Abstract
Purpose
To assess the pathological and the oncologic outcomes of the Prostate Cancer (PCa) missed by 6 and 12-core biopsy protocols by using a reference 21-core scheme.
Materials and methods
Between 2001 and 2009, all patients who had PCa detected in an initial 21-core TRUS biopsy scheme and were treated by a radical prostatectomy (RP) were included. Patients were sorted in 3 groups according to the diagnosis site: sextant (6 first cores; group 1), peripheral zone (12 first cores; group 2) or midline/transitional zone (after 21 cores; group 3). Demographics, pathological features in biopsy and RP specimens and follow-up after RP were analyzed. The 5-year progression free survival (PFS) was studied in the 3 groups.
Results
During the study period, 443 patients were included. Among them, 67 %, 23.7 % and 9.2% were respectively diagnosed in group 1, 2 and 3. Among PCa diagnosed in midline/transition zone cores, 42 % were intermediate or high risk. Unfavorable disease was more frequently reported in group 1 in terms of extraprostatic extension (p=0.001), high Gleason score (p=0.001) and progression (p=0.001). No significant difference was observed between group 2 and 3 in terms of pathological features in RP specimens and oncologic outcome. The 5-year PFS was 89.7% and not significantly different in patients diagnosed with a 12-core scheme compared to those diagnosed only with 21-core scheme (p=0.332).
Conclusions
Our findings emphasize that PCa diagnosed only in a 21-core protocol is at least as aggressive as PCa detected in a 12-core scheme. This study invalidates the widespread idea sustaining that cancers diagnosed by more than 12 biopsies are less aggressive.
Keywords: Prostate Biopsy, Prostate cancer, Diagnosis Yield
Introduction
Hodge’s sextant prostate biopsy scheme significantly increased cancer detection [1]. The systematic sextant biopsy protocol has been the standard procedure for detecting prostate cancer (PCa) for many years. Later, it has been established that the extended biopsy protocols detect more cancers [2–3]. It is not clear whether the increased detection rate is simply due to the additional biopsies or to the location from which the cores are taken [4]. Since then, 10, 12, 14, 18 and even 21-sample cores protocols have been reported [5]. In 2003, we reported a prospective study of a 21-core biopsy protocol. It showed a diagnostic yield of 42.5% without increasing morbidity [6]. To minimize false-negative results, saturation biopsy has been adopted in several centers, resulting in cancer detection rates approaching 40% [7].
Concerns have then arisen that increasing the number of cores may lead to the detection of clinically insignificant, low-risk cancers [8]. Nevertheless, the concern of over-detection must be weighed against the risk of missing clinically significant malignancy. In fact, saturation biopsies, which are now used as part of active surveillance protocols, have proved to provide helpful information about quantitative and qualitative histology to predict the clinical significance of prostate cancer [9–10]. Based on the systematic review of Eichler et al. [5], the EAU guidelines recommends at least 8 cores and consider protocols more than 12 cores are not significantly more conclusive [11].
However, the final pathology and the oncologic outcomes features of the potentially missed cancers by a restricted biopsy protocol in the same patient are lacking in the current literature. Furthermore, PCa diagnosed by extended protocols are likely to be less aggressive is a widely spread idea [8].
The objective of this study was to assess the pathological characteristics, on biopsy cores and final pathology, and the oncologic outcomes of missed PCa by the 6-/-12 and detected by 21-biopsy cores protocol.
Patients and methods
Patient Selection
All patients referred to our department for a suspected PCa because of an abnormal DRE and/or elevated serum PSA level, had an initial TRUS guided 21-biopsy core protocol. Inclusion criteria were patients who had a first positive 21-biopsy core for PCa followed by radical prostatectomy (RP) in our department between 2001 and 2009. We excluded patients treated with Finasteride, Dutasteride, and preoperative hormonal. Local ethics committee approval had been obtained.
TRUS Biopsy Protocol
The detailed protocol had already been published [6]. All participants underwent TRUS biopsies performed by one operator (ADLT).
Methods
As the protocol follows the same steps and the cores numbered and analyzed separately, patients were grouped into 3 groups as follows: PCa diagnosed after 6 cores (standard sextant biopsies; group 1) whatever the results of the additional cores; PCa diagnosed in the 6 additional posterolateral cores with negative sextant biopsies (12 cores; group 2) whatever the results from midline/transition zone cores; and PCa diagnosed only in midline/transition zone cores (21cores; group 3) with negative 12-core biopsy scheme. If a patient has different site of PCa, the patient is sorted according to the first positive site. For each group we assessed the general features of the patients and their clinical findings, the characteristics of the biopsy cores, the characteristics of the disease on the biopsy cores and the final pathology specimens after the radical prostatectomy. The 3 groups were compared. Then, groups 1 and 2 were pooled and their characteristics were compared with those from group 3.
Database, follow-up and statistical analysis
Mean follow-up was 51.3 (median: 42, range: 1–77.2) months. Data were collected prospectively into a database and were investigated retrospectively. Pathological data were collected from biopsy cores and from the pathological prostate specimen. Quantitative data were tested using a Student-t test or a Mann-Whitney test as appropriate. Qualitative data were compared using a Chi-square or a Fisher’s test as appropriate. A double-sided p value <0.05 was considered statistically significant. The 5-year progression free survival (PFS) was estimated according to Kaplan Meier’s model and survival curves were tested using a log-rank test. All data were analyzed using SPSS v.16.0 software (SPSS, Chicago, Illinois, USA).
Results
Among the selected patients, 443 patients completed the RP and follow-up in our department. The mean age was 63 years (range: 42–76). Table-1 shows the characteristics of the cohort. The final pathology after radical prostatectomy showed a localized and a locally advanced PCa in 60.7 % and 38.9 %, respectively (Table-2).
Table 1.
Patients’ and biopsy core characteristics.
| Patients, n | 443
|
||
|---|---|---|---|
| Min | Max | Mean | |
| Age (year) | 42 | 76 | 63 |
| BMI (Kg/m2) | 16 | 44 | 26.28 |
| PSA (ng/ml) | 1.1 | 67 | 10.25 |
| Length of BC (mm) | 12 | 20 | 12.13 |
| Nbr. of positive BC | 1 | 21 | 4 |
| Max. percentage of PCa on BC | 1 | 100 | 41.70 |
| Total length of PCa on BC (mm) | 0.6 | 200 | 18.80 |
BC: Biopsy Core, PCa: Prostate Cancer.
Table 2.
Tumor stage on pathology specimen.
| Stage | n | % |
|---|---|---|
| pT2a | 58 | 13.1 |
| pT2b | 13 | 2.9 |
| pT2c | 198 | 44.7 |
| pT3a | 115 | 26 |
| pT3b | 27 | 6.1 |
| pT4 | 30 | 6.8 |
| Subtotal | 441 | 99.5 |
| Missing | 2 | 0.5 |
| Total | 443 | 100 |
Of all, 297 (67 %), 105 (23.7 %) and 41 (9.2 %) were diagnosed after 6 (sextant; group 1), 12 (PZ; group 2) and 21 (midline and TZ; group 3) cores respectively. Among group 3 patients, 17 (42 %) were a high or intermediate risk according to the D’Amico classification and 10 (24 %) patients had 7 or more Gleason score PCa. Furthermore, among these extra-diagnosed patients, 40 (97%) had a serum PSA below 10 ng/ml
Table-3 compares the PCa characteristics in function of different diagnosis site. The percentage of high Gleason score (p=0.001), dominant Gleason grade 4–5 tumors on final pathology (p=0.001), extraprostatic extension (p=0.001) were significantly different between groups with a higher percentage of unfavorable disease in group 1. Gleason grade 4 on final pathology were found in 126 (43 %), 18 (17 %) and 8 (20 %) patients in group 1, 2 and 3 respectively.
Table 3.
Cancer characteristics diagnosed with 6, 12 and 21 biopsy cores.
| site
|
p | ||||
|---|---|---|---|---|---|
| 6 cores | 12 cores | 21 cores | |||
| D’Amico classification | Low | 139 (47) | 65 (62) | 24 (58) | 0.053 |
| Intermediate | 126 (42) | 33 (31) | 14 (35) | ||
| High | 32 (11) | 7 (7) | 3 (7) | ||
| GS on BC, n (%) | <7 | 179 (60) | 92 (88) | 31 (76) | 0.001 |
| 7 | 106 (36) | 13 (12) | 9 (22) | ||
| >7 | 12 (4) | 0 (0) | 1 (2) | ||
| pGrade 4, n (%) | No | 171 (57) | 87 (83) | 33 (80) | 0.001 |
| Yes | 126 (43) | 18 (17) | 8 (20) | ||
| 8 or more Gleason score on pathology | No | 258 (87) | 97 (92) | 38 (93) | 0.19 |
| Yes | 39 (13) | 8 (8) | 3 (7) | ||
| Caspule invasion, n (%) | No | 171 (57) | 81 (77) | 33 (80) | 0.001 |
| Yes | 126 (43) | 24 (23) | 8 (20) | ||
| SV invasion, n (%) | No | 268 (90) | 101 (96) | 39 (95) | 0.10 |
| Yes | 29 (10) | 4 (4) | 2 (5) | ||
| N+, n (%) | No | 287 (97) | 104 (99) | 41 (100) | 0.21 |
| Yes | 10 (3) | 1 (1) | 0 (0) | ||
| PM, n (%) | No | 215 (72) | 82 (78) | 31 (76) | 0.43 |
| Yes | 82 (28) | 23 (22) | 10 (24) | ||
| DRE, n (%) | T1C | 240 (81) | 93 (88) | 42 (98) | 0.22 |
| T2A | 54 (18) | 12 (12) | 1 (2) | ||
| T2C | 1 (0.3) | 0 (0) | 0 (0) | ||
| T3 | 2 (0.7) | 0 (0) | 0 (0) | ||
| Progression, n (%) | No | 244 (82) | 97 (92) | 37 (90) | 0.02 |
| Yes | 53 (18) | 8 (8) | 4 (10) | ||
BC: Biopsy core, GS: Gleason Score, N+: positive Node on pathology, PM: Positive Margins, DRE: Digital Rectal Examination, SV: Seminal Vesicle.
When the cancers diagnosed on 6 and 12 biopsy cores sites were pooled (groups 1 and 2) and compared with group 3, there were more positive biopsy cores, more percentage and length of cancer on the cores (p<0.005) in the former group. PCa diagnosed in group 1–2 were statistically more frequently unfavorable than those diagnosed in group 3 in terms of dominant grade 4–5 (p=0.02) and extraprostatic disease (p=0.01). No significant difference was observed concerning the others pathological parameters. Table-4 shows the whole comparison between groups 1–2 and group 3.
Table 4.
Cancer characteristics diagnosed with 12 and 21 biopsy cores.
| site
|
p | |||
|---|---|---|---|---|
| 12 cores | 21 cores | |||
| Age (year) | 63 | 63 | 0.70 | |
| BMI (Kg/m2) | 26.1 | 26.9 | 0.34 | |
| PSA (ng/ml) | 10.35 | 9.28 | 0.52 | |
| Length of BC (mm) | 12.15 | 11.98 | 0.64 | |
| Nbr. of positive BC | 5 | 1 | 0.001 | |
| Max. % of PCa on BC | 44.17 | 17.43 | 0.001 | |
| Length of PCa on BC (mm) | 5.8 | 2.1 | 0.001 | |
|
| ||||
| Damico classification | Low | 204 (51) | 24 (58) | 0.50 |
| Intermediate | 159 (39) | 14 (35) | ||
| High | 39 (10) | 3 (7) | ||
| GS on BC, n (%) | <7 | 271 (67) | 31 (76) | 0.42 |
| 7 | 119 (30) | 9 (22) | ||
| >7 | 12 (3) | 1 (2) | ||
| pGrade 4, n (%) | No | 258 (64) | 33 (80) | 0.02 |
| Yes | 144 (36) | 8 (20) | ||
| 8 or more Gleason score on pathology | No | 355 (88) | 38 (93) | 0.35 |
| Yes | 47 (12) | 3 (7) | ||
| Caspule invasion, n (%) | No | 252 (63) | 33 (80) | 0.018 |
| Yes | 150 (37) | 8 (20) | ||
| SV invasion, n (%) | No | 369 (92) | 39 (95) | 0.37 |
| Yes | 33 (8) | 2 (5) | ||
| N+, n (%) | No | 391 (97) | 41 (100) | 0.28 |
| Yes | 11 (3) | 0 (0) | ||
| PM, n (%) | No | 297 (74) | 31 (76) | 0.7 |
| Yes | 105 (26) | 10 (24) | ||
| DRE, n (%) | T1C | 333 (82) | 42 (98) | 0.1 |
| T2A | 66 (16) | 1 (2) | ||
| T2C | 1 (0.3) | 0 (0) | ||
| T3 | 2 (0.7) | 0 (0) | ||
| Progression, n (%) | No | 341 (85) | 37 (90) | 0.33 |
| Yes | 61 (15) | 4 (10) | ||
BC: Biopsy core, GS: Gleason Score, N+: positive Node on pathology, PM: Positive Margins, DRE: Digital Rectal Examination, SV: Seminal Vesicle.
During follow-up, the 5-year PFS in group 1, 2, and 3 patients were 78.9%, 89.7% and 89.7%, respectively (Figure1). The 5-year PFS in group 1 was significantly poorer than those reported in groups 2 and 3 (p=0.022). When groups 1 and 2 were pooled, difference did not reach significance as compared to group 3 with a 5- year PFS of 81.7% and 89.7%, respectively (log rank p =0.332).
Figure 1.
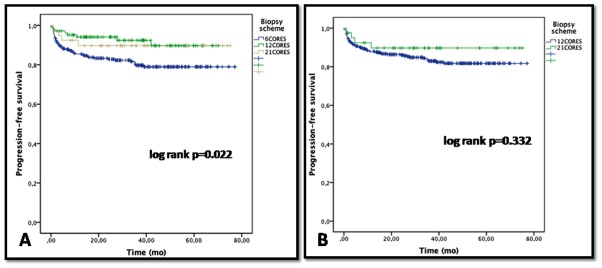
5-year PFS of patients diagnosed with 6, 12 and 21 biopsy cores (1A); 5-year PFS of patients diagnosed with 12 and 21 biopsy cores (1B).
Discussion
Biopsy core protocols showed a diagnosis rate increasing with the biopsy core number [12–13]. Since the cores were always taken in the same order and studied separately, we could use a study design we have already set in not systematically operated patients [14]. Each patient served as his own control. In this sense, we have previously shown that the biopsy core number was an important diagnostic tool in predicting the final pathological features of PCa [15–16].
The impact of “>10–12 biopsy protocols” on PCa detection rate remains very controversial. In a previous series, it has been shown that extended biopsy scheme could improve the prognostic assessment of PCa at initial diagnosis [17,18]. The addition of midline peripheral zone needle biopsies in the initial biopsy protocol is not supported by most of the studies [13–19]. However, a computer based modeling study showed 9.2 % were diagnosed in the midline/transition zone and half of these patients had an unfavorable disease [20].
Prognostic factors on biopsy and final were significantly different between the 3 groups, and thus, according to the diagnosis site. As the 6-biopsy core protocol is not recommended anymore [11], it appeared more relevant to us to compare the 12- and 21-biopsy cores groups. Accordingly, the capsule invasion, the presence of dominant Gleason grade 4 tumor, the percentage of cancer on biopsy cores, the maximum cancer length and the number of positive cores were also different in 12 and 21-biopsy protocols.
Considering the fact that limited morbidity is associated with the 21 biopsy core protocol [6] on one hand, and the aggressiveness of the extra diagnosed patients with the 21-core biopsy protocol on the other hand, we believe, this invalidates the widespread idea sustaining that cancers diagnosed by more than 12 biopsies are less aggressive. Therefore, we believe a 21-core TRUS biopsy protocol gives a better sampling of the prostate. We have previously shown that an extended biopsy scheme (>12 cores) improved the prostate detection rate in case of repeat biopsies but also in case of first set of biopsies [14,21]. Cancers missed by an initial 12-core scheme were also more aggressive according to biopsy parameters than those missed by an initial 21-core scheme [22]. Moreover, the prediction of pT3 disease in RP specimens is improved when the diagnosis of prostate cancer is based on an extended protocol [15]. The role of biopsy core number in reducing the reclassification in men eligible for active surveillance has also been emphasized [16]. Accordingly to this, we routinely perform this scheme in all patients referred for suspicion of localized PCa. However, some clinico-biological factors might be taken into account to better identify patients who will importantly benefit from an extended procedure, such the PSA level or the prostate volume [23].
The 5-years PFS study showed that patients diagnosed with 6 biopsies had a worse prognosis compared to those diagnosed 12 and 21 biopsy cores. However, comparing patients diagnosed by 12 and 21 biopsy cores did not show any difference. Thus, the extra-diagnosed cancers (detected only by a 21-core scheme) are at least as aggressive as those diagnosed with 12 biopsy cores. We are aware this result should be interpreted with little caution due the limited small size of group 3. A larger size of the study population and a longer follow-up should give more evidence to support our findings.
We would like to emphasize the limitations of our study. We used a prospective database but data were reviewed in a retrospective manner. The population represented the majority part of PCa patients who were diagnosed but some of them have been treated by radiotherapy or active surveillance, and thus, have not been included in the study because final pathology was not available. Besides, the final pathology shows that 30 patients had a pT4. This rose concern about the management of locally advanced PCa. Combined hormonal and radiation therapy seemed to be underused. We this is due to the prolonged study period. Recently, the locally advanced PCa is better defined in the guidelines and our practice has moved toward a less invasive approach.
Conclusion
Our findings emphasize PCa diagnosed only in a 21-core protocol are at least as aggressive as PCa detected in a 12-core scheme. This study invalidates the widespread idea sustaining that cancers diagnosed by more than 12 biopsies are less aggressive.
Footnotes
Conflict of interest statement: none.
References
- 1.Hodge KK, McNeal JE, Stamey TA. Ultrasound guided transrectal core biopsies of the palpably abnormal prostate. J Urol. 1989;142:66–70. doi: 10.1016/s0022-5347(17)38663-9. [DOI] [PubMed] [Google Scholar]
- 2.Norberg M, Egevad L, Holmberg L, et al. The sextant protocol for ultrasound- guided core biopsies of the prostate underestimates the presence of cancer. Urology. 1997;50:562–6. doi: 10.1016/S0090-4295(97)00306-3. [DOI] [PubMed] [Google Scholar]
- 3.Presti JC, Jr, O’Dowd GJ, Miller MC, et al. Extended peripheral zone biopsy schemes increase cancer detection rates and minimize variance in prostate specific antigen and age related cancer rates: results of a community multi-practice study. J Urol. 2003;169:125–9. doi: 10.1016/S0022-5347(05)64051-7. [DOI] [PubMed] [Google Scholar]
- 4.Scattoni V, Zlotta A, Montironi R, et al. Extended and saturation prostatic biopsy in the diagnosis and characterisation of prostate cancer: a critical analysis of the literature. Eur Urol. 2007;52:1309–22. doi: 10.1016/j.eururo.2007.08.006. [DOI] [PubMed] [Google Scholar]
- 5.Eichler K, Hempel S, Wilby J, et al. Diagnostic value of systematic biopsy methods in the investigation of prostate cancer: a systematic review. J Urol. 2006;175:1605–12. doi: 10.1016/S0022-5347(05)00957-2. [DOI] [PubMed] [Google Scholar]
- 6.de la Taille A, Antiphon P, Salomon L, et al. Prospective evaluation of a 21-sample needle biopsy procedure designed to improve the prostate cancer detection rate. Urology. 2003;61:1181–6. doi: 10.1016/s0090-4295(03)00108-0. [DOI] [PubMed] [Google Scholar]
- 7.Borboroglu PG, Comer SW, Riffenburgh RH, et al. Extensive repeat transrectal ultrasound guided prostate biopsy in patients with previous benign sextant biopsies. J Urol. 2000;163:158–62. [PubMed] [Google Scholar]
- 8.Djavan B, Remzi M, Marberger M. When to biopsy and when to stop biopsying. Urol Clin North Am. 2003;30:253–62. doi: 10.1016/s0094-0143(02)00188-x. [DOI] [PubMed] [Google Scholar]
- 9.Boccon-Gibod LM, de Longchamps NB, Toublanc M, et al. Prostate saturation biopsy in the reevaluation of microfocal prostate cancer. J Urol. 2006;176:961–3. doi: 10.1016/j.juro.2006.04.013. [DOI] [PubMed] [Google Scholar]
- 10.Epstein JI, Sanderson H, Carter HB, et al. Utility of saturation biopsy to predict insignificant cancer at radical prostatectomy. Urology. 2005;66:356–60. doi: 10.1016/j.urology.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 11.Heidenreich A, Bellmunt J, Bolla M, et al. EAU Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Treatment of Clinically Localised Disease. Eur Urol. 2010;59(1):61–71. doi: 10.1016/j.eururo.2010.10.039. [DOI] [PubMed] [Google Scholar]
- 12.Eskew LA, Bare RL, McCullough DL. Systematic 5 region prostate biopsy is superior to sextant method for diagnosing carcinoma of the prostate. J Urol. 1997;157:199–202. [PubMed] [Google Scholar]
- 13.Eskew LA, Woodruff RD, Bare RL, et al. Prostate cancer diagnosed by the 5 region biopsy method is significant disease. J Urol. 1998;160:794–6. doi: 10.1016/S0022-5347(01)62789-7. [DOI] [PubMed] [Google Scholar]
- 14.Guichard G, Larre S, Gallina A, et al. Extended 21-sample needle biopsy protocol for diagnosis of prostate cancer in 1000 consecutive patients. Eur Urol. 2007;52:430–5. doi: 10.1016/j.eururo.2007.02.062. [DOI] [PubMed] [Google Scholar]
- 15.Descazeaud A, Rubin M, Chemama S, et al. Saturation biopsy protocol enhances prediction of pT3 and surgical margin status on prostatectomy specimen. World J Urol. 2006;24:676–80. doi: 10.1007/s00345-006-0134-7. [DOI] [PubMed] [Google Scholar]
- 16.Ploussard G, Xylinnas E, Salomon L, et al. The role of biopsy core number in selecting prostate cancer patients for active surveillance. Eur Urol. 2009;56:891–8. doi: 10.1016/j.eururo.2009.07.053. [DOI] [PubMed] [Google Scholar]
- 17.Jones JS, Oder M, Zippe CD. Saturation prostate biopsy with periprostatic block can be performed in office. J Urol. 2002;168:2108–10. doi: 10.1016/S0022-5347(05)64307-8. [DOI] [PubMed] [Google Scholar]
- 18.Abouassaly R, Tan N, Moussa A, et al. Risk of prostate cancer after diagnosis of atypical glands suspicious for carcinoma on saturation and traditional biopsies. J Urol. 2008;180:911–4. doi: 10.1016/j.juro.2008.05.019. [DOI] [PubMed] [Google Scholar]
- 19.Terris MK, Wallen EM, Stamey TA. Comparison of mid-lobe versus lateral systematic sextant biopsies in the detection of prostate cancer. Urol Int. 1997;59:239–42. doi: 10.1159/000283071. [DOI] [PubMed] [Google Scholar]
- 20.Chen ME, Troncoso P, Johnston DA, et al. Optimization of prostate biopsy strategy using computer based analysis. J Urol. 1997;158:2168–75. doi: 10.1016/s0022-5347(01)68188-6. [DOI] [PubMed] [Google Scholar]
- 21.Campos-Fernandes JL, Bastien L, Nicolaiew N, et al. Prostate cancer 2010 [Google Scholar]
- detection rate in patients with repeated extended 21-sample needle biopsy. Eur Urol. 55:600–6. doi: 10.1016/j.eururo.2008.06.043. [DOI] [PubMed] [Google Scholar]
- 22.Ploussard G, Bastien L, Descazeaud A, et al. Extended biopsy protocol decreases prostate cancer incidence and risk of aggressive disease on repeated biopsies compared with initial standard procedure. Urol Int. 2010;84:147–52. doi: 10.1159/000277590. [DOI] [PubMed] [Google Scholar]
- 23.Scattoni V, Raber M, Abdollah F, et al. Biopsy schemes with the fewest cores for detecting 95% of the prostate cancers detected by a 24-core biopsy. Eur Urol. 2010;57:1–8. doi: 10.1016/j.eururo.2009.08.011. [DOI] [PubMed] [Google Scholar]


