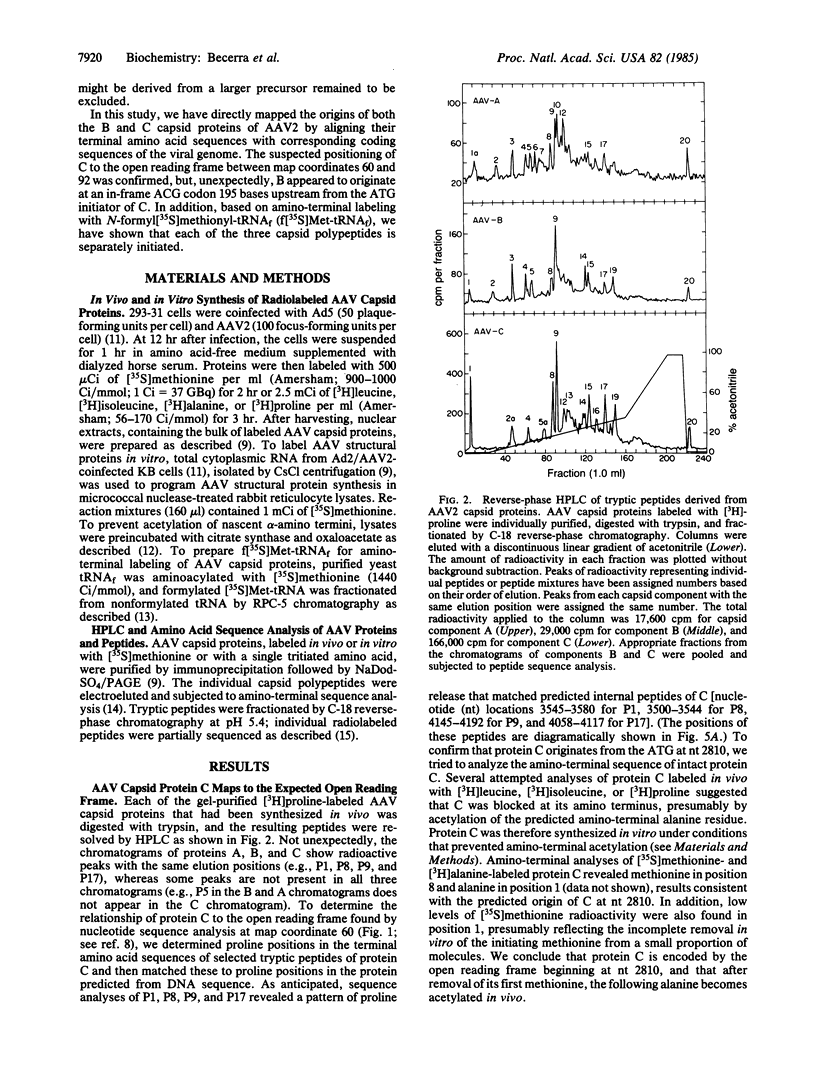
Abstract
The three major capsid proteins of adeno-associated virus type 2 (AAV2) virions are designated A, B, and C and have molecular sizes of 90, 72, and 60 kDa, respectively. These proteins are related, and genetic studies have shown they are encoded by a long open reading frame located in the right half of the genome. The coding capacity distal to the first ATG in this reading frame is only 503 amino acids (i.e., a protein about the size of protein C), but an open frame sequence devoid of ATG codons extends upstream for an additional 184 codons. Although the amino terminus of the C capsid protein is blocked, partial amino acid sequence analyses of peptides from C have confirmed that it is encoded within the portion of the reading frame distal to the first ATG at nucleotide (nt) location 2810. The amino terminus of the B capsid protein is not blocked, and its sequence begins with alanine. The triplet encoding this alanine lies 64 codons upstream from the initiation site for protein C and is immediately preceded by the threonine codon, ACG, at nt 2615. This ACG codon lies in the most favorable sequence context for protein synthesis initiation. All three AAV2 capsid proteins are labeled in vitro with formyl[35S]methionyl-tRNAf, indicating that synthesis of each protein is initiated independently. Our data suggest that the nt 2615 ACG codon directs the methionyl-tRNA-dependent initiation of the AAV2 B capsid protein. Proteins B and C may be synthesized from the same mRNA species and their relative abundance could be determined by the efficiencies of their respective initiation codons.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Buller R. M., Janik J. E., Sebring E. D., Rose J. A. Herpes simplex virus types 1 and 2 completely help adenovirus-associated virus replication. J Virol. 1981 Oct;40(1):241–247. doi: 10.1128/jvi.40.1.241-247.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buller R. M., Rose J. A. Characterization of adenovirus-associated virus-induced polypeptides in KB cells. J Virol. 1978 Jan;25(1):331–338. doi: 10.1128/jvi.25.1.331-338.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh H. P., Söll D., Khorana H. G. Studies on polynucleotides. LXVII. Initiation of protein synthesis in vitro as studied by using ribopolynucleotides with repeating nucleotide sequences as messengers. J Mol Biol. 1967 Apr 28;25(2):275–298. doi: 10.1016/0022-2836(67)90142-8. [DOI] [PubMed] [Google Scholar]
- Green M. R., Roeder R. G. Definition of a novel promoter for the major adenovirus-associated virus mRNA. Cell. 1980 Nov;22(1 Pt 1):231–242. doi: 10.1016/0092-8674(80)90171-3. [DOI] [PubMed] [Google Scholar]
- Green M. R., Roeder R. G. Transcripts of the adeno-associated virus genome: mapping of the major RNAs. J Virol. 1980 Oct;36(1):79–92. doi: 10.1128/jvi.36.1.79-92.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Housman D., Jacobs-Lorena M., Rajbhandary U. L., Lodish H. F. Initiation of haemoglobin synthesis by methionyl-tRNA. Nature. 1970 Aug 29;227(5261):913–918. doi: 10.1038/227913a0. [DOI] [PubMed] [Google Scholar]
- Janik J. E., Huston M. M., Rose J. A. Adeno-associated virus proteins: origin of the capsid components. J Virol. 1984 Nov;52(2):591–597. doi: 10.1128/jvi.52.2.591-597.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janik J. E., Huston M. M., Rose J. A. Locations of adenovirus genes required for the replication of adenovirus-associated virus. Proc Natl Acad Sci U S A. 1981 Mar;78(3):1925–1929. doi: 10.1073/pnas.78.3.1925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelmers A. D., Heatherly D. E. Columns for rapid chromatographic separation of small amounts of tracer-labeled transfer ribonucleic acids. Anal Biochem. 1971 Dec;44(2):486–495. doi: 10.1016/0003-2697(71)90236-3. [DOI] [PubMed] [Google Scholar]
- Kozak M. Comparison of initiation of protein synthesis in procaryotes, eucaryotes, and organelles. Microbiol Rev. 1983 Mar;47(1):1–45. doi: 10.1128/mr.47.1.1-45.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laughlin C. A., Westphal H., Carter B. J. Spliced adenovirus-associated virus RNA. Proc Natl Acad Sci U S A. 1979 Nov;76(11):5567–5571. doi: 10.1073/pnas.76.11.5567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodish H. G., Housman D., Jacobsen M. Initiation of hemoglobin synthesis. Specific inhibition by antibiotics and bacteriophage ribonucleic acid. Biochemistry. 1971 Jun 8;10(12):2348–2356. doi: 10.1021/bi00788a027. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- McPherson R. A., Rose J. A. Structural proteins of adenovirus-associated virus: subspecies and their relatedness. J Virol. 1983 May;46(2):523–529. doi: 10.1128/jvi.46.2.523-529.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mount S. M. A catalogue of splice junction sequences. Nucleic Acids Res. 1982 Jan 22;10(2):459–472. doi: 10.1093/nar/10.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oosterom-Dragon E. A., Anderson C. W. Polypeptide structure and encoding location of the adenovirus serotype 2 late, nonstructural 33K protein. J Virol. 1983 Jan;45(1):251–263. doi: 10.1128/jvi.45.1.251-263.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmiter R. D. Prevention of NH2-terminal acetylation of proteins synthesized in cell-free systems. J Biol Chem. 1977 Dec 25;252(24):8781–8783. [PubMed] [Google Scholar]
- Pirastu M., Saglio G., Chang J. C., Cao A., Kan Y. W. Initiation codon mutation as a cause of alpha thalassemia. J Biol Chem. 1984 Oct 25;259(20):12315–12317. [PubMed] [Google Scholar]
- Rose J. A., Berns K. I., Hoggan M. D., Koczot F. J. Evidence for a single-stranded adenovirus-associated virus genome: formation of a DNA density hybrid on release of viral DNA. Proc Natl Acad Sci U S A. 1969 Nov;64(3):863–869. doi: 10.1073/pnas.64.3.863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose J. A., Maizel J. V., Jr, Inman J. K., Shatkin A. J. Structural proteins of adenovirus-associated viruses. J Virol. 1971 Nov;8(5):766–770. doi: 10.1128/jvi.8.5.766-770.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srivastava A., Lusby E. W., Berns K. I. Nucleotide sequence and organization of the adeno-associated virus 2 genome. J Virol. 1983 Feb;45(2):555–564. doi: 10.1128/jvi.45.2.555-564.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thach R. E., Sundararajan T. A., Dewey K. F., Brown J. C., Doty P. Translation of synthetic messenger RNA. Cold Spring Harb Symp Quant Biol. 1966;31:85–97. doi: 10.1101/sqb.1966.031.01.015. [DOI] [PubMed] [Google Scholar]
- Vogelstein B., Gillespie D. Preparative and analytical purification of DNA from agarose. Proc Natl Acad Sci U S A. 1979 Feb;76(2):615–619. doi: 10.1073/pnas.76.2.615. [DOI] [PMC free article] [PubMed] [Google Scholar]




