Abstract
Background
Human saliva, a complex secretion that contains a mixture of inorganic and organic molecules, plays an essential role in the maintenance of oral health. Mucins are the major macromolecular component of the secretion and are considered the first line of defense for epithelial tissues. The aim of this study was to compare levels of mucins (MUC5B, MUC7, and MUC1) in saliva of young subjects with dental caries.
Material/Methods
All patients had DMF (decay/missing/filled) higher than value 0. Eight subjects with DMF=3 (control group) and 27 adolescents with DMF >11 (research group) were recruited for this study. Clinical evaluation procedures were oral examination, including tooth, periodontal, oral mucosal status, and collection of saliva samples. Saliva was collected for mucin assay. Enzyme-linked immunosorbent assay was used to quantitate MUC5B, MUC7, and MUC1.
Results
Our results indicate that adolescents with very high intensity of dental caries disease had increased levels of MUC1 and MUC5B. The membrane mucin MUC1 protein levels in the group with DMF>11 (research group) were higher compared to the group with DMF=3 (control group), and the increase was statistically significant (p=0.011). Similarly, secreted mucin MUC5B protein levels were higher (p=0.06) in the group with DMF>11 (research group). Although MUC7 protein levels were slightly reduced in symptomatic subjects, the decrease was statistically insignificant (p=0.918).
Conclusions
Our data suggest links between the production of mucins, especially MUC1 and MUC5B in saliva, and dental caries disease.
Keywords: Mucin-1, Mucin-5B, MUC7, Dental Caries Activity Tests
Background
Human saliva, a complex secretion that contains a mixture of inorganic and organic molecules, plays an essential role in the maintenance of oral health [1–3]. The tooth surface is protected by a film of salivary mucins and proline-rich glycoprotein [4]. Human saliva has been shown to contain at least 2 structurally and functionally distinct populations of secreted mucins: the high molecular weight (Mr>106 Da) polymeric, gel-forming population, MUC5B and the lower molecular weight (Mr 1.2–1.5×105 Da) non-polymerizing population MUC7 [5–10]. The secreted salivary mucins, MUC5B and MUC7, provide lubrication, protect oral tissue surfaces, and modulate oral microorganisms. Secreted mucins are considered the first line of defense for epithelial tissues since they act as physical barriers between extracellular milieu and the mucosal surface. Although they are components of the non-immune oral host defense system [11], they are only transient components of saliva and are swallowed soon after secretion [12,13].
It has been shown that the major and minor salivary glands, as well as oral epithelial cells, express the membrane-bound mucin MUC1 [13,14]. MUC1 is ubiquitously expressed in the epithelial cells lining the respiratory, reproductive, and gastrointestinal tracts. This mucin remains on the cell surface after secretion and is primarily involved in the protection of epithelial surfaces, acting as the second line of defense, and possibly in the mediation of intracellular signal processing [5,12]. The core protein of this glycoprotein is translated as a single polypeptide that is proteolytic cleaved into 2 subunits in endoplasmic reticulum [15]. The 2 fragments form a stable but noncovalent heterodimeric complex that remains associated during its post-translational processing and transport to the cell surface [16]. A portion of the membrane-associated MUC1 can be shed, presumably by the second proteolytic cleavage of the cytodomain [17]. In this way, MUC1 enters body fluids or layers on epithelial surfaces. Previous studies have reported that MUC1 is a critical element of the mucosal barrier to infection [18–20], and up-regulation of MUC1 follows infection with a bacterial pathogen [20]. It has also been suggested that the protective effects of MUC1 may be especially important in the oral cavity, where epithelial surfaces are constantly exposed to a variety of both pathogenic and commensal microbes [12,21]. In fact, up-regulation of MUC1 expression in oral epithelial cells has been reported to result from Porphyromonas gingivalis infection or increases in pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α [12]. There is also evidence that secreted mucin MUC2 and MUC5AC genes, which are not expressed in oral tissues, are up-regulated by pro-inflammatory mediators such as interleukin-1 beta and TNF alpha [22, 23]. Therefore, there is a possibility that expression of membrane (MUC1) and secreted mucins (MUC5B and MUC7) in the oral mucosal epithelial cells is affected by changes in pro-inflammatory cytokine levels in saliva.
The importance of salivary secreted mucins (MUC5B and MUC7) has been the focus of much research in the last 2 decades [2]. Significantly less attention has been paid to the role of transmembrane mucin MUC1. The aim of this study was to compare salivary MUC5B, MUC7, and MUC1 content in saliva of young people with dental caries.
Material and Methods
Study population
The adolescents (age 18 years) from a high school where students have a high prevalence of dental caries were chosen and served as the experimental group. The high caries rate of adolescents was examined using the caries intensity index DMF (decay/missing/filled; D+M+F/number of the examined). We failed to identify a control group with DMF=0. Eight adolescents with DMF=3 served as a control group and 27 adolescents with DMF>11 were the research group. The subjects in the control group had healthy periodontium and oral mucosa. Examination of patients’ dentition with regard to caries was carried out in a school surgery, in artificial light, using basic diagnostic instruments. The depth of gingival pouches and gingival hemorrhage were examined with a periodontal probe in individual sites in the teeth. Additionally, dental plaque and lesions on the mucus membrane of the oral cavity were evaluated. Saliva from all subjects was collected and the levels of mucins were recorded.
The research protocol was approved by the Committee for Ethics and Supervision on Human Research, Medical University of Białystok, with informed consent from the patients.
Salivary sample collection
Saliva was collected by a standard method. Samples from the subjects were collected between 9:00 and 11:00 a.m. All subjects abstained from eating and drinking for 2 h. Unstimulated whole saliva was collected for 10 min by a spitting method. Saliva samples were homogenized and clarified by centrifugation at 10.000 × g for 15 min at 4°C. The aliquots of clarified supernatants were stored at −70°C for the mucins measurements.
Analysis of salivary MUC7 and MUC5B
The high sensitivity assay kits (USCNK) were used to determine the levels of MUC5B and MUC7 in the saliva samples. The microtiter plates provided in the kits were pre-coated with a monoclonal antibody specific to MUC5B or MUC7. Standards and samples were added to the appropriate microtiter plate wells with a biotin-conjugated polyclonal antibody preparation specific for MUC5B or MUC7 and were incubated for 1 h at 37°C. After washing away any unbound substances, avidin conjugated to horseradish peroxidase was added to each microplate well and incubated. After another aspiration and washing step, a TMB substrate solution was added to each well. The enzyme-substrate reaction was terminated by the addition of a sulfuric acid solution and the color change was measured at a wavelength of 450 nm. The assay was performed in duplicate and the concentration of MUC5B and MUC7 in the samples was then determined by comparing the O.D. of the samples to the standard curve. The range of the standard curve was 0.625–40 ng/mL for MUC5B and 0.312–20 ng/mL for MUC7. The minimum detectable dose of human MUC5B was generally less than 0.247 ng/mL and MUC7 was less than 0.312 ng/mL.
Determination of membrane MUC1
Assay of MUC1 was done according to a previously published protocol [24]. Samples of human saliva were diluted in PBS to protein concentration 10 μg/mL and aliquots (50 μL) were coated onto microtiter plates (NUNC F96; Maxisorp, Roskilde, Denmark) at room temperature overnight. The wells were washed 3 times (100 μL) with PBS, pH 7.4, 0.05% Tween (PBS-T; washing buffer) between all ensuing steps. Unbound sites were blocked with 100 μL of 1% blocking reagent for ELISA (Roche Diagnostics, Mannheim, Germany) for 1 h. The wells were then incubated for 2 h with 100 μL of anti-MUC1 monoclonal antibody (anti-EMA, clone GP1.4; Sigma, St Louis, MO, USA) diluted in PBS-T/1% BSA (Sigma, St Louis, MO, USA) 1:500 (v/v). Next, plates were incubated (1 h) with 100 μL of secondary antibody, horseradish peroxidase conjugated anti-mouse IgG (Sigma, St Louis, MO, USA) diluted in PBS-T/1% BSA 1:1500 (v/v). Plates were washed 4 times in PBS and the colored reaction was developed by incubating with 2.2′-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid) (ABTS) – liquid substrate for horseradish peroxidase (Sigma, St Louis, MO, USA). Absorbance at 405 nm was measured after 30–45 min in an ELISA reader. As controls, wells with no saliva were used. The assay was performed in triplicate for saliva and the results are presented as the mean difference between the readings in optical density units (OD) in experimental wells and controls.
Statistical analysis
Normal distribution of data was assessed by the Shapiro-Wilk test. Since the data were not normally distributed, the Mann-Whitney nonparametric U test for unrelated results of MUC5B and MUC7 was used to compare differences between the groups. Data of MUC1 were normally distributed and the t test was used. All differences were considered significant at P<0.05. Correlations between mucins level and DMF, GI, and PI values in dental caries subjects were assessed by Spearman’s coefficients (level of significance, P<0.05). Statistical analysis was carried out using the Statistica 6.0.
Results
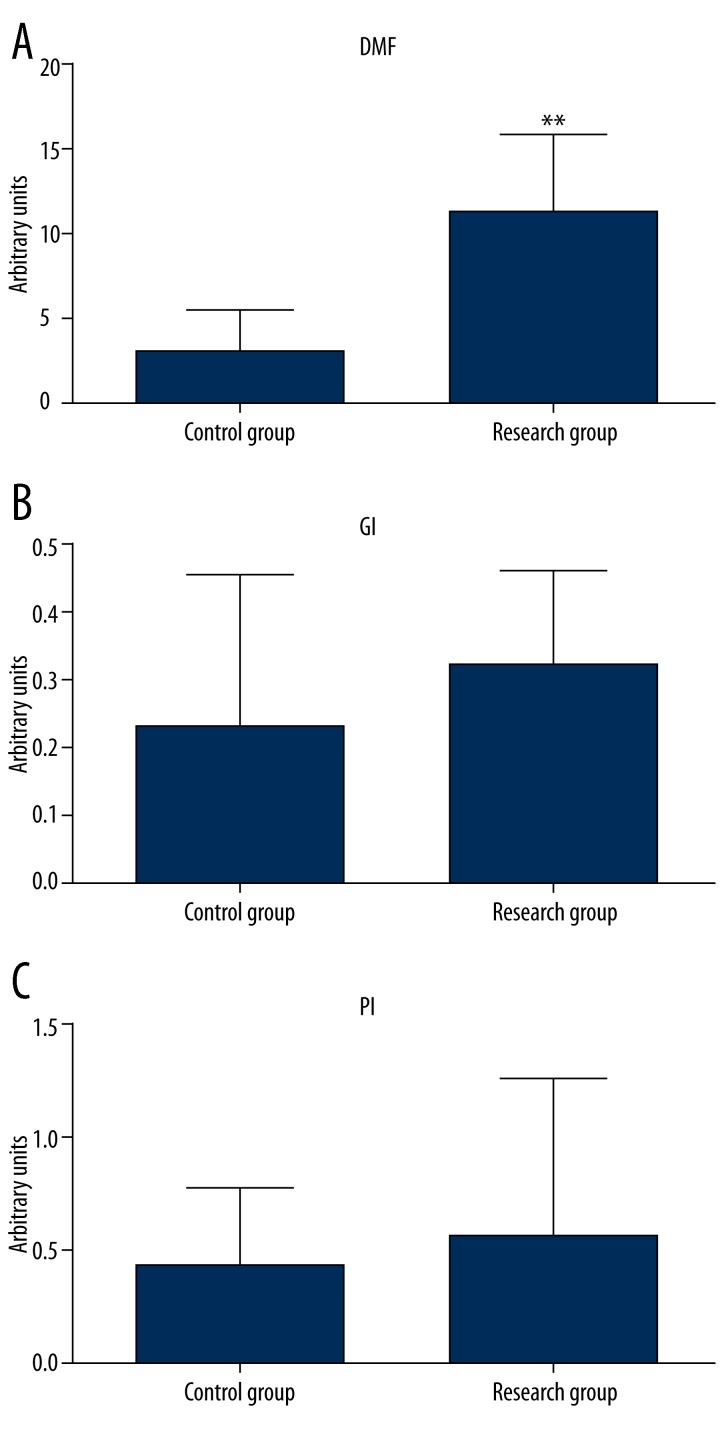
The index of intensity of caries/dental caries index - DMF was significantly higher (p=0.002) in the experimental group compared to the control group (Figure 1A). A mean value of Gingival Index 0.33 below the value of 1 indicates a mild form of gingivitis (Figure 1B). Gingivitis seems to be associated with dental plaque at the mean value of 0.53 PI Plaque Index/Periodontal Index (Figure 1C).
Figure 1.
Index of DMF, GI, PI in experimental subjects. ** P<0.05 – differences statistically significant between control and research group.
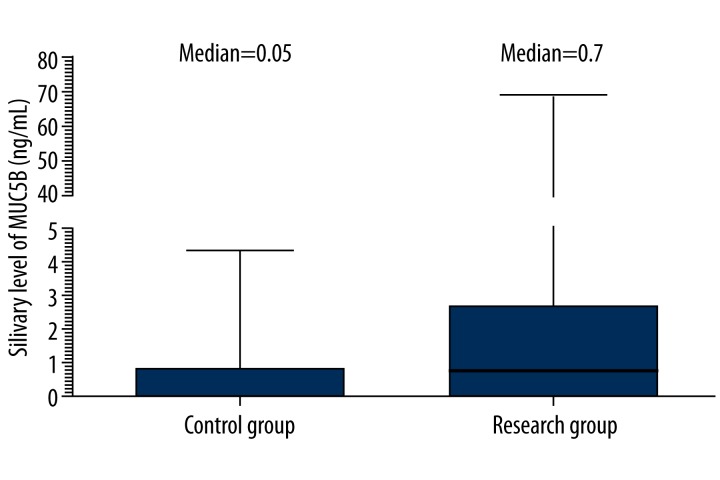
Results of MUC5B determination in groups are presented in Figure 2. Median of MUC5B saliva concentration in research group with DMF>11 was 0.7 ng/mL of saliva (25th–75th: 0.060–2.34) and was higher in comparison to patients with DMF=3 (control group) [0.05 ng/mL (25th–75th: 0.050–0.78)]. The difference was not statistically significant (p=0.06, Table 1).
Figure 2.
Comparison of expression patterns of MUC5B in subjects with DMF=3 (control group) and with DMF>11 (research group) using ELISA.
Table 1.
MUC5B, MUC7 and MUC1 levels in the whole unstimulated saliva from subjects with DMF=3 (control group) and subjects with DMF>11 (research group).
| Mucin | Control group (N=8) | Research group (N=27) | P |
|---|---|---|---|
| MUC5B (ng/mL) | 0.05 (0.05–0.78) | 0.70 (0.06–2.34) | 0.06 |
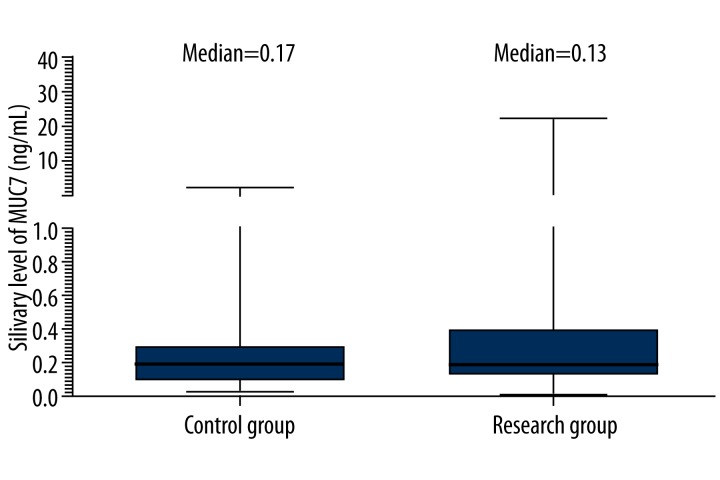
| MUC7 (ng/mL) | 0.17 (0.06–0.32) | 0.13 (0.06–0.42) | 0.918 |
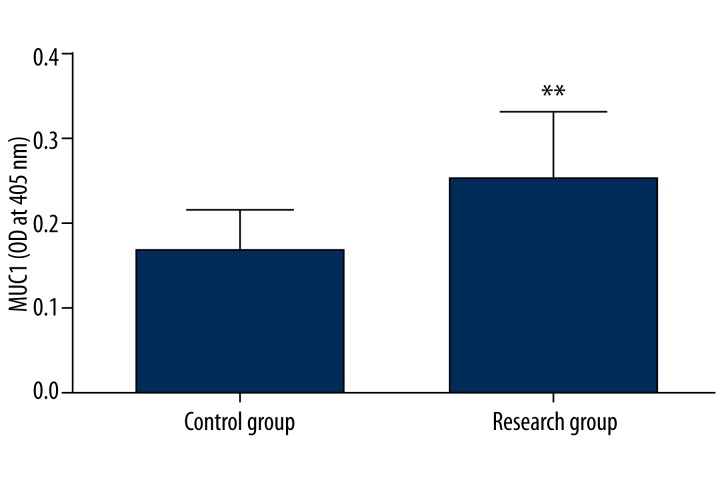
| MUC1 (OD at 405 nm) | 0.17±0.05 | 0.24*±0.08 | 0.011 |
For MUC5B and MUC7 values are given as the median with the range (25th–75th percentiles) given in parenthesis. For MUC1 values are given as the mean ±SD. Significant difference from healthy subjects: * p<0.05.
Results of MUC7 quantification are presented in Figure 3. The concentration of MUC7 in saliva of patients with DMF>11 ranged from 0.06 (25th percentile) to 0.42 (75th percentile) with the median 0.13 and was lower in comparison to subjects with DMF=3 [0.17 (25th–75th: 0.06–0.32)]. The difference between groups was not statistically significant (p=0.918, Table 1) due to variation between individuals in the level of MUC7.
Figure 3.
Comparison of expression patterns of MUC7 in subjects with DMF=3 (control group) and with DMF>11 (research group) using ELISA.
Figure 4 also shows the relative MUC1 transmembrane mucin levels (expressed as O.D. at 405 nm values) in saliva of experimental subjects. Significantly higher MUC1 level was found in subjects with DMF>11 (research group) in relation to subjects with DMF= 3 (control group) (p=0.011, Table 1). Statistical analysis showed a positive correlation between MUC1 levels and DMF in the research group (Table 2).
Figure 4.
Comparison of expression patterns of MUC1 in subjects with DMF=3 (control group) and with DMF>11 (research group) using ELISA. ** P<0.05 – differences statistically significant between control and research group.
Table 2.
Correlation coefficients calculated for mucins level versus DMF, GI, PI in dental caries subjects.
| Parameters | DMF | GI | PI |
|---|---|---|---|
| MUC5B | −0.09 | 0.13 | 0.09 |
| MUC7 | −0.19 | −0.1 | −0.07 |
| MUC1 | 0.41** | 0.07 | 0.03 |
P<0.05 statistically significant differences between adolescents with high intensity of caries.
Discussion
Dental caries, as well as obesity, is a growing epidemiological problem in adolescents [25]. There are many known predisposing factors for dental caries, especially injurious health behaviors and poor nutritional habits, which are based on a higher consumption of sweets and sweet beverages [26]. Recently, links between production of pro-inflammatory cytokines and dental caries process were also indicated [27]. Pro-inflammatory cytokines are markers of diseases associated with inflammation [28]. Saliva contains many more components that participate in the protection of the oral tissues (e.g. lysozyme, lactoferrin, lactoperoxidase, immunoglobulins, agglutinin, and mucins [4,29]. Research results clearly indicate that salivary mucins perform a variety of functions essential to maintaining a stable oral defense [11]. As a part of the enamel pellicle, mucins help initiate bacterial colonization by promoting the growth of benign commensal oral flora, forming a protective barrier and lubrication against excessive wear, providing a diffusion barrier against acid penetration, and limiting mineral egress from the tooth surface [11].
Our results indicated that high concentration of secreted mucin MUC5B predominates in people with severe caries. During caries development, a 14-fold increase in MUC5B concentration was determined in adolescents with DMF>11 and MUC7 mucin concentration was slightly decreased. Our data also demonstrate that MUC1 levels are increased in saliva of patients with very severe caries. Our research also showed a statistically significant positive correlation between MUC1 levels and DMF in a group with severe caries. There have been no studies aimed at the comparison of membrane mucin MUC1 levels in saliva of healthy subjects with those of people with caries. In a human study, MUC1 was detected as a component of the residual saliva that coats oral mucosal surfaces, meaning that MUC1 may aid in the hydration of the oral surface and may interact with oral bacteria [14,21].
Membrane-bound MUC1 mucin may form a scaffold or structural framework upon which the gel-forming mucin MUC5B assembles. The resulting membrane-bound and secreted mucins complex would further strengthen the epithelial protective barrier and increase the lifetime of secreted mucins in the oral cavity. The release of a MUC1 ectodomain to the saliva may cause this mucin to become an inefficient ‘scaffold’ maintaining secreted mucins. An increased content of MUC5B and increased ‘shedding’ of MUC1 mucin to the saliva may contribute to greater susceptibility to caries.
Conclusions
Our study demonstrated the possible connections between dental caries disease and the concentration of mucins, especially MUC1 and MUC5B, in saliva. The knowledge of functional properties of saliva, as well as those of its separate components, may permit a better assessment of dental caries susceptibility. Our data are based on a very small sample. Future research is essential to more completely characterize salivary components and their interactions and how these affect the caries process.
Acknowledgments
Agnieszka Gornowicz is a scholar of the project “I study, explore, commercialize – the UMB PhD fellowship program”. The project was co-financed by the European Union under the European Social Fund.
Footnotes
Source of support: This investigation was supported by research grant 134-29504F from the Medical University of Białystok, Poland
References
- 1.Fábián TK, Hermann P, Beck A, et al. Salivary defense proteins: their network and role in innate and acquired oral immunity. Int J Mol Sci. 2012;13:4295–320. doi: 10.3390/ijms13044295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Humphrey SP, Williamson RT. A review of saliva: normal composition, flow and function. J Prosthet Dent. 2001;85:162–69. doi: 10.1067/mpr.2001.113778. [DOI] [PubMed] [Google Scholar]
- 3.Patil PB, Patil BR. Saliva: a diagnostic biomarker of periodontal diseases. J Indian Soc Periodontol. 2011;15:310–17. doi: 10.4103/0972-124X.92560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Van Nieuw Amerongen A, Bolscher JGM, Veerman ECI. Salivary proteins: protective and diagnostic value in cariology? Caries Res. 2004;38:247–53. doi: 10.1159/000077762. [DOI] [PubMed] [Google Scholar]
- 5.Gendler SJ. MUC1: the renaissance molecule. J Mammary Gland Biol Neoplasia. 2001;6:339–53. doi: 10.1023/a:1011379725811. [DOI] [PubMed] [Google Scholar]
- 6.Hollingsworth MA, Swanson BJ. Mucins in cancer: Protection and control of the cell surface. Nat Rev Cancer. 2004;4:45–60. doi: 10.1038/nrc1251. [DOI] [PubMed] [Google Scholar]
- 7.Yonezawa S, Higashi M, Yamada N, et al. Mucins in human neoplasms: Clinical pathology, gene expression and diagnostic application. Pathol Int. 2011;61:697–716. doi: 10.1111/j.1440-1827.2011.02734.x. [DOI] [PubMed] [Google Scholar]
- 8.Veerman EC, van den Keybus PA, Valentijn-Benz M, Nieuw-Amerongen AV. Isolation of different high-Mr mucin species from human whole saliva. Biochem J. 1992;283:807–11. doi: 10.1042/bj2830807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ramasubbu N, Reddy MS, Bergey EJ, et al. Large-scale purification and characterization of the major phosphoproteins and mucins of human submandibular-sublingual saliva. Biochem J. 1991;280:341–52. doi: 10.1042/bj2800341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rousseau K, Wickstrom C, Whitehouse DB, et al. New monoclonal antibodies to non-glycosylated domains of the secreted mucins MUC5B and MUC7. Hybrid Hybridomics. 2003;22:293–99. doi: 10.1089/153685903322538818. [DOI] [PubMed] [Google Scholar]
- 11.Tabak LA. In defense of the oral cavity: structure, biosynthesis, and function of salivary mucins. Annu Rev Physiol. 1995;57:547–64. doi: 10.1146/annurev.ph.57.030195.002555. [DOI] [PubMed] [Google Scholar]
- 12.Li X, Wang L, Nunes DP, et al. Pro-inflammatory cytokines up regulate MUC1 gene expression in oral epithelial cells. J Dent Res. 2003;82:883–87. doi: 10.1177/154405910308201107. [DOI] [PubMed] [Google Scholar]
- 13.Offner DG, Troxler RF. Heterogeneity of high-molecular-weight human salivary mucins. Adv Dent Res. 2000;14:69–75. doi: 10.1177/08959374000140011101. [DOI] [PubMed] [Google Scholar]
- 14.Sengupta A, Valdramidou D, Huntley S, et al. Distribution of MUC1 in the normal human oral cavity is localized to the ducts of minor salivary glands. Arch Oral Biol. 2001;46:529–38. doi: 10.1016/s0003-9969(01)00010-3. [DOI] [PubMed] [Google Scholar]
- 15.Hilkens J, Buijs F. Biosynthesis of MAM-6, an epithelial sialomucin. Evidence for involvement of a rare proteolytic cleavage step in the endoplasmic reticulum. J Biol Chem. 1988;263:4215–22. [PubMed] [Google Scholar]
- 16.Ligtenberg MJ, Kruijshaar L, Buijs F, et al. Cell-associated episialin is a complex containing two proteins derived from a common precursor. J Biol Chem. 1992;267:6171–77. [PubMed] [Google Scholar]
- 17.Thathiah A, Blobel CP, Carson DD. Tumor necrosis factor-alpha converting enzyme/ADAM 17 mediates MUC1 shedding. J Biol Chem. 2003;278:3386–94. doi: 10.1074/jbc.M208326200. [DOI] [PubMed] [Google Scholar]
- 18.DeSousa MM, Surveyor GA, Price RE, et al. MUC1/episialin: a critical barrier in the female reproductive tract. J Reprod Immunol. 1999;45:127–58. doi: 10.1016/s0165-0378(99)00046-7. [DOI] [PubMed] [Google Scholar]
- 19.Kardon R, Price RE, Julian J, et al. Bacterial conjunctivitis in Muc1 null mice. Invest Ophthalmol Vis Sci. 1999;40:1328–35. [PubMed] [Google Scholar]
- 20.McAuley JL, Linden SK, Png CW, et al. MUC1 cell surface mucin is a critical element of the mucosal barrier to infection. J Clin Invest. 2007;117:2313–24. doi: 10.1172/JCI26705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pramanik R, Osailan SM, Challacombe SJ, et al. Protein and mucin retention on oral mucosal surfaces in dry mouth patients. Eur J Oral Sci. 2010;118:245–53. doi: 10.1111/j.1600-0722.2010.00728.x. [DOI] [PubMed] [Google Scholar]
- 22.Smirnova MG, Birchall JP, Pearson JP. TNF-alpha in the regulation of MUC5AC secretion: some aspects of cytokine-induced mucin hypersecretion on the in vitro model. Cytokine. 2000;12:1732–36. doi: 10.1006/cyto.2000.0763. [DOI] [PubMed] [Google Scholar]
- 23.Song KS, Lee WJ, Chung KC, et al. Interleukin-1 beta and tumor necrosis factor alpha induce MUC5AC overexpression through a mechanism involving ERK/p38 mitogen activated protein kinases-MSK-1CREB activation in human airway epithelial cells. J Biol Chem. 2003;278:23243–50. doi: 10.1074/jbc.M300096200. [DOI] [PubMed] [Google Scholar]
- 24.Radziejewska I, Borzym-Kluczyk M, Namiot Z, Stefańska E. Glycosylation of mucins present in gastric juice: the effect of Helicobacter pylori eradication treatment. Clin Exp Med. 2011;11:81–88. doi: 10.1007/s10238-010-0113-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sobieska M, Gajewska E, Kalmus G, Samborski W. Obesity, physical fitness, and inflammatory markers in Polish children. Med Sci Monit. 2014;20:493–500. doi: 10.12659/MSM.883959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lenander-Lumikari M, Loimaranta V. Saliva and dental caries. Adv Dent Res. 2000;14:40–47. doi: 10.1177/08959374000140010601. [DOI] [PubMed] [Google Scholar]
- 27.Gornowicz A, Bielawska A, Bielawski K, et al. Pro-inflammatory cytokines in saliva of adolescents with dental caries disease. Ann Agric Environ Med. 2012;19:711–16. [PubMed] [Google Scholar]
- 28.Yang D, Liu Z, Luo Q. Plasma ghrelin and pro-inflammatory markers in patients with obstructive sleep apnea and stable coronary heart disease. Med Sci Monit. 2014;20:251–56. doi: 10.12659/MSM.883874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Van Nieuw Amerongen A, Veerman ECI. Saliva – the defender of the oral cavity. Oral Dis. 2002;8:12–22. doi: 10.1034/j.1601-0825.2002.1o816.x. [DOI] [PubMed] [Google Scholar]