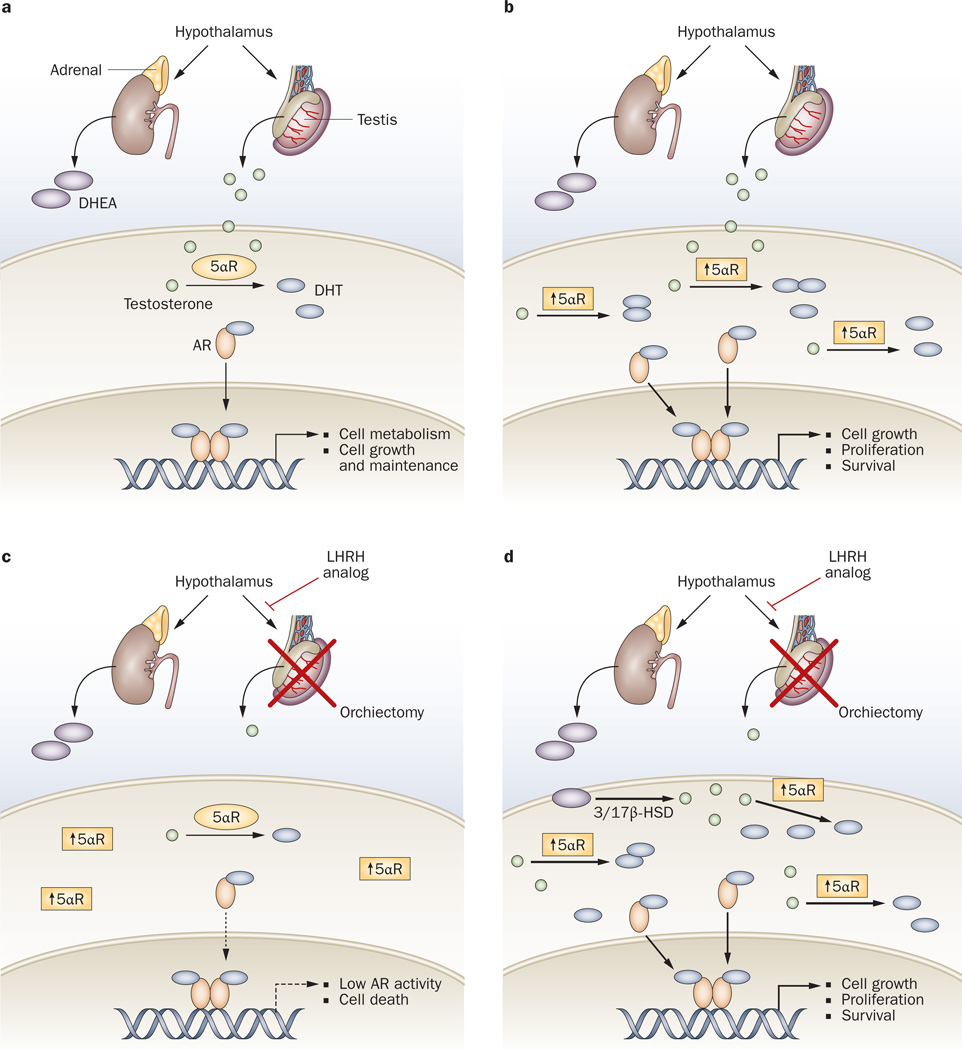
Testosterone is synthesized primarily in the testis (90–95%), with the remaining 5–10% produced from dehydroepiandrosterone (DHEA) released by the adrenal glands.5 Hormone ablation and castration therapies reduce circulating testosterone levels by up to 97%,45,46 but despite the low levels of circulating testosterone, intraprostatic androgen levels have been shown to be reduced by only 50% and 61% in two independent reports.47–50 In order to compensate for the low levels of circulating androgen, prostate cancer cells take advantage of the androgen precursor DHEA, which is synthesized and released by the adrenal glands in response to stimulation with ACTH (Figure 2). As prostate cancer cells express the enzymes necessary to convert DHEA to testosterone and then to DHT,51–53 CRPC cells can synthesize sufficient DHT to activate AR.54–56 Thus, de novo androgenesis could be an important mechanism leading to, or strongly contributing to, progression to CRPC and metastasis. Targeting the enzymes 5α-reductase or CYP17, key players in the intratumoral androgenesis, with specific inhibitors such as 5ARI or abiraterone, remains an attractive concept for the development of therapeutics to block AR activation via intratumoral production of DHT.
Figure 2. The role of DHT in different stages of prostate development and cancer.
a | During prostate development and homeostasis, testosterone diffuses into the prostate cell and is converted to DHT by 5α-reductase, which, via the AR, causes increased cell growth and metabolism. b | In locally advanced prostate cancer, increased activity of 5α-reductase results in an increase in DHT levels, which increases transcription of genes involved in growth, proliferation and survival of cancer cells. c | In patients undergoing castration therapy for prostate cancer, androgen depletion is achieved by chemical or surgical castration therapy, hormone analogues or orchiectomy. The resulting decrease in circulating testosterone levels results in lower intratumoral DHT levels, and, therefore, reduced AR activity and cancer cell death. d | In castration-resistant prostate cancer, DHT levels increase once again, as tumor cells express the enzymes required for DHT synthesis from adrenal DHEA. This generates a paracrine system that partially restores intratumoral DHT levels, resulting in a further wave of cancer cell proliferation and survival. Abbreviations: 3/17β-HSD, 3β-hydroxysteroid dehydrogenase and 17β-hydroxysteroid dehydrogenase; 5αR, 5α-reductase; AR, androgen receptor; DHEA, dehydroepiandrosterone; DHT, dihydrotestosterone; LHRH, luteinizing hormone-releasing hormone.
Inhibition of DHT synthesis
A functional androgen–AR pathway is required for the survival of CRPC cells, so blockade of DHT biosynthesis is of particular interest as a therapeutic target. The nonspecific inhibitor ketoconazole has been shown to abolish androgen synthesis, and it was postulated to be a useful therapeutic agent.57 However, although PSA and androgen levels initially decrease with ketoconazole therapy, the effect eventually reverses. Furthermore, ketoconazole is associated with several adverse effects such as hyponatremia, hyperkalemia, symptomatic adrenal insufficiency, hypothyroidism, lethargy and myelotoxicity.57,58 Abiraterone is a more specific inhibitor that irreversibly blocks the 17α-hydroxylase and C17,20-lyase enzymes.59,60 67% of patients with CRPC treated with abiraterone exhibit a 50% reduction in PSA levels.61 Adverse effects of abiraterone include hypertension, hypokalemia and fluid retention, and are due to the build-up of precursors (such as progesterone and 17α-progesterone) that are converted to corticosteroids.61–64
Inhibition of the 5α-reductase isoenzymes for prostate cancer prevention and treatment is particularly interesting, as all of the 5α-reductase enzyme isoforms are present in prostate cancer at elevated levels, correlating with the high DHT levels detected in prostate cancer tissue.65,66 Furthermore, 5ARIs have already been proven to be beneficial for the treatment of BPH.67–69 Finasteride and dutasteride are the two drugs that are currently being used in clinical practice to inhibit 5α-reductase.70–72 Both finasteride and dutasteride act as competitive substrates, forming strong ternary complexes with the 5α-reductase–NADPH complex, and are, therefore, considered to be irreversible inhibitors of the enzyme.12 Finasteride only inhibits 5α-reductase type 2. 5α-reductase converts finasteride to dihydrofinasteride, and, as it does not have an electron to transfer to NADP, a highly stable bisubstrate complex of dihydrofinasteride–NADP is formed with the enzyme, making it a potent inhibitor.73,74 However, finasteride is not a prodrug, because it does not need to be modified to block 5α-reductase. Finasteride binds the enzyme and forms a potent ternary complex (finasteride–NADP–5α-reductase). Although finasteride is modified by 5α-reductase, the modification is not required for it to bind and inhibit the enzyme. Unlike finasteride, dutasteride can inhibit both isoforms of 5α-reductase,75,76 and forms a ternary complex between the enzyme and NADPH, which exhibits a low dissociation constant, strongly inhibiting the enzyme.77
Treatment of prostate cancer cell lines with either finasteride or dutasteride results in the inhibition of cellular pathways involved in cell growth and proliferation, and cellular metabolism, and an increase in the activity of proapoptotic pathways.78–81 Interestingly, gene expression profiles of cells treated with dutasteride revealed expression of unconventional AR target genes, such as CASP7, CASP8, BNIP3, CDK8 and Skp2.79,81 It is possible, therefore, that 5ARIs affect other transcription factors, altering signaling pathways involved in cell growth and proliferation, apoptosis, metabolism or cell adhesion.81–83 Understanding these pathways could lead to the discovery of new therapeutic targets.
5α-reductase inhibitors in prostate cancer
The effects of finasteride and dutasteride have been intensively studied in men with BPH, who have elevated expression of 5α-reductase type 1 and type 2.12,84 Treatment with finasteride or dutasteride, alone or in combination with selective α1-adrenoceptor blockers such as doxazosin, terazosin or tamsulosin, resulted in a reduction in prostate volume and a decrease in urinary retention and urinary tract symptoms.71,72,85 Considering that deregulation of androgen–AR pathways is a major contributor to prostate cancer progression, it is reasonable to speculate that 5ARIs would also have beneficial effects in prostate cancer.
Clinical trials
The Prostate Cancer Prevention Trial
The Prostate Cancer Prevention Trial (PCPT)86 was the first large-scale study to investigate the role of finasteride in the context of prostate cancer development. The trial studied 18,882 men with a PSA level <3.0 ng/ml and a normal digital rectal exam (DRE). Patients were randomly assigned to receive daily doses of 5 mg finasteride or placebo. Finasteride treatment reduced the incidence of prostate cancer compared to the placebo group (18.4% versus 24.4%).86 Although the difference seems small, the number could be underestimated, as the characteristics of prostate cancer and the technicalities of performing biopsies (which are dependent on tumor size, prostate size and the number of biopsy cores in the sample)87,88 mean that the probability of detecting a tumor in a small prostate is higher than in a normal-sized prostate from patients within the placebo group. Treatment with 5ARIs decreased prostate size and increased the sensitivity of PSA and DRE for cancer detection.88 Furthermore, complications such as urinary tract infections (UTIs) were more common in the placebo group.86 Controversially, tumors found in patients in the finasteride group were of a higher grade than the tumors from patients in the placebo group. Gleason scores between 7 and 10 were found in 6.4% of the tumors in the finasteride group, and in only 5.1% of the placebo group. This observation still cannot be fully explained, although it is in accord with evidence suggesting that tumor grade inversely correlates with low androgen levels.89 It is also possible that finasteride has little or no effect on more aggressive tumors with high Gleason scores, and as 5ARI treatment causes the prostates of patients from this group to shrink, any tumor that is present can be detected more easily than in patients receiving placebo.
The REDUCE trial
Finasteride only inhibits the type 2 isoform of 5α-reductase, meaning that the type 1 isoform, which is also highly expressed in prostate cancer, remains active. It is, therefore, reasonable to consider that levels of DHT in the tumor could remain sufficiently high to affect the pathology of tumors in the finasteride group. The REDUCE trial was designed to study the effect of inhibiting both the type 1 and type 2 5α-reductase isoforms in a population at high risk of prostate cancer.6,88,90
Unlike the PCPT, the REDUCE trial studied patients with serum PSA levels of 2.5–10 ng/ml who had a negative biopsy at least 6 months before the study.88 Patients received either placebo or a daily dose of 0.5 mg dutasteride, with end points set at 2 and 4 years. In the first 2 years there were fewer tumors of Gleason score 5–7 in the dutasteride-treated group compared to those treated with placebo (12.9% versus 16.7%), but the number of tumors with Gleason scores 8–10 did not differ between groups. However, during years 3 and 4, tumors with Gleason score 8–10 were more common in the dutasteride-treated group than in the placebo group, with 12 cases and 1 case, respectively. It remains unclear whether the difference was due to the dutasteride treatment or the fact that, during the second year, a larger proportion of patients were removed from the control group of the study owing to detection of cancer, which could compensate for the difference in the number of more aggressive tumors observed at years 3 and 4. The REDUCE study showed an overall reduction in the number of tumors with Gleason score 5–6 in the group of patients receiving dutasteride versus placebo (19.9% compared to 25.1%, respectively). In addition, patients receiving dutasteride also benefitted from a reduction in BPH, as well as a decrease in urinary retention and incidence of UTIs.
5α-reductase in the clinic
The development of drugs that directly target the AR has proved to be useful for the treatment of prostate cancer. The use of AR antagonists or competitive agonists, such as flutamide, bicalutamide or nilutamide, shows a considerable benefit in blocking AR activity and decreasing tumor size and growth. However, these treatments can also lead to the development of CRPC. The affinity of these inhibitors for the AR is lower than that of DHT, and as intratumoral androgen synthesis increases, DHT out-competes the inhibitor, resulting in reactivation of the AR axis and progression to CRPC. It is, therefore, clear that we need to adopt multiple therapeutic approaches to target the activation of AR at different levels, including the use of 5ARIs to block conversion of testosterone to DHT.
The PCPT and REDUCE trials, both of which showed that finasteride and dutasteride decrease the risk of developing prostate cancer, support the importance of targeting DHT for therapeutic use. However, the additional finding of high Gleason score tumors in patients treated with 5ARIs raises some controversies regarding how these drugs should be used clinically. In that respect, other clinical trials are currently underway on the use of dutasteride in prostate cancer. The ARTS (Avodart® After Radical Therapy for Prostate Cancer) study91 is a phase II trial, comparing the use of 0.5 mg dutasteride versus placebo in patients with localized tumors and increasing PSA levels after either radical prostatectomy or radiotherapy. The study end point is PSA doubling after 2 years of treatment, and disease progression according to PSA levels and doubling time of ≤3 months or metastatic disease. The REDEEM (Reduction by Dutasteride of Clinical Progression Events in Expectant Management)92 trial is evaluating the use of 0.5 mg dutasteride daily in delaying the progression of prostate cancer from a localized to a more aggressive form. The patients enrolled in the study all have low-risk and low-grade localized tumors.
Other studies involve treatment with dutasteride in combination with other agents. The TARP (Therapy Assessed by Rising PSA)93 study is designed to compare the effect of dutasteride in combination with bicalutamide versus placebo and bicalut-amide in patients with CRPC. Another phase II study of 57 patients with CRPC has shown that addition of dutasteride to ketoconazole or hydrocortisone induces a decrease in PSA levels and an increase in median progression-free survival.94,95 Although DHT is the most potent activator of AR axis, some point mutations in AR make the tumor cells more responsive to stimulation by other steroids, including testosterone. Thus, concomitant treatment with drugs such as abiraterone may have additional beneficial effects. Neither the American Urological Association (AUA) nor the American Society of Clinical Oncology (ASCO) have produced guidelines for the use of these drugs in clinical practice; and instead, they suggest discussing the risks of using 5ARIs for treatment and prevention with the patient on a case-by-case basis.
Conclusions
Inappropriate activation of the androgen–AR axis is a major factor in the progression of prostate cancer. Since the initial discovery of the androgenic pathway in the 1940s, many advances have enhanced our understanding of androgen metabolism and the role of the AR in progression of prostatic diseases, and, based on this research, effective therapeutic approaches to treat BPH and prostate cancer have been implemented.
Prostate cancer represents both a health-related and a socioeconomic problem for today’s society, yet we still do not have an effective therapy that can control prostate cancer progression without the development of CRPC. This problem highlights the need for novel therapies. The use of 5ARIs for prevention and treatment of prostate cancer was a logical step, given the positive effects of these drugs in patients with BPH. Results from the clinical trials of finasteride and dutasteride are encouraging, but the possible adverse effects are worrying, emphasizing the need to further investigate the role of androgens in the prostate and their systemic effects.
Nevertheless, 5α-reductase inhibition does seem to have beneficial effects on prostate cancer incidence, and the role of these drugs in combination with other agents should be further evaluated for the treatment of CRPC.
Key points.
-
▪
Activation of the androgen receptor (AR) is crucial for tumor cell progression and survival of prostate cancer, and androgen deprivation therapy remains the main clinical approach in men with locally advanced tumors
-
▪
Current therapies incompletely suppress the androgen–AR axis, but a multiple therapeutic approach, targeting androgens and their receptor, has potential to improve clinical outcomes
-
▪
Treatment of prostate cancer cells with 5α-reductase inhibitors (5ARIs) inhibits cellular pathways regulating metabolism, cell growth and proliferation, triggering apoptosis and decreasing prostate size
-
▪
Although 5ARI treatment reduces the risk of developing prostate cancer, patients treated with these drugs have tumors with higher Gleason scores than those who receive placebo
-
▪
Use of 5ARIs to prevent and treat prostate cancer remains controversial, and further investigation is necessary to understand the presence of more-aggressive tumors in patients receiving these drugs
Review criteria.
Literature for this review was selected by searching PubMed for the following keywords: “prostate cancer”, “castration”, “androgen receptor”, “5 alpha-reductase”, “finasteride”, “dutasteride” and “androgenesis” alone or in combination. All papers selected were full text and in English. No limitations were used for year or period of publication. A number of references were identified from the reference lists of included papers.
Acknowledgments
The authors receive support from NIH grants CA121277, CA91956, and CA125747. D. J. Tindall receives support from the T. J. Martell Foundation, grant DK65236 and L. P. Nacusi is the recipient of the Mayo Clinic Endocrinology, Diabetes, and Metabolism training grant.
Footnotes
Competing interests
The authors declare no competing interests.
Author contributions
Both authors contributed equally to discussions of content, writing and editing of this article.
References
- 1.Jemal A, et al. Global cancer statistics. CA Cancer J. Clin. 2011;61:69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 2.Lange CA, Gioeli D, Hammes SR, Marker PC. Integration of rapid signaling events with steroid hormone receptor action in breast and prostate cancer. Annu. Rev. Physiol. 2007;69:171–199. doi: 10.1146/annurev.physiol.69.031905.160319. [DOI] [PubMed] [Google Scholar]
- 3.Nacusi LP, Tindall DJ. Androgen receptor abnormalities in castration-recurrent prostate cancer. Expert Rev. Endocrinol. Metab. 2009;4:417–422. doi: 10.1586/eem.09.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Russell DW, Wilson JD. Steroid 5 alpha-reductase: two genes/two enzymes. Annu. Rev. Biochem. 1994;63:25–61. doi: 10.1146/annurev.bi.63.070194.000325. [DOI] [PubMed] [Google Scholar]
- 5.Labrie F. Adrenal androgens and intracrinology. Semin. Reprod. Med. 2004;22:299–309. doi: 10.1055/s-2004-861547. [DOI] [PubMed] [Google Scholar]
- 6.Andriole G, et al. Dihydrotestosterone and the prostate: the scientific rationale for 5 alpha-reductase inhibitors in the treatment of benign prostatic hyperplasia. J. Urol. 2004;172:1399–1403. doi: 10.1097/01.ju.0000139539.94828.29. [DOI] [PubMed] [Google Scholar]
- 7.Imamoto T, et al. The role of testosterone in the pathogenesis of prostate cancer. Int. J. Urol. 2008;15:472–480. doi: 10.1111/j.1442-2042.2008.02074.x. [DOI] [PubMed] [Google Scholar]
- 8.Chatterjee B. The role of the androgen receptor in the development of prostatic hyperplasia and prostate cancer. Mol. Cell Biochem. 2003;253:89–101. doi: 10.1023/a:1026057402945. [DOI] [PubMed] [Google Scholar]
- 9.Tiwari A, Krishna NS, Nanda K, Chugh A. Benign prostatic hyperplasia: an insight into current investigational medical therapies. Expert Opin. Investig. Drugs. 2005;14:1359–1372. doi: 10.1517/13543784.14.11.1359. [DOI] [PubMed] [Google Scholar]
- 10.Dehm SM, Tindall DJ. Molecular regulation of androgen action in prostate cancer. J. Cell Biochem. 2006;99:333–344. doi: 10.1002/jcb.20794. [DOI] [PubMed] [Google Scholar]
- 11.Penning TM. New frontiers in androgen biosynthesis and metabolism. Curr. Opin. Endocrinol. Diabetes Obes. 2010;17:233–239. doi: 10.1097/MED.0b013e3283381a31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Aggarwal S, Thareja S, Verma A, Bhardwaj TR, Kumar M. An overview on 5 alpha-reductase inhibitors. Steroids. 2010;75:109–153. doi: 10.1016/j.steroids.2009.10.005. [DOI] [PubMed] [Google Scholar]
- 13.Imperato-McGinley J, Sanchez RS, Spencer JR, Yee B, Vaughan ED. Comparison of the effects of the 5 alpha-reductase inhibitor finasteride and the antiandrogen flutamide on prostate and genital differentiation: dose-response studies. Endocrinology. 1992;131:1149–1156. doi: 10.1210/endo.131.3.1324152. [DOI] [PubMed] [Google Scholar]
- 14.Pelletier G, Luu-The V, Huang XF, Lapointe H, Labrie F. Localization by in situ hybridization of steroid 5 alpha-reductase isozyme gene expression in the human prostate and preputial skin. J. Urol. 1998;160:577–582. [PubMed] [Google Scholar]
- 15.Rittmaster RS. 5 alpha-reductase inhibitors. J. Androl. 1997;18:582–587. [PubMed] [Google Scholar]
- 16.Silver RI, Russell DW. 5 alpha-reductase type 2 mutations are present in some boys with isolated hypospadias. J. Urol. 1999;162:1142–1145. doi: 10.1016/S0022-5347(01)68102-3. [DOI] [PubMed] [Google Scholar]
- 17.Walsh PC, et al. Familial incomplete male pseudohermaphroditism, type 2. Decreased dihydrotestosterone formation in pseudovaginal perineoscrotal hypospadias. N. Engl. J. Med. 1974;291:944–949. doi: 10.1056/NEJM197410312911806. [DOI] [PubMed] [Google Scholar]
- 18.Iehle C, et al. Differences in steroid 5 alpha-reductase iso-enzymes expression between normal and pathological human prostate tissue. J. Steroid Biochem. Mol. Biol. 1999;68:189–195. doi: 10.1016/s0960-0760(99)00030-8. [DOI] [PubMed] [Google Scholar]
- 19.Wilson JD, Griffin JE, Russell DW. Steroid 5 alpha-reductase 2 deficiency. Endocr. Rev. 1993;14:577–593. doi: 10.1210/edrv-14-5-577. [DOI] [PubMed] [Google Scholar]
- 20.Tamura K, et al. Molecular features of hormone-refractory prostate cancer cells by genome-wide gene expression profiles. Cancer Res. 2007;67:5117–5125. doi: 10.1158/0008-5472.CAN-06-4040. [DOI] [PubMed] [Google Scholar]
- 21.Uemura M, et al. Novel 5 alpha-steroid reductase (SRD5A3, type-3) is overexpressed in hormone-refractory prostate cancer. Cancer Sci. 2008;99:81–86. doi: 10.1111/j.1349-7006.2007.00656.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cantagrel V, et al. SRD5A3 is required for converting polyprenol to dolichol and is mutated in a congenital glycosylation disorder. Cell. 2010;142:203–217. doi: 10.1016/j.cell.2010.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li X, Chen C, Singh SM, Labrie F, Labrie F. The enzyme and inhibitors of 4-ene-3-oxosteroid 5 alpha-oxidoreductase. Steroids. 1995;60:430–441. doi: 10.1016/0039-128x(95)00021-h. [DOI] [PubMed] [Google Scholar]
- 24.Tindall DJ, Rittmaster RS. The rationale for inhibiting 5 alpha-reductase isoenzymes in the prevention and treatment of prostate cancer. J. Urol. 2008;179:1235–1242. doi: 10.1016/j.juro.2007.11.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhou ZX, Lane MV, Kemppainen JA, French FS, Wilson EM. Specificity of ligand-dependent androgen receptor stabilization: receptor domain interactions influence ligand dissociation and receptor stability. Mol. Endocrinol. 1995;9:208–218. doi: 10.1210/mend.9.2.7776971. [DOI] [PubMed] [Google Scholar]
- 26.Wright AS, Thomas LN, Douglas RC, Lazier CB, Rittmaster RS. Relative potency of testosterone and dihydrotestosterone in preventing atrophy and apoptosis in the prostate of the castrated rat. J. Clin. Invest. 1996;98:2558–2563. doi: 10.1172/JCI119074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wright AS, Douglas RC, Thomas LN, Lazier CB, Rittmaster RS. Androgen-induced regrowth in the castrated rat ventral prostate: role of 5 alpha-reductase. Endocrinology. 1999;140:4509–4515. doi: 10.1210/endo.140.10.7039. [DOI] [PubMed] [Google Scholar]
- 28.Wilson JD. The role of 5 alpha-reduction in steroid hormone physiology. Reprod. Fertil. Dev. 2001;13:673–678. doi: 10.1071/rd01074. [DOI] [PubMed] [Google Scholar]
- 29.Pratt WB, Toft DO. Steroid receptor interactions with heat shock protein and immunophilin chaperones. Endocr. Rev. 1997;18:306–360. doi: 10.1210/edrv.18.3.0303. [DOI] [PubMed] [Google Scholar]
- 30.Smith DF, Toft DO. Minireview: the intersection of steroid receptors with molecular chaperones: observations and questions. Mol. Endocrinol. 2008;22:2229–2240. doi: 10.1210/me.2008-0089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Elbi C, et al. Molecular chaperones function as steroid receptor nuclear mobility factors. Proc. Natl Acad. Sci. USA. 2004;101:2876–2881. doi: 10.1073/pnas.0400116101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Agoulnik IU, Weigel NL. Co-activator selective regulation of androgen receptor activity. Steroids. 2009;74:669–674. doi: 10.1016/j.steroids.2009.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Heemers HV, Tindall DJ. Androgen receptor (AR) coregulators: a diversity of functions converging on and regulating the AR transcriptional complex. Endocr. Rev. 2007;28:778–808. doi: 10.1210/er.2007-0019. [DOI] [PubMed] [Google Scholar]
- 34.Heemers HV, Tindall DJ. Unraveling the complexities of androgen receptor signaling in prostate cancer cells. Cancer Cell. 2009;15:245–247. doi: 10.1016/j.ccr.2009.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Huggins C, Hodges CV. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. 1941. J. Urol. 2002;168:9–12. doi: 10.1016/s0022-5347(05)64820-3. [DOI] [PubMed] [Google Scholar]
- 36.Harris WP, Mostaghel EA, Nelson PS, Montgomery B. Androgen deprivation therapy: progress in understanding mechanisms of resistance and optimizing androgen depletion. Nat. Clin. Pract. Urol. 2009;6:76–85. doi: 10.1038/ncpuro1296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Crawford ED, et al. A controlled trial of leuprolide with and without flutamide in prostatic carcinoma. N. Engl. J. Med. 1989;321:419–424. doi: 10.1056/NEJM198908173210702. [DOI] [PubMed] [Google Scholar]
- 38.Denis L. European Organization for Research and Treatment of Cancer (EORTC) prostate cancer trials, 1976–1996. Urology. 1998;51:50–57. doi: 10.1016/s0090-4295(98)00076-4. [DOI] [PubMed] [Google Scholar]
- 39.Eisenberger MA, et al. Bilateral orchiectomy with or without flutamide for metastatic prostate cancer. N. Engl. J. Med. 1998;339:1036–1042. doi: 10.1056/NEJM199810083391504. [DOI] [PubMed] [Google Scholar]
- 40.Agoulnik IU, Weigel NL. Androgen receptor action in hormone-dependent and recurrent prostate cancer. J. Cell Biochem. 2006;99:362–372. doi: 10.1002/jcb.20811. [DOI] [PubMed] [Google Scholar]
- 41.Debes JD, Tindall DJ. Mechanisms of androgen-refractory prostate cancer. N. Engl. J. Med. 2004;351:1488–1490. doi: 10.1056/NEJMp048178. [DOI] [PubMed] [Google Scholar]
- 42.Dehm SM, Tindall DJ. Regulation of androgen receptor signaling in prostate cancer. Expert Rev. Anticancer Ther. 2005;5:63–74. doi: 10.1586/14737140.5.1.63. [DOI] [PubMed] [Google Scholar]
- 43.Taplin ME. Drug insight: role of the androgen receptor in the development and progression of prostate cancer. Nat. Clin. Pract. Oncol. 2007;4:236–244. doi: 10.1038/ncponc0765. [DOI] [PubMed] [Google Scholar]
- 44.Pienta KJ, Bradley D. Mechanisms underlying the development of androgen-independent prostate cancer. Clin. Cancer Res. 2006;12:1665–1671. doi: 10.1158/1078-0432.CCR-06-0067. [DOI] [PubMed] [Google Scholar]
- 45.Oefelein MG. Time to normalization of serum testosterone after 3-month luteinizing hormone-releasing hormone agonist administered in the neoadjuvant setting: implications for dosing schedule and neoadjuvant study consideration. J. Urol. 1998;160:1685–1688. [PubMed] [Google Scholar]
- 46.Tran C, et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science. 2009;324:787–790. doi: 10.1126/science.1168175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Belanger B, et al. Comparison of residual C-19 steroids in plasma and prostatic tissue of human, rat and guinea pig after castration: unique importance of extratesticular androgens in men. J. Steroid Biochem. 1989;32:695–698. doi: 10.1016/0022-4731(89)90514-1. [DOI] [PubMed] [Google Scholar]
- 48.Mizokami A, et al. The adrenal androgen androstenediol is present in prostate cancer tissue after androgen deprivation therapy and activates mutated androgen receptor. Cancer Res. 2004;64:765–771. doi: 10.1158/0008-5472.can-03-0130. [DOI] [PubMed] [Google Scholar]
- 49.Nishiyama T, Hashimoto Y, Takahashi K. The influence of androgen deprivation therapy on dihydrotestosterone levels in the prostatic tissue of patients with prostate cancer. Clin. Cancer Res. 2004;10:7121–7126. doi: 10.1158/1078-0432.CCR-04-0913. [DOI] [PubMed] [Google Scholar]
- 50.Yoon FH, et al. Testosterone recovery after prolonged androgen suppression in patients with prostate cancer. J. Urol. 2008;180:1438–1444. doi: 10.1016/j.juro.2008.06.029. [DOI] [PubMed] [Google Scholar]
- 51.Mitamura K, et al. Identification of dehydroepiandrosterone metabolites formed from human prostate homogenate using liquid chromatography-mass spectrometry and gas chromatography-mass spectrometry. J. Chromatogr. A. 2002;961:97–105. doi: 10.1016/s0021-9673(02)00134-6. [DOI] [PubMed] [Google Scholar]
- 52.Nakamura Y, et al. In situ androgen producing enzymes in human prostate cancer. Endocr. Relat. Cancer. 2005;12:101–107. doi: 10.1677/erc.1.00914. [DOI] [PubMed] [Google Scholar]
- 53.Pelletier G. Expression of steroidogenic enzymes and sex-steroid receptors in human prostate. Best Pract. Res. Clin. Endocrinol. Metab. 2008;22:223–228. doi: 10.1016/j.beem.2008.02.004. [DOI] [PubMed] [Google Scholar]
- 54.Locke JA, et al. A novel communication role for CYP17A1 in the progression of castration-resistant prostate cancer. Prostate. 2009;69:928–937. doi: 10.1002/pros.20940. [DOI] [PubMed] [Google Scholar]
- 55.Locke JA, et al. Androgen levels increase by intratumoral de novo steroidogenesis during progression of castration-resistant prostate cancer. Cancer Res. 2008;68:6407–6415. doi: 10.1158/0008-5472.CAN-07-5997. [DOI] [PubMed] [Google Scholar]
- 56.Montgomery RB, et al. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 2008;68:4447–4454. doi: 10.1158/0008-5472.CAN-08-0249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Small EJ, et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583) J. Clin. Oncol. 2004;22:1025–1033. doi: 10.1200/JCO.2004.06.037. [DOI] [PubMed] [Google Scholar]
- 58.Kruit WH, Stoter G, Klijn JG. Effect of combination therapy with aminoglutethimide and hydrocortisone on prostate-specific antigen response in metastatic prostate cancer refractory to standard endocrine therapy. Anticancer Drugs. 2004;15:843–847. doi: 10.1097/00001813-200410000-00004. [DOI] [PubMed] [Google Scholar]
- 59.Potter GA, Barrie SE, Jarman M, Rowlands MG. Novel steroidal inhibitors of human cytochrome P45017 alpha (17 alpha-hydroxylase-C17, 20-lyase): potential agents for the treatment of prostatic cancer. J. Med. Chem. 1995;38:2463–2471. doi: 10.1021/jm00013a022. [DOI] [PubMed] [Google Scholar]
- 60.Rowlands MG, et al. Esters of 3-pyridylacetic acid that combine potent inhibition of 17 alpha-hydroxylase/C17, 20-lyase (cytochrome P45017 alpha) with resistance to esterase hydrolysis. J. Med. Chem. 1995;38:4191–4197. doi: 10.1021/jm00021a008. [DOI] [PubMed] [Google Scholar]
- 61.Reid AH, Attard G, Barrie E, de Bono JS. CYP17 inhibition as a hormonal strategy for prostate cancer. Nat. Clin. Pract. Urol. 2008;5:610–620. doi: 10.1038/ncpuro1237. [DOI] [PubMed] [Google Scholar]
- 62.Yap TA, Carden CP, Attard G, de Bono JS. Targeting CYP17: established and novel approaches in prostate cancer. Curr. Opin. Pharmacol. 2008;8:449–457. doi: 10.1016/j.coph.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 63.Attard G, et al. Phase I clinical trial of a selective inhibitor of CYP17, abiraterone acetate, confirms that castration-resistant prostate cancer commonly remains hormone driven. J. Clin. Oncol. 2008;26:4563–4571. doi: 10.1200/JCO.2007.15.9749. [DOI] [PubMed] [Google Scholar]
- 64.Attard G, et al. Selective inhibition of CYP17 with abiraterone acetate is highly active in the treatment of castration-resistant prostate cancer. J. Clin. Oncol. 2009;27:3742–3748. doi: 10.1200/JCO.2008.20.0642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Smith CM, Ballard SA, Worman N, Buettner R, Masters JR. 5 alpha-reductase expression by prostate cancer cell lines and benign prostatic hyperplasia in vitro. J. Clin. Endocrinol. Metab. 1996;81:1361–1366. doi: 10.1210/jcem.81.4.8636334. [DOI] [PubMed] [Google Scholar]
- 66.Negri-Cesi P, et al. Presence of 5 alpha-reductase isozymes and aromatase in human prostate cancer cells and in benign prostate hyperplastic tissue. Prostate. 1998;34:283–291. doi: 10.1002/(sici)1097-0045(19980301)34:4<283::aid-pros6>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 67.Vaughan D, et al. Long-term (7 to 8-year) experience with finasteride in men with benign prostatic hyperplasia. Urology. 2002;60:1040–1044. doi: 10.1016/s0090-4295(02)01971-4. [DOI] [PubMed] [Google Scholar]
- 68.Evans HC, Goa KL. Dutasteride. Drugs Aging. 2003;20:905–918. doi: 10.2165/00002512-200320120-00005. [DOI] [PubMed] [Google Scholar]
- 69.Clark RV, et al. Marked suppression of dihydrotestosterone in men with benign prostatic hyperplasia by dutasteride, a dual-5 alpha-reductase inhibitor. J. Clin. Endocrinol. Metab. 2004;89:2179–2184. doi: 10.1210/jc.2003-030330. [DOI] [PubMed] [Google Scholar]
- 70.Gilling PJG, et al. Efficacy of dutasteride and finasteride for the treatment of benign prostate hyperplasia:results of the 1-year Enlarged Prostate International Comparator Study (EPICS) BJU Int. 2005;95(Suppl. 1):1–38. [Google Scholar]
- 71.Kirby RS, et al. Efficacy and tolerability of doxazosin and finasteride, alone or in combination, in treatment of symptomatic benign prostatic hyperplasia: the Prospective European Doxazosin and Combination Therapy (PREDICT) trial. Urology. 2003;61:119–126. doi: 10.1016/s0090-4295(02)02114-3. [DOI] [PubMed] [Google Scholar]
- 72.Nickel JC. Comparison of clinical trials with finasteride and dutasteride. Rev. Urol. 2004;6(Suppl. 9):S31–S39. [PMC free article] [PubMed] [Google Scholar]
- 73.Faller B, Farley D, Nick H. Finasteride: a slow-binding 5 alpha-reductase inhibitor. Biochemistry. 1993;32:705–710. doi: 10.1021/bi00072a028. [DOI] [PubMed] [Google Scholar]
- 74.Bull HG, et al. Mechanism-based inhibition of human steroid 5α-reductase by finasteride: enzyme-catalyzed formation of NADP–dihydrofinasteride, a potent bisubstrate analog inhibitor. J. Am. Chem. Soc. 1996;118:2359–2365. [Google Scholar]
- 75.Bakshi RK, et al. 4-Aza-3-oxo-5 alpha-androst-1-ene-17 beta-N-arylcarboxamides as dual inhibitors of human type 1 and type 2 steroid 5 alpha-reductases. Dramatic effect of N.-aryl substituents on type 1 and type 2 5 alpha-reductase inhibitory potency. J. Med. Chem. 1995;38:3189–3192. doi: 10.1021/jm00017a001. [DOI] [PubMed] [Google Scholar]
- 76.Roehrborn CG, Boyle P, Nickel JC, Hoefner K, Andriole G. Efficacy and safety of a dual inhibitor of 5-alpha-reductase types 1 and 2 (dutasteride) in men with benign prostatic hyperplasia. Urology. 2002;60:434–441. doi: 10.1016/s0090-4295(02)01905-2. [DOI] [PubMed] [Google Scholar]
- 77.Stuart JD, et al. Pharmacokinetic parameters and mechanisms of inhibition of rat type 1 and 2 steroid 5 alpha-reductases: determinants for different in vivo activities of GI198745 and finasteride in the rat. Biochem. Pharmacol. 2001;62:933–942. doi: 10.1016/s0006-2952(01)00728-6. [DOI] [PubMed] [Google Scholar]
- 78.Harris GS, Kozarich JW. Steroid 5 alpha-reductase inhibitors in androgen-dependent disorders. Curr. Opin. Chem. Biol. 1997;1:254–259. doi: 10.1016/s1367-5931(97)80017-8. [DOI] [PubMed] [Google Scholar]
- 79.Schmidt LJ, Murillo H, Tindall DJ. Gene expression in prostate cancer cells treated with the dual 5 alpha-reductase inhibitor dutasteride. J. Androl. 2004;25:944–953. doi: 10.1002/j.1939-4640.2004.tb03166.x. [DOI] [PubMed] [Google Scholar]
- 80.Andriole GL, et al. Effect of the dual 5 alpha-reductase inhibitor dutasteride on markers of tumor regression in prostate cancer. J. Urol. 2004;172:915–919. doi: 10.1097/01.ju.0000136430.37245.b9. [DOI] [PubMed] [Google Scholar]
- 81.Schmidt LJ, et al. Effects of the 5 alpha-reductase inhibitor dutasteride on gene expression in prostate cancer xenografts. Prostate. 2009;69:1730–1743. doi: 10.1002/pros.21022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Mendelsohn LG. Prostate cancer and the androgen receptor: strategies for the development of novel therapeutics. Prog. Drug Res. 2000;55:213–233. doi: 10.1007/978-3-0348-8385-6_6. [DOI] [PubMed] [Google Scholar]
- 83.Bonaccorsi L, et al. Prostate cancer: a model of integration of genomic and non-genomic effects of the androgen receptor in cell lines model. Steroids. 2008;73:1030–1037. doi: 10.1016/j.steroids.2008.01.028. [DOI] [PubMed] [Google Scholar]
- 84.Thomas LN, et al. 5 alpha-reductase type 1 immunostaining is enhanced in some prostate cancers compared with benign prostatic hyperplasia epithelium. J. Urol. 2003;170:2019–2025. doi: 10.1097/01.ju.0000091804.20183.81. [DOI] [PubMed] [Google Scholar]
- 85.Lepor H, et al. The efficacy of terazosin, finasteride, or both in benign prostatic hyperplasia. Veterans Affairs Cooperative Studies Benign Prostatic Hyperplasia Study Group. N. Engl. J. Med. 1996;335:533–539. doi: 10.1056/NEJM199608223350801. [DOI] [PubMed] [Google Scholar]
- 86.Thompson IM, et al. The influence of finasteride on the development of prostate cancer. N. Engl. J. Med. 2003;349:215–224. doi: 10.1056/NEJMoa030660. [DOI] [PubMed] [Google Scholar]
- 87.Serfling R, et al. Quantifying the impact of prostate volumes, number of biopsy cores and-5 alpha-reductase inhibitor therapy on the probability of prostate cancer detection using mathematical modeling. J. Urol. 2007;177:2352–2356. doi: 10.1016/j.juro.2007.01.116. [DOI] [PubMed] [Google Scholar]
- 88.Andriole GL, et al. Effect of dutasteride on the risk of prostate cancer. N. Engl. J. Med. 2010;362:1192–1202. doi: 10.1056/NEJMoa0908127. [DOI] [PubMed] [Google Scholar]
- 89.Ishikawa S, Soloway MS, Van der Zwaag R, Todd B. Prognostic factors in survival free of progression after androgen deprivation therapy for treatment of prostate cancer. J. Urol. 1989;141:1139–1142. doi: 10.1016/s0022-5347(17)41193-1. [DOI] [PubMed] [Google Scholar]
- 90.Andriole G, et al. Chemoprevention of prostate cancer in men at high risk: rationale and design of the reduction by dutasteride of prostate cancer events (REDUCE) trial. J. Urol. 2004;172:1314–1317. doi: 10.1097/01.ju.0000139320.78673.2a. [DOI] [PubMed] [Google Scholar]
- 91.Schroder FH, et al. Can. dutasteride delay or prevent the progression of prostate cancer in patients with biochemical failure after radical therapy? Rationale and design of the Avodart after Radical Therapy for Prostate Cancer Study. BJU Int. 2009;103:590–596. doi: 10.1111/j.1464-410X.2009.08373.x. [DOI] [PubMed] [Google Scholar]
- 92.Fleshner N, et al. Delay in the progression of low-risk prostate cancer: rationale and design of the Reduction by Dutasteride of Clinical Progression Events in Expectant Management (REDEEM) trial. Contemp. Clin. Trials. 2007;28:763–769. doi: 10.1016/j.cct.2007.05.006. [DOI] [PubMed] [Google Scholar]
- 93.Sartor O, Gomella LG, Gagnier P, Melich K, Dann R. Dutasteride and bicalutamide in patients with hormone-refractory prostate cancer: the Therapy Assessed by Rising PSA (TARP) study rationale and design. Can. J. Urol. 2009;16:4806–4812. [PubMed] [Google Scholar]
- 94.Taplin ME, et al. Phase II study of androgen synthesis inhibition with ketoconazole, hydrocortisone, and dutasteride in asymptomatic castration-resistant prostate cancer. Clin. Cancer Res. 2009;15:7099–7105. doi: 10.1158/1078-0432.CCR-09-1722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Sartor O, et al. Activity of dutasteride plus ketoconazole in castration-refractory prostate cancer after progression on ketoconazole alone. Clin. Genitourin. Cancer. 2009;7:E90–E92. doi: 10.3816/CGC.2009.n.030. [DOI] [PubMed] [Google Scholar]