Abstract
The investigations on sources and viability of stem cells are important as stem cell transplantation has shown promising results in diseases like leukemias and lymphomas. Umbilical cord blood samples were collected in a closed aseptic system. The samples were diluted with phosphate buffered saline, treated with ficol and centrifuged at 15,000 rpm for the recovery of progenitor stem cells.The stem cells were cryopreserved with different media Containing DMSO, patient’s serum and human albumin.The viability of the cells was studied by dye exclusion method. Suitable media are necessary for optimal cryoprotection and prevention of cryoinjury DMSO is essential for improved cryopreservation and post-thaw quality. The addition of a protein additive also provides a protective effect. The medium containing DMSO, DMEM and patient’s serum proved to be the most effective for cryopreservation and viability as high as 82.4% was achieved after one year. The unique findings of the present study are that the addition of patient’s serum enhances the cryoprotection and cord blood stem cells can be stored at -20°C for the duration up to two months instead of the requirement of storage at ultralow temperature at -186°C.
Keywords: UCB, Viability, Stem cells, Media
Introduction
In view of the rapidly growing potential of stem cell therapies for a number of diseases like leukemia, lymphomas, alzheimers disease, muscular disorders, spinal cord injuries, juvenile diabetes and many other genetic and degenerative diseases(1,2,3) the investigations on analysis of the sources and viability of stem cells are highly relevant.
Main sources of stem cells are bone marrow, adult peripheral blood, embryos and umbilical cord blood. The
retrieval of stem cells from bone marrow involves deep and invasive technique. The numbers of stem cells present in adult peripheral blood are too low and large volumes of blood may be required for sufficient numbers of stem cells. There are ethical issues associated with the use of embryonic stem cells. Umbilical cord blood is a rich source of the stem cells.(4) Both term and pre term cord bloods contain significantly higher number of early and committed stem cells.(5) Umbilical cord blood stem cells are more sensitive to ex vivo expansion and less immunoreactive.(6,7) Hence, instead of being discarded as a biological waste, the umbilical cord blood can be preserved as a source of stem cells. Present study deals with cryopservation and viability testing of cord blood stem cells using different media and conditions.
Mateirals and Methods
Umbilical cord blood samples, UCB collection bags, DMSO, DMEM, Human albumin, Heparin, Cryovials, Cryobox, Micro tubes, Micropipettes, Haemocytometer, serowaterbath etc. Each observation represents the mean of ten replicates.
Collection and processing of Umbilical Cord Blood
Umbilical Blood was collected by puncturing the umbilical vein of full term deliveries in UCB collection bags with anticoagulant after taking the prior consent under aseptic conditions. The samples were collected at a Govt. recognized hospital. Umbilical cord samples were diluted with three volumes of phosphate buffered saline. Diluted blood was layered over 12.5 ml. of ficol in 50 ml. centrifuge tubes and centrifuged at 15000 rpm for 25 minutes.
The MNC (Mononuclear cell) layer was separated, washed twice with PBS and centrifuged at 15000 rpm for 5 minutes and resuspended in 1ml. PBS. The cells were counted using haemocytometer. The viability of the cell was checked before cryopreservation.
The cord blood samples were cryopreserved with three different media: Medium 1, Medium 2 and Medium 3.
Composition of different cryoprotectants used was as follows:
Medium 1 Patient’s serum and DMSO in a ratio of 9.5: 0.5 Medium 2 Patient’s serum, DMSO and DMEM in a ratio o 3:1:1 Medium 3 Normal saline, DMSO and Human albumin (20%) in the ratio of 1:0.28:0.75.
Cryopreservation
Cryovials were labeled properly. MNC suspension was divided into three portions. MNC cell suspension was then mixed with cryoprotectant Medium 1, Medium 2 and Medium 3 respectively at final approximate cell concentrations of 1×106 cells per ml. Samples were cooled by gradually decreasing the temperature using rate control freezer at a freezing rate of 1° C/minute till -40°C and then samples were frozen at -86°C. Some aliquots of the samples cryopreserved in different media were stored at -20°C but most of the aliquots were maintained at -86°C
Thawing
Cryopreserved stem cells were thawed immediately thawed at 37°C using the serowaterbath.
Viability Testing
Viability of cryopreserved stem cells stored at room temperature, 4°C, -20°C and t minus 86°C was examined. Viability of the cells was checked by trypan blue dye exclusion test % Viability = White cell×100/White cells + blue cells
Results
Volume of umbilical cord blood ranged from 100 to 150 ml. Total number of mononuclear cells in UCB was in the range of 3.5×106 to 4.1×108 per ml. before cryopreservation.
The viability of the cord blood stem cells was assessed before and after cryopreservation. Progenitor cells remain viable in untreated anticoagulant added UCB for three days at 4°C.
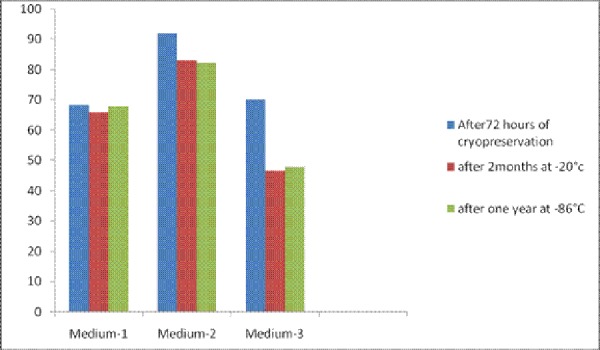
The viability of stem cell deteriorates quickly with time. After 24 hours a significant proportion stem cells become nonviable (Table1). Therefore, cord blood samples were processed for cryopreservation within two to four hours after collection. With medium 1; comprising of patient’s serum and DMSO the viability of cord blood cells was 68.4% after 72 hours of cryopreservation, 66% after two months of storage at -20°C subsequent to cryopreservation and 68% when the cells were preserved at -86°C for one year subsequent to cryopreservation. (figure 1) When medium-2; comprising of a mixture of patient’s serum, DMSO and DMEM was used as a cryoprotectant the viability of cord blood cells was 92% after 72 hours of cryopreservation, 83% after two months of storage at -20°C subsequent to cryopreservation and 82.4% when the cord blood cells were maintained at minus 86°C for one year after cryopreservation (figure 1). When medium 3; comprising of DMSO, human albumin and normal saline was used the viability of cord blood cells was 70.2% after 72 hours of cryopreservation. The viability of cells maintained at -20°C after two months of cryopreservation was 46.6%. The viability was reduced to 48% when the cells were maintained at -86°C for one year after cryopreservation (figure 1). Significantly high percentage of viability was achieved with cryoprotectant medium 2 even after one year of storage at -86°C.
Table 1.
Viability of cord blood cells before Cryopreservation at 4°C
| Time after UCB Collection | Percentage Viability |
| 2 hours | 98.6±2.5 |
| 24 hours | 83.9±3.6 |
| 48 hours | 70.9±4.4 |
| 72 hours | 56.6±4.5 |
Figure I.
Discussion
Umbilical cord blood (UCB) is a very useful source of precious primitive hematopoietic stem cells (HSC) and progenitor cells. It exists in limitless supply and can be safely used for reconstituting the hematopoietic system in patients with malignant and nonmalignant disorders treated with myeloablative therapy. UCB cells possess an enhanced capacity for progenitor cell proliferation and self-renewal in vitro.(8) In order to avoid microbial contamination closed collection system was used for collect the umbilical cord blood by adopting simple venipuncture followed by gravity drainage into a standard sterile anti-coagulant-filled blood bag. After aliquots have been removed for routine testing, the units were cryopreserved. Stem cells can be successfully stored at low temperature of -86°C and ultra low temperature of -186°C for later use after a prolonged period. Cells when subjected to extremely low temperature suffer from cryo-injury and get damaged due to thermal shock, membrane shearing forces and formation of free radicals.
Suitable media are therefore, necessary for optimal cryoprotection and prevention of cryoinjury. Stem cells were processed and resuspended in cryoprotectant media which protect the cell from damaging factors, at low temperatures.
DMSO is essential for improved cryopreservation and post-Lhaw quality. The addition of a protein additive also provides a protective effect. When the patient’s serum was used as a source of protein additive to DMSO (the constituents of Medium-1) significant viability of 68% was achieved after one year of cryopreservation when the samples were stored at -86°C instead of being stored at -186°C.The inclusion of DMEM as a source of additional nutrients further enhanced our medium. With DMSO, patient’s serum and DMEM as constituents of Medium-2; the viability of cord blood cells was raised to 83% after one year of cryopreservation when the samples were maintained at -86°C instead of being stored at -186°C. Subsequent to cryopreservation with Medium-2 samples could even be maintained at minus 20°C for two months with viability as high as 83%.
The sample cryopreserved with Medium-2 also exhibited good post thaw viability. The cord blood stem cells were viable for four hours after thawing with viability as high as74.6% at room temperature. The cocktail of cryoprotectant (Medium 2) consisting of patient’s serum, DMSO and DMEM was found to be most effective for cryopreservation as indicated by a high level of post thaw viability of 92% immediately after crypreservation. This medium was capable of maintaining an appreciable viability of 74.6% when the samples were kept at room temperature for four hours after thawing and this medium proved to be most effective in protecting the cells against cryo-injury and the most suitable medium for long term cryopreservation even after one year. The unique findings of the present study are (1) the addition of patient’s serum enhances the cryoprotection and (2)With our medium cord blood stem cells can be stored at -20°C for the duration up to two months instead of the requirement of storage at ultralow temperature at minus186°C.
Table 2.
Viability cord blood stem cells after cryopreservation The viability of stem cells preserved in different cryoprotectant media for 72hours was tested immediately after thawing
| Cryoprotectant Medium | Percentage Viability immediately after thawing | Percentage Viability room temperature four hours after thawing |
| Medium–1. | 68.4±4.2 | 62.0±4.6 |
| Medium--2. | 92.0±4.8 | 74.6±2.2 |
| Medium--3 | 70.2±3.6 | 49.0±2.8 |
Table 3.
Long-term Viability of cord blood stem cells stored at -20°C after cryopreservation The viability of cord blood stem cells maintained at -20 °C after cryopreservation was examined immediately after thawing.
| Cryoprotectant Medium | Percentage Viability |
| Medium–1. | 66.0±4.4 |
| Medium–2. | 83±4.6 |
| Medium--3 | 46.6±3.6 |
Table 4.
Long-term Viability of cord blood stem cells stored at -86°C after one year of cryopreservation
| Cryoprotectant Medium | Percentage Viability |
| Medium–1. | 68.0±4.6 |
| Medium–2. | 82.4±2.6 |
| Medium–3 | 48±4.2 |
Acknowledgements
Author is deeply indebted to Dr. Deepti Saigal, Senior Resident, Safdarjung Hospital for helping in the collection of umbilical cord blood samples.
References
- (1).Gahrton G, Bjorkstrand B.(2000). “Progress in haematopoietic stem cell transplantation for multiple myeloma”. J Intern Med 248 (3): 185-201 [DOI] [PubMed] [Google Scholar]
- (2).Lindvall O.(2003). “Stem cells for cell therapy in Parkinson’s disease”. Pharmacol Res 47 (4): 279?87 [DOI] [PubMed] [Google Scholar]
- (3).Goldman S, Windrem M.(2006). “Cell replacement therapy in neurological disease”. Philos Trans R Soc Lond B Biol Sci 361 (1473): 1463-75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- (4).Smith S, Broxmeyer HE.1986. The influence of oxygen tension on the long-term growth in vitro of haematopoietic progenitor cells from human cord blood. British Journal Haematology 63(1):29-34 [DOI] [PubMed] [Google Scholar]
- (5).Nakahata T, Ogawa M.1982. Hemopoietic colony-forming cells in umbilical cord blood with extensive capability to generate mono- and multipotential hemopoietic progenitors. Journal of Clinical Investigation 70(6): 1324-1328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Sirchia G, Rebulla P.1999. Placental/umbilical cord blood transplantation. Haematologica 84(8):738-747 [PubMed] [Google Scholar]
- (7).Thierry D, Hervatin F, Traineau R, Brossard Y, Stark R, Benbunan M, Gluckman E: 1992hematopoietic progenitor cells in cord blood. Bone Marrow Transplant 9:101. [PubMed] [Google Scholar]
- (8).Mitchell S. Cairo and, Wagner J.E: 1997Review article: Placental and/or Umbilical Cord Blood: An Alternative Source of Hematopoietic Stem Cells for Transplantation Blood, Vol. 90 No. l2 (December15), 1997: pp. 4665-4678 [PubMed] [Google Scholar]


