Abstract
The present study evaluates the immune response of memory CD4+ and CD8+ T cells from patients following a natural Vaccinia virus (VACV) infection. A total of 42 individuals were involved in the study being: 22 previously infected individuals (vaccinated or not against smallpox) and 20 non-infected individuals (vaccinated or not). A short-term in vitro stimulation with UV-inactivated VACV of whole blood cells was performed. Our study showed that previously infected individuals have a lower percentage of CD4+ T cells expressing lymph-node homing receptors (CD4+CD62L+CCR7+) and higher percentage of memory CD4+ T cells subsets (CD4+CD45ROHigh) when compared with non-infected subjects, after in vitro viral stimulation. We also showed that infected individuals presented higher percentages of CD4+ and CD8+ memory T lymphocytes expressing IFN-γ when compared to non-infected individuals. We verified that the percentage of CD4+ and CD8+ T memory cells expressing TNF-α was higher in infected and non-infected vaccinated subjects when compared with non-infected unvaccinated individual. We also observed that previously infected individuals have higher percentages of CD8+ T cells expressing lymph-node homing receptors (CCR7+ and CD62L+) and that the memory T cells expressing IFN-γ and TNF-α were at higher percentages in the whole blood cells from infected and non-infected vaccinated individuals, when compared to unvaccinated non-infected subjects. Thus, our findings suggest that CD4+ and CD8+ T cells are involved in the immune memory response against Vaccinia virus natural infection.
Keywords: Vaccinia virus, Poxviridae infections, Infection, Zoonoses, Smallpox vaccine
1. Introduction
Vaccinia virus (VACV) is the causative agent of a zoonotic infection that affects cattle and humans in many regions of Brazil, since 1969 [1]. The occurrence of these outbreaks is interesting as some individuals have no immunity against this infection and there are no immunological studies on the duration of immunity after natural human VACV infection. Most of the studies have evaluated the duration of immunity after smallpox vaccination [2–8], but there is little information about the immunity against a natural Smallpox and VACV human infection [7,9].
Smallpox vaccine induces strong CD4+ and CD8+ T cell responses that peak at 2–4 weeks post-immunization and decreasing to form a stable memory population of T cells thereafter [5,10]. Previous studies demonstrated the importance of the CD4+ T cell compartment and have shown that long-term cellular memory to VACV predominantly involves CD4+ Th1 IFN-γ producing cells [3,11,12] and have observed a reduction over time of VACV-specific memory T cells in previously vaccinated individuals that retained a predominant CD4+ memory T cell response [5,11]. The CD8+ T cell memory population seems to decline faster than memory CD4+ T cells [3]. These studies investigated the CD4+ and CD8+ memory T cells against the Orthopoxvirus in some individuals after smallpox infection [9,13] or in vaccinated individuals [5,9,11,12]. In humans, two broad memory T cell subsets (CD4+ or CD8+) have been delineated: central memory (TCM) and effector memory (TEM) T cells [14–16]. TCM are CD45RO+ memory cells that constitutively express CCR7 and CD62L (TCMCD45RO+CCR7+CD62L+), home to the lymph nodes, produce low levels of IFN-γ and have limited effector function, but they proliferate and become effector cells upon secondary stimulation [17–22]. By contrast TEM are memory cells that have lost the constitutive expression of CCR7, are heterogeneous for CD62L (TEMCD45RO+CCR7−CD62L−/+), home to peripheral tissues, and can rapidly produce effector cytokines such as IFN-γ upon antigenic stimulation [17–19,22]. T memory cells are not classified just by the expression of the surface markers CD45RO, CD62L and CCR7. It is also necessary to evaluate the production of effector cytokines, such as IFN-γ. TEM expresses high levels of activation markers (such as CD45RO), lose expression of the chemokine receptor CCR7, and acquire the capacity to produce IFN-γ and to kill target cells. In contrast, TCM cells retain CCR7 expression, and fail to acquire cytotoxic function, although produce low levels of IFN-γ [17–22].
The importance of the role of CD4+ and CD8+ T cells in the control of VACV infection, in the induction and maintenance of long-term memory is well known [23], but the phenotype of these T cell subsets was not described. In the present study, we evaluated the phenotype of CD4+ and CD8+ T cells expressing activation markers (CD45RO), lymph node-homing receptors L-selectin (CD62L) and CCR7 and production of cytokines (IFN-γ and TNF-α) after in vitro stimulation with UV-inactivated VACV following a natural Vaccinia virus infection after a zoonotic outbreak that occurred in Brazil in 2005.
2. Participants, materials, and methods
2.1. Study population
In 2005 a zoonotic outbreak of VACV infection occurred in different areas in Minas Gerais state, Brazil. During the outbreak, blood from these individuals and from cattle were collected and sent to the Virus Laboratory of the Federal University of Minas Gerais (UFMG) and to the Immunology Laboratory of the Oswaldo Cruz Foundation (FIOCRUZ) as a part of the outbreak investigation to confirm the infection with Vaccinia virus. For confirmation of the infection some experiments were performed as previously described by virus isolation, phylogenetic analysis, plaque-reduction neutralization test and IgG ELISA [24]. As an extension of the outbreak investigation, this study was initiated to assess immune responses 5 years post-exposure. The same individuals were recruited and specimens collected to investigate the profile of CD4 and CD8 memory T cells. A total of 42 individuals were considered in these analyses. The studied group consisted of 22 infected human patients and 20 non-infected and healthy individuals. Importantly, out of 22 individuals previously infected, 12 were vaccinated against smallpox in the past, as confirmed by visualization of a typical vaccination scar in their left arm. Of the 20 non-infected individuals, 10 were vaccinated against smallpox in the past, as confirmed by visualization of a typical vaccination scar in their left arm and by data on the vaccination card; 10 were unvaccinated (control group) (Table 1). All patients were previously examined by a physician and those presenting apparent clinical signs of any other disease, infectious or not, were not included in the study. All study participants provided a written informed consent following the guidelines of the Ethics Committee of the Federal University of Minas Gerais.
Table 1.
Characterization of the population involved in the study.
| Groups | Vaccination status | Number of individuals studied | Mean age (min–max) | Gender (n) |
|
|---|---|---|---|---|---|
| Male | Female | ||||
| Previously infecteda | Vaccinatedb | 12 | 56 (42–68) | 12 | 0 |
| Unvaccinated | 10 | 24.5 (18–34) | 10 | 0 | |
| Non-infected | Vaccinatedc | 10 | 46.4 (39–62) | 7 | 3 |
| Unvaccinated | 10 | 29.2 (28–31) | 6 | 4 | |
These individuals were infected with Vaccinia virus during outbreaks that occurred in 2005 in different farms in the state of Minas Gerais, Brazil.
Importantly, out of 22 individuals previously infected, 12 were vaccinated against smallpox in the past, as confirmed by visualization of a typical vaccination scar in their left arm.
Of the 20 individuals non-infected, 10 were vaccinated against smallpox in the past, as confirmed by visualization of a typical vaccination scar in their left arm and data from vaccination card.
2.2. Viruses and cells
Vero cells were used for virus replication, titration and UV inactivated viral test [25]. A thin layer of viral stocks, at a distance of 15 cm, was exposed for 5–10 min to UV radiation at a wavelength of 280 nm. UV irradiated viruses that were unable to form plaques were considered to be UV inactivated.
2.3. Cell phenotype analysis
In vitro short-term cultures of whole blood samples were performed. Whole blood was stimulated in vitro with UV-inactivated VACV for 6 h at 37 °C and 5% CO2. Approximately 1 × 104 PFU of Vaccinia virus strain WR was added per 1 × 106 leucocytes. Control cultures were maintained in culture media for the same period of time. Cultured cells were washed in FACS buffer and stained with monoclonal antibodies against CD4, CD8, CD45RO, CCR7, and CD62L. Cell preparation was incubated in the dark for 30 min at room temperature. Following incubation, erythrocytes were lysed, the cells washed and fixed in FACS fix solution. A total of 100,000 events/tube were acquired using a FACScalibur flow cytometer (Becton Dickinson). The CELLQuestTM software was used for data acquisition and the FLOW JO Version 7.5.5 software for analysis.
2.4. Intracellular cytokine staining (ICCS)
Whole blood was stimulated in vitro with UV-inactivated VACV as described above. During the last 4 h of culture, Brefeldin A (10 mg/mL) was added to the cultures. Cultured cells were washed and stained with monoclonal antibodies against the different cell-surface markers, as described above. The cells were then fixed in FACS fix solution. Finally, the cells were incubated and stained intracellularly with monoclonal antibodies against IFN-γ and TNF-α PE. A total of 100,000 events/tube were acquired using a FACScalibur flow cytometer (BD Biosciences, USA). Samples were analyzed using the FlowJo software (Tree Star – Version 7.5.5).
2.5. FACS analysis of surface markers and intracellular cytokine
Lymphocytes were analyzed for their intracellular cytokine expression patterns and frequencies as well as for cell surface markers using the FlowJo software (Tree Star – Version 7.5.5), as described in Supplementary Fig. S1. First we performed the identification of peripheral lymphocytes population in diagram of FSC × SSC (Fig. S1A). Second we made dot plots of FL-1 × FL-4, displaying the frequency of CD4+CD45RO+ cells. The CD4+CD45ROLow (R1) and CD4+CD45ROHigh (R2) populations were sorted using the indicated sorting gates (Fig. S1B). The same strategy was used to select the CD4+CD62L+ subpopulations. Dot plots of FL-1 × FL-4 were made displaying the frequency of CD4+CD62L+ cells. The CD4+CD62Lneg (R1), CD4+CD62LLow (R2) and CD4+CD62LHigh (R3) populations were sorted using the indicated sorting gates (Fig. S1C). Finally, we made dot plots of FL-4 × FL-2, displaying the frequency of CD4+CD45ROLowIFN-γ+, CD4+CD45ROHighIFN-γ+, CD4+CD62LnegIFN-γ+, CD4+CD62LLowIFN-γ+ and CD4+CD62LHighIFN-γ+ (Fig. S1D). Limits for the quadrant markers were always set based on negative populations and isotype controls.
2.6. Statistical analysis
Analyses were performed using GraphPad Prism version 4.0 software (GraphPad Software Inc., USA). The nonparametric Mann–Whitney U test was used to compare stimulated and non-stimulated cultures in each of the four groups. The Kruskal–Wallis test was used to compare the four clinical groups, followed by Dunn's test to compare all stimulated and non-stimulated cultures. Differences were considered significant when a p value of less than 0.05 was obtained.
3. Results
3.1. Previous vaccination leads to a mild form of VACV infection
Some of the infected individuals were previously vaccinated against smallpox, so we evaluated the clinical differences that occurred between the vaccinated and unvaccinated individuals. For this purpose we applied and analyzed a questionnaire previously validated by our group. In unvaccinated individuals, the mean age was 24.5 years with a minimum of 18 years and a maximum of 34. However, in patients previously vaccinated, the mean age was 56 years, with a minimum of 42 and a maximum of 68. All subjects studied were male milkers and the average time worked in this function was 18 years in the unvaccinated individuals and 32 in vaccinated patients. None of these individuals reported having prior infection by VACV. It was observed that the clinical symptoms (fever, headache, muscle pain, nausea, lymphadenopathy, headache, and others) occurred in a milder form in the vaccinated individuals. Analysis of the clinical symptoms showed that in unvaccinated patients the mean was six symptoms, ranging from 4 to 7; in the vaccinated individual the mean was 2.5 symptoms, ranging from 1 to 7. As for the features of the lesions, the vaccinated individuals had an average of 2.5 injuries and the unvaccinated individuals an average of eight lesions. In relation to the place of occurrence of these lesions, in the vaccinated group they concentrated largely on the hands (100%) and small events in other regions (17%) (arms, nose and legs), while in the unvaccinated, the primary site of involvement were the hands (100%), however, it also occurred in other regions (40%) (face, neck and legs). Assessing the days of leave of absence due to illness it was found that 25% of the vaccinated group had an average of 10 days off and in the unvaccinated 60% of individuals with an average of 15 days of sick leave.
3.2. Previously infected individuals have lower percentages of CD4+ T cells expressing limph-node homing receptors cells but higher percentages of CD4 effectors T cells and CD8 T cells expressing lymph-node homing receptors
The mean percentage of T lymphocytes (CD3+) and its subsets (CD4+ and CD8+) (Table 2) was not different when the four groups were compared. In order to evaluate the CD4 and CD8 T cells phenotype, we used lymph node-homing receptors, L-selectin (CD62L) and CCR7 and activation markers (CD45RO, IFN-γ, TNF-α). Our data showed that the expression of CCR7 on CD4+ T cells, was significantly lower in the infected groups – vaccinated (p < 0.05) and unvaccinated (p < 0.01) – after in vitro stimulation (Fig. 1A) when compared with non-infected (vaccinated and unvaccinated). Despite this observation, the percentage of CCR7 negative CD4 T cells (CD4+CCR7−) was significantly higher, in the infected groups, vaccinated (p < 0.05) and unvaccinated (p < 0.01), after in vitro stimulation (Fig. 1B), when compared with non-infected (vaccinated and unvaccinated). The results also showed that previously infected individuals had higher percentage of CD4+ T memory cells (CD4+CD62LnegCCR7−) when compared with non-infected subjects (vaccinated and unvaccinated) (Fig. 1C). Our data also showed that the expression of CD45ROHigh by CD4+ T cells (memory lymphocytes) was statistically higher in the infected groups – vaccinated (p < 0.01) and unvaccinated (p < 0.05) – after in vitro stimulation with VACV, when compared to non-infected individuals (vaccinated and unvaccinated). In the non-infected individuals, there were no increases in the percentage of CD4 memory T cells (Fig. 1D).
Table 2.
Mean percentage of T cells (CD3+, CD4+, CD8+) on the whole blood from individuals infected (vaccinated and unvaccinated) and noninfected (vaccinated and unvaccinated) by Vaccinia virus, after in vitro stimulation with UV-inactivated VACV.
| Groups | Cell phenotype |
|||
|---|---|---|---|---|
| CD3+ (%)a | CD4+ (%)a | CD8+ (%)a | ||
| Non-infected | Unvaccinated | 71.62 ± 6.31 | 42.9 ± 5.36 | 23.368 ± 5.29 |
| Vaccinated | 65.18 ± 8.19 | 45.66 ± 8.78 | 19.828 ± 2.82 | |
| Infected | Unvaccinated | 76.28 ± 7,61 | 40.9 ± 6.33 | 22.367 ± 5.04 |
| Vaccinated | 72.18 ± 5.63 | 45.95 ± 7.08 | 20.903 ± 4.46 | |
There were no statistically significant differences in the mean percentage of T cells (CD3+, CD4+, CD8+) when the four groups were compared.
Fig. 1.
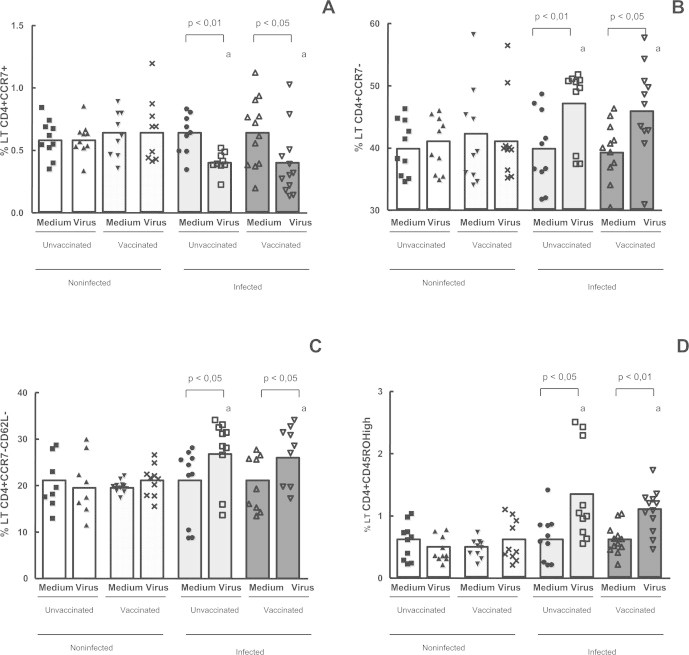
Evaluation of memory and naïve TCD4+ cells in the whole blood cells from individuals previously infected or not by zoonotic Vaccinia virus. Whole blood cells from patients infected or not with zoonotic Vaccinia virus were stimulated in the presence of UV-inactivated VACV or mock-treated (medium). After stimulus, cells were labeled with mouse anti-human CD4, CD62L, CCR7 and CD45 antibodies. The percentages of CD4+ CCR7+ (A); CD4+ CCR7− (B); CD4+ CCR7−CD62L− and CD4+ CD45ROHigh T cell subsets were determined by flow citometry. Statistical significance (P values) is presented on the graphs. *a means different from non-infected unvaccinated, stimulated cultures.
On the other hand the expression of CCR7 in CD8+ T cells was higher in the infected groups (vaccinated and unvaccinated) when compared with non-infected (vaccinated and unvaccinated) subjects, after viral stimulation (Fig. 2A). The results also show that infected (vaccinated and unvaccinated) individuals have a higher percentage of CD8+ T cells expressing lymph node-homing receptors (CD8+CD62L+CCR7+) when compared with non-infected (vaccinated and unvaccinated) individuals (Fig. 2B). Furthermore, when we analyzed the subpopulation of CD8+CD45ROHigh memory T cells, we observed that there were a statistically higher percentage of these cells only in the infected groups (vaccinated and unvaccinated) (p < 0.05) after in vitro stimulation with VACV, when compared with non-infected (vaccinated and unvaccinated) patients.
Fig. 2.
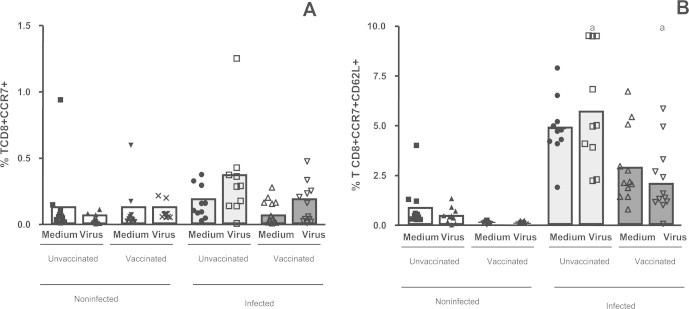
Evaluation of naïve and memory TCD8+ cells in the whole blood cells from individuals previously infected or not by zoonotic Vaccinia virus. Whole blood cells from patients infected or not with zoonotic Vaccinia virus were stimulated in the presence of UV-inactivated VACV or mock-treated (medium). After stimulus, cells were labeled with mouse anti-human CD8, CD62L, CCR7 antibodies. The percentages of CD8+ CCR7+ (A) and CD8+ CCR7+CD62L+ (B) T cell subsets were determined by flow citometry. Statistical significance (P values) is presented on the graphs. *a means different from non-infected unvaccinated, stimulated cultures.
3.3. Vaccinia virus exposure leads to higher percentages of IFN-γ and TNF-α expression by CD4+ T cells and its subsets (CD4+CD45RO+ and CD4+CD62Lneg)
As it is well established that Vaccinia virus infection induces strong cellular responses capable of secreting effectors cytokines such as IFN γ and TNF-α, we evaluated the expression of these markers. In this study the expression levels of IFN-γ and TNF-α on CD4+ T lymphocytes were evaluated and the results showed that expression of IFN-γ and TNF-α on CD4+ T cells were significantly higher in the infected (vaccinated and unvaccinated) individuals and in the non-infected vaccinated subjects, after in vitro stimulation with VACV, when compared with non-infected unvaccinated patients (Fig. 3A and B).
Fig. 3.
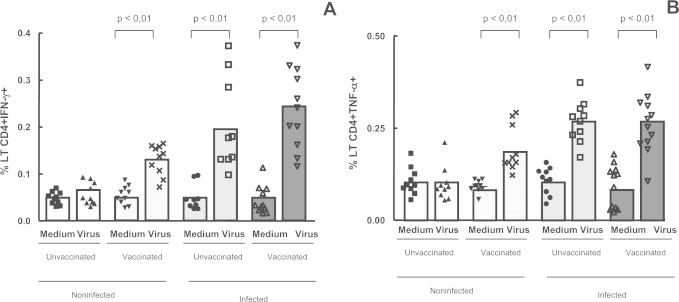
Intracellular IFN-γ and TNF-α detection in the whole blood cells from individuals previously infected or not by zoonotic Vaccinia virus. Whole blood cells from patients infected or not with zoonotic Vaccinia virus were stimulated in the presence of UV-inactivated VACV or mock-treated (medium). After stimulus, cells were labeled with mouse anti-human CD4 antibodies and intracellular staining was used to measure the percentage of positive IFN-γ and TNF-α CD4 T cells. The percentages of CD4+ IFN-γ + (A) and CD4+ TNF-α+ (B) T cell subsets were determined by flow citometry. Statistical significance (P values) is presented on the graphs. *a means different from non-infected unvaccinated, stimulated cultures.
Following the identification of CD4+ T cells expressing IFN-γ and TNF-α, the intracytoplasmatic presence of these cytokines in CD4+CD45RO+, CD4+CD62L− (activated cells) and CD4CD62LLow T cells was evaluated in the four groups, in the presence of in vitro stimulation by UV-inactivated VACV. The mean percentage T CD4 lymphocytes co-expressing CD45RO and IFN-γ showed that only the subjects who had been previously infected – both vaccinated (p < 0.001) and unvaccinated (p < 0.01) – presented a significantly higher percentages of CD4+CD45RO+ and CD4+CD45ROHigh T memory cells expressing IFN-γ (Fig. 4A and B), when compared with non-infected (vaccinated and unvaccinated) individuals. We compared in Fig. 4C and D the production of IFN-γ by CD4 T cells that lost the selectin, CD62L, to those cells that appear to regain the marker (CD62LLow), those called TCM cells. Expression levels of IFN-γ were measured on CD4+CD62Lneg T cells, and a significant increase of these cells was detected only in the infected individuals – vaccinated (p < 0.01) and unvaccinated (p < 0.01), after culture in the presence of UV-inactivated VACV, when compared with non-infected (vaccinated and unvaccinated) patients (Fig. 4C). Analysis of IFN-γ on CD4+CD62L+ and CD4+CD62LLow T lymphocytes showed that there were significantly higher percentages of this CD4+ T cell subset only in the infected (vaccinated and unvaccinated) groups, after in vitro stimulation with VACV, when compared with non-infected (vaccinated and unvaccinated) patients (Fig. 4D).
Fig. 4.
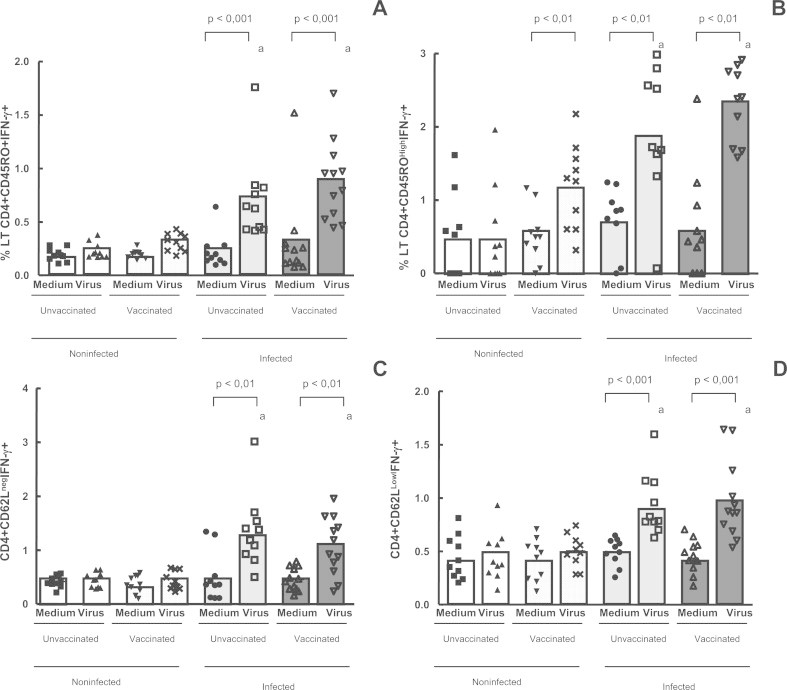
Intracellular IFN-γ detection in CD4+ T memory cells in the whole blood cells from individuals previously infected or not by zoonotic Vaccinia virus. Whole blood cells from patients infected or not with zoonotic Vaccinia virus were stimulated in the presence of UV-inactivated VACV or mock-treated (medium). After stimulus, cells were labeled with mouse anti-human CD4, CD45RO and CD62L antibodies and intracellular staining was used to measure the percentage of positive IFN-γ cells. The percentages of CD4+CD45RO+IFN-γ+ (A); CD4+CD45ROHighIFN-γ+ (B); CD4+CD62LnegIFN-γ+ (C) and CD4+CD62LowIFN-γ+ T cell subsets were determined by flow citometry. Statistical significance (P values) is presented on the graphs. *a means different from non-infected unvaccinated, stimulated cultures.
Analysis of the expression of TNF-α on CD4+CD45RO+ and CD4+CD45ROLow T memory cells showed that there were significantly higher percentages of these cells in the non-infected vaccinated individuals (p < 0.05) and in the infected (vaccinated and unvaccinated) subjects (p < 0.01), when compared with non-infected unvaccinated patients (Fig. 5A), after viral stimulation. The mean percentage of CD45ROHighCD4+TNFα+ T memory cells was lower in the non-infected unvaccinated group (p < 0.01) and were not different when we compared the other groups (Fig. 5B).
Fig. 5.
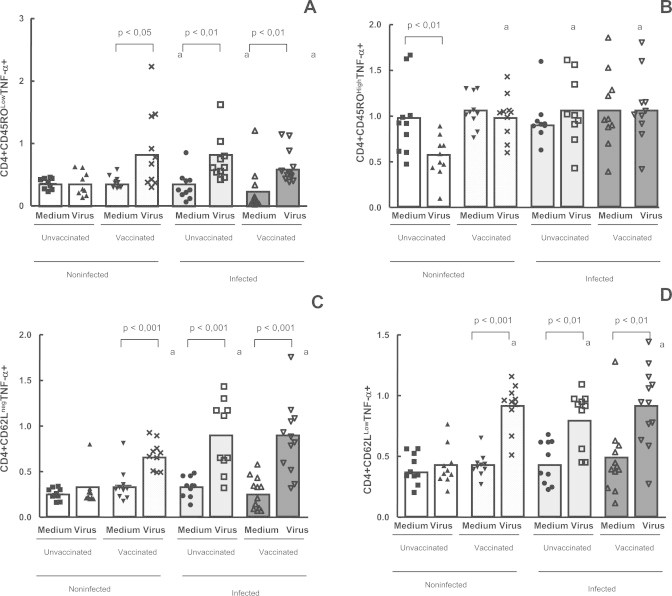
Evaluation of CD4+ TNF-α+ T memory cells, using intracellular staining in the whole blood cells from individuals previously infected or not by zoonotic Vaccinia virus. Whole blood cells from patients infected or not with zoonotic Vaccinia virus were stimulated in the presence of UV-inactivated VACV or mock-treated (medium). After stimulus, cells were labeled with mouse anti-human CD4, CD45RO and CD62L antibodies and intracellular staining was used to measure the percentage of positive TNF-α+ cells. The percentages of CD4+CD45ROLow+ TNF-α+ (A); CD4+CD45ROHighTNF-α+ (B); CD4+CD62Lneg TNF-α+ (C) and CD4+CD62LowTNF-α+ T-cell subsets were determined by flow citometry. Statistical significance (P values) is presented on the graphs. *a means different from non-infected unvaccinated, stimulated cultures.
In this study we compared the expression of TNF-α by CD4 effectors T cells that lost the selectin, CD62L, to those cells that appear to regain the marker. Expression levels of TNF-α on CD4+CD62Lneg and CD4+CD62LLow T effectors cells showed a significantly higher (p < 0.05) percentage of these cells in the infected (vaccinated and unvaccinated) groups and in the non-infected vaccinated individuals, after culture in the presence of UV-inactivated VACV, but not in the non-infected unvaccinated group (Fig. 5C and D).
3.4. Previous Vaccinia virus exposure leads to higher percentages of IFN-γ and TNF-α by CD8+ memory (CD8+CD45RO+) and lymph-node homing (CD8+CD62L+) T cells
Following the identification of CD8+ T cells expressing lymph-node homing receptors and activations markers, we evaluated the intracytoplasmatic expression of the effectors cytokines IFN-γ and TNF-α by these cells, in the groups of non-infected and infected individuals (vaccinated and unvaccinated), after in vitro stimulation by UV-inactivated VACV.
The data showed that only the infected individuals – both vaccinated (p < 0.001) and unvaccinated (p < 0.01) – presented a significantly higher percentage of IFN-γ producing activated CD8+ T cells (CD8+CD45RO+IFN-γ+ and CD8+CD45ROLowIFN-γ+), after in vitro stimulation (Fig. 6A and B). Analysis of CD8+ CD45ROHigh T cells expressing IFN-γ did not show any significant difference when we compare the percentage of these cells in all four groups, after in vitro stimulation (data not shown). We also analyzed the CD8+CD62L+ expressing IFN-γ and we observed a significantly higher percentage (p < 0.05) of these cells in the infected subjects and in the non-infected vaccinated individuals, after culture in the presence of UV-inactivated VACV, but not in the non-infected unvaccinated group (Fig. 6C). Analysis of CD8+CD62L+ and CD8+CD62LHigh T cells expressing TNF-α showed that a significantly higher (p < 0.05) percentage of these cells was observed in the infected subjects and in the non-infected vaccinated individuals, after viral stimulation, but not in the non-infected unvaccinated group (Fig. 7A and B). Furthermore the analysis of CD8+ activated cells showed that the CD8+CD45RO+ T effectors cells expressing TNF-α were at higher percentages only in the infected individuals, when we compared the stimulated cultures (Fig. 7C).
Fig. 6.
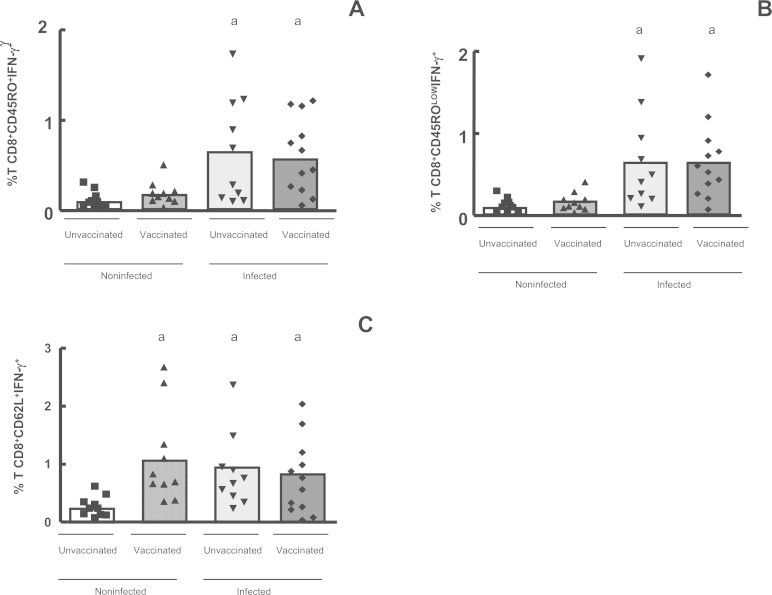
Intracellular IFN-γ detection in CD8+ memory and naïve T cells in the whole blood cells from individuals previously infected or not by zoonotic Vaccinia virus, after viral stimulation. Whole blood cells from patients infected or not with zoonotic Vaccinia virus were stimulated in the presence of UV-inactivated VACV. After stimulus, cells were labeled with mouse anti-human CD8, CD45RO and CD62L antibodies and intracellular staining was used to measure the percentage of positive IFN-γ cells. The percentages of CD8+CD45RO+IFN-γ+ (A); CD8+CD45ROLowIFN-γ+ (B); and CD8+CD62L+IFN-γ+ (C) T cell subsets were determined by flow citometry. Statistical significance (P values) is presented on the graphs. *a means different from non-infected unvaccinated, stimulated cultures.
Fig. 7.
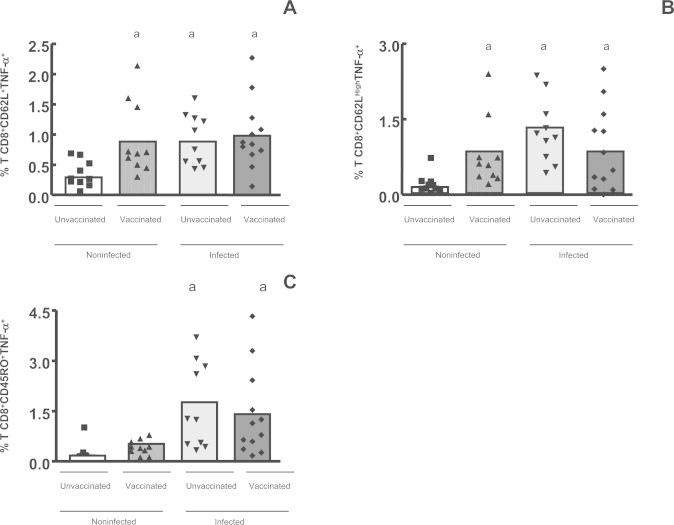
Intracellular TNF-α+ detection in CD8+ naïve and memory T cells in the whole blood cells from individuals previously infected or not by zoonotic Vaccinia virus, after viral stimulation. Whole blood cells from patients infected or not with zoonotic Vaccinia virus were stimulated in the presence of UV-inactivated VACV. After stimulus, cells were labeled with mouse anti-human CD8, CD45RO and CD62L antibodies and intracellular staining was used to measure the percentage of positive TNF-α+ cells. The percentages of CD8+CD62L+TNF-α+ (A); CD8+CD62LowTNF-α+ (B) and CD8+CD45RO+TNF-α+ (C) T-cell subsets were determined by flow citometry. Statistical significance (P values) is presented on the graphs. *a means different from non-infected unvaccinated, stimulated cultures.
4. Discussion
In this study the phenotype of CD4+ and CD8+ memory T cells was described following a natural Vaccinia virus infection and compared with the profile of CD4+ and CD8+ T memory cells induced by smallpox vaccination. Some differences were detected comparing CD4 and CD8 T cells, but the data clearly indicated that these cells are involved in the immune response to the VACV infection. In the present study, 12 (54%) of the 22 individuals previously infected by Vaccinia virus received a previous (childhood) smallpox vaccination. Despite their vaccination status, they acquired the VACV infection, but developed a milder form. A possible explanation for this fact is that Vaccinia virus infection occurred long after the vaccination, at least 30 years before the recent infection, suggesting that previous exposure to the antigen induced partial protection that was reflected on a milder symptoms. However, at this point it is not possible to elucidate what mechanisms are involved in this process. Karem and colleagues correlated the absence of complete smallpox vaccine-derived protection during the Monkeypox outbreak to the longer interval in time between vaccination and the outbreak [26]. Corroborating with the data presented in this work, previous studies with human Monkeypox infection indicated that smallpox vaccination was not able to prevent the infection, but it seems that Monkeypox infection in humans that received a smallpox vaccine manifested in a milder form [27].
In this present study, we used lymph node-homing receptors, L-selectin (CD62L) and CCR7, and activation markers (CD45ROHigh) to describe the lymph-node homing capability and the effectors functions of CD4+ and CD8+ T-cells. Several markers have been identified to allow for the differentiation of naive and memory T cells. Human naive cells are CD45RO−, CCR7+ and CD62LHigh while memory cells (TEM) are CD45RO+, CCR7− and CD62L−. However, some memory cells appear to regain CD62L and CCR7, especially TCM cells [17–22,28,29]. Here the CD4+ T cell memory subsets were evaluated and the data indicated that infected individuals (vaccinated or not) have higher percentages of T memory cells (CD4+CCR7−CD62L− and CD4+CD45ROHigh) when compared to non-infected individuals (both vaccinated and unvaccinated). The percentage of CD4+ T lymph-node homing T cells (CD4+CCR7+CD62L+) did not differ between the four groups analyzed. However, when we analyzed the CD8+ T cells, our results showed that previously infected individuals (vaccinated or not) have higher percentage of CD8+ naive and/or TCM cells (CD8+CCR7+ and CD8+CCR7+CD62L+) when compared with non-infected individuals (vaccinated and unvaccinated). It is also important to note is that these cells co-express IFN-γ. Similarly, a recent study identified antigen-specific CD8+ T cells with a central memory phenotype. These cells were CD44High and CD62L+ virus-specific CD8+ T cells that constituted approximately 10% of all antigen-specific T cells and expressed high levels of CCR7 [30]. Our data suggest that effector memory is predominantly composed by CD4+ T cells and these cells are at higher levels only in previously infected individuals, since this increase was not observed in non-infected vaccinated and unvaccinated patients.
In order to further study the population of CD4+ and CD8+ effectors memory T cells, in vitro stimulation was performed with UV-inactivated VACV and intracellular cytokine staining to identify virus-specific CD4+ and CD8+ T cells that expressed either IFN-γ or TNF-α. We observed that the percentage of CD4+ T cells expressing IFN-γ- was higher in individuals previously infected by Vaccinia virus (vaccinated and unvaccinated) and in non-infected vaccinated when compared with non-infected unvaccinated subjects. Puissant-Lubrano and colleagues also found that the frequency of VACV-specific IFN-γ−producing cells and of VACV-specific proliferating cells in previously vaccinated volunteers was significantly higher than that of unvaccinated healthy volunteers [12]. In the present study, it was noted that the CD4+ IFN-γ-producing T cells were composed of CD4+CD45RO+ (CD4+CD45ROLow and CD4+CD45ROHigh), CD4+CD62Low and CD4+CD62L− T cells. It was also detected that just the infected individuals (vaccinated and unvaccinated) presented higher percentages of CD4+CD62L− and CD4+CD45ROHigh T cells co-expressing IFN-γ-which are the effector-memory T cells (TEM). In the non-infected individuals (vaccinated and unvaccinated), this increase in the mean percentage of CD4 memory lymphocytes expressing IFN-γ was not observed. Our data reinforce the idea that cellular memory to VACV predominantly involves CD4+ Th1 IFN-γ producing cells, as previously described [9,31,32]. In our studies, we also observed that recent infection by VACV induced a more significant CD4 effector memory response when we compared previously infected individuals (vaccinated and unvaccinated) to non-infected vaccinated patients.
Similarly, an increase on the percentage of the CD8+ memory T cells (CD8+CD45ROHigh) was detected only in individuals previously infected by Vaccinia virus (vaccinated and unvaccinated), but not in the non-infected (vaccinated or not) subjects. Similar results were observed in the CD4 compartment where an increase in the percentage of the CD4+CD45RO+ memory T cells expressing IFN-γ+ was identified only in the previously infected individuals (vaccinated and unvaccinated), after in vitro stimulation with UV-inactivated VACV, when compared with non-infected (vaccinated and unvaccinated) patients. Analysis of T CD8+CD62L+ memory cells expressing IFN-γ showed an increase in the percentage of these cells in individuals previously exposed to Vaccinia virus (infected vaccinated or not and non-infected vaccinated), but not in the non-infected unvaccinated group, after viral stimulation. In the present study, it was observed that the CD8+ IFN-γ-producing T cells were composed of CD8+CD45RO+, CD8+CD45ROLow, and CD8+CD62L+T cells. These results suggest that previous smallpox vaccination during childhood does not lead to a complete protection against VACV infection, but there is still a residual immunity. We also noted that previous infection leads to a more significant CD8 memory response when we compared previously infected individuals (vaccinated and unvaccinated) to non-infected vaccinated patients.
Moreover, it was demonstrated that CD4+ T cells from individuals who were once exposed to Vaccinia virus (either by smallpox vaccination or Vaccinia virus infection) were able to produce TNF-α. Our data showed that the Vaccinia-specific TNF-α producing T cells were composed mainly of CD4+CD45ROLow, CD4+CD62L− and CD4+CD62LLow. Other studies have already demonstrated that smallpox vaccination induces TNF-α expression by CD4+ memory T cells [5,12,13]. Similar results were observed when CD8+ T memory cells (CD8+CD62L+ and CD8+CD62LHigh) co-expressing TNF-α were analyzed. The results showed an increase in the percentage of these cells in individuals previously exposed to VACV (either by infection or vaccination) but not in the non-infected unvaccinated group. Our data also demonstrated an increase in the percentage of the CD8+CD45RO+TNF-α+ memory T cells only in the previously infected individuals (vaccinated and unvaccinated), when compared with non-infected (vaccinated and unvaccinated) individuals. It is important to note that in this study both CD4+ and CD8+ T memory cells were expressing TNF-α. One study, using cell cultures and ICCS, demonstrated that in response to VACV infection, the large majority of the cytokine secreting cells produce only TNF-α (52–95% depending on the clone) [23]. Our results corroborates with this study, since we detected that CD4 and CD8 T memory cells are expressing TNF-α, an important molecule involved in the resistance against VACV infections.
Finally, our findings suggest that vaccinated individuals, even long after vaccination for smallpox, maintain a residual beneficial immune response to the virus. It induces partial long-term protection that in our study was clearly reflected on the fact that milder symptoms were developed and on the presence of memory cells. Our study, however, has some limitations such as the size of the analyzed group, which comprises 42 individuals, being 22 Vaccinia virus infected individuals and 20 non-infected individuals. As we previously described in another study the notification of human Poxvirus infections is not mandatory in Brazil, and identification of patients is done through active random search [24]. This fact impaired our ability to enroll a larger number of patients and we cannot elucidate at this time the mechanisms involved in the immune memory response against Vaccinia virus infection.
5. Conclusions
In this study we observed that previous smallpox vaccination does not lead to a complete protection against VACV infection, since previously vaccinated individuals acquired the infection; however it lead to partial protection that is reflected on a milder clinical form. This fact is probably related to the fact that only previously infected individuals had higher percentages of CD4 and CD8 T memory cells when compared to non-infected subjects, vaccinated or not. Furthermore, it was found that CD4+ and CD8+ T memory cells expressing IFN-γ+ are at higher percentages only in previously infected individuals that may lead to faster elimination of the virus.
Financial support
CAPES/CNPQ/FAPEMIG.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial-No Derivative Works License, which permits non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
Supplementary Material
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.rinim.2013.10.002.
Appendix. Supplementary Materials
FACS analysis. (A) First we performed the identification of peripheral lymphocytes population in diagram of FSC × SSC. (B) Second we made dot plots of FL-1 × FL-4, displaying the frequency of CD4+CD45RO+ cells. The CD4+CD45ROLow (R1) and CD4+CD45ROHigh (R2) populations were sorted using the indicated sorting gates. (C) The same strategy was used to select the CD4+CD62L+ subpopulations. Dot plots of FL-1 × FL-4 were made displaying the frequency of CD4+CD62L+ cells. The CD4+CD62Lneg (R1), CD4+CD62LLow (R2) and CD4+CD62LHigh (R3) populations were sorted using the indicated sorting gates. (D) Finally, we made dot plots of FL-4 × FL-2, displaying the frequency of CD4+CD45ROLowIFN-γ+, CD4+CD45ROHighIFN-γ+, CD4+CD62LnegIFN-γ+, CD4+CD62LLowIFN-γ+ and CD4+CD62LHighIFN-γ+.
REFERENCES
- 1.Trindade GS, Lobato ZI, Drumond BP, Leite JA, Trigueiro RC, Guedes MI. Short report: isolation of two Vaccinia virus strains from a single bovine vaccinia outbreak in rural area from Brazil: implications on the emergence of zoonotic orthopoxviruses. Am J Trop Med Hyg. 2006;75(3):486–490. [PubMed] [Google Scholar]
- 2.Amanna IJ, Carlson NE, Slifka MK. Duration of humoral immunity to common viral and vaccine antigens. N Engl J Med. 2007;357:1903–1915. doi: 10.1056/NEJMoa066092. [DOI] [PubMed] [Google Scholar]
- 3.Amara R R, Nigam P, Sharma S, Liu J, Bostik V. Long-lived poxvirus immunity, robust CD4 help, and better persistence of CD4 than CD8 T cells. J Virol. 2004;78:3811–3816. doi: 10.1128/JVI.78.8.3811-3816.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crotty S, Ahmed R. Immunological memory in humans. Semin Immunol. 2004;16:197–203. doi: 10.1016/j.smim.2004.02.008. [DOI] [PubMed] [Google Scholar]
- 5.Hammarlund E, Lewis MW, Hansen SG, Strelow LI, Nelson JA, Sexton GJ, Hanifin JM, Slifka MK. Duration of antiviral immunity after smallpox vaccination. Nat Med. 2003;9:1131–1137. doi: 10.1038/nm917. [DOI] [PubMed] [Google Scholar]
- 6.Putz MM, Alberini I, Midgley CM, Manini I, Montomoli E, Smith GL. Prevalence of antibodies to Vaccinia virus after smallpox vaccination in Italy. J Gen Virol. 2005;86:2955–2960. doi: 10.1099/vir.0.81265-0. [DOI] [PubMed] [Google Scholar]
- 7.Taub DD, Ershler WB, Janowski M, Artz A, Key ML, McKelvey J, Muller D, Moss B, Ferrucci L, Duffey PL, Longo DL. Immunity from smallpox vaccine persists for decades: a longitudinal study. Am J Med. 2008;121:1058–1064. doi: 10.1016/j.amjmed.2008.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Demkowicz WE, Jr, Littaua RA, Wang J, Ennis FA. Human cytotoxic T-cell memory: long-lived responses to vaccinia virus. J Virol. 1996;70(4):2627–2631. doi: 10.1128/jvi.70.4.2627-2631.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sivapalasingam S, Kennedy JS, Borkowsky W, Valentine F, Zhan MX, Pazoles P, Paolino A, Ennis FA, Steigbigel NH. Immunological memory after exposure to Variola virus, Monkeypox virus, and Vaccinia virus. J Infect Dis. 2007;195:1151–1159. doi: 10.1086/512161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Amanna IJ, Slifka MK, Crotty S. Immunity and immunological memory following smallpox vaccination. Immunol Rev. 2006;211:320–337. doi: 10.1111/j.0105-2896.2006.00392.x. [DOI] [PubMed] [Google Scholar]
- 11.Combadiere B, Boissonnas A, Carcelain G, Lefranc E, Samri A, Bricaire F, Debre P, Autran B. Distinct time effects of vaccination on long-term proliferative and IFN-γ–producing T cell memory to smallpox in humans. J Exp Med. 2004;199(11):1585–1593. doi: 10.1084/jem.20032083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Puissant-Lubrano B, Bossi P, Gay F, Crance JM, Bonduelle O, Garin D, Bricaire F, Autran B, Combadière B. Control of Vaccinia virus skin lesions by long-term-maintained IFN-gamma+TNF-alpha+ effector/memory CD4+ lymphocytes in humans. J Clin Invest. 2010;120(5):1636–1644. doi: 10.1172/JCI38506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hammarlund E, Lewis MW, Hanifin JM, Mori Mi, Koudelka CW, Slifka MK. Antiviral immunity following Smallpox Virus infection: a case-control study. J Virol. 2010;84(24):12754–12760. doi: 10.1128/JVI.01763-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999;401:708–712. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
- 15.Masopust D, Vezys V, Marzo AL, Lefrancois L. Preferential localization of effector memory cells in nonlymphoid tissue. Science. 2001;291:2413–2417. doi: 10.1126/science.1058867. [DOI] [PubMed] [Google Scholar]
- 16.Reinhardt RL, Khoruts R, Merica A, Zell T, Jenkins MK. Visualizing the generation of memory CD4 T cells in the whole body. Nature. 2001;410:101–105. doi: 10.1038/35065111. [DOI] [PubMed] [Google Scholar]
- 17.Lanzavecchia A, Sallusto F. Progressive differentiation and selection of the fittest in the immune response. Nat Rev Immunol. 2002;2(12):982–987. doi: 10.1038/nri959. [DOI] [PubMed] [Google Scholar]
- 18.Lanzavecchia A, Sallusto F. Understanding the generation and function of memory T cell subsets. Curr Opin Immunol. 2005;17(3):326–332. doi: 10.1016/j.coi.2005.04.010. [DOI] [PubMed] [Google Scholar]
- 19.Geginat J, Sallusto F, Lanzavecchia A. Cytokine-driven proliferation and differentiation of human naïve, central memory and effector memory CD4+ T cells. Pathol Biol (Paris) 2003;51(2):64–66. doi: 10.1016/s0369-8114(03)00098-1. [DOI] [PubMed] [Google Scholar]
- 20.Campbell JJ, Bowman EP, Murphy K, Youngman KR, Siani MA, Thompson DA. 6-C-kine (SLC), a lymphocyte adhesion-triggering chemokine expressed by high endothelium, is an agonist for the MIP-3beta receptor CCR7. J Cell Biol. 1998;141:1053–1059. doi: 10.1083/jcb.141.4.1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Forster R, Schubel A, Breitfeld D, Kremmer E, Renner-Muller I, Wolf E. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell. 1999;99:23–33. doi: 10.1016/s0092-8674(00)80059-8. [DOI] [PubMed] [Google Scholar]
- 22.Sallusto F, Geginat J, Lanzavecchia A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol. 2004;22:745–763. doi: 10.1146/annurev.immunol.22.012703.104702. [DOI] [PubMed] [Google Scholar]
- 23.Judkowski V, Bunying A, Ge F, Appel JR, Law K, Sharma A, Raja-Gabaglia C, Norori P, Santos RG, Giulianotti MA, Slifka MK, Douek DC, Graham BS, Pinilla C. GM-CSF production allows the identification of immunoprevalent antigens recognized by human CD4+ T cells following smallpox vaccination. PLoS One. 2011;6(9):e24091. doi: 10.1371/journal.pone.0024091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Silva Gomes JA, de Araújo FF, de Souza Trindade G, Quinan BR, Drumond BP, Ferreira JM, Mota BE, Nogueira ML, Kroon EG, Santos Abrahão J, Côrrea-Oliveira R, da Fonseca FG. Immune modulation in primary Vaccinia virus zoonotic human infections. Clin Dev Immunol. 2012;2012:974067. doi: 10.1155/2012/974067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Campos MAS, Kroon EG. Critical period for irreversible block of Vaccinia virus replication. Rev Microbiol. 1993;24:104–110. [Google Scholar]
- 26.Karem KL, Reynolds M, Hughes C, Braden Z, Nigam P, Crotty S, Glidewell J, Ahmed R, Amara R, Damon IK. Monkeypox-induced immunity and failure of childhood smallpox vaccination to provide complete protection. Clin Vaccine Immunol. 2007;14:1318–1327. doi: 10.1128/CVI.00148-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Heymann DL, Szczeniowski M, Esteves K. Re-emergence of monkeypox in Africa: a review of the past six years. Br Med Bull. 1998;54(3):693–702. doi: 10.1093/oxfordjournals.bmb.a011720. [DOI] [PubMed] [Google Scholar]
- 28.Bunce C, Bell EB. CD45RC isoforms define two types of CD4 memory T cells, one of which depends on persisting antigen. J Exp Med. 1997;185:767–776. doi: 10.1084/jem.185.4.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bradley LM, Duncan DD, Tonkonogy SL, Swain SL. Characterization of antigen-specific CD4+ effector T cells in vivo: immunization results in a transient population of MEL-14-, CD45RB- helper cells that secretes Il-2, IL-3 and IL-4. J Exp Med. 1991;174:547–559. doi: 10.1084/jem.174.3.547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Laouar A, Manocha M, Haridas V, Manjunath N. Concurrent generation of effector and central memory CD8 T cells during vaccinia virus infection. PLoS One. 2008;3(12):e4089. doi: 10.1371/journal.pone.0004089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Combadiere B, Boissonnas A, Carcelain G, Lefranc E, Samri A, Bricaire F, Debre P, Autran B. Distinct Time Effects of Vaccination on Long-Term Proliferative and IFN-γ-producing T Cell Memory to Smallpox in Humans. J. Exp Med. 2004;199(11):1585–1593. doi: 10.1084/jem.20032083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Puissant-Lubrano B, Bossi P, Gay F, Crance JM, Bonduelle O, Garin D, Bricaire F, Autran B, Combadière B. Control of vaccinia virus skin lesions by long-term-maintained IFN-gamma+TNF-alpha+ effector/memory CD4+ lymphocytes in humans. J Clin Invest. 3 May 2010;120(5):1636–1644. doi: 10.1172/JCI38506. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
FACS analysis. (A) First we performed the identification of peripheral lymphocytes population in diagram of FSC × SSC. (B) Second we made dot plots of FL-1 × FL-4, displaying the frequency of CD4+CD45RO+ cells. The CD4+CD45ROLow (R1) and CD4+CD45ROHigh (R2) populations were sorted using the indicated sorting gates. (C) The same strategy was used to select the CD4+CD62L+ subpopulations. Dot plots of FL-1 × FL-4 were made displaying the frequency of CD4+CD62L+ cells. The CD4+CD62Lneg (R1), CD4+CD62LLow (R2) and CD4+CD62LHigh (R3) populations were sorted using the indicated sorting gates. (D) Finally, we made dot plots of FL-4 × FL-2, displaying the frequency of CD4+CD45ROLowIFN-γ+, CD4+CD45ROHighIFN-γ+, CD4+CD62LnegIFN-γ+, CD4+CD62LLowIFN-γ+ and CD4+CD62LHighIFN-γ+.


