Abstract
Immunoglobulins with germline sequences occur in invertebrates and vertebrates and are named naturally occurring autoantibodies (NAbs). NAbs may target foreign antigens, self- or altered self-components and are part of the normal immunoglobulin repertoire. Accumulating evidence indicates that naturally occurring antibodies can act as systemic surveillance molecules, which tag, damaged or stressed cells, invading pathogens and toxic cellular debris for elimination by the immune system. In addition to acting as detecting molecules, certain types of NAbs actively signal in different cell types with a broad range of responses from induction of apoptosis in cancer cells to stimulation of remyelination in glial cells. This review emphasizes functions and characteristics of NAbs with focus on remyelination-promoting mouse and human antibodies. Human remyelination-promoting NAbs are potential therapeutics to combat a wide spectrum of disease processes including demyelinating diseases like multiple sclerosis. We will highlight the identified glycosphingolipid (SL) antigens of polyreactive remyelination-promoting antibodies and their proposed mechanism(s) of action. The nature of the identified antigens suggests a lipid raft-based mechanism for remyelination-promoting antibodies with SLs as most essential raft components. However, accumulating evidence also suggests involvement of other antigens in stimulation of remyelination, which will be discussed in the text.
Keywords: autoantibodies, apoptosis, demyelination, multiple sclerosis, proliferation, TMEV animal model
Natural antibodies
Soon after natural antibodies were documented by Avrameas [1–3] and Notkins [4–7], it became evident that naturally occurring autoantibodies (NAbs) are part of the human innate immunoglobulin repertoire [2,8].
NAbs utilize germline-encoded genes directed against foreign antigens, self- and altered self-structures [1] and are present in newborns without stimulation by foreign antigens [9]. NAbs are polyreactive by definition with few or no somatic mutations in the VH and VL antibody regions, which are necessary for high affinity binding of a single antigen. NAbs of the IgM isotype are found in invertebrates and vertebrates. High levels of IgG and lower amounts of IgM and IgA NAbs are detected in higher vertebrates [10]. In general, NAbs bind their antigen with rather low affinity but high avidity [11], which describes the combined synergistic strength of multiple bond interactions rather than the sum of bonds between antigen and antibody. In contrast, conventional antibodies, typically of the IgG isotype, undergo affinity maturation and contain somatic mutations to ensure high-affinity antigen binding.
Accumulating evidence categorize NAbs as natural systemic surveillance molecules that tag damaged cells and foreign pathogens for elimination by the immune system through opsonization or antibody-dependent cytotoxicity. In addition to their molecular-tag function, some NAbs can actively signal in different cell types including cancer and brain cells. The ability of certain NAbs to detect and sometimes induce apoptosis in tumor cells may play an important function in tumor surveillance [12–15]. In mice, another class of NAbs, termed remyelination-promoting antibodies, actively promotes repair in demyelinated spinal cord lesions [16–18].
Properties of B cells producing NAbs
Natural antibodies are produced by different mature B-cell subsets. These include B1 cells that constitutively produce natural antibodies, most often of the IgM isotype followed by IgG and IgA, marginal zone-B lymphocytes and splenic follicular B2 cells [19,20]. B1 cells constitutively secrete antibodies without prior immune activation [21,22]. These immunoglobulins are termed naturally occurring antibodies because they may be present during fetal life without antigenic exposure. In contrast to mature follicular B cells (B2 cells), which are typically responsible for T-cell mediated immune responses, mature B1 cells and marginal-zone B cells mediate innate immune responses with rapid responses to non-protein antigens. Natural antibodies from B1 cells are often of germline origin and unaffected by somatic hypermutation [23].
Polyreactive NAbs
Immunologists had some difficulties in accepting the existence of polyreactive monoclonal antibodies. The problem originates from the immunological paradigm that antibodies engage antigens with high-affinity in a monospecific manner [24]. Ever since, antibodies are associated with high affinity and (mono) specificity. This point of view is supported by the fact that polyreactive antibodies with germline origin are mostly ineffective as detecting agents in cell biological or biochemical settings where promiscuous binding of antibodies is commonly equalized with ‘non-specific binding’.
The establishment of natural antibody occurrence in physiological conditions by Avrameas [1–3] and Notkins [4–7] led to greater acceptance of the polyreactivity of antibodies in general. Antibodies are designated polyreactive when they bind sufficiently to more than one unrelated self or foreign antigen. Sufficient proof of monospecificity, on the other hand, is commonly accepted in arrays with >104 antigens. In the past, antibody monospecificity had not been tested to this degree. It remains to be seen to what extent previously classified monospecific antibodies would pass today’s criteria in antigen arrays of higher than 104 antigens.
Polyreactivity of a single immunoglobulin was formally proven in monoclonal antibodies produced by i) hybridoma technology, ii) Epstein-Barr transfected B lymphocytes and iii) from patients with B-cell malignancies [1,25,26] with polyreactive antibodies of the IgM, IgG and IgA isotype [27]. Polyreactivity of antibodies has been highly conserved in evolution and can even be found in sharks [28]. The affinity of polyreactive NAbs to their different antigens can vary by a factor 1000 and is, in general, lower (Kd = 10−3–10−7 mol/l) than affinity-maturated monospecific antibodies (Kd = 10−7–10−11 mol/l) (Table 1) [29].
Table 1.
Properties of polyreactive versus monospecific monoclonal antibodies.
| Property | Polyreactive monoclonal antibody | Monospecific monoclonal antibody |
|---|---|---|
| Antigen | Many structurally divers and unrelated antigens | Single antigen |
| Affinity | Low (Kd:10−4–10−7) | High (Kd:10−7–10−11) |
| Sequence | Germline or near germline with few somatic mutations, no affinity maturation | Somatically mutated, affinity maturated |
| Number of potentially allowed conformations of antigen-binding pocket | More than one (conformational selection) or (induced fit hypothesis) | Only one (lock and key fit mechanism) |
| Immunoglobulin subtype | Mainly IgM, also IgA and IgG | IgG, IgM, IgA |
| Half-life time | IgM: ~8 h; IgG: ~10 h; IgA: ~8 h | IgM:~35 h; IgG: ~280 h; IgA: ~26 h |
Kd: Dissociation constants.
The half-life of a polyreactive natural antibody in vivo is significantly shorter than a monospecific one (Table 1). The rapid serum clearance of polyreactive monoclonal antibodies is likely due to binding to multiple endogenous antigens.
Because linear amino acid sequence analysis could not explain differences in antigen specificity of polyreactive and monospecific antibodies, it became evident that 3D structure of the immunoglobulin is responsible for differences in specificity and affinity between both subtypes. The structural basis of polyreactivity relates to the properties of the variable domains, particularly of the heavy chain [30,31]. Accordingly, transferring the CDR3 region of the heavy Ig chain from a polyreactive to a monospecific antibody induces polyreactivity [32]. Supporting evidence comes from studies showing that single amino acid replacements in the CDRH3 region of a polyreactive antibody are sufficient to create a monospecific antibody [33,34]. Interestingly, so far no differences in the conformation, amino acid chain length or sequence can be detected in the CDRH3 regions of monospecific vs polyreactive antibodies [29]. In addition, a single amino acid replacement outside the antigen-binding pocket is sufficient to abolish polyreactivity [26]. This indicates that not only the paratope but the whole variable domain is essential for polyreactivity.
Of note, there is a higher degree in glycosylation of polyreactive monoclonal antibodies relative to monospecific monoclonal antibodies [35]. Bulky carbohydrate moieties attached to the variable regions of immunoglobulins contribute to the protein conformation [36]. Glycosylation of the immunoglobulin’s variable domains can interfere with its ability to target its antigen [37,38]; this may be an alternative explanation for a higher degree of variability in the antigen-antibody interaction as seen with polyreactive antibodies compared with monoreactive antibodies.
Remyelination-promoting antibodies
The focus in the following section is on NAbs that stimulate remyelination in different animal models of multiple sclerosis (MS).
Remyelination-promoting antibodies are a subclass of NAbs
All identified remyelination-promoting antibodies were of germline origin or near germline with few somatic mutations, thus having the cardinal features of physiologic natural autoantibodies. So far, all identified remyelination-promoting antibodies with NAb features are of the IgM isotype (with the exception of high-affinity anti-Lingo IgG antibodies, which stimulate remyelination in rodents but do not have NAb features). In addition, all remyelination-promoting antibodies with known antigens are polyreactive, which is the result of their rather flexible antigen-binding site typical for NAbs. Of note, all remyelination-promoting antibodies with identified antigens bind to at least one or multiple sphingolipids, which are glycosylated lipids with ceramide backbone and essential lipid-raft components. Only the hydrophilic carbohydrate moiety of the sphingolipids is exposed to the cell surface and, therefore, detectable by antibodies. This emphasizes the carbohydrate moiety and neglects the lipid backbone as the essential part of the antigen.
In summary, remyelination-promoting antibodies show all cardinal features of NAbs and represent a subclass of NAbs.
Discovery of remyelination-promoting antibodies
The first successful attempt to stimulate remyelination using NAbs was performed in the Theiler’s murine encephalomyelitis virus (TMEV)-induced model of demyelination [39]. We immunized TMEV-infected SJL mice with spinal cord homogenates (SCH) of normal mice to stimulate a polyclonal antibody response directed against a variety of CNS antigens including myelin components. Instead of an expected exacerbation of the disease course, mice immunized with SCH showed four-times’ higher levels of remyelination than non-immunized mice. Whole antisera [40] or purified immunoglobulins [41] raised against CNS antigens increased remyelination to a similar extent in the same animal model. These findings demonstrated for the first-time a beneficial effect of antibodies in stimulating CNS remyelination. In order to raise a more specific (monoclonal) antibody response toward a single antigen, we screened hybridomas made from B-cells of SCH-immunized mice for their ability to induce remyelination in chronically demyelinated mice. Two monoclonal mouse antibodies of the IgM isotype (SCH79.08 and SCH94.03) proved effective in promoting remyelination [42]. Both antibodies (SCH79.08 and SCH94.03) were able to target mature oligodendrocytes (OLs) in vitro. In all following attempts to identify mouse or human remyelination-promoting antibodies, we used the antibodies ability to bind to OL-lineage cells as first selection criteria. This was a necessary but not sufficient criterion to identify remyelination-promoting antibodies. Very few antibodies stimulated remyelination in vivo without showing a cell-surface reactivity toward OLs [43]. Subsequent studies using four well-known mouse monoclonal IgMs (O4, HNK-1, A2B5 and O1) [44–46] resulted in their classification as promoters of CNS remyelination in vivo [47]. Based on this information, our laboratory employed a novel strategy to identify monoclonal remyelination-promoting antibodies of human origin from sera of patients with monoclonal gammopathies. Cerebellar slice cultures were used in addition to cultured OLs for preliminary screening [17]. This resulted in the identification of two serum-derived human remyelination-promoting antibodies (sHIgM22 and sHIgM46). To make a recombinant antibody (rHIgM22), we obtained the IgMs (HIgM22) variable region-coding DNA sequence through protein sequencing and cloned it into an IgM expression vector [48,49]. We confirmed identical properties of the recombinant IgM relative to its serum-derived counterpart to stimulate remyelination in vivo and to bind to OLs in vitro. rHIgM22 has recently been approved by the FDA for Phase I, multi-center, double-blind, randomized, placebo-controlled, dose-escalation study designed to evaluate safety, tolerability, pharmacokinetics, and immunogenicity of single IV administrations of rHIgM22 in patients with all clinical presentations of MS.
Identified mouse and human remyelination-promoting IgMs stimulate remyelination after TMEV- and lysolecithin-induced demyelination in mice [16–18,42,47,50–52]. A single bolus injection of monoclonal human IgM, rHIgM22, is sufficient to promote repair of demyelinated lesions [17,18]. Antibodystimulated remyelination was shown by electron microscopy and immunohistochemically [16,17,42,47].
Characterization of remyelination-promoting antibodies
The dissociation constants (Kd) of the remyelination-promoting IgMs O4 and O1 to sulfatide (O4) and galactosylceramide (O1) were recently determined (2.4 × 10−9 mol/l (O1) and 2.2 × 10−9 mol/l (O4)) and are unusually high for polyreactive natural antibodies (Table 1) [53]. A study by Paz Soldan et al. demonstrated that all tested remyelination-promoting IgMs induce a Ca2+-influx in astrocytes (GFAP+), oligodendrocyte progenitor cells (OPCs) and immature OLs [43]. IgM-mediated effects in astrocytes and OLs were, however, independent from each other, based on different signaling mechanisms and based on different Ca2+ pools (ER-stored Ca2+ for astrocytes and extracellular Ca2+ for OLs) [43]. In summary, all well-characterized remyelination-promoting antibodies are of germline origin, belong to the IgM isotype and induce calcium influx into astrocytes, OPCs and immature OLs.
Common antigens & mechanistic theories for remyelination-promoting antibodies
All remyelination-promoting antibodies with known antigens are polyreactive. The ganglioside-binding antibody A2B5 targets several sialylated glycosphingolipids (SLs) due to their similar carbohydrate epitope [44,54,55]. HNK-1 recognizes the SL 3-sulfoglucuronyl paragloboside (SGPG) [45,56,57] as well as the carbohydrate epitope of the glycoproteins MAG and P0 [57]. The mouse IgM O1 binds to galactocerebroside and similar SLs [58,59], whereas O4 targets sulfated galactocerebroside (sulfatide), seminolipid, the unknown proligodendroblast antigen (POA) and cholesterol [58–61]. SLs and cholesterol are essential components of lipid rafts, which act as signaling platforms at the level of the plasma membrane in cells. Lipid rafts may enable or disable interactions between many different cell-surface receptors (proteins) to transduce extracellular signals over the plasma membrane into the cytoplasmic space (general lipid-raft concept). Pentameric IgM molecules can bind and cluster up to 10 antigens at a time on different cells and <10 antigens on a single cell due to steric hindrance/tension within the IgM molecule. We hypothesize that IgMs targeting SLs stabilize existing rafts or stimulate the formation of new lipid rafts at the plasma membrane, thereby enhancing the effects of extracellular stimuli via existing cellular signaling pathways (lipid-raft hypothesis).
Alternatively, remyelination-promoting IgMs may be involved in the opsonization of cellular debris and dead or apoptotic cells in a lesion site. Remyelination-promoting antibodies 94.03 and 79.08 [47], O1 and the human sulfatide-binding IgM DS1F8 [62] prominently stain filaments in astrocytes or HeLa cells, which are identified as microtubule-like structures [62]. Binding to intracellular filamentous structures seems to be common with antibodies targeting galactosylceramide and sulfatide [46,63,64]. This may indicate epitope similarities between OL-specific SLs present at the cell surface and in internal pools relative to intracellular cytoskeletal proteins detected by polyreactive antibodies. The ability of certain remyelination-promoting antibodies to target both membrane lipids and attached cytoskeletal proteins may significantly facilitate lesion clearance by the immune system and help to repopulate demyelinated areas.
A more recent study shifted our focus from SLs to glycoproteins as an alternative class of antigens for remyelination-promoting antibodies. As mentioned earlier, the carbohydrate, but not the lipid moiety of sphingolipids, is accessible to antibodies and may be sufficient for antibody binding when linked to either a lipid or protein backbone. The relatively simple carbohydrate structure on sphingolipids compared to the complex glycosylation found on many glycoproteins suggests that carbohydrate building blocks responsible for antibody binding can be found on glycoproteins as well. Inoko et al. (2010) demonstrated binding of the remyelination-promoting IgM A2B5 to a novel set of brain-derived glycoproteins as shown by Western blots [65]. It is well accepted that A2B5 binds to c-series gangliosides GT3 and GQ1c. However, c-series gangliosides and their O-acetyl derivatives are temporarily expressed in embryonic stages in the CNS but seldom during adulthood in vertebrates [66–70]. Cerebellar stellate neurons are the only known exception in the adult human CNS that express c-series gangliosides [71]. This raises the question whether remyelination-promotion in adult mice by A2B5 is mediated through binding to SLs or glycoproteins [47].
In summary, all remyelination-promoting IgM mAbs bind to SLs and potentially to glycoproteins at the cell surface of OPCs/OLs, and some bind to neurons. It remains elusive whether effects seen in vivo and in vitro are mediated through binding to cell-surface SLs, glycoproteins and/or internal cytoskeletal structures.
Responsive OL differentiation stages for IgM-stimulated lesion repair in brain & spinal cord
The remyelination-promoting mouse IgMs O4, O1 and A2B5 are commonly used markers for specific differentiation stages in OPCs and/or OLs [44–46,55–57]. However, the targeted differentiation stages vary strongly between different remyelination-promoting IgMs from OPCs (A2B5), late OPCs and immature OLs (O4), immature OLs (O1) to mature MBP-positive OLs (rHIgM22, 79.08). This would suggest that basically all OPC/OL-differentiation stages are responsive to IgM-stimulated repair in spinal-cord lesions. Overwhelming evidence demonstrates, however, that OPCs – but not surviving mature OLs – are responsible for remyelination in vivo [72–77]. Stimulation of remyelination by remyelination-promoting antibodies may be mediated by OPCs within the OL-lineage. This implies that: i) the SL antigens galactosylceramide (O1) and most likely sulfatide (O4) are not involved in antibody-stimulated remyelination due to the temporal mismatch between antigen expression and the responsive OPC stage involved in remyelination, ii) Identification of the late OL antigens for rHIgM22 (unknown) and 79.08 (MBP) may be unimportant for the same reason. With the latter possibility, the antigens are sufficiently expressed already at earlier stages during OL development but undetectable by immunocytochemistry. In support of these hypotheses, we demonstrated stimulation of OPC proliferation by rHIgM22 in a PDGF-dependent manner [78]. rHIgM22 had no detectable effects in mature (MBP+) OLs, which are strongly positive for rHIgM22 in immunocytochemistry. In addition, all remyelination-promoting antibodies stimulated calcium influx into OPCs but not into mature OLs [43]. The expression of SL sulfatide is associated with the immature OL stage but not with the late-OPC stage. However, expression of the unidentified pro-oligodendroblast antigen (POA) for O4 precedes the expression of sulfatide, which may explain the temporal mismatch between expression of the O4 antigen(s) and the fact that OPCs are mediators for remyelination. The POA antigen may be a sulfated glycoprotein.
In summary, there is a strong temporal mismatch between the expression of the antigens galactosylceramide (O1), sulfatide (O4), MBP (79.08), the unknown antigen for rHIgM22 and the OPC stage as the responsible developmental stage of OLs for remyelination. We conclude that other antigens on more immature cells other than the named myelin markers are responsible for remyelination in vivo and effects seen in vitro. The nature of those unidentified antigens remains unclear.
Low amounts of rHIgM22 are effective to stimulate remyelination in TMEV-infected animals
The effective dose of rHIgM22 to stimulate spinal-cord remyelination in TMEV-infected mice is as low as 500 ng per mouse when administered ip. in a single bolus injection. Remyelination was completed 5 weeks after the IgM injection. Surprisingly, higher antibody doses (up to 1000-fold) were not more effective in stimulating remyelination [18]. rHIgM22-mediated remyelination was detectable at 1 and 3 weeks after administration into chronically demyelinated mice; however, changes were not yet statistically significant [18]. In addition, multiple dosing of TMEV-infected mice with human antibody rHIgM22 was not more effective than a single dose of antibody. This may be the result of a possible immune response in mice toward human antibodies 5 weeks after the first exposure to rHIgM22.
The half-life of human rHIgM22 in infected and uninfected mice was ~15 h with no difference in serum clearance between both groups. This resulted in an 8.6-fold decrease of antibody detectable in serum at the 48 h timepoint [18].
In summary, rHIgM22 is a potent therapeutic in mice, which acts within a short window of time and causes longlasting tissue repair. It also suggests a molecular and cellular memory effect (i.e., with newly synthesized cells or stable changes of cellular behavior) long after endocytosis and destruction of the antibody.
The integrity of the IgM molecule is required to stimulate remyelination
So far, all attempts to manipulate the integrity of the pentameric rHIgM22 molecule chemically or enzymatically have degraded its capability to remyelinate. Cleavage of the IgM molecule results in unstable pseudo-IgGs that spontaneously reassemble into a tetrameric isoform. The tetrameric isoform resulted in identical levels of (spontaneous) remyelination as PBS-treated mice, which was significantly lower when compared with animals receiving intact pentameric rHIgM22 molecule (unpublished data). This may either suggest that antigen clustering on a single living cell, clustering of multiple cells or antigen clustering of cellular debris by the IgM molecule is essential for its cellular effect.
Remyelination-promoting IgMs have access to demyelinated lesions
Although debate continues concerning antibody access to the brain (i.e., limited brain access of high-molecular weight proteins; specific antibody-transporter systems via transferrin receptor-mediated transflux; age-related blood–brain barrier (BBB) changes [79], endogenous factors affecting BBB [80]), the concept has generated new potential therapeutics to treat demyelinating diseases including anti-Lingo IgGs and remyelination-promoting IgMs. Direct evidence of IgM access to the brain comes from a MRI study using antibodies labeled with ultra-small superparamagnetic iron oxide particles (USPIO). 3D T2-weighted imaging of spinal cords demonstrated co-localization of rHIgM22 with demyelinating lesions in mice spinal cords when injected into the tail vein of Theiler’s virus-infected mice [81]. No IgM accumulation in the CNS was apparent in non-infected animals or animals without demyelination [81]. In addition, dysfunction of the BBB is a typical feature of MS [82,83] and has been demonstrated in different presentations of the disease including chronic-progressive MS [84–86] and acute MS plaques [85]. BBB breakdown in MS allows high molecular-weight serum proteins, including fibrinogen (340 kDa), vitronectin and fibronectin (~440 kDa), to enter and accumulate within the lesion area [87–93]. Therefore, it is plausible to accept IgM access to demyelinated brain lesions as well in the human situation.
Remyelination-promoting antibodies in EAE
The well-characterized IgMs O4 and O1 promote remyelination in the TMEV model [47,94] comparable to the degree induced by rHIgM22 [16–18], and polyclonal human IgM [17]. In stark contrast to results obtained from the TMEV model, multiple dosing with the mouse IgM O4 causes a more severe disease phenotype in acute PLP-induced experimental autoimmune encephalomyelitis (EAE) [95]. Confirming the discrepancy between different animal models of MS, we demonstrate rHIgM22-stimulated promotion of remyelination in TMEV-and lysolecithin-demyelinated mice [16–18], but not in EAE (unpublished data). Unlike O4, rHIgM22 does not exacerbate the disease course in EAE (unpublished data). The precise reason( s) for the opposite outcome in different animal models of MS are unknown. However, EAE is an animal model of brain inflammation with relatively little demyelination compared to the TMEV model. Drugs effective in EAE commonly target the immune system and modulate the inflammatory response. Remyelination-promoting antibodies have little or no effect on the immune response but directly target cells within the demyelinated lesion to stimulate remyelination. It is, therefore, not surprising that remyelination-promoting antibodies do not affect remyelination in EAE in the absence of substantial demyelination. Because these autoantibodies do not exacerbate an autoimmune disease in animals, they are unlikely to exacerbate autoimmune disease in humans.
Proposed mechanism(s) of action of remyelination-promoting IgMs
This section will concentrate on general mechanistic aspects of mouse and human remyelination-promoting IgMs with further focus on rHIgM22 in clinical trials.
Mouse and human remyelination-promoting IgMs stimulate repair in TMEV- and lysolecithin-demyelinated mice [16–18,42,47,50–52]. To date, the precise mechanism of action is still unknown. Two main hypotheses have been proposed: i) the direct hypothesis proposes antibodies recognize OPCs and promote the synthesis of new myelin; ii) the indirect hypothesis proposes antibodies activate either immune cells or cell types other than cells of the OL lineage within the CNS, which, in turn, stimulate OPCs or OLs (i.e., by secreting remyelination-promoting factors).
Support exists for both hypotheses. Evidence supporting the direct hypothesis is derived from the observation that remyelination-promoting IgMs target OLs in culture, isolated myelin and myelin tracks in cerebellar slice cultures [16–18,42,43,47,50,94,96]. However, fluorescence microscopy, when used as an approach to detect rHIgM22 binding to glial cells qualitatively, detected binding only to mature OLs but not to OPCs, astrocytes or microglia. Biological effects were, however, detectable only in OPCs. Therefore, we conclude that fluorescence microscopy is not sufficiently conclusive to determine relevant cell types involved in rHIgM22-mediated effects.
All tested human and mouse remyelination-promoting IgMs induce calcium influx in cells of the OL-lineage, which suggests activation of intracellular signaling pathways potentially important for remyelination [43,94]. In addition, rHIgM22 inhibited OL differentiation and apoptotic signaling in enriched OPC cultures in vitro [97]. Inhibition of myelin-sheath formation in OLs and Schwann cells through IgMs in vitro has been previously shown with the mouse remyelination-promoting IgM O4 [98], the Ranscht-IgG targeting the sphingolipids galactosylceramide and sulfatide [60,99] and polyclonal IgM preparations [100].
Evidence supporting the indirect hypothesis is derived from the observation that remyelination-promoting IgMs stimulate Ca2+ signaling in GFAP-positive astrocytes [43]. The astrocytic response to IgMs is immediate and precedes the oligodendrocyte response [43]. However, the underlying mechanisms of calcium influxes into astrocytes and OPCs are very different and do not depend on each other [43]. The identification of an OPC-signaling complex responsible for rHIgM22-mediated actions, including PDGFα receptor [97], suggests involvement of the astroglial growth factor PDGF in rHIgM22-mediated actions in OPCs.
Most important, IgM-mediated OPC proliferation was detectable only in cultures containing substantial amounts of astrocytes, microglia and OPCs (mixed glial cultures) but not in highly enriched OPC populations [78]. Secreted astrocytic or microglial factors or direct cellular contact between OPCs and other glia seems to be essential for the proliferative response. In addition, we demonstrated rHIgM22-mediated activation of the PDGF receptor in oligodendrocytes. Because PDGF is secreted by astrocytes and possibly microglia [101,102] but not oligodendrocyte-lineage cells, it demonstrates an involvement of cells other than OLs in IgM-mediated OPC proliferation and, possibly, remyelination. In an analogy to rHIgM22, polyclonal IgM preparations inhibit OPC differentiation in mixed glial but not isolated OPC cultures [100].
It appears that three cell types (OPCs, microglia and astrocytes) are required for IgM-stimulated proliferation of OPCs in vitro. The growth factor PDGF and potentially other secreted microglial and astrocytic factors are important mediators for this effect. However, it has not been determined whether rHIgM22 directly stimulates glial cells other than OPCs (i.e., to produce more growth factors) where IgMbinding to OPCs may not be essential for stimulation of OPC proliferation. Alternatively, rHIgM22-mediated calcium signaling in astrocytes may not affect OPC behavior with unchanged secreted astrocytic factors or cellular components necessary for physical contact with OPCs by rHIgM22. Instead, binding of rHIgM22 to OPCs only may be the necessary prerequisite for stimulation of OPC proliferation in vitro or lesion repair in vivo.
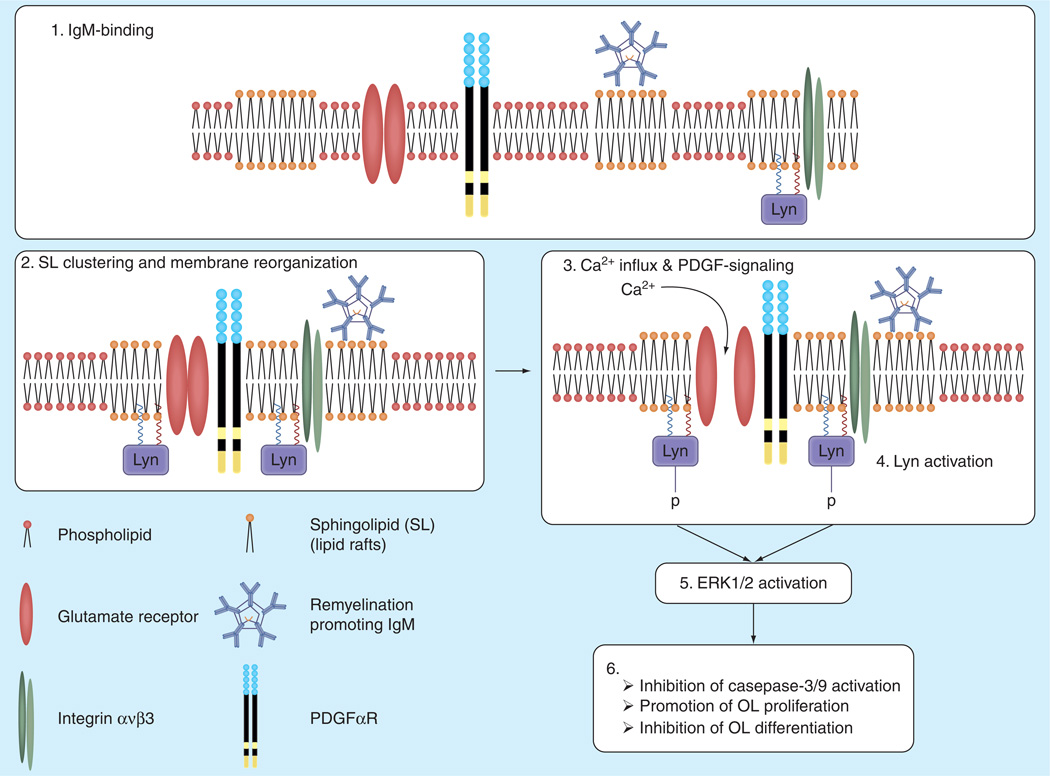
So far, supporting data exist for the latter hypothesis only. No IgM-mediated signal transductions other than calcium influx could be detected in astrocytic cultures [97] or in isolated microglia [97]. In contrast, remyelination-promoting IgMs, including rHIgM22, bind to OL-lineage cells, with rHIgM22 activating Lyn, ERK1 and ERK2 in OPCs leading to inhibition of OPC differentiation and reduced apoptotic signaling (Figure 1) [97]. We also identified the signaling complex in OPCs consisting of integrin αvβ3, Lyn and the OPC marker PDGFαR responsible for rHIgM22-mediated effects [97]. Involvement of PDGFαR not only demonstrated that OPCs mediate the IgM effects but also suggested a central role for the astrocytic ligand, PDGF, in rHIgM22-stimulated OPC proliferation. Inhibition of the PDGFR kinase in OPCs prevented antibody-stimulated OPC proliferation in mixed glial cultures (Figure 1) [78]. In our most recent model (Figure 1), remyelination-promoting IgMs may directly act on OPCs by lowering the threshold for PDGF produced in low quantity in the in vivo situation [103]. This model is supported by the fact that rHIgM22 activates the PDGF receptor in OPCs [78].
Figure 1. Proposed mechanism of action of rHIgM22 in oligodendrite progenitor cells.
Evidence suggests that rHIgM22 targets and clusters SLs at the level of the plasma membrane in OL-lineage cells through its pentameric IgM structure. SLs are essential lipid raft components and IgM-mediated SL clustering may induce and stabilize certain cell surface domains. Membrane reorganization may bring together components of the identified signaling complex including integrin αvβ3, PDGFαR and Lyn kinase. IgM-stimulated activation of kinases Lyn and ERK1 and ERK2 is required for anti-apoptotic signaling and inhibition of OL differentiation. It is undetermined whether Lyn and ERK activation are essential for rHIgM22-stimulated OPC proliferation. Factors from cells other than OPCs are most likely essential for this effect (e.g., PDGF, FGF-2).
OL: Oligodendrocytes; OPCs: Oligodendrocyte progenitor cells; SLs: Glycosphingolipids.
It has not been determined whether PDGF is crucial for IgM-mediated remyelination in vivo. Detectable PDGF levels in vivo in the embryonic brain are below 1 ng/ml [103]. This amount of PDGF is not sufficient by itself to stimulate OPC survival [104,105] or proliferation [106] in vitro. We conclude additional factors are necessary to activate the PDGF response in vivo [107].
In addition, PDGF-concentrations in demyelinated lesions may be different from the surrounding tissue. In fact, evidence from a viral-demyelination model demonstrates up-regulated PDGF expression within the lesion and primarily associated with astrocytes but not neurons [108]. In a chemical-demyelination model, mice overexpressing PDGF have higher levels of proliferating OPCs and less OPC apoptosis in the lesion area [109]. This may indicate involvement of PDGF in OPC proliferation and survival to stimulate remyelination in vivo.
However, in order to make a definitive statement regarding the indirect or direct function of astrocytes in IgM-stimulated OPC proliferation in vitro and lesion repair in vivo, experiments involving IgM-stimulated remyelination in CST−/− (O4) or CGT−/− (O1) animals may be necessary to rule out whether IgM-binding to astrocytes, and potentially microglia, but not to OPCs is sufficient for lesion repair in vivo.
Remyelination-promoting antibodies & their potential beneficial or deleterious effects in the PNS
Elevated levels of certain immunoglobulins, termed autoantibodies targeting SLs and the myelin protein MAG, are sometimes found in peripheral neuropathies [110–114]. The constant association of anti-SL antibodies with PNS dysimmune neuropathies supports a pathogenic link between anti-SL antibodies and neuropathy (e.g., Guillain-Barre syndrome [GBS], Miller Fisher syndrome [MFS], neuropathy associated with IgM monoclonal gammopathy [PN+IgM], and chronic inflammatory demyelinating polyneuropathy [CIDP]). This includes immunoglobulins of the isotypes IgG, IgA and IgM. Antibodies to the glycoprotein MAG and >20 different SLs have been associated with chronic and acute peripheral neuropathic syndromes including GM1, GM1b, GD1a, GalNAc-GD1a (acute motor axonal neuropathy), GQ1b, GD3, GD1b, GT1a (sensory variants GBS) [114] and sulfatide (GBS, CIDP) [115].
Interestingly, antibodies associated with peripheral neuropathies can bind to similar or identical antigens as remyelination-promoting antibodies (anti-sulfatide [O4] [112, 115–118]; anti-SGPG, anti-MAG [HNK1]) [113, 119–121]. Both categories of antibodies can also be of IgM isotype, which seems to be essential for the remyelination-promoting antibody rHIgM22. Some antibodies associated with peripheral neuropathies may act through complement fixation [122], which results in pore formation and cellular destruction after plasma-membrane binding. In contrast to these antibodies, the remyelination-promoting antibody rHIgM22 does not fix complement and does not target Schwann cells or peripheral nerves (unpublished data). Toxicology studies in primates and rodents using 1000-fold higher amounts of rHIgM22 than the therapeutic dose demonstrated no pathological effects in the PNS. Lack of antibody binding to peripheral nerves, however, suggests that rHIgM22 does not stimulate remyelination in the PNS.
Unlike rHIgM22, remyelination-promoting antibodies O4 (sulfatide), O1 (galactosylceramide) and HNK1 (anti-SGPG, MAG) do bind to cell-surface antigens on Schwann cells. It has not been determined whether these antibodies stimulate remyelination in the PNS. Surprisingly, antibodies associated with peripheral neuropathies target the same antigens as the mentioned promotors of remyelination (see above). The different outcome of various antibodies targeting identical antigens on myelinating OLs vs Schwann cells raises the question whether antibodies associated with peripheral neuropathies actively exacerbate the disease course or merely represent a bystander effect. Generation of anti-sphingolipid antibodies may occur after the axonal and myelin destruction due to increasing amounts of cellular debris.
In contrast to their suggested involvement in peripheral neuropathies, gangliosides promote neurite outgrowth and regeneration both in vivo and in vitro [123,124]. Clinical trials using purified gangliosides in neuromuscular disorders did not show sufficient efficacy but also did not give rise to an immune response followed by increased pathology [125,126]. Intramuscular doses of purified bovine ganglioside mixtures were used therapeutically for years throughout Europe, and it is widely believed that these protein-free sphingolipids are not antigenic and do not elicit immune-mediated side-effects. Experimentally, several studies failed to show that gangliosides enhance autoimmune demyelination in the PNS [127–130] and did not induce neurological signs of neuropathies or neuropathological changes [131–133]. In addition, passive transfer of anti-GM1 antibodies failed to transfer the disease [134–136].
Given the number of different diseases covered under the umbrella of ‘peripheral neuropathies’ and their individual complexity, it is extremely difficult to extrapolate which antibodies actively participate in the pathogenesis of different neuropathies based on clinical studies using immunosuppressive and immunomodulatory drugs. However, the efficacy of current immune therapies such as rituximab, prednisolone and cyclophosphamide in neuropathies with anti-MAG IgM antibodies remains unproven [137]. Plasma exchange (PE) seems to be effective in patients with paraproteinemic neuropathies associated with high IgG or IgA levels but not IgM levels [138]. Similarly, corticosteroids, when administered in monotheraphy, were not effective in IgM-associated neuropathies [139]. In support of suggested differences between different antibody isotypes in paraproteinemic neuropathies, IgM-associated distal demyelinating symmetric neuropathies respond rather poorly to immunosuppressive therapy [140]. The reported differences between immunoglobulins of the IgM vs other isotypes in paraproteinemic neuropathies may be due to completely different pathophysiological mechanisms. Apparently, elimination or suppression of the IgM molecule has little or no impact on the disease course and challenges direct involvement of the antibody in the pathophysiological mechanism.
All of this argues against the hypothesis that antibodies targeting SLs are involved in the pathogenesis of peripheral neuropathies.
Whether remyelination-promoting antibodies stimulate remyelination in the PNS remains undetermined. It seems unlikely, however, that remyelination-promoting antibodies O4, O1 and HNK-1 exacerbate the disease course in chronic inflammatory demyelinating polyneuropathies, which is an exclusion criterion in the rHIgM22 clinical trials. It is puzzling that, different IgM antibodies targeting identical SLs have very different outcomes on myelinating OLs compared to Schwann cells. They may lead either to remyelination in the CNS or cellular destruction in the PNS. Clinical results from IgM-associated neuropathies using immunomodulatory or immunosuppressive treatments suggest little or no direct involvement of IgMs in the pathological mechanism of the disease. It could be argued, however, that disease-associated anti-sulfatide IgMs and possibly other IgMs may stimulate remyelination in the CNS similar to the IgM O4 but are ineffective in the PNS. There is no direct evidence suggesting that disease-associated IgM antibodies differ from remyelination-promoting antibodies when targeting identical antigens and are of the same isotype. Mechanistic data indicates that rHIgM22 requires the astrocytic growth factors PDGF and likely FGF-2 to stimulate OPC proliferation and to reduce levels of apoptotic signaling in vitro [78,97]. No rHIgM22-mediated effects could be observed in isolated OPC cultures. This suggests that the cellular microenvironment (astrocytes and possibly microglia) is essential for rHIgM22-mediated effects in the CNS. It is unclear whether the microenvironment provided by Schwann cells and possibly other cells in the PNS support IgM-mediated stimulation of remyelination by rHIgM22 or possibly other remyelination-promoting antibodies.
In brief, it is very unclear whether remyelination-promoting antibodies have an impact on remyelination in the PNS. However, we tend to be rather skeptical because of the presence of disease-associated antibodies with potentially very similar properties compared to remyelination-promoting antibodies and lack of sufficient remyelination in the PNS in those cases. In addition, it remains elusive whether the cellular microenvironment present at the lesion site in the PNS resembles the microenvironment in CNS lesions, which is required for rHIgM22-mediated stimulation of remyelination.
Expert commentary & five-year view
The human-remyelination promoting IgM rHIgM22 recently entered Phase I clinical trial for MS patients. rHIgM22 shows efficacy in animal models of MS when administered as a single bolus injection. These results suggest a rather catalytic mechanism for rHIgM22 and possibly other remyelination-promoting antibodies in vivo with minimal amounts of antibodies required; this is in contrast to blocking or neutralizing antibodies, where stoichiometric ratios between antigen and antibody are necessary. BBB permeability is a necessity for all neurotherapeutics injected peripherally. Accumulating evidence suggests that <0.1% of the original amount of antibody administered crosses the BBB in different animal models of neurological diseases. No significant differences in BBB permeability were detected between antibodies of the IgG and IgM isotype. The low amount of antibody necessary for stimulation of remyelination (rHIgM22) favors NAbs as a potential therapeutic for neurological and other disorders. Due to the complexity of MS, with multiple inhibitory factors present in demyelinated lesions, we expect combination therapies to be most successful. In order to find the most potent treatment, there clearly is a need to develop better pre-screening tests as well as animal models that more closely mimic human disease. The choice of animal model will preselect certain classes of molecules or otherwise exclude potentially successful therapeutics from human trials. The progress made over the last years in the characterization of the lesion microenvironment, including inhibitory molecules on the one hand and the use of NAbs in the treatment of human diseases on the other provides reasons for optimism.
Key issues.
Naturally occurring autoantibodies (NAbs) are of germline origin with little or no somatic mutations. They are often polyreactive and bind with rather low affinity to one or multiple structurally unrelated antigens.
NAbs are part of the innate immunity and involved in tissue homeostasis, defense against pathogens, tumor surveillance and stimulation of brain repair.
NAbs often bind to carbohydrate epitopes on glycosphingolipids (SLs) and, potentially, proteins in normal, apoptotic or cancer cells.
Remyelination-promoting antibodies belong to the group of natural occurring antibodies.
Most known remyelination-promoting antibodies are of the IgM isotype and bind to myelin and oligodendrocytes. Their molecular targets are cell surface SLs and, to some extent, cytoskeletal proteins.
Repair of brain lesions by remyelination-promoting antibodies may be mediated through increased lesion clearance with IgM-tagged myelin debris ready for opsonization or through lipid-raft formation in oligodendrocyte progenitor cells with higher levels of PDGF-receptor activation.
Stimulation of oligodendrocyte progenitor proliferation in vitro by remyelination-promoting antibodies (rHIgM22) requires the presence of astrocytes and microglial cells.
The remyelination-promoting antibody rHIgM22 stimulates remyelination in Theiler’s murine encephalomyelitis virus-infected mice after a single minimal dose of 500 ng per animal.
The integrity of the IgM molecule appears to be essential for stimulation of remyelination in vivo.
Remyelination-promoting antibodies show efficacy in multiple sclerosis animal models with extensive (chronic) demyelination (Theiler’s virus-induced demyelination) but not in primarily immune-mediated animal models (experimental autoimmune encephalomyelitis).
Acknowledgments
This work was supported by grants from the NIH (R01 GM092993, R01 NS048357 and R21 NS073684) and the National Multiple Sclerosis Society (CA 1060A). This work was also supported by a High-Impact Pilot and Feasibility Award (HIPFA) and Novel Methodology Award (NMDA) from the Mayo Clinic Center for Translational Science Activities (CTSA) and Mayo Clinic CTSA grant number UL1 TR000135 from the National Center for Advancing Translational Science (NCATS), a component of the National Institutes of Health (NIH). The authors also acknowledge with thanks support from the Applebaum, Hilton, Peterson and Sanford Foundations, the Minnesota Partnership Award for Biotechnology and Medical Genomics and the McNeilus family.
Footnotes
Financial & competing interests disclosure
The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
References
- 1.Avrameas S. Natural autoantibodies: from ‘horror autotoxicus’ to ‘gnothi seauton’. Immunol. Today. 1991;12(5):154–159. doi: 10.1016/S0167-5699(05)80045-3. [DOI] [PubMed] [Google Scholar]
- 2.Coutinho A, Kazatchkine MD, Avrameas S. Natural autoantibodies. Curr. Opin. Immunol. 1995;7(6):812–818. doi: 10.1016/0952-7915(95)80053-0. [DOI] [PubMed] [Google Scholar]
- 3.Dighiero G, Lymberi P, Mazie JC, et al. Murine hybridomas secreting natural monoclonal antibodies reacting with self antigens. J. Immunol. 1983;131(5):2267–2272. [PubMed] [Google Scholar]
- 4.Haspel MV, Onodera T, Prabhakar BS, Horita M, Suzuki H, Notkins AL. Virus-induced autoimmunity: monoclonal antibodies that react with endocrine tissues. Science. 1983;220(4594):304–306. doi: 10.1126/science.6301002. [DOI] [PubMed] [Google Scholar]
- 5.Haspel MV, Onodera T, Prabhakar BS, et al. Multiple organ-reactive monoclonal autoantibodies. Nature. 1983;304(5921):73–76. doi: 10.1038/304073a0. [DOI] [PubMed] [Google Scholar]
- 6.Prabhakar BS, Saegusa J, Onodera T, Notkins AL. Lymphocytes capable of making monoclonal autoantibodies that react with multiple organs are a common feature of the normal B cell repertoire. J. Immunol. 1984;133(6):2815–2817. [PubMed] [Google Scholar]
- 7.Satoh J, Prabhakar BS, Haspel MV, Ginsberg-Fellner F, Notkins AL. Human monoclonal autoantibodies that react with multiple endocrine organs. N. Engl. J. Med. 1983;309(4):217–220. doi: 10.1056/NEJM198307283090405. [DOI] [PubMed] [Google Scholar]
- 8.Lacroix-Desmazes S, Kaveri SV, Mouthon L, et al. Self-reactive antibodies (natural autoantibodies) in healthy individuals. J. Immunol. Methods. 1998;216(1–2):117–137. doi: 10.1016/s0022-1759(98)00074-x. [DOI] [PubMed] [Google Scholar]
- 9.Merbl Y, Zucker-Toledano M, Quintana FJ, Cohen IR. Newborn humans manifest autoantibodies to defined self molecules detected by antigen microarray informatics. J. Clin. Invest. 2007;117(3):712–718. doi: 10.1172/JCI29943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Avrameas S, Selmi C. Natural autoantibodies in the physiology and pathophysiology of the immune system. J. Autoimmun. 2013;41:46–49. doi: 10.1016/j.jaut.2013.01.006. [DOI] [PubMed] [Google Scholar]
- 11.Avrameas S, Ternynck T. The natural autoantibodies system: between hypotheses and facts. Mol. Immunol. 1993;30(12):1133–1142. doi: 10.1016/0161-5890(93)90160-d. [DOI] [PubMed] [Google Scholar]
- 12.Brandlein S, Rauschert N, Rasche L, et al. The human IgM antibody SAM-6 induces tumor-specific apoptosis with oxidized low-density lipoprotein. Mol. Cancer Ther. 2007;6(1):326–333. doi: 10.1158/1535-7163.MCT-06-0399. [DOI] [PubMed] [Google Scholar]
- 13.Pohle T, Brandlein S, Ruoff N, Muller-Hermelink HK, Vollmers HP. Lipoptosis: tumor-specific cell death by antibody-induced intracellular lipid accumulation. Cancer Res. 2004;64(11):3900–3906. doi: 10.1158/0008-5472.CAN-03-3149. [DOI] [PubMed] [Google Scholar]
- 14.Rauschert N, Brandlein S, Holzinger E, Hensel F, Muller-Hermelink HK, Vollmers HP. A new tumor-specific variant of GRP78 as target for antibody-based therapy. Lab. Invest. 2008;88(4):375–386. doi: 10.1038/labinvest.2008.2. [DOI] [PubMed] [Google Scholar]
- 15.Vollmers HP, Brandlein S. Natural antibodies and cancer. N. Biotechnol. 2009;25(5):294–298. doi: 10.1016/j.nbt.2009.03.016. [DOI] [PubMed] [Google Scholar]
- 16.Bieber AJ, Warrington A, Asakura K, et al. Human antibodies accelerate the rate of remyelination following lysolecithin-induced demyelination in mice. Glia. 2002;37(3):241–249. doi: 10.1002/glia.10033. [DOI] [PubMed] [Google Scholar]
- 17.Warrington AE, Asakura K, Bieber AJ, et al. Human monoclonal antibodies reactive to oligodendrocytes promote remyelination in a model of multiple sclerosis. Proc. Natl Acad. Sci. USA. 2000;97(12):6820–6825. doi: 10.1073/pnas.97.12.6820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Warrington AE, Bieber AJ, Ciric B, Pease LR, Van Keulen V, Rodriguez M. A recombinant human IgM promotes myelin repair after a single, very low dose. J. Neurosci. Res. 2007;85(5):967–976. doi: 10.1002/jnr.21217. [DOI] [PubMed] [Google Scholar]
- 19.Roy B, Shukla S, Lyszkiewicz M, et al. Somatic hypermutation in peritoneal B1b cells. Mol. Immunol. 2009;46(8–9):1613–1619. doi: 10.1016/j.molimm.2009.02.026. [DOI] [PubMed] [Google Scholar]
- 20.Tumas-Brundage KM, Notidis E, Heltemes L, Zhang X, Wysocki LJ, Manser T. Predominance of a novel splenic B cell population in mice expressing a transgene that encodes multireactive antibodies: support for additional heterogeneity of the B cell compartment. Int. Immunol. 2001;13(4):475–484. doi: 10.1093/intimm/13.4.475. [DOI] [PubMed] [Google Scholar]
- 21.Baumgarth N, Tung JW, Herzenberg LA. Inherent specificities in natural antibodies: a key to immune defense against pathogen invasion. Springer Semin. Immunopathol. 2005;26(4):347–362. doi: 10.1007/s00281-004-0182-2. [DOI] [PubMed] [Google Scholar]
- 22.Holodick NE, Tumang JR, Rothstein TL. Immunoglobulin secretion by B1 cells: differential intensity and IRF4-dependence of spontaneous IgM secretion by peritoneal and splenic B1 cells. Eur. J. Immunol. 2010;40(11):3007–3016. doi: 10.1002/eji.201040545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kantor AB, Merrill CE, Herzenberg LA, Hillson JL. An unbiased analysis of V(H)-D-J(H) sequences from B-1a, B-1b, and conventional B cells. J. Immunol. 1997;158(3):1175–1186. [PubMed] [Google Scholar]
- 24.Landsteiner K. The Specificity of Serological Reactions. MA, USA: Harvard University Press; 1947. [Google Scholar]
- 25.Casali P, Notkins AL. CD5+ B lymphocytes, polyreactive antibodies and the human B-cell repertoire. Immunol. Today. 1989;10(11):364–368. doi: 10.1016/0167-5699(89)90268-5. [DOI] [PubMed] [Google Scholar]
- 26.Dighiero G, Guilbert B, Avrameas S. Naturally occurring antibodies against nine common antigens in humans sera. II. High incidence of monoclonal Ig exhibiting antibody activity against actin and tubulin and sharing antibody specificities with natural antibodies. J. Immunol. 1982;128(6):2788–2792. [PubMed] [Google Scholar]
- 27.Quan CP, Berneman A, Pires R, Avrameas S, Bouvet JP. Natural polyreactive secretory immunoglobulin A autoantibodies as a possible barrier to infection in humans. Infect. Immun. 1997;65(10):3997–4004. doi: 10.1128/iai.65.10.3997-4004.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Marchalonis JJ, Adelman MK, Robey IF, Schluter SF, Edmundson AB. Exquisite specificity and peptide epitope recognition promiscuity, properties shared by antibodies from sharks to humans. J. Mol. Recognit. 2001;14(2):110–121. doi: 10.1002/jmr.527. [DOI] [PubMed] [Google Scholar]
- 29.Notkins AL. Polyreactivity of antibody molecules. Trends Immunol. 2004;25(4):174–179. doi: 10.1016/j.it.2004.02.004. [DOI] [PubMed] [Google Scholar]
- 30.Crouzier R, Martin T, Pasquali JL. Heavy chain variable region, light chain variable region, and heavy chain CDR3 influences on the mono- and polyreactivity and on the affinity of human monoclonal rheumatoid factors. J. Immunol. 1995;154(9):4526–4535. [PubMed] [Google Scholar]
- 31.Ditzel HJ, Itoh K, Burton DR. Determinants of polyreactivity in a large panel of recombinant human antibodies from HIV-1 infection. J. Immunol. 1996;157(2):739–749. [PubMed] [Google Scholar]
- 32.Ichiyoshi Y, Casali P. Analysis of the structural correlates for antibody polyreactivity by multiple reassortments of chimeric human immunoglobulin heavy and light chain V segments. J. Exp. Med. 1994;180(3):885–895. doi: 10.1084/jem.180.3.885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Martin T, Crouzier R, Weber JC, Kipps TJ, Pasquali JL. Structure-function studies on a polyreactive (natural) autoantibody. Polyreactivity is dependent on somatically generated sequences in the third complementarity-determining region of the antibody heavy chain. J. Immunol. 1994;152(12):5988–5996. [PubMed] [Google Scholar]
- 34.Polymenis M, Stollar BD. Critical binding site amino acids of anti-Z-DNA single chain Fv molecules. Role of heavy and light chain CDR3 and relationship to autoantibody activity. J. Immunol. 1994;152(11):5318–5329. [PubMed] [Google Scholar]
- 35.Fernandez C, Alarcon-Riquelme ME, Sverremark E. Polyreactive binding of antibodies generated by polyclonal B cell activation. II. Crossreactive and monospecific antibodies can be generated from an identical Ig rearrangement by differential glycosylation. Scand. J. Immunol. 1997;45(3):240–247. doi: 10.1046/j.1365-3083.1997.d01-398.x. [DOI] [PubMed] [Google Scholar]
- 36.Leibiger H, Wustner D, Stigler RD, Marx U. Variable domain-linked oligosaccharides of a human monoclonal IgG: structure and influence on antigen binding. Biochem. J. 1999;338(Pt 2):529–538. [PMC free article] [PubMed] [Google Scholar]
- 37.Leung SO, Dion AS, Pellegrini MC, et al. Effect of VK framework-1 glycosylation on the binding affinity of lymphoma-specific murine and chimeric LL2 antibodies and its potential use as a novel conjugation site. Int. J. Cancer. 1995;60(4):534–538. doi: 10.1002/ijc.2910600418. [DOI] [PubMed] [Google Scholar]
- 38.Wright A, Tao MH, Kabat EA, Morrison SL. Antibody variable region glycosylation: position effects on antigen binding and carbohydrate structure. EMBO J. 1991;10(10):2717–2723. doi: 10.1002/j.1460-2075.1991.tb07819.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rodriguez M, Oleszak E, Leibowitz J. Theiler’s murine encephalomyelitis: a model of demyelination and persistence of virus. Crit. Rev. Immunol. 1987;7(4):325–365. [PubMed] [Google Scholar]
- 40.Rodriguez M, Lennon VA, Benveniste EN, Merrill JE. Remyelination by oligodendrocytes stimulated by antiserum to spinal cord. J. Neuropathol. Exp. Neurol. 1987;46(1):84–95. doi: 10.1097/00005072-198701000-00008. [DOI] [PubMed] [Google Scholar]
- 41.Rodriguez M, Lennon VA. Immunoglobulins promote remyelination in the central nervous system. Ann. Neurol. 1990;27(1):12–17. doi: 10.1002/ana.410270104. [DOI] [PubMed] [Google Scholar]
- 42.Miller DJ, Sanborn KS, Katzmann JA, Rodriguez M. Monoclonal autoantibodies promote central nervous system repair in an animal model of multiple sclerosis. J. Neurosci. 1994;14(10):6230–6238. doi: 10.1523/JNEUROSCI.14-10-06230.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Paz Soldan MM, Warrington AE, Bieber AJ, et al. Remyelination-promoting antibodies activate distinct Ca2+ influx pathways in astrocytes and oligodendrocytes: relationship to the mechanism of myelin repair. Mol. Cell Neurosci. 2003;22(1):14–24. doi: 10.1016/s1044-7431(02)00018-0. [DOI] [PubMed] [Google Scholar]
- 44.Eisenbarth GS, Walsh FS, Nirenberg M. Monoclonal antibody to a plasma membrane antigen of neurons. Proc. Natl Acad. Sci. USA. 1979;76(10):4913–4917. doi: 10.1073/pnas.76.10.4913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ilyas AA, Quarles RH, Brady RO. The monoclonal antibody HNK-1 reacts with a human peripheral nerve ganglioside. Biochem. Biophys. Res. Commun. 1984;122(3):1206–1211. doi: 10.1016/0006-291x(84)91220-8. [DOI] [PubMed] [Google Scholar]
- 46.Sommer I, Schachner M. Monoclonal antibodies (O1 to O4) to oligodendrocyte cell surfaces: an immunocytological study in the central nervous system. Dev. Biol. 1981;83(2):311–327. doi: 10.1016/0012-1606(81)90477-2. [DOI] [PubMed] [Google Scholar]
- 47.Asakura K, Miller DJ, Pease LR, Rodriguez M. Targeting of IgMkappa antibodies to oligodendrocytes promotes CNS remyelination. J. Neurosci. 1998;18(19):7700–7708. doi: 10.1523/JNEUROSCI.18-19-07700.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mitsunaga Y, Ciric B, Van Keulen V, et al. Direct evidence that a human antibody derived from patient serum can promote myelin repair in a mouse model of chronic-progressive demyelinating disease. FASEB J. 2002;16(10):1325–1327. doi: 10.1096/fj.01-0994fje. [DOI] [PubMed] [Google Scholar]
- 49.Warrington AE, Bieber AJ, Ciric B, Pease LR, Van Keulen V, Rodriguez M. A recombinant human IgM promotes myelin repair after a single, very low dose. J. Neurosci. Res. 2007;85(5):967–976. doi: 10.1002/jnr.21217. [DOI] [PubMed] [Google Scholar]
- 50.Asakura K, Miller DJ, Murray K, Bansal R, Pfeiffer SE, Rodriguez M. Monoclonal autoantibody SCH94.03, which promotes central nervous system remyelination, recognizes an antigen on the surface of oligodendrocytes. J. Neurosci. Res. 1996;43(3):273–281. doi: 10.1002/(SICI)1097-4547(19960201)43:3<273::AID-JNR2>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 51.Asakura K, Pogulis RJ, Pease LR, Rodriguez M. A monoclonal autoantibody which promotes central nervous system remyelination is highly polyreactive to multiple known and novel antigens. J. Neuroimmunol. 1996;65(1):11–19. doi: 10.1016/0165-5728(95)00175-1. [DOI] [PubMed] [Google Scholar]
- 52.Pavelko KD, van Engelen BG, Rodriguez M. Acceleration in the rate of CNS remyelination in lysolecithin-induced demyelination. J. Neurosci. 1998;18(7):2498–2505. doi: 10.1523/JNEUROSCI.18-07-02498.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wittenberg NJ, Im H, Xu X, et al. High-affinity binding of remyelinating natural autoantibodies to myelin-mimicking lipid bilayers revealed by nanohole surface plasmon resonance. Anal. Chem. 2012;84(14):6031–6039. doi: 10.1021/ac300819a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dubois C, Manuguerra JC, Hauttecoeur B, Maze J. Monoclonal antibody A2B5, which detects cell surface antigens, binds to ganglioside GT3 (II3 (NeuAc)3LacCer) and to its 9-O-acetylated derivative. J. Biol. Chem. 1990;265(5):2797–2803. [PubMed] [Google Scholar]
- 55.Fredman P, Magnani JL, Nirenberg M, Ginsburg V. Monoclonal antibody A2B5 reacts with many gangliosides in neuronal tissue. Arch. Biochem. Biophys. 1984;233(2):661–666. doi: 10.1016/0003-9861(84)90492-2. [DOI] [PubMed] [Google Scholar]
- 56.Burger D, Perruisseau G, Simon M, Steck AJ. Comparison of the N-linked oligosaccharide structures of the two major human myelin glycoproteins MAG and P0: assessment of the structures bearing the epitope for HNK-1 and human monoclonal immunoglobulin M found in demyelinating neuropathy. J. Neurochem. 1992;58(3):854–861. doi: 10.1111/j.1471-4159.1992.tb09335.x. [DOI] [PubMed] [Google Scholar]
- 57.Burger D, Simon M, Perruisseau G, Steck AJ. The epitope(s) recognized by HNK-1 antibody and IgM paraprotein in neuropathy is present on several N-linked oligosaccharide structures on human P0 and myelin-associated glycoprotein. J. Neurochem. 1990;54(5):1569–1575. doi: 10.1111/j.1471-4159.1990.tb01206.x. [DOI] [PubMed] [Google Scholar]
- 58.Schachner M. Cell type-specific surface antigens in the mammalian nervous system. J. Neurochem. 1982;39(1):1–8. doi: 10.1111/j.1471-4159.1982.tb04694.x. [DOI] [PubMed] [Google Scholar]
- 59.Singh H, Pfeiffer SE. Myelin-associated galactolipids in primary cultures from dissociated fetal rat brain: biosynthesis, accumulation, and cell surface expression. J. Neurochem. 1985;45(5):1371–1381. doi: 10.1111/j.1471-4159.1985.tb07202.x. [DOI] [PubMed] [Google Scholar]
- 60.Bansal R, Pfeiffer SE. Reversible inhibition of oligodendrocyte progenitor differentiation by a monoclonal antibody against surface galactolipids. Proc. Natl Acad. Sci. USA. 1989;86(16):6181–6185. doi: 10.1073/pnas.86.16.6181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bansal R, Stefansson K, Pfeiffer SE. Proligodendroblast antigen (POA), a developmental antigen expressed by A007/ O4-positive oligodendrocyte progenitors prior to the appearance of sulfatide and galactocerebroside. J. Neurochem. 1992;58(6):2221–2229. doi: 10.1111/j.1471-4159.1992.tb10967.x. [DOI] [PubMed] [Google Scholar]
- 62.Kirschning E, Jensen K, Dubel S, Rutter G, Hohenberg H, Will H. Primary structure of the antigen-binding domains of a human oligodendrocyte-reactive IgM monoclonal antibody derived from a patient with multiple sclerosis. J. Neuroimmunol. 1999;99(1):122–130. doi: 10.1016/s0165-5728(99)00118-6. [DOI] [PubMed] [Google Scholar]
- 63.Sakakibara K, Momoi T, Uchida T, Nagai Y. Evidence for association of glycosphingolipid with a colchicine-sensitive microtubule-like cytoskeletal structure of cultured cells. Nature. 1981;293(5827):76–78. doi: 10.1038/293076a0. [DOI] [PubMed] [Google Scholar]
- 64.Zalc B, Monge M, Dupouey P, Hauw JJ, Baumann NA. Immunohistochemical localization of galactosyl and sulfogalactosyl ceramide in the brain of the 30-day-old mouse. Brain Res. 1981;211(2):341–354. doi: 10.1016/0006-8993(81)90706-x. [DOI] [PubMed] [Google Scholar]
- 65.Inoko E, Nishiura Y, Tanaka H, et al. Developmental stage-dependent expression of an alpha2,8-trisialic acid unit on glycoproteins in mouse brain. Glycobiology. 2010;20(7):916–928. doi: 10.1093/glycob/cwq049. [DOI] [PubMed] [Google Scholar]
- 66.Dubois C, Magnani JL, Grunwald GB, et al. Monoclonal antibody 18B8, which detects synapse-associated antigens, binds to ganglioside GT3 (II3 (NeuAc)3LacCer) J. Biol. Chem. 1986;261(8):3826–3830. [PubMed] [Google Scholar]
- 67.Hirabayashi Y, Hirota M, Matsumoto M, Tanaka H, Obata K, Ando S. Developmental changes of C-series polysialogangliosides in chick brains revealed by mouse monoclonal antibodies M6704 and M7103 with different epitope specificities. J. Biochem. 1988;104(6):973–979. doi: 10.1093/oxfordjournals.jbchem.a122593. [DOI] [PubMed] [Google Scholar]
- 68.Hirabayashi Y, Hirota M, Suzuki Y, Matsumoto M, Obata K, Ando S. Developmentally expressed O-acetyl ganglioside GT3 in fetal rat cerebral cortex. Neurosci. Lett. 1989;106(1–2):193–198. doi: 10.1016/0304-3940(89)90225-5. [DOI] [PubMed] [Google Scholar]
- 69.Rosner H, Greis C, Henke-Fahle S. Developmental expression in embryonic rat and chicken brain of a polysialoganglioside-antigen reacting with the monoclonal antibody Q 211. Brain Res. 1988;470(2):161–171. doi: 10.1016/0165-3806(88)90234-9. [DOI] [PubMed] [Google Scholar]
- 70.Rosner H, Sonnentag U, Meiri K. Developmental changes of growth cone gangliosides of the postnatal rat cerebrum. Neuroreport. 1993;4(10):1207–1210. [PubMed] [Google Scholar]
- 71.Heffer-Lauc M, Cacic M, Serman D. C-series polysialogangliosides are expressed on stellate neurons of adult human cerebellum. Glycoconj. J. 1998;15(4):423–426. doi: 10.1023/a:1006938221704. [DOI] [PubMed] [Google Scholar]
- 72.Carroll WM, Jennings AR. Early recruitment of oligodendrocyte precursors in CNS demyelination. Brain. 1994;117(Pt 3):563–578. doi: 10.1093/brain/117.3.563. [DOI] [PubMed] [Google Scholar]
- 73.Gensert JM, Goldman JE. Endogenous progenitors remyelinate demyelinated axons in the adult CNS. Neuron. 1997;19(1):197–203. doi: 10.1016/s0896-6273(00)80359-1. [DOI] [PubMed] [Google Scholar]
- 74.Horner PJ, Power AE, Kempermann G, et al. Proliferation and differentiation of progenitor cells throughout the intact adult rat spinal cord. J. Neurosci. 2000;20(6):2218–2228. doi: 10.1523/JNEUROSCI.20-06-02218.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Watanabe M, Toyama Y, Nishiyama A. Differentiation of proliferated NG2-positive glial progenitor cells in a remyelinating lesion. J. Neurosci. Res. 2002;69(6):826–836. doi: 10.1002/jnr.10338. [DOI] [PubMed] [Google Scholar]
- 76.Windrem MS, Roy NS, Wang J, et al. Progenitor cells derived from the adult human subcortical white matter disperse and differentiate as oligodendrocytes within demyelinated lesions of the rat brain. J. Neurosci. Res. 2002;69(6):966–975. doi: 10.1002/jnr.10397. [DOI] [PubMed] [Google Scholar]
- 77.Zhang SC, Ge B, Duncan ID. Adult brain retains the potential to generate oligodendroglial progenitors with extensive myelination capacity. Proc. Natl Acad. Sci. USA. 1999;96(7):4089–4094. doi: 10.1073/pnas.96.7.4089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Watzlawik JO, Warrington AE, Rodriguez M. PDGF is required for remyelination-promoting igm stimulation of oligodendrocyte progenitor cell proliferation. PLoS ONE. 2013;8(2):e55149. doi: 10.1371/journal.pone.0055149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kleine TO, Hackler R, Zofel P. Age-related alterations of the blood-brain-barrier (bbb) permeability to protein molecules of different size. Z. Gerontol. 1993;26(4):256–259. [PubMed] [Google Scholar]
- 80.Argaw AT, Asp L, Zhang J, et al. Astrocyte-derived VEGF-A drives blood-brain barrier disruption in CNS inflammatory disease. J. Clin. Invest. 2012;122(7):2454–2468. doi: 10.1172/JCI60842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pirko I, Ciric B, Gamez J, et al. A human antibody that promotes remyelination enters the CNS and decreases lesion load as detected by T2-weighted spinal cord MRI in a virus-induced murine model of MS. FASEB J. 2004;18(13):1577–1579. doi: 10.1096/fj.04-2026fje. [DOI] [PubMed] [Google Scholar]
- 82.Broman T. Blood-Brain Barrier Damage in Multiple Sclerosis Supravital Test-Observations. Acta Neurol. Scand. Suppl. 1964;40(Suppl. 10):21–14. doi: 10.1111/j.1600-0404.1964.tb04718.x. [DOI] [PubMed] [Google Scholar]
- 83.McQuaid S, Cunnea P, McMahon J, Fitzgerald U. The effects of blood-brain barrier disruption on glial cell function in multiple sclerosis. Biochem. Soc. Trans. 2009;37(Pt 1):329–331. doi: 10.1042/BST0370329. [DOI] [PubMed] [Google Scholar]
- 84.Claudio L, Raine CS, Brosnan CF. Evidence of persistent blood-brain barrier abnormalities in chronic-progressive multiple sclerosis. Acta Neuropathol. 1995;90(3):228–238. doi: 10.1007/BF00296505. [DOI] [PubMed] [Google Scholar]
- 85.Gay D, Esiri M. Blood-brain barrier damage in acute multiple sclerosis plaques. An immunocytological study. Brain. 1991;114(Pt 1B):557–572. doi: 10.1093/brain/114.1.557. [DOI] [PubMed] [Google Scholar]
- 86.Kwon EE, Prineas JW. Blood-brain barrier abnormalities in longstanding multiple sclerosis lesions. An immunohistochemical study. J. Neuropathol. Exp. Neurol. 1994;53(6):625–636. doi: 10.1097/00005072-199411000-00010. [DOI] [PubMed] [Google Scholar]
- 87.Filippi M, Rocca MA, Martino G, Horsfield MA, Comi G. Magnetization transfer changes in the normal appearing white matter precede the appearance of enhancing lesions in patients with multiple sclerosis. Ann. Neurol. 1998;43(6):809–814. doi: 10.1002/ana.410430616. [DOI] [PubMed] [Google Scholar]
- 88.Kermode AG, Thompson AJ, Tofts P, et al. Breakdown of the blood-brain barrier precedes symptoms and other MRI signs of new lesions in multiple sclerosis. Pathogenetic and clinical implications. Brain. 1990;113(Pt 5):1477–1489. doi: 10.1093/brain/113.5.1477. [DOI] [PubMed] [Google Scholar]
- 89.Marik C, Felts PA, Bauer J, Lassmann H, Smith KJ. Lesion genesis in a subset of patients with multiple sclerosis: a role for innate immunity? Brain. 2007;130(Pt 11):2800–2815. doi: 10.1093/brain/awm236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sobel RA. The extracellular matrix in multiple sclerosis lesions. J. Neuropathol. Exp. Neurol. 1998;57(3):205–217. doi: 10.1097/00005072-199803000-00001. [DOI] [PubMed] [Google Scholar]
- 91.Sobel RA, Chen M, Maeda A, Hinojoza JR. Vitronectin and integrin vitronectin receptor localization in multiple sclerosis lesions. J. Neuropathol. Exp. Neurol. 1995;54(2):202–213. doi: 10.1097/00005072-199503000-00007. [DOI] [PubMed] [Google Scholar]
- 92.Sobel RA, Mitchell ME. Fibronectin in multiple sclerosis lesions. Am. J. Pathol. 1989;135(1):161–168. [PMC free article] [PubMed] [Google Scholar]
- 93.Vos CM, Geurts JJ, Montagne L, et al. Blood-brain barrier alterations in both focal and diffuse abnormalities on postmortem MRI in multiple sclerosis. Neurobiol. Dis. 2005;20(3):953–960. doi: 10.1016/j.nbd.2005.06.012. [DOI] [PubMed] [Google Scholar]
- 94.Howe CL, Bieber AJ, Warrington AE, Pease LR, Rodriguez M. Antiapoptotic signaling by a remyelination-promoting human antimyelin antibody. Neurobiol. Dis. 2004;15(1):120–131. doi: 10.1016/j.nbd.2003.09.002. [DOI] [PubMed] [Google Scholar]
- 95.Kanter JL, Narayana S, Ho PP, et al. Lipid microarrays identify key mediators of autoimmune brain inflammation. Nat. Med. 2006;12(1):138–143. doi: 10.1038/nm1344. [DOI] [PubMed] [Google Scholar]
- 96.Mitsunaga Y, Ciric B, Van Keulen V, et al. Direct evidence that a human antibody derived from patient serum can promote myelin repair in a mouse model of chronic-progressive demyelinating disease. Faseb. J. 2002;16(10):1325–1327. doi: 10.1096/fj.01-0994fje. [DOI] [PubMed] [Google Scholar]
- 97.Watzlawik J, Holicky E, Edberg DD, et al. Human remyelination promoting antibody inhibits apoptotic signaling and differentiation through Lyn kinase in primary rat oligodendrocytes. Glia. 2010;58(15):1782–1793. doi: 10.1002/glia.21048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bansal R, Winkler S, Bheddah S. Negative regulation of oligodendrocyte differentiation by galactosphingolipids. J. Neurosci. 1999;19(18):7913–7924. doi: 10.1523/JNEUROSCI.19-18-07913.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ranscht B, Wood PM, Bunge RP. Inhibition of in vitro peripheral myelin formation by monoclonal anti-galactocerebroside. J. Neurosci. 1987;7(9):2936–2947. doi: 10.1523/JNEUROSCI.07-09-02936.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Stangel M, Bernard D. Polyclonal IgM influence oligodendrocyte precursor cells in mixed glial cell cultures: implications for remyelination. J. Neuroimmunol. 2003;138(1–2):25–30. doi: 10.1016/s0165-5728(03)00087-0. [DOI] [PubMed] [Google Scholar]
- 101.Pringle N, Collarini EJ, Mosley MJ, Heldin CH, Westermark B, Richardson WD. PDGF A chain homodimers drive proliferation of bipotential (O-2A) glial progenitor cells in the developing rat optic nerve. EMBO J. 1989;8(4):1049–1056. doi: 10.1002/j.1460-2075.1989.tb03472.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Silberstein FC, De Simone R, Levi G, Aloisi F. Cytokine-regulated expression of platelet-derived growth factor gene and protein in cultured human astrocytes. J. Neurochem. 1996;66(4):1409–1417. doi: 10.1046/j.1471-4159.1996.66041409.x. [DOI] [PubMed] [Google Scholar]
- 103.van Heyningen P, Calver AR, Richardson WD. Control of progenitor cell number by mitogen supply and demand. Curr. Biol. 2001;11(4):232–241. doi: 10.1016/s0960-9822(01)00075-6. [DOI] [PubMed] [Google Scholar]
- 104.Colognato H, Baron W, Avellana-Adalid V, et al. CNS integrins switch growth factor signalling to promote target-dependent survival. Nat. Cell Biol. 2002;4(11):833–841. doi: 10.1038/ncb865. [DOI] [PubMed] [Google Scholar]
- 105.Frost EE, Buttery PC, Milner R, ffrench-Constant C. Integrins mediate a neuronal survival signal for oligodendrocytes. Curr. Biol. 1999;9(21):1251–1254. doi: 10.1016/s0960-9822(99)80506-5. [DOI] [PubMed] [Google Scholar]
- 106.Baron W, Shattil SJ, ffrench-Constant C. The oligodendrocyte precursor mitogen PDGF stimulates proliferation by activation of alpha(v)beta3 integrins. EMBO J. 2002;21(8):1957–1966. doi: 10.1093/emboj/21.8.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Baron W, Colognato H, ffrench-Constant C. Integrin-growth factor interactions as regulators of oligodendroglial development and function. Glia. 2005;49(4):467–479. doi: 10.1002/glia.20132. [DOI] [PubMed] [Google Scholar]
- 108.Redwine JM, Armstrong RC. In vivo proliferation of oligodendrocyte progenitors expressing PDGFalphaR during early remyelination. J. Neurobiol. 1998;37(3):413–428. doi: 10.1002/(sici)1097-4695(19981115)37:3<413::aid-neu7>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- 109.Vana AC, Flint NC, Harwood NE, Le TQ, Fruttiger M, Armstrong RC. Platelet-derived growth factor promotes repair of chronically demyelinated white matter. J. Neuropathol. Exp. Neurol. 2007;66(11):975–988. doi: 10.1097/NEN.0b013e3181587d46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ilyas AA, Quarles RH, MacIntosh TD, et al. IgM in a human neuropathy related to paraproteinemia binds to a carbohydrate determinant in the myelin-associated glycoprotein and to a ganglioside. Proc. Natl Acad. Sci U.S.A. 1984;81(4):1225–1229. doi: 10.1073/pnas.81.4.1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ilyas AA, Willison HJ, Quarles RH, et al. Serum antibodies to gangliosides in Guillain-Barre syndrome. Ann. Neurol. 1988;23(5):440–447. doi: 10.1002/ana.410230503. [DOI] [PubMed] [Google Scholar]
- 112.Latov N. Pathogenesis and therapy of neuropathies associated with monoclonal gammopathies. Ann. Neurol. 1995;37(Suppl. 1):S32–S42. doi: 10.1002/ana.410370705. [DOI] [PubMed] [Google Scholar]
- 113.Quarles RH, Weiss MD. Autoantibodies associated with peripheral neuropathy. Muscle Nerve. 1999;22(7):800–822. doi: 10.1002/(sici)1097-4598(199907)22:7<800::aid-mus2>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- 114.Willison HJ, Yuki N. Peripheral neuropathies and anti-glycolipid antibodies. Brain. 2002;125(Pt 12):2591–2625. doi: 10.1093/brain/awf272. [DOI] [PubMed] [Google Scholar]
- 115.Carpo M, Meucci N, Allaria S, et al. Anti-sulfatide IgM antibodies in peripheral neuropathy. J. Neurol. Sci. 2000;176(2):144–150. doi: 10.1016/s0022-510x(00)00342-7. [DOI] [PubMed] [Google Scholar]
- 116.Dabby R, Weimer LH, Hays AP, Olarte M, Latov N. Antisulfatide antibodies in neuropathy: clinical and electrophysiologic correlates. Neurology. 2000;54(7):1448–1452. doi: 10.1212/wnl.54.7.1448. [DOI] [PubMed] [Google Scholar]
- 117.Ferrari S, Morbin M, Nobile-Orazio E, et al. Antisulfatide polyneuropathy: antibody-mediated complement attack on peripheral myelin. Acta Neuropathol. 1998;96(6):569–574. doi: 10.1007/s004010050937. [DOI] [PubMed] [Google Scholar]
- 118.Quattrini A, Corbo M, Dhaliwal SK, et al. Anti-sulfatide antibodies in neurological disease: binding to rat dorsal root ganglia neurons. J. Neurol. Sci. 1992;112(1–2):152–159. doi: 10.1016/0022-510x(92)90145-b. [DOI] [PubMed] [Google Scholar]
- 119.Chassande B, Leger JM, Younes-Chennoufi AB, et al. Peripheral neuropathy associated with IgM monoclonal gammopathy: correlations between M-protein antibody activity and clinical/ electrophysiological features in 40 cases. Muscle Nerve. 1998;21(1):55–62. doi: 10.1002/(sici)1097-4598(199801)21:1<55::aid-mus8>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- 120.Latov N. Antibodies to glycoconjugates in neuropathy and motor neuron disease. Prog. Brain Res. 1994;101:295–303. doi: 10.1016/s0079-6123(08)61957-3. [DOI] [PubMed] [Google Scholar]
- 121.Weiss MD, Dalakas MC, Lauter CJ, Willison HJ, Quarles RH. Variability in the binding of anti-MAG and anti-SGPG antibodies to target antigens in demyelinating neuropathy and IgM paraproteinemia. J. Neuroimmunol. 1999;95(1–2):174–184. doi: 10.1016/s0165-5728(98)00247-1. [DOI] [PubMed] [Google Scholar]
- 122.Kaida K, Kusunoki S. Immune-mediated neuropathy and anti-glycolipid antibodies. Brain Nerve. 2013;65(4):413–423. [PubMed] [Google Scholar]
- 123.Gorio A, Vitadello M. Ganglioside prevention of neuronal functional decay. Prog. Brain Res. 1987;71:203–208. doi: 10.1016/s0079-6123(08)61825-7. [DOI] [PubMed] [Google Scholar]
- 124.Ledeen RW. Biology of gangliosides: neuritogenic and neuronotrophic properties. J. Neurosci. Res. 1984;12(2–3):147–159. doi: 10.1002/jnr.490120204. [DOI] [PubMed] [Google Scholar]
- 125.Bradley WG, Badger GJ, Tandan R, et al. Double-blind controlled trials of Cronassial in chronic neuromuscular diseases and ataxia. Neurology. 1988;38(11):1731–1739. doi: 10.1212/wnl.38.11.1731. [DOI] [PubMed] [Google Scholar]
- 126.Horowitz SH. Ganglioside (Cronassial) therapy in diabetic neuropathy. Adv. Exp. Med. Biol. 1984;174:593–600. doi: 10.1007/978-1-4684-1200-0_50. [DOI] [PubMed] [Google Scholar]
- 127.Ledeen RW, Oderfeld-Nowak B, Brosnan CF, Cervone A. Gangliosides offer partial protection in experimental allergic neuritis. Ann. Neurol. 1990;27(Suppl):S69–S74. doi: 10.1002/ana.410270718. [DOI] [PubMed] [Google Scholar]
- 128.Oderfeld-Nowak B, Brosnan C, Cervone A, Oderfeld J, Ledeen RW. Gangliosides improve the outcome of experimental allergic neuritis (EAN) Acta Neurobiol. Exp. (Wars) 1990;50(4–5):495–504. [PubMed] [Google Scholar]
- 129.Ponzin D, Menegus AM, Kirschner G, Nunzi MG, Fiori MG, Raine CS. Effects of gangliosides on the expression of autoimmune demyelination in the peripheral nervous system. Ann. Neurol. 1991;30(5):678–685. doi: 10.1002/ana.410300508. [DOI] [PubMed] [Google Scholar]
- 130.Zielasek J, Kolb H. Ganglioside therapy of type I diabetes: enhancement of hyperglycemia in the low dose streptozotocin model. Life Sci. 1992;51(1):49–52. doi: 10.1016/0024-3205(92)90217-d. [DOI] [PubMed] [Google Scholar]
- 131.Dasgupta S, Li D, Yu RK. Lack of apparent neurological abnormalities in rabbits sensitized by gangliosides. Neurochem. Res. 2004;29(11):2147–2152. doi: 10.1007/s11064-004-6888-7. [DOI] [PubMed] [Google Scholar]
- 132.Ilyas AA, Chen ZW. Lewis rats immunized with GM1 ganglioside do not develop peripheral neuropathy. J. Neuroimmunol. 2007;188(1–2):34–38. doi: 10.1016/j.jneuroim.2007.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Lopez PH, Villa AM, Sica RE, Nores GA. High affinity as a disease determinant factor in anti-GM(1) antibodies: comparative characterization of experimentally induced vs disease-associated antibodies. J. Neuroimmunol. 2002;128(1–2):69–76. doi: 10.1016/s0165-5728(02)00139-x. [DOI] [PubMed] [Google Scholar]
- 134.Hadden RD, Gregson NA, Gold R, Willison HJ, Hughes RA. Guillain-Barre syndrome serum and anti-Campylobacter antibody do not exacerbate experimental autoimmune neuritis. J. Neuroimmunol. 2001;119(2):306–316. doi: 10.1016/s0165-5728(01)00390-3. [DOI] [PubMed] [Google Scholar]
- 135.Sheikh KA, Zhang G, Gong Y, Schnaar RL, Griffin JW. An anti-ganglioside antibody-secreting hybridoma induces neuropathy in mice. Ann. Neurol. 2004;56(2):228–239. doi: 10.1002/ana.20173. [DOI] [PubMed] [Google Scholar]
- 136.Yuki N, Susuki K, Koga M, et al. Carbohydrate mimicry between human ganglioside GM1 and Campylobacter jejuni lipooligosaccharide causes Guillain-Barre syndrome. Proc. Natl Acad. Sci. USA. 2004;101(31):11404–11409. doi: 10.1073/pnas.0402391101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Rojas-Garcia R, Gallardo E, Illa I. Paraproteinemic neuropathies. Presse Med. 2013;42(6 Pt 2):e225–e234. doi: 10.1016/j.lpm.2013.02.329. [DOI] [PubMed] [Google Scholar]
- 138.Dyck PJ, Low PA, Windebank AJ, et al. Plasma exchange in polyneuropathy associated with monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 1991;325(21):1482–1486. doi: 10.1056/NEJM199111213252105. [DOI] [PubMed] [Google Scholar]
- 139.Nobile-Orazio E, Meucci N, Baldini L, Di Troia A, Scarlato G. Long-term prognosis of neuropathy associated with anti-MAG IgM M-proteins and its relationship to immune therapies. Brain. 2000;123(Pt 4):710–717. doi: 10.1093/brain/123.4.710. [DOI] [PubMed] [Google Scholar]
- 140.Mygland A, Monstad P. Chronic acquired demyelinating symmetric polyneuropathy classified by pattern of weakness. Arch. Neurol. 2003;60(2):260–264. doi: 10.1001/archneur.60.2.260. [DOI] [PubMed] [Google Scholar]