Abstract
Both behavioral and neuroimaging evidence support a female advantage in the perception of human faces. Here we explored the possibility that this relationship may be partially mediated by female sex hormones by investigating the relationship between the brain’s response to faces and the use of oral contraceptives, as well as the phase of the menstrual cycle. First, functional magnetic resonance images were acquired in 20 young women [10 freely cycling and 10 taking oral contraception (OC)] during two phases of their cycle: mid-cycle and menstruation. We found stronger neural responses to faces in the right fusiform face area (FFA) in women taking oral contraceptives (vs freely cycling women) and during mid-cycle (vs menstruation) in both groups. Mean blood oxygenation level-dependent response in both left and right FFA increased as function of the duration of OC use. Next, this relationship between the use of OC and FFA response was replicated in an independent sample of 110 adolescent girls. Finally in a parallel behavioral study carried out in another sample of women, we found no evidence of differences in the pattern of eye movements while viewing faces between freely cycling women vs those taking oral contraceptives. The imaging findings might indicate enhanced processing of social cues in women taking OC and women during mid-cycle.
Keywords: menstrual cycle; oral contraception (OC); fMRI, fusiform face area (FFA); eye movements
INTRODUCTION
Robust sex differences exist in face perception. A meta-analysis exploring sex differences in the development of facial-emotion recognition showed that the origins of this female advantage might be present already in the first year of life (McClure, 2000). Both behavioral and neuroimaging studies agree that the recognition of facial emotions and the related neural architecture continue to develop throughout childhood and adolescence (Herba and Philips, 2004) and that the peak in emotion recognition is reached in young adulthood (Sullivan et al., 2007). While the presence of sex differences in emotion recognition in the first year of life suggests a possible role of genes and/or prenatal exposure to androgens, it seems that postnatal sex hormones might play a role in face perception as well (e.g. Derntl et al., 2008). Sex differences in face perception and emotion recognition vary in their magnitude. Women outperform men in face detection, the very first stage of face perception (Cohen’s d= 0.91; McBain et al., 2009). Women also outperform men in accuracy and speed of recognizing emotions in faces (Cohen’s d= 0.3–0.4; Kirouac and Dore, 1985; Rahman et al., 2004; Hall and Matsumoto, 2004; Hampson et al., 2006), including situations when emotions are displayed at lower intensities (Montagne et al., 2005). This female advantage in emotion recognition was predicted by both dwell time and number of fixations to the eyes when scanning the face (Hall et al., 2010).
Sex differences in neuroimaging findings parallel these behavioral differences in face perception. Schulte-Rüther et al. (2008) reported that during perception of emotional faces, women (vs men) had higher blood oxygenation level-dependent (BOLD) response in face processing regions, such as the right superior temporal sulcus (STS). A large study of typically developing adolescents (518 males and 592 females) found that females watching emotionally ambiguous faces had a stronger BOLD response than males in a number of cortical regions, including the fusiform face area (FFA) (Tahmasebi et al., 2012), a region selectively involved in the perception of faces (Kanwisher et al., 1997). As mentioned above, sex hormones might, in part, explain the presence of the above sex differences. As estrogen enhances performance on sexually dimorphic tasks that favor women (e.g. verbal memory) and impairs performance on tasks that favor men (Hampson and Kimura, 1988; Sandres et al., 2002), estrogen might also modulate face perception. Miyahira et al. (2000) found that sex differences in exploratory eye movements emerge alongside hormonal changes following puberty and disappear following menopause, thus suggesting that some (general) aspects of visual information processing might be regulated by sex hormones.
In this study we wanted to explore whether variations in sex hormones influence brain response to faces. Menstrual cycle and the use of oral contraception (OC) provide an opportunity to study such effects given the fluctuations in sex hormones associated with both. Since estrogen and progesterone levels are higher during mid-cycle than menstruation, we hypothesized that women would have higher BOLD response to faces in the face processing network in general, and in the FFA in particular, during mid-cycle. We also predicted that women taking OC, and therefore having higher levels of estrogen and progesterone due to exogenous hormones, would have higher BOLD response to faces compared with freely cycling women. In addition, we also explored whether the duration of OC use modulates the FFA response in a dose-related manner.
We replicate these findings in a sample of adolescent females, thus exploring whether the effects of OC on FFA BOLD response can be detected as early as adolescence. Given the expected dose effect of OC use duration, we predicted that the effect of OC on BOLD response in FFA would be most likely smaller in adolescents compared with adult women. Finally, we tested whether such effects of sex hormones on brain response to faces might be reflected in the pattern of eye movements scanning the face. Therefore, we conducted an eye-tracking study using the same face stimuli in another sample of women; we predicted that women using OC might show longer fixations to the eye region.
EXPERIMENT I: fMRI IN YOUNG WOMEN
Materials and Methods
Participants
Twenty healthy women between 18 and 29 years of age were recruited at the University of Nottingham (UK): 10 freely cycling and 10 taking OC. The three types of OC used (Ovranette, Microgynon and Femodene) had the same amount of Ethinyl Estradiol (30 µg) and slightly different amounts of progestin (150 µg of Levonogestrel in Ovranette and Microgynon and 75 µg of Gestogene in Femodene). Duration of OC varied between 3 and 53 months (mean = 20.56 months, s.d. = 16.89). There was no significant difference between the age of freely cycling women (mean = 20.44 years, s.d. = 2.69) and women taking OC [mean = 22.0 years, s.d. = 3.26; t(18) = 1.12; P= 0.28]. Participants’ consent was obtained according to the Declaration of Helsinki and approved by the Ethics Committee of the Medical School at the University of Nottingham.
Design
In order to schedule the MR visits, each participant filled a brief questionnaire about their menstrual cycle (average length of cycle, average length of menstruation, date of last menstruation, brand and duration of OC use), and tracked basal body temperature for one menstrual cycle prior to scanning. This information was taken into account when scheduling the subsequent visits. Basal body temperature was measured orally right after awakening so that the ∼0.5°C increase in temperature just prior mid-cycle could be detected. All 20 participants provided a blood sample and took part in a structural MRI session four times: (i) menstruation (Day 5 ± 2 days); (ii) follicular phase (Day 11 ± 2 days); (iii) mid-cycle (Day 15 ± 2 days); and (iv) late luteal phase (Day 28 ± 2 days). The structural MRI session included T1-weighted (T1W) images, magnetic transfer ratio (MTR) and diffusion tensor imaging (DTI). In addition, functional MRI (fMRI) data were collected twice: at menstruation and mid-cycle. Phases of the menstrual cycle were counterbalanced across the two visits. At the first visit, six women from each group (pill and no pill) were scanned during menstruation and four women from each group were scanned during mid-cycle (and vice versa at the second visit).
Sex hormones
Blood samples were collected between 9 am and 10:30 am and serum was analyzed at clinical hematology laboratory at the Queen’s Medical Centre, Nottingham, UK. Levels of 17-β oestradiol were obtained via the ADVIA Centaur Estradiol assay (Siemens) and levels of progesterone via ADVIA Centaur Progesterone assay (Siemens). Both immunoassays used direct chemiluminescent technology and the ADVIA Centaur System (Siemens).
The ADVIA Centaur estradiol assay is a competitive immunoassay method employing a sheep anti-E2 monoclonal antibody labeled with acridium ester. Functional sensitivity is 0.019 ng/ml and specificity is high. Cross reactivity with estradiol derivatives, estriol, estrone, testosterone and testosterone derivatives was <0.5%. Cross reactivity with ethinyl estradiol was 0.037%. Analytical measurement range was 0.0118–3 ng/ml.
The ADVIA Centaur progesterone assay employs acridinium ester-labeled mouse monoclonal anti-progesterone antibody in the Little Reagent. Sensitivity of the ADVIA Centaur Progesterone assay was 0.21 ng/ml and the specificity was high. Cross-reactivity was 0.95% for corticosterone, 0.46% for pregnenolone, 0.31% for 17-α-hydroxyprogesterone, 0.08% for 11-deoxycorticosterone and was not detectable for cortisol, testosterone, aldosterone, androstenediol, 11-deoxycortisol, danazol, prednisolone, 17-β-estradiol, estrone, estriol, clomiphene and bromocryptine. Analytical measuring range was 0.21– 60 ng/ml.
Magnetic resonance imaging: acquisition
Scanning was carried out on a 1.5T Philips scanner. We acquired TIW 1-mm isotropic images and BOLD T2*-weighted gradient-echo, echo-planar images (EPI; matrix size 64 × 64, TE = 50 ms, TR = 3000 ms, 180 volumes, voxel size 4 × 4 × 4 mm3). Each EPI image covered the whole brain and consisted of 32 axial slices.
Functional paradigm: the face task
Participants passively viewed black-and-white videoclips of faces with ambiguous (e.g. twitching their nose, blinking their eyes, opening their mouth) and angry expressions (Grosbras and Paus, 2006). In both videoclips the gaze of the actor is direct and forward. This paradigm is identical to the one used in the Tahmasebi et al. (2012) study of typically developing adolescents. Each condition/block lasted for 18 s and included seven to eight videoclips of faces with either ambiguous or angry expression. Non-biological motion (moving circles) was used as a control condition. Henceforth, the term ‘angry condition’ refers to the ‘angry-control’ contrast and the term ‘ambiguous condition’ refers to the ‘ambiguous-control’ contrast.
fMRI: preprocessing and analysis
All brain-imaging data were processed with fMRI Expert Analysis Tool (FEAT), FSL version 4.1. (FMRIB Software Library, www.fmrib.ox.ac.uk/fsl). Pre-processing consisted of motion correction using MCFLIRT, spatial smoothing using Gaussian kernel of FWHM 8 mm and high-pass filter of 100. fMRI were registered to each participant’s T1-weighted images that were, in turn, registered to the standard space (152-MNI brain, 2 mm) using non-linear registration.
Outcomes of nonlinear registration and MCFLIRT motion-correction were checked. Imaging data affected by translations larger than 2 mm and rotations >2° (0.035 rad) were flagged; this was the case for two women in each group (one session each). For these two images, a confounder variable identifying volumes of no interest was created by fsl_motion_outliers (FSL 4.1.1), added to the design of first level analysis, and new outputs of first level analysis for these two subjects were created.
Functional data were analyzed using a 2 (pill: yes, no) by 2 (phase: menstruation, mid-cycle) analysis of variance (ANOVA). Cluster-based thresholding was used to correct for multiple comparisons: First, images were thresholded voxelwise at z= 1.7. Second, random field theory was applied to identify clusters that are big enough for the z= 1.7 to ensure an overall (family-wise) P < 0.05. The minimal cluster size of the group analysis output (resampled to 2 × 2 × 2 mm) for this P-threshold was 252 voxels.
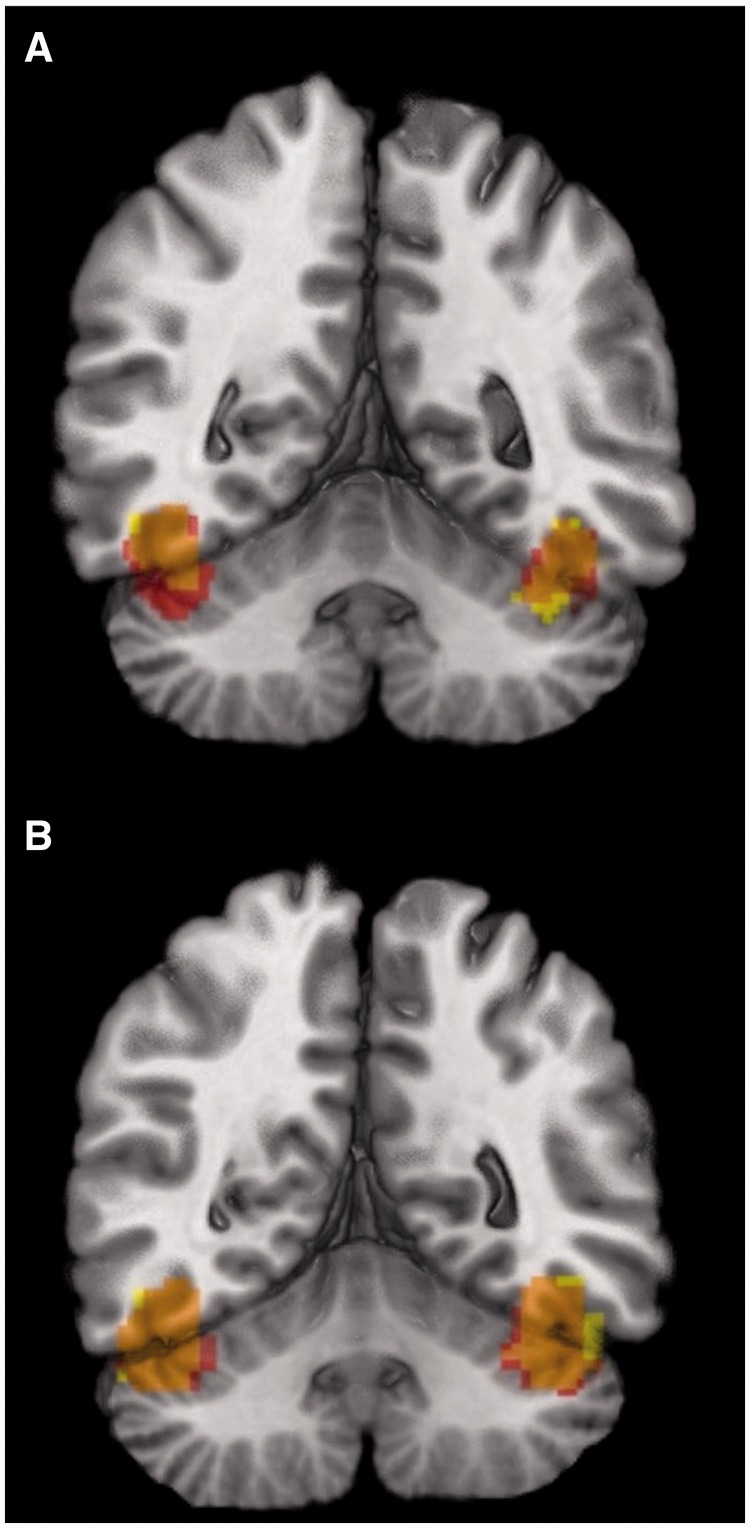
In order to explore the effect of OC duration on mean BOLD response in FFA, the voxelwise approach was complemented by a region-of-interest (ROI) analysis focusing on the FFA. Similar to Tahmasebi et al. (2012), masks for right and left FFA were created separately for ambiguous and angry condition in the following way: First, we created thresholded images of z-statistics (z> 2.3) for the following four groups: (i) OC during menstruation; (ii) OC during mid-cycle; (iii) freely cycling during menstruation and (iv) freely cycling during mid-cycle. Second, an intersection with Tahmasebi et al.’s (2012) FFA mask was created for each of these four images. Third, a union of these four intersections was created. This was repeated separately for the ambiguous and angry contrasts, respectively (Figure 3A). Mean BOLD response in these unionized masks was calculated by Featquery (FSL 4.1.1) and its relationship with OC duration was assessed.
Fig. 3.
(A) Experiment I — unionized FFA masks created for angry (red) and ambiguous (yellow) condition. (Their intersection is in orange). (B) Experiment II — unionized FFA masks created for angry (red) and ambiguous (yellow) condition. (Their intersection is in orange).
EXPERIMENT II: fMRI IN ADOLESCENT GIRLS
Methods and materials
Participants
A total of 110 adolescent girls were recruited in the context of a European multi-site study Imagen (Schumann et al., 2010). All girls were between 13.5 and 15.5 years of age and post-menarcheal (94 in Tanner stage 4, 16 in Tanner stage 5). Local ethics boards approved the study protocol; the parents and adolescents provided written informed consent and assent, respectively. While the whole Imagen study contains data for 2117 adolescents (1041 females), only 55 female adolescents were taking OC. We matched these 55 adolescent girls taking OC with 55 freely cycling adolescent girls by age (in months), Tanner pubertal stage and the acquisition site. Information about the type of OC and the length of use was not collected. Possible group differences in personality traits were assessed with NEO-FFI (Costa and McCrae, 1989).
MRI: acquisition and task
Scanning was carried out on 3T scanners from four different manufacturers (Siemens at four sites, Philips at two sites, General Electric and Bruker at one site, respectively). This report utilizes T1-weighted images (TE = 2.8 ms, TR = 2300 ms, voxel size 1 × 1 × 1 mm3) and BOLD T2*-weighted gradient-echo, EPI (matrix size 64 × 64, TE = 30 ms, TR = 2200 ms, 160 volumes, voxel size 3.4 × 3.4 × 3.4 mm3). Participants viewed the same stimuli as in Experiment I, namely the black-and-white videoclips of faces with ambiguous and angry expressions (Grosbras and Paus, 2006).
Functional paradigm: preprocessing and analysis
Preprocessing steps were identical to those of Experiment I, with the exception that spatial smoothing used Gaussian kernel of FWHM 7 mm (in accordance with the voxel size of the raw images). No imaging data were affected by head motion larger than 2 mm [translations larger than 2 mm and/or rotations >2° (0.035 rad)].
Voxelwise analysis used an unpaired t-test design to explore the effect of pill on BOLD response to faces. Similarly to Experiment I, the correction for multiple comparisons was done using cluster-based thresholding: First, voxelwise images were thresholded at z= 1.7, and second, random field theory was applied to identify clusters that are big enough for the z= 1.7 to ensure an overall (family-wise) P < 0.05.
A ROI analysis focused on the FFA. As described above (Experiment I), masks for right and left FFA were created separately for ambiguous and angry condition in the following way: First, we created thresholded images of z-statistics (z> 2.3) for the following two groups: (i) OC and (ii) freely cycling girls (n= 55 for each group). Second, an intersection with Tahmasebi et al.’s (2012) FFA mask was created for each of these two images. Third, a union of these two intersections was created. This was repeated separately for the ambiguous and angry contrasts, respectively (Figure 3B). Mean BOLD response in these unionized masks was calculated by Featquery (FSL 4.1.1).
EXPERIMENT I: fMRI IN YOUNG WOMEN
Results
Information about the length of menstrual cycle and length of menses for both pill and no-pill group are provided in Table 1.
Table 1.
Length of menstrual cycle and length of menses in pill and no pill group
| Freely cycling Mean (s.d.) | Pill Mean (s.d.) | |
|---|---|---|
| Length of menstrual cycle (days) | 26.9 (2.46) | 28 (0) |
| Length of menses (days) | 5.35 (1.11) | 4.65 (1.33) |
Sex hormones
Levels of estrogen and progesterone (means and standard deviations) during menstruation and mid-cycle in women from the pill and no pill group are provided in Table 2. For estradiol, a two-way repeated measures ANOVA showed main effects of Pill [F(1,36) = 11.59, P< 0.01, Cohen’s d= −0.9] and Phase [F(1,36) = 7.23, P= 0.01, Cohen’s d= −0.68], and a Pill × Phase interaction (F(1,36) = 9.6, P< 0.01). Student-t post hoc analyses showed that estradiol levels were higher in freely cycling women during mid-cycle compared with menstruation (t = 4.09, P< 0.002), higher in freely cycling women than in OC women during mid-cycle (t = −4.6, P< 0.001) but not during menstruation (t = −0.22, P= 0.83). For progesterone, a two-way repeated measures ANOVA showed main effect of Pill [F(1,36) = 6.74, P= 0.01, Cohen’s d= −0.7] and a Pill × Phase interaction [F(1,36) = 4.55, P= 0.04]. Student-t post hoc analyses showed that progesterone levels were higher in freely cycling women during mid-cycle compared with menstruation (t = 2.9, P= 0.01), higher in freely cycling than in OC women during mid-cycle (t = −3.34, P< 0.01), but not during menstruation (t = −0.33; P= 0.75).
Table 2.
Levels of estrogen and progesterone (means and standard deviations) during menstruation and mid-cycle in women from the pill and no pill group
| Freely cycling, menstruation Mean (s.d.) | Freely cycling, mid-cycle Mean (s.d.) | Pill, menstruation Mean (s.d.) | Pill, mid-cycle Mean (s.d.) | |
|---|---|---|---|---|
| Estradiol (pmol/L) | 144.67 (65.23) | 597.68 (479.32) | 120.71 (96.32) | 88.63 (42.5) |
| Progesterone (nmol/L) | 3.87 (1.23) | 12.04 (12.43) | 2.95 (1.07) | 2.63 (1.0) |
fMRI: voxelwise analysis
A two-way ANOVA showed significant main effects of Pill, Phase and an interaction between the Pill and Phase (Tables 3–8, Figure 1A and B). In the pill group (vs no pill group), we observed a stronger BOLD response in the right FFA to both ambiguous and angry faces. When watching angry faces, a stronger BOLD response in right FFA was also observed in the mid-cycle (vs menstruation) phase.
Table 3.
Voxelwise approach — main effect of contraceptive pill: pill (+) vs no pill (−) in ambiguous face condition
| No. | Label | Hemisphere | Lobe | Voxels | X | Y | Z | z-value |
|---|---|---|---|---|---|---|---|---|
| 1. | FFA | Right | Occipital | 6024 | 40 | −54 | −20 | 8.62 |
| 2. | Middle temporal | Left | Temporal | 9125 | −50 | −44 | 0 | 7.97 |
| 3. | Paracentral lobule | Right | Frontal | 3817 | 2 | −30 | 66 | − 4.51 |
Table 4.
Voxelwise approach — main effect of contraceptive pill: pill (+) vs no pill (−) in angry face condition
| No. | Label | Hemisphere | Lobe | Voxels | X | Y | Z | z-value |
|---|---|---|---|---|---|---|---|---|
| 1. | FFA | Right | Occipital | 12091 | 38 | −54 | −22 | 7.51 |
Table 5.
Voxelwise approach — main effect of phase: mid-cycle (+) vs menstruation (−) in ambiguous face condition
| No. | Label | Hemisphere | Lobe | Voxels | X | Y | Z | z-value |
|---|---|---|---|---|---|---|---|---|
| 1. | STS | Right | Temporal | 4087 | 50 | −48 | 6 | 6.4 |
| 2. | IFG | Left | Frontal | 6100 | −50 | 20 | 18 | 4.97 |
| 3. | IFG | Right | Frontal | 3201 | 54 | 20 | 24 | 4.63 |
| 4. | Ling. gyr. | Right | Occipital | 9613 | 2 | −86 | −6 | −5.15 |
Table 6.
Voxelwise approach — main effect of phase: mid-cycle (+) vs menstruation (−) in angry face condition
| No. | Label | Hemisphere | Lobe | Voxels | X | Y | Z | z-value |
|---|---|---|---|---|---|---|---|---|
| 1. | FFA | Right | Occipital | 6093 | 40 | −62 | −26 | 6.35 |
| 2. | IFG | Left | Frontal | 2816 | −50 | 34 | 8 | 4.86 |
| 3. | Middle temporal | Left | Temporal | 3331 | −58 | −58 | 4 | 4.13 |
Table 7.
Voxelwise approach — Pill by Phase interaction (no pill mid-cycle and pill menstruation > pill mid-cycle and no pill menstruation) in ambiguous face condition
| No. | Label | Hemisphere | Lobe | Voxels | X | Y | Z | z-value |
|---|---|---|---|---|---|---|---|---|
| 1. | Middle occipital | Left | Occipital | 8419 | −24 | −92 | 12 | 5.63 |
| 2. | Middle frontal | Left | Frontal | 4013 | −36 | 64 | 10 | 3.79 |
Table 8.
Voxelwise approach — Pill by Phase interaction (no pill mid-cycle and pill menstruation > pill mid-cycle and no pill menstruation) in angry face condition
| No. | Label | Hemisphere | Lobe | Voxels | X | Y | Z | z-value |
|---|---|---|---|---|---|---|---|---|
| 1. | Middle occipital | Left | Occipital | 11 893 | −24 | −90 | 14 | 5.87 |
Fig. 1.
(A) Effect of OC (pill vs freely cycling). The left image shows the effect of pill during ambiguous face condition, the right image shows the effect of pill during angry face condition. (B) Effect of phase (mid-cycle vs menstruation). The left image shows the effect of phase during ambiguous face condition, the right image shows the effect of phase during angry face condition. (C) Effect of pill duration on mean BOLD response. The left image shows the effect of pill duration on left FFA during ambiguous condition, the right image shows the effect of pill duration on left FFA during angry condition. Each women is represented by a number. Data obtained in menstruation and mid-cycle phase are represented by squares and circles, respectively.
fMRI: ROI analysis of the FFA response
A two-way repeated measures ANOVA showed significant effect of Pill use on the mean BOLD response in the right FFA (Figure 2A, Tables 9 and 10). There was no effect of Phase and no interaction between the Pill and Phase in the left or right FFA. Mean BOLD response in the right FFA was higher in the pill group (vs no pill group) in both ambiguous [F(1,36) = 5.18, P= 0.03, Cohen’s d= 0.69] and angry condition [F(1,36) = 4.51, P= 0.04, Cohen’s d= 0.66]. Mean BOLD response in the left FFA did not show any significant differences between the pill and no pill group in the ambiguous [F(1,36) = 1.58, P= 0.22] or angry condition (F(1,36) = 0.79, P= 0.38). These results are consistent with those of the voxelwise analyses reported above.
Fig. 2.
(A) Experiment I — effect of pill and cycle phase on BOLD response in FFA in young women. (B) Experiment II — effect of pill on BOLD response in FFA in adolescent females.
Table 9.
Experiment I, ROI approach — effect of pill and cycle phase on mean BOLD response in FFA (mean and s.d.).
| Freely, mid-cycle Mean (s.d.) | Freely, menstruation Mean (s.d.) | Pill, mid-cycle Mean (s.d.) | Pill, menstruation Mean (s.d.) | |
|---|---|---|---|---|
| Right FFA, ambiguous condition | 0.22 (0.28) | 0.03 (0.41) | 0.43 (0.22) | 0.25 (0.27) |
| Right FFA, angry condition | 0.23 (0.31) | 0.02 (0.52) | 0.47 (0.13) | 0.27 (0.39) |
| Left FFA, ambiguous condition | 0.11 (0.32) | 0.23 (0.22) | 0.27 (0.24) | 0.29 (0.32) |
| Left FFA, angry condition | 0.15 (0.49) | 0.18 (0.27) | 0.37 (0.19) | 0.15 (0.44) |
Table 10.
Experiment I, ROI approach — effect of contraceptive pill (pill vs no pill) on mean BOLD response in FFA, as assessed with two-way repeated measures ANOVA
| Mask | F | P |
|---|---|---|
| Right FFA, ambiguous condition | 5.18 | 0.03 |
| Right FFA, angry condition | 4.51 | 0.04 |
| Left FFA, Ambiguous condition | 1.58 | 0.22 |
| Left FFA, Angry condition | 0.79 | 0.38 |
A three-way repeated measures ANOVA was used to assess the effect of Pill, Cycle and Condition on BOLD response in the right FFA; it confirmed the significant effect of Pill (F = 4.23, P= 0.05) and Phase (F = 5.06, P= 0.04) and showed no effect of Condition (F = 0.11, P= 0.75) or any condition-related interaction (Pill × Condition: F = 0.14, P= 0.71; Phase × Condition: F = 0.12, P= 0.73; Pill × Phase × Condition: F = 0.006, P= 0.94). A three-way repeated-measures ANOVA assessing the effects of Pill, Phase and Condition on BOLD response in the left FFA showed a Phase × Condition interaction (F = 5.35, P= 0.03) and as expected, no effect of Pill (F = 0.91, P= 0.35), Phase (F = 0.02, P= 0.89), Condition (F= 0.12, P= 0.73), or any other condition-related interaction (Pill × Condition: F= 0.01, P= 0.92; Pill × Phase × Condition: F= 1.21, P= 0.29).
fMRI: ROI analysis of the amygdala response
Even though the voxelwise results showed no effect of Pill, Phase or Pill × Phase interaction on BOLD response in amygdala, we have conducted an ROI analysis of the amygdala response upon reviewer’s suggestion and we report these results in supplementary material.
Sex hormones and FFA BOLD response
We also explored effects of estrogen and progesterone on BOLD response in the right FFA using a three-way repeated-measures ANOVA, which examined the effect of Estrogen (log-transformed values), Pill and Phase on BOLD response in right FFA. This analysis revealed a significant three-way interaction in both the ambiguous (F= 23.79, P< 0.0001) and angry condition (F= 12.79, P= 0.001). We explored this interaction using two-way ANOVA examining the effects of Pill and Estrogen separately for menstruation and mid-cycle. No significant effects were found in mid-cycle. During menstruation, the interaction between Estrogen and Pill was significant (Ambiguous: F= 18.69, P= 0.0005; Angry: F= 1.49, P= 0.01): the BOLD response increased as a function of estrogen in freely cycling women [Ambiguous: t(9) = 2.8, P= 0.02; Angry: t(9) = 2.24, P= 0.06) but decreased as a function of estrogen in OC women [Ambiguous: t(9) = −3.28, P= 0.005; Angry: t(9) = −0.89, P= 0.4]. No significant effects of progesterone were observed.
Duration of OC use
One woman from the pill group (n= 10) had to be excluded from this analysis because of missing information about OC use-duration.
As shown in Table 11, mean BOLD response in the left FFA increased as a function of OC duration in both the ambiguous [F(1,14) = 6.2, P= 0.03, R2 = 0.29] and angry condition [F(1,14) = 5.1, P= 0.04, R2 = 0.22; Figure 1C]. While mean BOLD response in the right FFA was also increasing as a function of OC duration during the angry condition [F(1,14) = 5.7, P= 0.03, R2 = 0.13], no effect was found in the ambiguous condition [F(1,14) = 2.48, P= 0.14]. Note, however, that these results did not survive correction for multiple (four) comparisons.
Table 11.
Experiment I, ROI approach — results of two-way repeated measures ANOVA exploring the effect of OC duration, phase and their interaction on FFA BOLD
| Mask | F | P | R2 | F (corrected for age) | P (corrected for age) | R2 (corrected for age) |
|---|---|---|---|---|---|---|
| Right FFA, ambiguous condition | 2.48 | 0.14 | 0.26 | 0.94 | 0.35 | 0.32 |
| Right FFA, angry condition | 5.7 | 0.03 | 0.45 | 2.66 | 0.13 | 0.51 |
| Left FFA, ambiguous condition | 6.2 | 0.03 | 0.34 | 4.87 | 0.046 | 0.34 |
| Left FFA, angry condition | 5.1 | 0.04 | 0.38 | 3.72 | 0.076 | 0.38 |
Here we are reporting the F and R2 values for the effect of OC duration on mean BOLD response.
Given a negative trend between Pill Duration and Age (r = −0.44, P= 0.07), we also checked possible effects of Age on the FFA response to faces; none were significant.
EXPERIMENT II: fMRI IN ADOLESCENT GIRLS
Results
fMRI: voxelwise analysis
The pill vs freely cycling contrast showed one significant cluster in the right caudate region (z= 3.71; x = 12, y = 2, z= 16) in the angry condition and no significant clusters in the ambiguous condition. The freely vs pill contrast showed one significant cluster in the right thalamus region (z= 3.87; x = 6, y = −12, z= 14) in the angry condition and no significant clusters in the ambiguous condition.
fMRI: ROI analysis of the FFA response
Mean BOLD response in the left FFA was higher in the Pill group (vs No Pill group) during the ambiguous condition [t(108) = 2.57, P= 0.012, Cohen’s d= 0.49] and remained significant after correcting for multiple comparisons. Mean BOLD response in the left FFA during angry condition and right FFA during both ambiguous and angry condition did not show any significant difference when comparing adolescent girls in the pill vs no pill group [left FFA angry: t(108) = 1.05, P= 0.3; right FFA ambiguous: t(108) = 1.46, P= 0.15; right FFA angry: t(108) = 0.41, P= 0.68]. These results are illustrated in Figure 2B.
A two-way repeated-measures ANOVA examining the effect of Pill, Condition and Pill × Condition interaction on BOLD response in the left FFA confirmed the main effect of Pill (F= 4.0, P= 0.048) and showed no effect of Condition (F= 0.88, P= 0.35) or Pill × Condition interaction (F= 1.59, P= 0.21). A two-way repeated-measures ANOVA examining the effect of Pill, Condition and their interaction on BOLD response in the right FFA showed no significant results (Pill: F= 0.97, P= 0.33; Condition: F= 0.001, P= 0.98; Pill × Condition: F= 0.85, P= 0.36). Adding Laterality to the whole model and testing it with a three-way ANOVA confirmed the effect of Pill (F= 7.33, P= 0.007) and showed no effect of Laterality (F= 2.5, P= 0.11), Condition (F= 0.13, P= 0.72) or any related interaction (Pill × Laterality: F= 0.7, P= 0.4; Pill × Condition: F= 0.17, P= 0.68, Pill × Laterality × Condition: F= 0.04, P= 0.84) on the mean BOLD response in FFA.
fMRI: ROI analysis of the amygdala response
Upon reviewer’s suggestion we have conducted ROI analysis of the amygdala response also in Experiment II and we report these results in supplementary material.
Personality: NEO-FFI
No significant differences were found in the personality profiles of OC vs freely cycling adolescent girls: neuroticism [OC: mean = 23.51, s.d. = 8.00; freely cycling: mean = 25.25, s.d. = 6.84; t(108) = −1.23, P= 0.22], extraversion [OC: mean = 30.311, s.d. = 5.27; freely cycling: mean = 28.96, s.d. = 5.58; t(108) = 1.3, P= 0.2], openness [OC: mean = 25.58, s.d. = 5.48; freely cycling: mean = 27.16, s.d. = 5.63; t(108) = −1.49, P= 0.14], agreeableness [OC: mean = 29.29, s.d. = 5.88; freely cycling: mean = 29.4, s.d. = 4.79; t(108) = −0.11, P= 0.92] and conscientiousness [OC: mean = 28.38, s.d. = 7.03; freely cycling: = 28, s.d. = 6.96; t(108) = 0.29, P= 0.78].
EXPERIMENT III: SCANNING EYE MOVEMENTS
Methods and materials
In order to assess whether the above group differences in the FFA response to faces might be explained by a different pattern of eye movements while scanning the face, we conducted a behavioral eye-tracking study in another sample of women. Morris et al. (2007) experimentally manipulated scanpath during face viewing and showed that atypical scanpaths (i.e. less fixations to the eye region) are associated with lower BOLD response in the FFA.
Participants
Twenty healthy women between the ages 18 and 29 years (mean = 24.5 years, s.d. = 3.07) were recruited from the Baycrest volunteer database and Toronto area: 10 freely cycling and 10 taking OC. Participants’ consent was obtained according to the Declaration of Helsinki and approved by the Ethics Committee at Baycrest. The five types of OC used (Alesse, Cyclen, Diane, Marvelon and Yasmine) have 20–35 µg of Ethinyl Estradiol and different progestin substances (200 µg of Cyprosterone in Diane, 150 µg of Desogestrel in Marvelon, 300 µg of Drospirenone in Yasmine, 100 µg of Levonogestrel in Alesse and 250 µg of Norgestimate in Cyclen). Duration of OC ranged from 3 to 180 months (mean = 47.9 months, s.d. = 56.25). There was no significant relationship between OC duration and age (r = 0.35, P= 0.33). Participants were tested during the second, third or fourth day after the end of their period. This timing was chosen to maximize the difference in estrogen levels between the pill and freely cycling women; while women taking OC were already receiving artificial estrogens, freely cycling women had still very low levels of endogenous estrogens.
Procedure
An eye-tracking system (EyeLink II) was used to record eyes movements of the participants while they were watching the angry and ambiguous face videoclips used in Experiment I. Participants were sitting 91 cm from the monitor and were viewing 8-cm long faces under a visual angle of 5°; this corresponds to the angular size employed in Experiment I. Chin rest was used to minimize head movements. The eye-tracker was calibrated before each 18-s block of videoclips.
EXPERIMENT III: SCANNING EYE MOVEMENTS
Results
A mixed model exploring the effects of OC use (OC, freely cycling), Type of Face (ambiguous, angry), and Face Area viewed (interest areas: eyes, nose and mouth) on dwell time explained 53% of variability in the data (Adj R2 = 0.53) and showed main effects of Face Type [F(1,18) = 90.93, P< 0.0001], Interest Area [F(2,36) = 29.76, P< 0.0001] and an interaction between the two [F(2,36) = 42.28, P< 0.0001]; the dwell times were longer while viewing ambiguous (vs angry) faces and when looking at the eyes (vs nose vs mouth), with the latter effect more pronounced while viewing ambiguous faces. There was no main effect of Pill [F(1,18) = 0.79, P= 0.39] or any pill-related interactions [Pill × Face Type: F(2,36) = 0.02, P= 0.88; Pill × Interest area: F(2,36) = 3.1, P= 0.06; and Pill × Face Type × Interest Area: F(2,36) = 1.14, P= 0.33].
Discussion
Results of the voxelwise analysis showed that women taking OC compared with freely cycling women, as well as women during mid-cycle compared with menstruating women, had stronger BOLD response to faces in the right FFA, and several other brain regions (e.g. IFG, STS and middle temporal gyrus). This finding is consistent with those of Dietrich et al. (2001) who scanned women performing a verbal task and mental rotation tasks at different phases of menstrual cycle and showed that female sex hormones had a profound effect on the magnitude, but not lateralization or localization, of brain responses associated with these tasks. The fact that these pill/phase-related effects reported in our study were most pronounced in the right FFA is also consistent with our previous observation, made in a large group of adolescents using the same face paradigm, of the largest sex difference in the brain response to faces (female > male) being present the right FFA (Tahmasebi et al., 2012).
Focusing on the FFA, we also found that the mean BOLD response in this cortical region increased as a function of OC use duration; women who used OC longer had a stronger BOLD response in the left FFA while viewing both ambiguous and angry faces, and in the right FFA while viewing angry faces. Even though these effects of OC duration did not survive correction for multiple comparisons, they are suggestive of a dose–response relationship between the OC use and the brain response to faces.
As expected, plasma levels of estradiol were significantly higher in the freely cycling women compared with women taking OC (but note the high inter-individual variations in hormone levels). It should be noted, however, that this observation concerns endogenous sex hormones only (describerd later) and, as such, can be potentially misleading with regards to the interpretation of our findings. Standard assays are designed to measure these naturally occurring (endogenous) forms of estradiol. Contraceptive steroids, however, show only a limited cross-reactivity with the antiserum used in the standard assays (Hampson and Young, 2007). Therefore blood samples processed with the standard assays do not reflect the overall levels of female sex-steroids (Hampson and Young, 2007). But artificial sex steroids (ethinyl estradiol) contained in OC are biologically active: ‘ethinyl estradiol binds to the estrogen receptor complex and enters the nucleus activating DNA transcription of genes involved in estrogenic cellular responses’ (Medical Dictionary, 2011). OC provides constant levels of estrogen and progestin in the blood and thus suppresses the pulsatile release of FSH and LH from the anterior pituitary; during the pill-active phase, women taking OC are exposed daily to a high influx of ethinyl estradiol. The daily peak plasma concentrations of ethinyl estradiol in women taking OCs with 30 µg of ethinyl estradiol range from 125 to 168 pg/ml (van der Heuvel et al., 2005), corresponding to 459–616 pmol/l (Society for Biomedical Diabetes Research, 2010). Since participants in Experiments I and II were taking OC with 30 µg and between 20 and 35 µg of ethinyl estradiol, respectively, their daily peak plasma levels of ethinyl estradiol were likely to be in a similar range as reported by van der Heuvel et al. (2005) and measured in freely cycling women in the current study (estradiol) during ovulation (597.68 pmol/l).
An indirect evidence of higher estrogen levels in OC women comes from studies exploring the effects of estrogen on tasks that are known to show a female ‘advantage’, such as episodic (Herlitz and Rehnman, 2008) or verbal (Kimura, 1999) memory, or female ‘disadvantage’, such as spatial skills (Geary and DeSoto, 2001). Rosenberg (2002) showed that women taking OC reached better verbal scores but worse spatial scores compared with freely cycling women. Worse performance on visuospatial task in OC vs freely cycling women was also reported by Wharton et al. (2008). Mordecai et al. (2008) reported that OC users showed enhanced verbal memory during the active pill phase, while freely cycling women did not show any difference. We have shown clear sex differences using the face task employed here: female adolescents have a stronger FFA response to ambiguous faces than male adolescents (Tahmasebi et al., 2012). Thus, we observe a similar directionality in the FFA response to faces across sex (female > male), cycle (mid-cycle > menstruation) and OC (pill > no pill). Nonetheless, whether estradiol is the common element binding together these three phenomena remains to be tested in future studies.
Interpretation of the observed correlations between (endogenous) estradiol and BOLD in right FFA is challenging. First of all, the fact that these hormone–brain relationships hold only when the estrogen levels are very low (menstruation) suggests a fast saturation of the hormonal effects. Second, the opposite direction of this relationship in the two groups might reflect either the different state (‘plasticity’—described later) of the system in the long-term users of OC and/or the reciprocal relationship between endogenous and synthetic hormones in OC women. The latter could be related, for example, to the variation in the rate of EE metabolism: slow metabolizers of EE may still have relatively high levels of EE—and therefore low levels of (endogenous) E—in their system during menstruation and, as such, the observed inverse relationship between E and BOLD may, in fact, reflect positive relationship between EE (not measured) and BOLD.
While the voxelwise results of Experiment II did not replicate findings from Experiment I, results of the ROI analysis of Experiment II showed an enhanced BOLD response in FFA in OC compared with freely cycling adolescent girls, thus replicating results obtained in Experiment I in a sample of adolescent girls. Note that we designed Experiment II as a replication of the FFA effects observed in Experiment I; voxelwise analysis was added only for completeness of the reporting. The effect of pill on FFA BOLD response was in the medium range for both samples, but still slightly higher in the adults compared with adolescents. The fact that the group differences in the FFA response to faces are smaller in the adolescent sample, as compared with the young women, is consistent with the overall shorter duration of the OC use in the adolescent sample. Smaller effect size in the sample of adolescents might be also related to the fact that the adolescent girls were not tested during a particular phase of their cycle, which could have added noise to the pill effect. As shown in Figure 2B, both right and left FFA showed the same direction for the effect of pill (pill > freely) on BOLD response and a three-way ANOVA examining the effects of pill, condition and laterality on the FFA BOLD response showed an effect of pill but no effect of laterality or any laterality-related interaction.
Results from Experiment III showed that women taking OC did not differ from freely cycling women in their pattern of exploratory eye-movements while viewing the same videoclips of faces employed in Experiment I. Although Dalton et al. (2005) showed that persons with autism who made fewer fixations on the eyes of a displayed face had a lower BOLD response in FFA than those who made more fixations on the eyes, and Morris et al. (2007) manipulated scanpaths within healthy individuals and found that ‘atypical’ scanpaths of faces were associated with lower BOLD response in FFA, both authors agree that under free-viewing condition healthy participants did not show any correlation between scanpath and FFA response. If not the pattern of eye movements, what else might underlie the observed group differences in the FFA response?
It is possible that women taking OC pay closer attention to the cues carried by the face when processing, for example, the biologically significant regions of the face, such as the person’s eyes and their direction (gaze). It is known, for example, that attention to visual cues increases activity in extrastriate visual cortex and anterior temporal region (Lane et al., 1999). Kastner et al. (1999) demonstrated that directing attention to a particular location, and expecting the occurrence of visual stimuli at that location, increases BOLD response of human visual cortex even in the absence of visual stimulation. Palermo and Rhodes (2007) supported these results and showed that the neural responses in face-selective cortex are larger for attended compared with ignored faces. Petrovic et al. (2008) showed that during evaluation of conditioned faces oxytocin modulated BOLD response in the fusiform gyrus and amygdala. Given the modulatory role of estrogens on oxytocin (Silber et al., 1987; Stock et al., 1994; McCarthy, 1995), future research might consider the possible effect of oxytocin on attention and the possibility that estrogen effects on brain response to faces might be mediated by oxytocin.
We do not know whether the effect of OC on brain response to faces has any behavioral consequences. It is possible that women taking OC might demonstrate a better detection and/or recognition of faces in tasks where sex differences between males and females have been described (Kirouac and Dore, 1985; Hall and Matsumoto, 2004; Rahman et al., 2004; Montagne et al., 2005; Hampson et al., 2006; McBain et al., 2009). Since these studies report small to medium effect size for differences between males and females, it is very likely that the effects of OC would be only small and a larger sample size would be necessary to detect them.
While replicating the effect of pill on BOLD response in FFA in two independent samples (Experiment I and II) is strength of this study, using a third sample for collecting purely behavioral data is a limitation. Conducting a multimodal study that would record scanpaths during the fMRI task and had a bigger sample size would be ideal. Nonetheless, we used the same recruitment criteria (women, 18–29 years old, of white Caucasian background) and the same stimuli (Grosbras and Paus, 2006) in Experiment I and III.
Use of OCs exposes women to exogenous estrogen and progesterone, suppresses the release of FSH and LH and lowers levels of endogenous estrogens and progesterones. Use of OCs also lowers levels of testosterone (Graham et al., 2007; Hietala et al., 2007). In the current study, both the pill vs no pill and the mid-cycle vs menstruation contrasts showed a higher BOLD response in the right FFA, suggesting a similarity in the modulation of the brain response to faces in this region between the two hormonal states. But the exact mechanisms underlying such a similarity are unknown and may include, for example, OC-related variations in other hormones (e.g. low levels of testosterone) or long-term effects of OC at the level of the brain (see below).
Future studies may explore, for example, the effect of progestin-only pills on brain response to faces; this would allow us to ascertain whether the observed effects are related to the progestin part of the combined oral contraceptives used by women included in the present study or whether they might be related to the stimulating effects of estradiol. It would be also helpful to measure levels of androgens. But the exact mechanisms underlying effects of sex hormones are best addressed in experimental models. In this context, it is of interest to note the work of Follesa et al. (2001) who showed that long-term exposure to OC decreases levels of progesterone and its metabolite allopregnanolone in the rat brain, and in turn, increases expression of GABAa subunits in the cerebral cortex. Similarly, Smith (1994) reviewed several mechanisms by which estradiol might amplify neuronal excitability, including those involving excitatory (e.g. synthesis, degradation and release of glutamate) and inhibitory (reduction of GABA) neurotransmission. It is unclear, however, how such effects combine over time and whether the net outcome of the long-term use of OC is an increase or decrease in cortical excitability. The positive relationship between the duration of OC use and the FFA response to faces observed in our study suggests that the net effect of OC might be that of a higher excitability; this hypothesis can be explored, for example, in studies of cortical excitability with paired-pulse transcranial magnetic stimulation (Wassermann et al., 2008).
Conclusion
This study examined the effects of female sex hormones on BOLD response to faces. Women taking OC (vs freely cycling women) and women during mid-cycle (vs menstruation), were found to have a stronger BOLD response in the right FFA (and several other cortical regions). Stronger BOLD response to faces in the FFA was also found when comparing OC and freely cycling adolescent girls. The behavioral study suggests that these group differences are not related to the pattern of exploratory eye movements while viewing faces. The mean BOLD response in FFA also increased as a function of OC use duration, supporting the possibility of a long-term plasticity-like adaptation related to the use of OC.
MEMBERS OF THE IMAGEN CONSORTIUM
Institute of Psychiatry, King’s College, London, UK: G. Schumann, P. Conrod, L. Reed, G. Barker, S. Williams, E. Loth, M. Struve, A. Lourdusamy, S. Costafreda, A. Cattrell, C. Nymberg, L. Topper, L. Smith, S. Havatzias, K. Stueber, C. Mallik, T-K. Clarke, D. Stacey, C. Peng Wong, H. Werts, C. Andrew, S. Desrivieres, F. Carvalho; Department of Psychiatry and Psychotherapy, Campus Charité Mitte, Charité – Universitätsmedizin Berlin: A. Heinz, J. Gallinat, I. Häke, N. Ivanov, A. Klär, J. Reuter, C. Palafox, C. Hohmann, C. Schilling, K. Lüdemann, A. Romanowski, A. Ströhle, E. Wolff, M. Rapp; Physikalisch-Technische Bundesanstalt, Berlin, Germany: B. Ittermann, R. Brühl, A. Ihlenfeld, B. Walaszek, F. Schubert; Institute of Neuroscience, Trinity College, Dublin, Ireland: H. Garavan, C. Connolly, J. Jones, E. Lalor, E. McCabe, A. Ní Shiothcháin, R. Whelan; Department of Psychopharmacology, Central Institute of Mental Health, Mannheim, Germany: R. Spanagel, F. Leonardi-Essmann, W. Sommer; Department of Cognitive and Clinical Neuroscience, Central Institute of Mental Health, Mannheim, Germany: H. Flor, F. Nees; Department of Child and Adolescent Psychiatry, Central Institute of Mental Health, Mannheim, Germany: T. Banaschewski, L. Poustka, S. Steiner; Department of Addictive Behaviour and Addiction Medicine: K. Mann, M. Buehler, S. Vollstaedt-Klein; Department of Genetic Epidemiology in Psychiatry, Central Institute of Mental Health, Mannheim, Germany: M. Rietschel, E. Stolzenburg, C. Schmal; Schools of Psychology, Physics and Biomedical Sciences, University of Nottingham, UK: T. Paus, P. Gowland, N. Heym, C. Lawrence, C. Newman, Z. Pausova; Technische Universitaet Dresden, Germany: M. Smolka, T. Huebner, S. Ripke, E. Mennigen, K. Muller, V. Ziesch; Department of Systems Neuroscience, University Medical Center Hamburg-Eppendorf, Hamburg, Germany: C. Büchel, U. Bromberg, T. Fadai, L. Lueken, J. Yacubian, J. Finsterbusch; Institut National de la Santé et de la Recherche Médicale, Service Hospitalier Frédéric Joliot, Orsay, France: J-L. Martinot, E. Artiges, F. Gollier Briand, J. Massicotte, N. Bordas, R. Miranda, Z. Bricaud, Marie L. Paillère Martinot, N. Pionne-Dax, M. Zilbovicius, N. Boddaert, A. Cachia, J-F. Mangin; Neurospin, Commissariat à l'Energie Atomique, Paris, France: J-B. Poline, A. Barbot, Y. Schwartz, C. Lalanne, V. Frouin, B. Thyreau; Behavioural and Clinical Neurosciences Institute, Department of Experimental Psychology, University of Cambridge, UK: J. Dalley, A. Mar, T. Robbins, N. Subramaniam, D. Theobald, N. Richmond, M. de Rover, A. Molander, E. Jordan.
Supplementary Material
Acknowledgments
Experiment I was supported by the University of Nottingham. Experiment II was supported by IMAGEN, which receives research funding from the ‘European Community’s Sixth Framework Programme’ (grant number LSHM-CT-2007-037286) and the MRC program grant ‘Developmental pathways into adolescent substance abuse’ (grant number 93558). Experiment III was supported by Rotman Research Institute at Baycrest. This article reflects only the author’s views and the Community is not liable for any use that may be made of the information contained therein.
REFERENCES
- Costa PT, Jr, McCrae RR. NEO-PI/FFI Manual Supplement. Odessa, FL: Psychological Assessment Resources; 1989. [Google Scholar]
- Derntl B, Windischberger C, Robinson S, et al. Facial emotion recognition and amygdala activation are associated with menstrual cycle phase. Psychoneuroendocrinology. 2008;33:1031–40. doi: 10.1016/j.psyneuen.2008.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalton KM, Nacewicz BM, Johnstone T, et al. Gaze fixation and the neural circuitry of face processing in autism. Nature Neuroscience. 2005;8(4):519–26. doi: 10.1038/nn1421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dietrich T, Krings T, Neulen J, et al. Effects of blood estrogen level on cortical activation patterns during cognitive activation as measured by fuctional MRI. Neuroimage. 2001;13(3):425–32. doi: 10.1006/nimg.2001.0703. [DOI] [PubMed] [Google Scholar]
- Follesa P, Concas A, Porcu P, et al. Role of allopregnanolone in regulation of GABA(A) receptor plasticity during long-term exposure to and withdrawal from progesterone. Brain Research Reviews. 2001;37(1–3):81–90. doi: 10.1016/s0165-0173(01)00125-4. [DOI] [PubMed] [Google Scholar]
- Geary DC, DeSoto MC. Sex differences in spatial abilities among adults from the United States and China. Evolution and Cognition. 2001;7(2):172–7. [Google Scholar]
- Graham CA, Bancroft J, Doll HA, Greco T, Tanner A. Does oral contraceptive-induced reduction in free testosterone adversely affect the sexuality or mood in women? Psychoneuroendocrinology. 2007;32(3):246–55. doi: 10.1016/j.psyneuen.2006.12.011. [DOI] [PubMed] [Google Scholar]
- Grosbras MH, Paus T. Brain networks involved in viewing angry hands or faces. Cerebral Cortex. 2006;16(8):1087–96. doi: 10.1093/cercor/bhj050. [DOI] [PubMed] [Google Scholar]
- Hall JK, Hutton SB, Morgan MJ. Sex differences in scanning faces: does attention to the eyes explain female superiority in facial expression recognition? Cognition and Emotion. 2010;24(4):629–37. [Google Scholar]
- Hall JA, Matsumoto D. Gender differences in judgments of multiple emotions from facial expressions. Emotion. 2004;4(2):201–6. doi: 10.1037/1528-3542.4.2.201. [DOI] [PubMed] [Google Scholar]
- Hampson E, Kimura D. Reciprocal effects of hormonal fluctuations on human motor and perceptual-spatial skills. Behavioral Neuroscience. 1988;102(3):456–9. doi: 10.1037//0735-7044.102.3.456. [DOI] [PubMed] [Google Scholar]
- Hampson E, Vanandres S, Mullin L. A female advantage in the recognition of emotional facial expressions: test of an evolutionary hypothesis. Evolution and Human Behavior. 2006;27(6):401–16. [Google Scholar]
- Hampson E, Young EA. Chapter 4: methodological issues in the study of hormone-behavior relations in humans: understanding and monitoring the menstrual cycle. In: Becker JB, Berkley KJ, Geary N, Hampson E, Herman JP, Young E, editors. Sex Differences in the Brain: From Genes to Behavior. New York: Oxford University Press Inc; 2008; 2007. [Google Scholar]
- Herba C, Phillips M. Annotation: development of facial expression recognition from childhood to adolescence: behavioural and neurological perspectives. Journal of Child Psychology and Psychiatry. 2004;45(7):1185–98. doi: 10.1111/j.1469-7610.2004.00316.x. [DOI] [PubMed] [Google Scholar]
- Herlitz A, Rehnman J. Sex differences in episodic memory. Current Directions in Psychological Science. 2008;17(1):52–6. [Google Scholar]
- Hietala M, Sandberg T, Borg A, Olsson H, Jenstrom H. Testosterone levels in relation to oral contraceptive use and the androgen receptor CAG and GGC length polymorphisms in healthy young women. Human Reproduction. 2007;22(1):83–91. doi: 10.1093/humrep/del318. [DOI] [PubMed] [Google Scholar]
- Kanwisher N, McDermott J, Chun MM. The fusiform face area: a module in human extrastriate cortex specialized for face perception. The Journal of Neuroscience. 1997;17(11):4302–11. doi: 10.1523/JNEUROSCI.17-11-04302.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kastner S, Pinsk MA, De Weerd P, Desimone R, Ungerleider LG. Increased activity in human visual cortex during directed attention in the absence of visual stimulation. Neuron. 1999;22:751–61. doi: 10.1016/s0896-6273(00)80734-5. [DOI] [PubMed] [Google Scholar]
- Kimura D. Sex and Cognition. Cambridge: MIT Press; 1999. [Google Scholar]
- Kirouac G, Doré FY. Accuracy of the judgment of facial expression of emotions as a function of sex and level of education. Journal of Nonverbal Behavior. 1985;9:3–7. [Google Scholar]
- Lane RD, Chua PM, Dolan RJ. Common effects of emotional valence, arousal and attention on neural activation during visual processing of pictures. Neuropsychologia. 1999;37:989–97. doi: 10.1016/s0028-3932(99)00017-2. [DOI] [PubMed] [Google Scholar]
- McBain R, Norton D, Chen Y. Females excel at basic face perception. Acta Psychologica. 2009;130(2):168–73. doi: 10.1016/j.actpsy.2008.12.005. [DOI] [PubMed] [Google Scholar]
- McCarthy MM. Estrogen modulation of oxytocin and its relation to behavior. Advances in Experimental Medicine and Biology. 1995;395:235–45. [PubMed] [Google Scholar]
- McClure EB. A Meta-analytic review of sex differences in facial expression processing and their development in infants, children, and adolescents. Psychological Bulletin. 2000;126(3):424–53. doi: 10.1037/0033-2909.126.3.424. [DOI] [PubMed] [Google Scholar]
- Medical Dictionary (2011). Available at http://de.dict.md/definition/estradiol, accessed 20 November 2012.
- Miyahira A, Morita K, Yamaguchi H, Nonaka K, Maeda H. Gender differences of exploratory eye movements: a life span study. Life Sciences. 2000;68(5):569–77. doi: 10.1016/s0024-3205(00)00963-2. [DOI] [PubMed] [Google Scholar]
- Montagne B, Kessels RPC, Frigerio E, de Haan EHF, Perrett DI. Sex differences in the perception of affective facial expressions: do men really lack emotional sensitivity? Cognitive Processing. 2005;6(2):136–41. doi: 10.1007/s10339-005-0050-6. [DOI] [PubMed] [Google Scholar]
- Mordecai KL, Rubin LH, Maki PM. Effects of menstrual cycle phase and oral contraceptive use on verbal memory. Hormones and Behavior. 2008;54(2):286–93. doi: 10.1016/j.yhbeh.2008.03.006. [DOI] [PubMed] [Google Scholar]
- Morris JP, Pelphrey KA, McCarthy G. Controlled scanpath variation alters fusiform face activation. Social Cognitive and Affective Neuroscience. 2007;2(1):31–8. doi: 10.1093/scan/nsl023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palermo R, Rhodes G. Are you always on my mind? A review of how face perception and attention interact. Neuropsychologia. 2007;45(1):75–92. doi: 10.1016/j.neuropsychologia.2006.04.025. [DOI] [PubMed] [Google Scholar]
- Petrovic P, Kalisch R, Singer T, Dolan RJ. Oxytocin attenuates affective evaluations of conditioned faces and amygdala activity. Journal of Neuroscience. 2008;28(26):6607–15. doi: 10.1523/JNEUROSCI.4572-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman Q, Wilson GD, Abrahams S. Sex, sexual orientation, and identification of positive and negative facial affect. Brain and Cognition. 2004;54(3):179–85. doi: 10.1016/j.bandc.2004.01.002. [DOI] [PubMed] [Google Scholar]
- Rosenberg L, Park S. Verbal and spatial functions across the menstrual cycle in healthy young women. Psychoneuroendocrinology. 2002;27:835–41. doi: 10.1016/s0306-4530(01)00083-x. [DOI] [PubMed] [Google Scholar]
- Sandres G, Sjodin M, de Chastelaine M. On the elusive nature of sex differences in cognition: hormonal influences contributing to within-sex variation. Archives of Sexual Behavior. 2002;31:145–52. doi: 10.1023/a:1014095521499. [DOI] [PubMed] [Google Scholar]
- Schulte-Ruther M, Markowitch HJ, Shah JN, Fink GR, Piefke M. Gender differences in brain networks supporting empathy. NeuroImage. 2008;42:393–403. doi: 10.1016/j.neuroimage.2008.04.180. [DOI] [PubMed] [Google Scholar]
- Schumann G, Loth E, Banaschewski T, et al. The IMAGEN study: reinforcement-related behaviour in normal brain function and psychopathology. Molecular Psychiatry. 2010;15(12):1128–39. doi: 10.1038/mp.2010.4. [DOI] [PubMed] [Google Scholar]
- Silber M, Almkvist O, Lrsson B, Stock S, Uvnas-Moberg K. The effect of oral contraceptive pills on levels of oxytocin in plasma and on cognitive functions. Contraception. 1987;36(6):641–50. doi: 10.1016/0010-7824(87)90037-0. [DOI] [PubMed] [Google Scholar]
- Smith SS. Female sex steroid hormones: from receptors to networks to performance – actions on the sensorimotor system. Progress in Neurobiology. 1994;44:55–86. doi: 10.1016/0301-0082(94)90057-4. [DOI] [PubMed] [Google Scholar]
- Society for Biomedical Diabetes Research (2010). SI Unit Conversion Calculator. Retrieved from http://www.socbdr.org/rds/authors/unit_tables_conversions_and_genetic_dictionaries/e5196/index_en.html, accessed 20 November 2012.
- Stock S, Karlsson R, von Schoultz B. Serum profiles of oxytocin during oral contraceptive treatment. Gynecological Endocrinology. 1994;8(2):121–6. doi: 10.3109/09513599409058033. [DOI] [PubMed] [Google Scholar]
- Tahmasebi A, Artiges E, Banaschewski T, et al. Creating probabilistic maps of the face network in the adolescent brain: a multi-centre functional MRI study. Human Brain Mapping. 2012;33(4):938–57. doi: 10.1002/hbm.21261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan S, Ruffman T, Hutton SB. Age differences in emotion recognition skills and the visual scanning of emotion faces. Journals of Gerontology: Series B. 2007;62(1):53–60. doi: 10.1093/geronb/62.1.p53. [DOI] [PubMed] [Google Scholar]
- Van den Heuvel MW, van Bragt AJ, Alnabawy AK, Kaptein MC. Comparison of ethinylestradiol pharmacokinetics in three hormonal contraceptive formulations: the vaginal ring, the transdermal patch and an oral contraceptive. Contraception. 2005;72(3):168–74. doi: 10.1016/j.contraception.2005.03.005. [DOI] [PubMed] [Google Scholar]
- Wassermann E, Epstein CM, Ziemann U, Walsh V, Paus T, Lisanby SH, editors. The Oxford Handbook of Transcranial Stimulation. Oxford: Oxford University Press; 2008. [Google Scholar]
- Wharton W, Hirshman E, Merritt P, Doyle L, Paris S, Gleason C. Oral contraceptives and androgenicity: influences on visuospatial task performace in younger individuals. Experimental and Clinical Psychopharmacology. 2008;16(2):156–64. doi: 10.1037/1064-1297.16.2.156. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.