Abstract
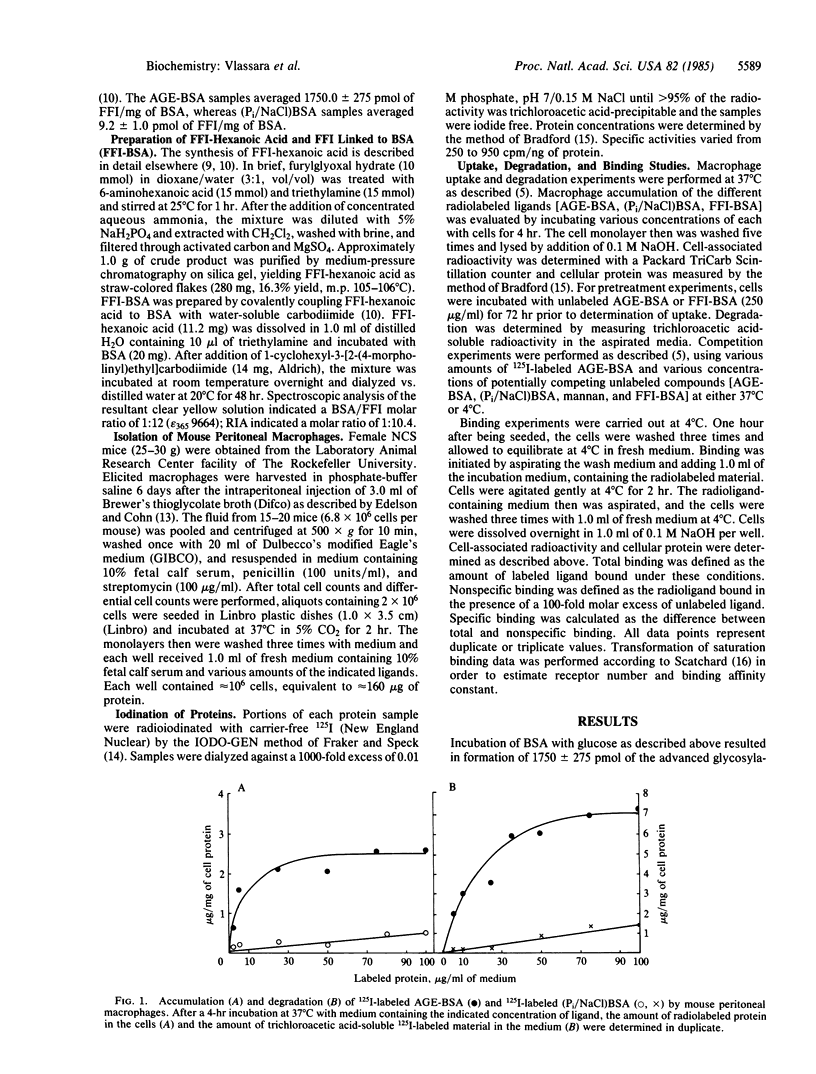
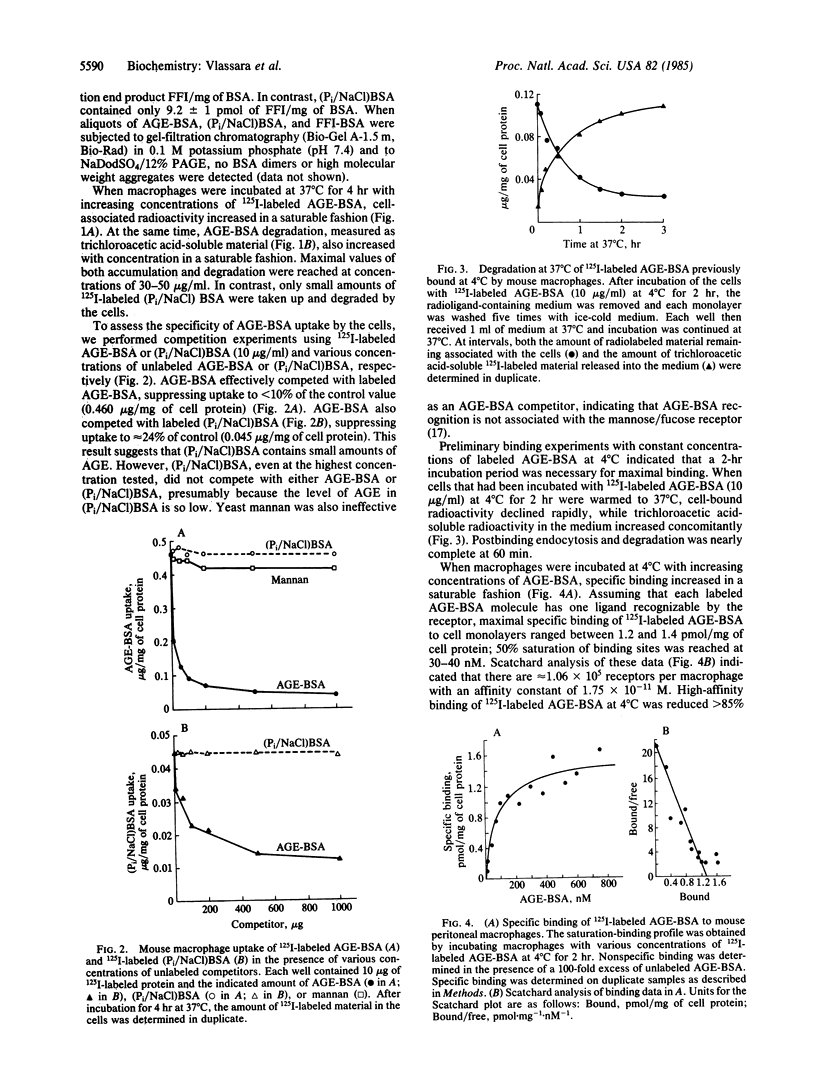
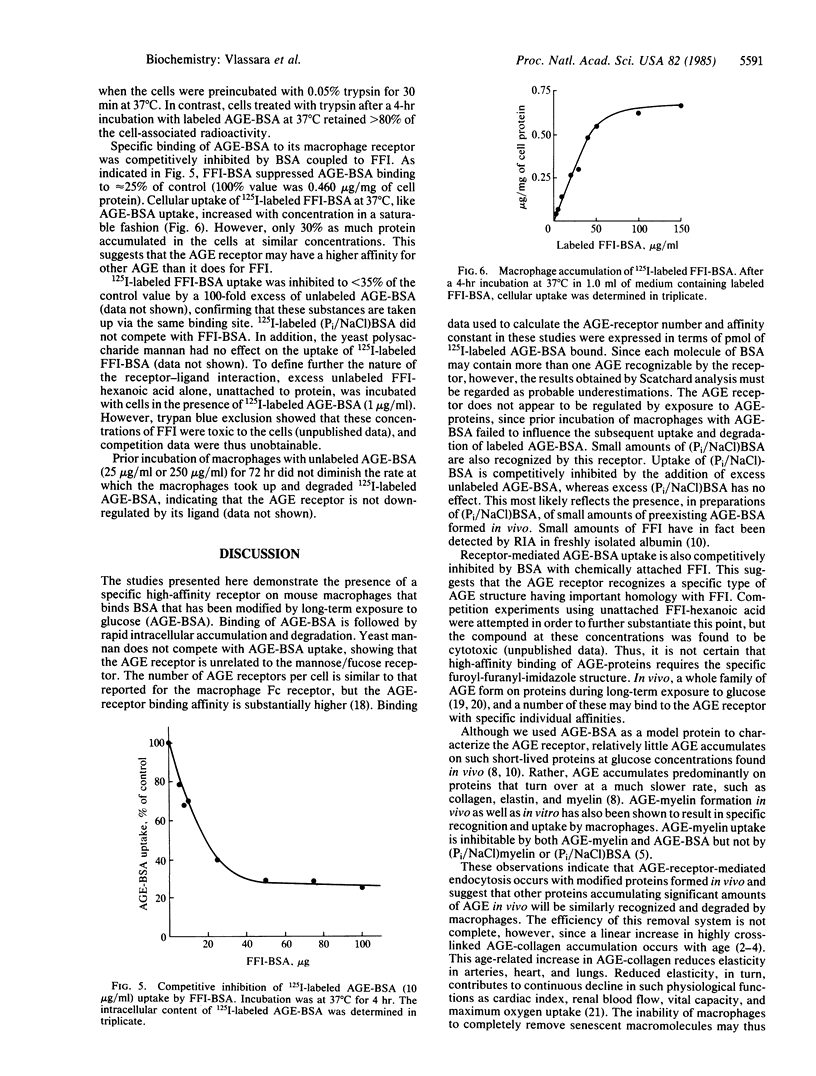
Proteins that have been modified by long-term exposure to glucose accumulate advanced glycosylation end products (AGE) as a function of protein age. In these studies, we have characterized the interaction of AGE-protein with mouse peritoneal macrophages, using AGE-modified bovine serum albumin (AGE-BSA, prepared by incubation with glucose) as a probe. AGE-BSA was specifically bound to cells at 4 degrees C and was taken up and degraded at 37 degrees C; these processes were concentration dependent and saturable. Competition experiments with AGE-BSA, BSA incubated with phosphate-buffered saline rather than glucose, and yeast mannan demonstrated that macrophages specifically recognize AGE on proteins by a receptor that is completely distinct from the mannose/fucose receptor. Scatchard analysis of AGE-BSA binding data indicated that there are approximately 1.06 X 10(5) receptors per macrophage, with an affinity constant of 1.75 X 10(-11) M. Specific binding of AGE-BSA to the macrophage receptor was competitively inhibited by BSA that had been chemically coupled to a synthetic analogue of the specific AGE, 2-(2-furoyl)-4(5)-(2-furanyl)-1H-imidazole (FFI-BSA). FFI-BSA was also taken up by macrophages in a concentration-dependent, saturable manner. Prior incubation of macrophages with AGE-BSA failed to influence the subsequent uptake and degradation of added AGE-BSA. Thus, the AGE receptor does not appear to be down-regulated by exposure to AGE-proteins. Results from these studies suggest that AGE could act in vivo as a specific signal for recognition and degradation of senescent macromolecules. Incomplete removal of AGE-proteins by macrophages may ultimately give rise to some of the physiologic changes that occur with normal aging.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Brownlee M., Vlassara H., Cerami A. Nonenzymatic glycosylation and the pathogenesis of diabetic complications. Ann Intern Med. 1984 Oct;101(4):527–537. doi: 10.7326/0003-4819-101-4-527. [DOI] [PubMed] [Google Scholar]
- Fraker P. J., Speck J. C., Jr Protein and cell membrane iodinations with a sparingly soluble chloroamide, 1,3,4,6-tetrachloro-3a,6a-diphrenylglycoluril. Biochem Biophys Res Commun. 1978 Feb 28;80(4):849–857. doi: 10.1016/0006-291x(78)91322-0. [DOI] [PubMed] [Google Scholar]
- Imber M. J., Pizzo S. V., Johnson W. J., Adams D. O. Selective diminution of the binding of mannose by murine macrophages in the late stages of activation. J Biol Chem. 1982 May 10;257(9):5129–5135. [PubMed] [Google Scholar]
- Koenig R. J., Cerami A. Hemoglobin A Ic and diabetes mellitus. Annu Rev Med. 1980;31:29–34. doi: 10.1146/annurev.me.31.020180.000333. [DOI] [PubMed] [Google Scholar]
- Monnier V. M., Kohn R. R., Cerami A. Accelerated age-related browning of human collagen in diabetes mellitus. Proc Natl Acad Sci U S A. 1984 Jan;81(2):583–587. doi: 10.1073/pnas.81.2.583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mortensen H. B., Christophersen C. Glucosylation of human haemoglobin a in red blood cells studied in vitro. Kinetics of the formation and dissociation of haemoglobin A1c. Clin Chim Acta. 1983 Nov 15;134(3):317–326. doi: 10.1016/0009-8981(83)90370-4. [DOI] [PubMed] [Google Scholar]
- Pongor S., Ulrich P. C., Bencsath F. A., Cerami A. Aging of proteins: isolation and identification of a fluorescent chromophore from the reaction of polypeptides with glucose. Proc Natl Acad Sci U S A. 1984 May;81(9):2684–2688. doi: 10.1073/pnas.81.9.2684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- REYNOLDS T. M. CHEMISTRY OF NONENZYMIC BROWNING. I. THE REACTION BETWEEN ALDOSES AND AMINES. Adv Food Res. 1963;12:1–52. doi: 10.1016/s0065-2628(08)60005-1. [DOI] [PubMed] [Google Scholar]
- Reynolds T. M. Chemistry of nonenzymic browning. II. Adv Food Res. 1965;14:167–283. doi: 10.1016/s0065-2628(08)60149-4. [DOI] [PubMed] [Google Scholar]
- Schnider S. L., Kohn R. R. Effects of age and diabetes mellitus on the solubility and nonenzymatic glucosylation of human skin collagen. J Clin Invest. 1981 Jun;67(6):1630–1635. doi: 10.1172/JCI110198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnider S. L., Kohn R. R. Glucosylation of human collagen in aging and diabetes mellitus. J Clin Invest. 1980 Nov;66(5):1179–1181. doi: 10.1172/JCI109950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unkeless J. C. The presence of two Fc receptors on mouse macrophages: evidence from a variant cell line and differential trypsin sensitivity. J Exp Med. 1977 Apr 1;145(4):931–945. doi: 10.1084/jem.145.4.931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlassara H., Brownlee M., Cerami A. Accumulation of diabetic rat peripheral nerve myelin by macrophages increases with the presence of advanced glycosylation endproducts. J Exp Med. 1984 Jul 1;160(1):197–207. doi: 10.1084/jem.160.1.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlassara H., Brownlee M., Cerami A. Nonenzymatic glycosylation of peripheral nerve protein in diabetes mellitus. Proc Natl Acad Sci U S A. 1981 Aug;78(8):5190–5192. doi: 10.1073/pnas.78.8.5190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlassara H., Brownlee M., Cerami A. Recognition and uptake of human diabetic peripheral nerve myelin by macrophages. Diabetes. 1985 Jun;34(6):553–557. doi: 10.2337/diab.34.6.553. [DOI] [PubMed] [Google Scholar]


