Abstract
The purpose of this study was to compare the clinical efficacy and safety of vancomycin to those of teicoplanin for the treatment of adult patients with health care-associated methicillin-resistant Staphylococcus aureus (HA-MRSA) bacteremia. A multicenter observational study was prospectively conducted in 15 teaching hospitals in Korea between February 2010 and July 2011. Adult patients (≥18 years old) with HA-MRSA bacteremia who were initially treated with vancomycin (VAN) (n = 134) or teicoplanin (TEC) (n = 56) were enrolled. Clinical and microbiological responses and drug-related adverse events were compared between the two treatment groups using univariate and multivariate logistic regression analyses. The vancomycin and teicoplanin MICs were determined by Etest. The MRSA-related mortality, duration of fever, and duration of MRSA bacteremia in the treatment groups were not significantly different. There was no significant difference in the occurrence of drug-related adverse events. Among the 190 MRSA isolates, the VAN MICs ranged from 0.5 to 2 μg/ml (MIC50 and MIC90, 1.5 μg/ml), and the TEC MIC ranged from 0.5 to 8 μg/ml (MIC50, 3 μg/ml; MIC90, 6 μg/ml). In multivariate analyses, the antibiotic type (vancomycin or teicoplanin) was not associated with treatment outcomes. This study indicates that teicoplanin is an effective and safe alternative to vancomycin for the treatment of HA-MRSA bacteremia.
INTRODUCTION
Nosocomial bloodstream infections represent a major clinical challenge in many health care institutions worldwide, despite laborious and costly infection control efforts. Health care-associated methicillin-resistant Staphylococcus aureus (HA-MRSA) bacteremia has imposed a distinctly high burden on medical expenses and has caused considerable morbidity and mortality (1, 2).
Vancomycin (VAN) has widely been used for the treatment of MRSA infection over the past decades. Increasingly, however, therapeutic failures with VAN have been reported (3). There is also growing evidence of bacteremia caused by MRSA isolates with an increased VAN MIC (4, 5). The VAN therapeutic monitoring guidelines in 2009 recommended more aggressive VAN dosing schemes, targeting VAN serum trough concentrations of 15 to 20 mg/liter for MRSA bacteremia (6). Similarly, optimizing the pharmacokinetics of VAN to achieve an area under the curve (AUC)/MIC ratio of ≥211 has been shown to predict more favorable treatment outcomes in cases of MRSA-associated complicated bacteremia (7). However, if the MRSA strains' MIC is ≥2 μg/ml, conventional intermittent dosing might not achieve this ratio. Rather, it may increase nephrotoxicity (8).
Teicoplanin (TEC) is a glycopeptide antibiotic with an antibacterial spectrum similar to that of VAN but is less toxic at daily doses of less than 800 mg (9, 10). It has a long half-life (45 to 70 h), permitting once-daily dosing (11), and may enhance the intracellular killing of bacteria by phagocytes (12). TEC is commonly used for MRSA infections in Europe, while its use has not yet been approved in the United States. TEC has been used as an alternative agent for MRSA infections; however, there is a limited number of studies that have evaluated the clinical efficacy of TEC in patients with HA-MRSA bacteremia (13, 14).
The purpose of this prospective observational study was to compare the clinical efficacy and safety of VAN to those of TEC for the treatment of adult patients with HA-MRSA bacteremia.
MATERIALS AND METHODS
Study design and patients.
A prospective, multicenter observational study was conducted in 15 teaching hospitals in the Republic of Korea over an 18-month period from February 2010 to July 2011. The subjects comprised hospitalized adult patients (≥18 years) with HA-MRSA bacteremia who were initially treated with VAN (n = 134) or TEC (n = 56) and who were followed until death or hospital discharge. Only the first episode of HA-MRSA bacteremia and the first blood isolate of MRSA per patient that was susceptible to both VAN and TEC were included for analysis. Patients with polymicrobial bacteremia were excluded in order for this study to evaluate the impact of antibiotic therapy for MRSA bacteremia specifically.
A loading dose of VAN (1 g every 12 h) or TEC (400 mg every 12 h) was administered for an initial 24 h or 36 h, respectively, and then followed by daily maintenance doses of each drug that were adjusted to the patient's renal function, if needed (15). In the 11 participating hospitals (73.3%) that ran the therapeutic drug monitoring (TDM) practices for VAN, the TDM-guided VAN dosing was performed, targeting serum trough levels between 15 and 20 μg/ml. None of the participating hospitals ran the TDM for TEC. During the study period, there were no other standardized interventions for the management of MRSA bacteremia, and physicians treated the patients according to routine medical practice.
The study protocol was approved prior to study initiation by the institutional review boards at each participating hospital. As this observational study required no deviation from routine medical practice, the boards waived the need for informed consent.
Definitions.
MRSA bacteremia was considered present if one or more blood cultures had positive results and if the clinical signs and course were consistent with MRSA infection (16).
The primary source of infection, based on the organs affected, was classified as one of the following: lower respiratory tract, intra-abdominal area, genitourinary tract, skin and soft tissue, bone and joint, central nervous system, and catheter. The origin of infection was considered unknown in cases of positive blood cultures without primary infection at another body site (16).
MRSA bacteremia was categorized epidemiologically as health care associated or nosocomial. Community-onset MRSA bacteremia within ≤48 h of hospital admission was considered health care associated if, during the preceding 12 months, the patient had any of the following: admission to other hospitals or health care facilities for more than 2 days, surgery, dialysis, specialized home care, care received at day hospitals, or permanent indwelling catheters. Patients defined as having community-acquired infections were excluded from this study. Infections occurring in patients after 48 h of hospital admission were considered nosocomial.
The duration of bacteremia after VAN or TEC treatment was calculated as the number of days from the start of MRSA treatment to the day the first negative blood culture was drawn. Sepsis, severe sepsis, and septic shock were defined according to the standard criteria (17). The community-acquired phenotype for the MRSA isolates was defined as being susceptible to clindamycin, erythromycin, and ciprofloxacin (18, 19).
The primary endpoint was clinical failure, defined as a composite of mortality attributable to MRSA bacteremia, microbiological failure, and/or persistent fever, except drug fever. Mortality attributable to MRSA bacteremia was defined as positive blood cultures for MRSA, persistent fever, and no other definite causes of death. Microbiological failure was defined as positive blood cultures for MRSA ≥7 days from the index culture under VAN or TEC therapy. Persistent fever was defined as ≥38.0°C for ≥7 days after the commencement of VAN or TEC treatment.
Variables.
Physicians or research coordinators of the participating hospitals entered the clinical data for each patient into a standardized web-based case report form. An infectious disease doctor at the coordinating center checked the entered data and supported the study sites by sending queries throughout the study period. The parameters collected for this analysis included demographic characteristics, comorbid medical conditions, including Charlson's comorbidity index (20), factors predisposing to infections, primary source of MRSA bacteremia, acute physiology and chronic health evaluation II (APACHE II) score (21) or Pitt's bacteremia score (22) at the onset of MRSA bacteremia, diagnosis of severe sepsis or septic shock, hospital mortality, and microbiological data.
Microbiological tests.
Bacterial identification and antibiotic susceptibility were performed at each study site using a Vitek II (bioMérieux, Hazelwood, MO) or MicroScan Pos Combo panel type 6 system (Baxter Diagnostics, West Sacramento, CA). All MRSA isolates from participating hospitals were sent to the coordinating center. All isolates received were immediately stored at −70°C until August 2012, when microbiologic tests were performed all at once. The VAN and TEC MICs for all 190 MRSA isolates were further determined by the Etest (bioMérieux, Marcy l'Etoile, France) at the coordinating center according to the manufacturer's instructions.
Statistical analysis.
For comparisons between groups of continuous independent variables that were normally distributed, the two-sample Student's t test was used. For comparisons of continuous independent variables that were not normally distributed, the Mann-Whitney U test was used. Summaries of the continuous variables were expressed as medians and interquartile ranges (IQR). Independent categorical variables were described using count (proportion), and comparisons between groups were made using the Pearson's chi-squared test or Fisher's exact test.
In the univariate analysis, the VAN and TEC MICs were evaluated as continuous variables as well as categorical variables. The cutoff values of the VAN MICs and the TEC MICs were determined with an analysis using the chi-squared automatic interaction detector (CHAID) decision tree algorithm, to predict treatment outcome in the respective treatment group. The cutoff values of the VAN MICs and the TEC MICs drawn from the CHAID algorithm were 1.5 μg/ml and 4.0 μg/ml, respectively.
Multivariate logistic regression analyses using the backward stepwise variable selection based on the LR statistic were used to examine the impact of multiple independent predictors on the clinical failure as a dependent variable. Trauma, renal diseases, hepatic diseases, pneumonia, Pitt's bacteremia score, C-reactive protein, acute renal injury, duration of fever or bacteremia after VAN or TEC treatment, and antibiotic type were evaluated as independent variables for multivariable logistic regression analysis if such independent variables were predictors of clinical failure at the 10% significance level. Hosmer-Lemeshow goodness-of-fit tests were performed to evaluate the models. Internal accuracy obtained by leave-one-out cross-validation was used to evaluate the performance of a predictive model. All tests were 2-tailed, and a P value <0.05 was considered statistically significant. All of the analyses were performed with IBM SPSS Statistics version 20.0 (IBM Corporation, Armonk, NY), R 2.15.2 (The R Foundation for Statistical Computing, Vienna, Austria), and SAS 9.2 (SAS Institute Inc., Cary, NC).
RESULTS
Patients and clinical characteristics.
During the study period, 426 patients with HA-MRSA bacteremia were enrolled from the participating hospitals. Patients who were given antibiotics with no activity against MRSA isolates (n = 81) and 49 patients from whom MRSA isolates were not collected were excluded from the analysis. Patients who initially received other antibiotics before VAN or TEC (n = 96) and who received VAN or TEC for <3 days (n = 10) were also excluded. Eventually, 190 patients with HA-MRSA bacteremia who were initially treated with VAN (n = 134) or TEC (n = 56) for ≥3 days were included in this study.
The demographic and baseline characteristics of the 190 patients are listed in Table 1. Of these, 158 patients (83.2%) had nosocomial infections and 128 (67.4%) were male. The median age was 66 years (IQR, 51 to 74 years). The univariate analyses determined that there were no significant differences in sex, age, and category of infection between the VAN and TEC treatment groups (Table 1).
TABLE 1.
Demographic and baseline characteristics of 190 patients with MRSA bacteremia according to treatment group and outcomea
| Variable | All (n = 190) | Treatment group |
Treatment outcome |
||||
|---|---|---|---|---|---|---|---|
| Vancomycin (n = 134) | Teicoplanin (n = 56) | P | Success (n = 112, 58.9%) | Failure (n = 78, 41.1%) | P | ||
| No. (%) receiving vancomycin | 134 (70.5) | 134 (100) | 0 | <0.001 | 85 (75.9) | 49 (62.8) | 0.052 |
| No. (%) of males | 128 (67.4) | 92 (68.7) | 36 (64.3) | 0.558 | 78 (69.6) | 50 (64.1) | 0.423 |
| Median age, yrs (IQR) | 66 (51–73) | 64.5 (51–73) | 68 (51.5–74) | 0.521 | 65 (52–72) | 67.5 (50–76) | 0.508 |
| No. (%) with time of bacteremia | |||||||
| ≤48 h | 42 (25.6) | 35 (26.1) | 7 (23.3) | 0.752 | 28 (26.9) | 14 (23.3) | 0.612 |
| >48 h | 122 (74.4) | 99 (73.9) | 23 (76.7) | 76 (73.1) | 46 (76.7) | ||
| No. (%) with category of infection | |||||||
| Health care associated | 32 (16.8) | 23 (17.2) | 9 (16.1) | 0.854 | 20 (17.9) | 12 (15.4) | 0.654 |
| Nosocomial | 158 (83.2) | 111 (82.8) | 47 (83.9) | 92 (82.1) | 66 (84.6) | ||
| Comorbid illness | |||||||
| No. (%) with cardiovascular disease | 97 (51.1) | 65 (48.5) | 32 (57.1) | 0.278 | 54 (48.2) | 43 (55.1) | 0.348 |
| No. (%) with central nervous system disease | 47 (24.7) | 33 (24.6) | 14 (25.0) | 0.957 | 31 (27.7) | 16 (20.5) | 0.260 |
| No. (%) with malignancy | 57 (30.0) | 47 (35.1) | 10 (17.9) | 0.018 | 36 (32.1) | 21 (26.9) | 0.440 |
| No. (%) with trauma | 18 (9.5) | 12 (9.0) | 6 (10.7) | 0.706 | 5 (4.5) | 13 (16.7) | 0.005 |
| No. (%) with renal disease | 39 (20.5) | 28 (20.9) | 11 (19.6) | 0.845 | 18 (16.1) | 21 (26.9) | 0.068 |
| No. (%) with hepatic disease | 20 (10.5) | 17 (12.7) | 3 (5.4) | 0.133 | 16 (14.3) | 4 (5.1) | 0.043 |
| No. (%) with respiratory disease | 23 (12.1) | 16 (11.9) | 7 (12.5) | 0.914 | 10 (8.9) | 13 (16.7) | 0.108 |
| No. (%) with solid organ or bone marrow transplant | 4 (2.1) | 3 (2.2) | 1 (1.8) | 1.000 | 3 (2.7) | 1 (1.3) | 0.645 |
| No. (%) with metabolic disease | 66 (34.7) | 48 (35.8) | 18 (32.1) | 0.627 | 34 (30.4) | 32 (41.0) | 0.129 |
| No. (%) with HIV infection | 2 (1.1) | 2 (1.5) | 0 | 1.000 | 1 (0.9) | 1 (1.3) | 1.000 |
| No. (%) with hematologic disease | 30 (15.8) | 26 (19.4) | 4 (7.1) | 0.035 | 19 (17.0) | 11 (14.1) | 0.595 |
| No. (%) with gastrointestinal bleeding | 8 (4.2) | 5 (3.7) | 3 (5.4) | 0.695 | 3 (2.7) | 5 (6.4) | 0.276 |
| Charlson's comorbidity index, median (IQR) | 2 (1–4) | 3 (1–4) | 1 (0–3) | 0.008 | 2 (0–4) | 2 (1–4) | 0.693 |
| No. (%) with primary source of bacteremia | |||||||
| Catheter-related infection | 91 (47.9) | 70 (52.2) | 21 (37.5) | 0.064 | 59 (52.7) | 32 (41.0) | 0.114 |
| Pneumonia | 28 (14.7) | 19 (14.2) | 9 (16.1) | 0.737 | 10 (8.9) | 18 (23.1) | 0.007 |
| Surgical wound infection | 19 (10.0) | 13 (10.4) | 5 (8.9) | 0.750 | 13 (11.6) | 6 (7.7) | 0.376 |
| Bone and joint infection | 11 (5.8) | 10 (7.5) | 1 (1.8) | 0.179 | 5 (4.5) | 2 (2.6) | 0.702 |
| Intra-abdominal infection | 10 (5.3) | 3 (2.2) | 7 (12.5) | 0.008 | 3 (2.7) | 4 (5.1) | 0.448 |
| Urinary tract infection | 7 (3.7) | 3 (2.2) | 4 (7.1) | 0.198 | 7 (6.2) | 4 (5.1) | 1.000 |
| Skin and soft tissue infection | 7 (3.7) | 5 (3.7) | 2 (3.6) | 1.000 | 1 (0.9) | 3 (3.8) | 0.307 |
| Cardiovascular infection | 4 (2.1) | 2 (1.5) | 2 (3.6) | 0.583 | 5 (4.5) | 5 (6.4) | 0.743 |
| Central nervous system infection | 1 (0.5) | 1 (0.7) | 0 | 1.000 | 0 | 1 (1.3) | 0.411 |
| Head and neck infection | 1 (0.5) | 0 | 1 (1.8) | 0.295 | 1 (0.9) | 0 | 1.000 |
| Unknown | 11 (5.8) | 7 (5.2) | 4 (7.1) | 0.734 | 8 (7.1) | 3 (3.8) | 0.530 |
| Clinical severity at the onset of MRSA bacteremia | |||||||
| No. (%) with fever (≥38.0°C) | 142 (74.7) | 104 (77.6) | 38 (67.9) | 0.158 | 90 (80.4) | 52 (66.7) | 0.033 |
| No. (%) with SIRS | 189 (99.5) | 133 (99.3) | 56 (100) | 1.000 | 111 (99.1) | 78 (100) | 1.000 |
| No. (%) with development of severe sepsis or septic shock | 66 (34.7) | 43 (32.1) | 23 (41.1) | 0.236 | 31 (27.7) | 35 (44.9) | 0.014 |
| Pitt's bacteremia score [median (IQR)] | 1 (0–3) | 1 (0–3) | 1 (0–4) | 0.542 | 1 (0–3) | 2 (0–3) | 0.286 |
| APACHE II score [median (IQR)] | 17 (12–21) | 15 (12–21) | 19 (13–23) | 0.211 | 17 (11–21) | 17 (13–21) | 0.667 |
| No. (%) with APACHE II score of ≥20 | 42 (33.3) | 23 (32.9) | 19 (33.9) | 0.899 | 22 (30.6) | 20 (37.0) | 0.445 |
| No. (%) with complicated condition | |||||||
| Foreign body retention | 8 (4.2) | 7 (5.2) | 1 (1.8) | 0.440 | 7 (6.2) | 1 (1.3) | 0.144 |
| Infective endocarditis | 5 (2.6) | 4 (3.0) | 1 (1.8) | 1.000 | 3 (2.7) | 2 (2.6) | 1.000 |
| Metastatic infectionsb | 11 (5.8) | 9 (6.7) | 2 (3.6) | 0.512 | 8 (7.1) | 3 (3.8) | 0.530 |
| Laboratory findings at the onset of MRSA bacteremia | |||||||
| C-reactive protein (mg/liter) | 10.8 (4.8–22.1) | 11.1 (5.2–20.2) | 9.9 (4.1–23.0) | 0.914 | 9.2 (3.9–17.4) | 14.3 (6.8–24.4) | 0.039 |
| No. (%) with hematocrit of <30% | 98 (51.6) | 66 (49.3) | 32 (57.1) | 0.321 | 53 (47.3) | 45 (57.7) | 0.159 |
| No. (%) with platelet count of <100,000/μl | 41 (21.6) | 30 (22.4) | 11 (19.6) | 0.675 | 22 (19.6) | 19 (24.4) | 0.437 |
| No. (%) with albumin of <3.0 g/dl | 84 (44.2) | 56 (41.8) | 28 (50.0) | 0.299 | 49 (43.8) | 35 (44.9) | 0.878 |
| No. (%) with total bilirubin of ≥2.0 mg/dl | 37 (19.5) | 27 (20.1) | 10 (17.9) | 0.716 | 20 (17.9) | 17 (21.8) | 0.500 |
| No. (%) with creatinine of ≥2.0 mg/dl | 52 (27.4) | 37 (27.6) | 15 (26.8) | 0.907 | 33 (29.5) | 19 (24.4) | 0.272 |
| No. (%) with serum sodium of <130.0 mmol/liter | 17 (8.9) | 15 (11.2) | 2 (3.6) | 0.093 | 13 (11.6) | 4 (5.1) | 0.124 |
HIV, human immunodeficiency virus; SIRS, systemic inflammatory response syndrome; IQR, interquartile range; APACHE II, acute physiology and chronic health evaluation II; SD, standard deviation; MRSA, methicillin-resistant Staphylococcus aureus.
Sites of metastatic infections include bones and joints, the epidural space, intervertebral disks, heart valves, and intra-abdominal organs.
The most common source of MRSA bacteremia was catheter-related infections (47.9%), followed by pneumonia (14.7%), surgical wounds (10.0%), and bone and joint infections (5.8%). The univariate analyses revealed no significant differences in the primary source of infection between the 2 treatment groups, except for intra-abdominal infections (Table 1).
The median Charlson comorbidity index was 2 (IQR, 1 to 4), and univariate analyses determined that the VAN group had a significantly higher Charlson comorbidity index than the TEC group. In particular, underlying malignancy and hematologic diseases were significantly more common in the VAN group than the TEC group (Table 1). Sixty-six patients (34.7%) had severe sepsis or septic shock, and the median APACHE II score at the onset of HA-MRSA bacteremia was 17 (IQR, 12 to 21). There was no significant difference in the APACHE II score of HA-MRSA bacteremia between the two treatment groups (Table 1).
Microbiological characteristics.
All 190 MRSA isolates underwent microbiological analysis. The VAN MIC range was 0.5 to 2 μg/ml, and the VAN MIC50 and MIC90 were both 1.5 μg/ml. The TEC MIC range was 0.5 to 8 μg/ml, and the TEC MIC50 and MIC90 were 3 μg/ml and 6 μg/ml, respectively. Distribution of the VAN and TEC MICs and the antibiotic phenotype among the MRSA isolates, categorized by treatment group and treatment outcome, are shown in Table 2. In a total of 190 patients analyzed, the VAN or TEC MICs were not associated with clinical failure.
TABLE 2.
Microbiological characteristics of 190 patients with MRSA bacteremia according to treatment group and outcome
| Variable | All (n = 190) | Treatment group |
Treatment outcome |
||||
|---|---|---|---|---|---|---|---|
| Vancomycin (n = 134) | Teicoplanin (n = 56) | P | Success (n = 112, 58.9%) | Failure (n = 78, 41.1%) | P | ||
| MIC, μg/ml [median (IQR)] | |||||||
| Vancomycin | 1.5 (1.0–1.5) | 1.5 (1.0–1.5) | 1.0 (1.0–1.5) | <0.001 | 1.5 (1.0–1.5) | 1.5 (1.0–1.5) | 0.324 |
| Teicoplanin | 3.0 (2.0–4.0) | 3.0 (2.0–4.0) | 3.0 (2.0–3.0) | 0.039 | 3.0 (2.0–4.0) | 3.0 (2.0–4.0) | 0.476 |
| No. (%) with vancomycin MIC of ≥1.5 μg/ml | 106 (55.8) | 87 (64.9) | 19 (33.9) | 0.001 | 67 (59.8) | 39 (50.0) | 0.180 |
| No. (%) with teicoplanin MIC of MIC ≥ 4 μg/ml | 64 (33.7) | 53 (39.6) | 11 (19.6) | 0.008 | 42 (37.5) | 22 (28.2) | 0.182 |
| No. (%) with CA-MRSAa phenotype (18, 19) | 31 (16.3) | 25 (18.7) | 6 (10.7) | 0.101 | 21 (18.8) | 10 (12.8) | 0.199 |
CA-MRSA, community-acquired methicillin-resistant Staphylococcus aureus.
When the influences of the VAN MICs on clinical outcomes in the VAN-treated group were evaluated, a VAN MIC of ≥1.5 μg/ml was the significant risk factor for in-hospital mortality (VAN MIC, <1.5 μg/ml versus ≥1.5 μg/ml; 19.1% [9/47] versus 41.4% [36/87]; P = 0.009) but not for clinical failure (40.4% [19/47] versus 34.5% [30/87]; P = 0.495). In the TEC-treated group, a TEC MIC of ≥4 μg/ml in the TEC group was not significantly associated with treatment failure (TEC MIC, <4 μg/ml versus ≥4 μg/ml; 53.3% [24/45] versus 45.5% [5/11]; P = 0.639) or in-hospital mortality (40.0% [18/45] versus 27.3% [3/11]; P = 0.508).
The proportion of MRSA isolates with phenotypic expression of community-acquired MRSA was 16.3% (31/190) and was not significantly different between the 2 treatment groups (Table 2).
Treatment outcomes.
The overall all-cause in-hospital mortality and MRSA-related mortality were 34.7% (66/190) and 14.7% (28/190), respectively. There were no significant differences in the all-cause in-hospital mortality and MRSA-related mortality between the treatment groups. After the commencement of VAN or TEC therapy, a significant difference was not exhibited for the duration of fever and MRSA bacteremia between the treatment groups (Table 3). The median durations of VAN and TEC treatment showed no significant differences (median, [IQR], 14 days [IQR, 9 to 23 days] versus 13 days [IQR, 8 to 21 days]; P = 0.239).
TABLE 3.
Antibiotic treatment outcomes and related adverse events for 190 patients with health care-associated MRSA bacteremiaa
| Variable | Treatment group |
Treatment outcome |
|||||
|---|---|---|---|---|---|---|---|
| All (n = 190) | Vancomycin (n = 134) | Teicoplanin (n = 56) | P | Success (n = 112, 58.9%) | Failure (n = 78, 41.1%) | P | |
| Antibiotic treatment | |||||||
| No. (%) with interval from culture to VAN or TEC treatment of ≥48 h | 64 (41.8) | 42 (39.3) | 22 (47.8) | 0.324 | 38 (40.9) | 26 (43.3) | 0.762 |
| Duration of VAN or TEC treatment (days), median (IQR) | 14 (3–23) | 14 (9–23) | 13 (8–21) | 0.239 | 14 (9–21) | 13 (8–24) | 0.995 |
| Clinical response | |||||||
| Duration of bacteremia after VAN or TEC treatment (days), median (IQR) | 1 (0–2) | 1 (0–2) | 0 (0–1) | 0.254 | 0 (1–0) | 1 (0–7) | <0.001 |
| No. (%) with bacteremia ≥7 days after VAN or TEC treatment | 20 (10.9) | 15 (11.7) | 5 (9.1) | 0.601 | 0 | 20 (25.6) | <0.001 |
| Duration of fever after VAN or TEC treatment (days), median (IQR) | 4 (2–7) | 4 (2–6) | 5 (2–11) | 0.084 | 3 (2–5) | 8 (4–17) | <0.001 |
| No. (%) with fever ≥7 days after VAN or TEC treatment | 51 (29.5) | 29 (24.0) | 22 (42.3) | 0.015 | 0 | 51 (67.1) | <0.001 |
| No. (%) with drug-related adverse events during treatment) | 36 (18.9) | 28 (20.9) | 8 (14.3) | 0.289 | 19 (17.0) | 17 (21.8) | 0.403 |
| No. (%) with acute renal injury | 17 (8.9) | 14 (10.4) | 3 (5.4) | 0.262 | 6 (5.4) | 11 (14.1) | 0.038 |
| No. (%) with hepatotoxicity | 3 (1.6) | 1 (0.7) | 2 (3.6) | 0.208 | 3 (2.7) | 0 | 0.270 |
| No. (%) with bone marrow toxicity | 10 (5.3) | 8 (6.0) | 2 (3.6) | 0.726 | 8 (7.1) | 2 (2.6) | 0.202 |
| No. (%) with fever | 8 (4.2) | 6 (4.5) | 2 (3.6) | 1.000 | 5 (4.5) | 3 (3.8) | 1.000 |
| No. (%) with rash | 1 (0.5) | 1 (0.7) | 0 | 1.000 | 0 | 1 (1.3) | 0.411 |
| No. (%) with change of initial antibiotics | 36 (18.9) | 20 (14.9) | 16 (28.6) | 0.029 | 17 (15.2) | 19 (24.4) | 0.112 |
| Outcome | |||||||
| No. of days of hospital stay after bacteremia, median (IQR) | 23 (11–49) | 23 (12–49) | 25 (9–52) | 0.706 | 23 (10–47) | 24 (12–53) | 0.867 |
| No. (%) with in-hospital mortality | 66 (34.7) | 45 (33.6) | 21 (37.5) | 0.605 | 27 (24.1) | 39 (50.0) | <0.001 |
| No. (%) with mortality attributable to MRSA | 28 (14.7) | 18 (13.4) | 10 (17.4) | 0.433 | 0 | 28 (35.9) | <0.001 |
IQR, interquartile range; VAN, vancomycin; TEC, teicoplanin; MRSA, methicillin-resistant Staphylococcus aureus.
In total, 36 patients (18.9%) received alternative drugs due to poor clinical response (n = 23), drug-related adverse events (n = 12), or other reasons (n = 4). In the VAN group, VAN was switched with alternative antibiotics in 20 patients (14.9%): TEC (n = 11), linezolid (n = 6), tigecycline (n = 2), or levofloxacin plus rifampin (n = 1). In the TEC group, 16 patients (28.6%) received alternative antibiotics: VAN (n = 9), linezolid (n = 4), an aminoglycoside (n = 1), clindamycin (n = 1), or trimethoprim-sulfamethoxazole plus rifampin (n = 1).
The median duration of VAN in alternation from VAN to TEC (n = 11) and vice versa (n = 9) was 9 days (IQR, 6 to 17 days) and 10 days (IQR, 7 to 13 days), respectively.
There was no significant difference in the occurrence of drug-related adverse events between the 2 treatment groups (20.9% [28/134] versus 14.3% [8/56], P = 0.289) (Table 3). Among the 8 patients who received alternative glycopeptides due to drug-related adverse events, cross-reactivity was not observed between VAN and TEC. One patient with VAN-induced acute kidney injury developed TEC-induced neutropenia.
In the multiple logistic regression modeling, the antibiotic type (VAN or TEC) was not an independent risk factor for clinical failure in the patients with HA-MRSA bacteremia, regardless of variable selection (Table 4). The statistically significant factors associated with clinical failure included Pitt's bacteremia score (odds ratio [OR], 1.51; 95% confidence interval [CI], 1.10 to 2.06), acute renal injury (OR, 15.99; 95% CI, 1.81 to 141.16), duration of fever (OR, 1.77; 95% CI, 1.36 to 2.32), and duration of bacteremia (OR, 1.76; 95% CI, 1.34 to 2.31) (Table 4). The P values for the Hosmer-Lemeshow goodness-of-fit test were greater than 0.05. Hence, there is no significant evidence of lack of fit for any of the final models.
TABLE 4.
Multivariable logistic regression analysis of risk factors associated with clinical failure in the 190 patients with MRSA bacteremiaa
| Independent variable | Multivariate logistic regression analysis without variable selection |
Multivariate logistic regression with backward variable selection based on LR |
||
|---|---|---|---|---|
| OR (95% CI for OR) | P | OR (95% CI for OR) | P | |
| Antibiotic type (vancomycin) | 0.73 (0.18, 2.98) | 0.666 | ||
| Trauma (yes) | 7.38 (0.85, 63.77) | 0.069 | ||
| Renal disease (yes) | 1.62 (0.39, 6.76) | 0.509 | ||
| Hepatic disease (yes) | 1.09 (0.12, 10.21) | 0.939 | ||
| Pneumonia (yes) | 1.94 (0.43, 8.78) | 0.391 | ||
| Pitt's bacteremia score | 1.60 (1.13, 2.26) | 0.008 | 1.51 (1.10, 2.06) | 0.010 |
| C-reactive protein | 1.00 (0.99, 1.02) | 0.740 | ||
| Acute renal injury (yes) | 18.41 (1.76, 192.26) | 0.015 | 15.99 (1.81, 141.16) | 0.013 |
| Duration of fever (days) | 1.78 (1.34, 2.37) | <0.001 | 1.77 (1.36, 2.32) | <0.001 |
| Duration of bacteremia (days) | 1.83 (1.34, 2.48) | <0.001 | 1.76 (1.34, 2.31) | <0.001 |
LR, Logistic regression analysis; OR, odds ratio; 95% CI, 95% confidence interval.
Leave-one-out cross-validation was performed to assess the predictive accuracy of each final model. The AUCs for the clinical failure model were greater than 0.90 for both the raw data set and leave-one-out cross-validation. For this mode, the sensitivity, specificity, positive predictive value, and negative predictive value obtained with an optimal cutoff point were greater than 0.80 (Fig. 1; Table 5).
FIG 1.
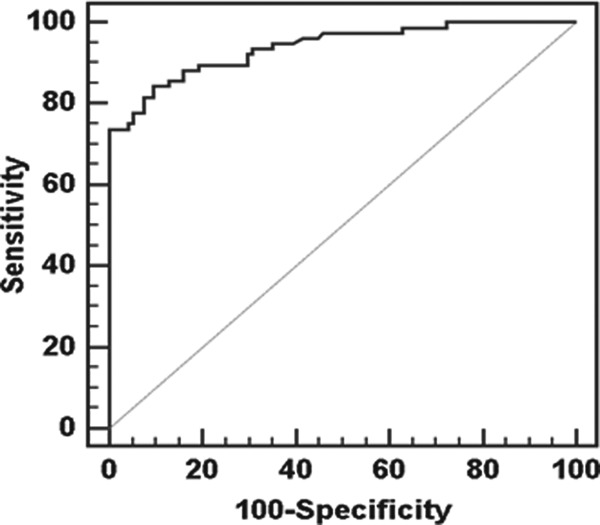
Receiver operating characteristic curve for clinical failure obtained using the predictive probability of multivariate logistic regression model and validation results.
TABLE 5.
Validation results for the clinical failure variablea
| Validation | AUC | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) |
|---|---|---|---|---|---|
| Validation for raw data set | 0.939 (0.903–0.974) | 84.2 (74.0–91.6) | 90.4 (82.6–95.5) | 87.7 (78.1–93.5) | 87.6 (79.1–94.1) |
| Leave-one-out cross-validation | 0.926 (0.876–0.961) | 80.3 (69.5–88.5) | 91.5 (83.9–96.3) | 88.4 (78.4–94.9) | 85.1 (76.6–91.5) |
All values except AUC are optimal values in each final model. AUC, area under the curve; PPV, positive predictive value; NPV, negative predictive value; MRSA, methicillin-resistant Staphylococcus aureus.
DISCUSSION
This multicenter prospective study compared the clinical efficacy and safety of VAN versus TEC for the treatment of adult patients with HA-MRSA bacteremia in hospital settings where MRSA prevalence was about 70% (23). This study found that TEC has efficacy and safety comparable to those of VAN for the treatment of HA-MRSA bacteremia.
The in-hospital mortality rate of HA-MRSA bacteremia in the VAN and TEC treatment groups of this study were 33.6% and 37.5%, respectively, which is comparable to the range of 14% to 60% reported previously in other studies (3, 24–26). Based on the Charlson comorbidity index or Pitt's bacteremia score, the clinical severity of the infections of the patients in our study was comparable to that of health care-associated and community-acquired MRSA infections (14, 26). In addition, 4 risk factors for clinical failure of HA-MRSA bacteremia, namely, Pitt's bacteremia score, acute renal injury, duration of fever, and bacteremia, were not different from those reported previously (26–29).
In this study, the type of glycopeptide, i.e., VAN or TEC, was not the risk factor associated with clinical failure. Meta-analysis studies have reported that there were no differences in clinical cure, microbiological cure, and mortality between VAN and TEC treatments for Gram-positive infections, including bacteremia, pneumonia, febrile neutropenia, and skin and soft tissue infections (10, 30, 31). However, studies on the comparative efficacy of VAN versus TEC against MRSA bacteremia (13, 32, 33) are still limited. Liu et al. (33) demonstrated that TEC was as efficacious as VAN in terms of treatment success rate for MRSA bacteremia (TEC group, 85% [17/20], versus VAN group, 75% [15/20]; P = 0.69). On the other hand, Huang and Hsu (32) reported that there was no statistically significant difference in the hospital mortality rate (42% versus 47%) and microbiological failure rate (34% versus 40%) between the VAN group (n = 36) and the TEC group (n = 15) among patients with MRSA infective endocarditis.
In this study, there was no significant difference in the occurrence of drug-related adverse events between the VAN and TEC treatment groups. In meta-analysis studies, the incidence of total drug-related adverse events, including nephrotoxicity and red man syndrome, was lower with TEC (10, 30, 31). This discrepancy might have resulted from the closed TDM of VAN in our study, carried out according to recent clinical practice guidelines (34). On the other hand, TEC was administered as directed in the package insert because TDM of TEC is not routinely available.
In this study, the use of an alternative agent, i.e., switching from VAN to TEC or vice versa, was common in the patients with HA-MRSA bacteremia due to the reimbursement system in Korea. VAN was replaced with TEC as an alternative agent, or vice versa, in 20 (14.9%) and 16 patients (28.6%), respectively. Lin et al. (13) reported no significant difference in 30-day mortality among 3 treatment groups of elderly patients with persistent MRSA bacteremia (VAN versus TEC versus VAN/TEC alternately, 59.6% [65/109] versus 50.0% [7/14] versus 65.5% [19/29]). They also reported that alternation between VAN and TEC treatment was not more effective than either VAN or TEC treatment alone (13). However, the appropriateness of this alternative therapy needs to be evaluated in prospective randomized controlled trials. Antibiotics such as linezolid or daptomycin as promising salvage agents or a novel strategy of combined antibiotic treatment should be considered for better treatment outcomes of HA-MRSA bacteremia (27, 35).
In our study, the adverse cross-reactions between VAN and TEC were not remarkable, although a limited number of cases were evaluated. Previous studies have reported that the alternate use of TEC in cases of VAN intolerance was associated with a high incidence of drug-related adverse events, most notably neutropenia (36, 37). Therefore, the potential cross-reactivity between these 2 glycopeptides remains to be clarified in future studies.
In recent meta-analysis studies, VAN MICs of ≥1.5 μg/ml or ≥2.0 μg/ml are associated with increased mortality as well as clinical failure among patients with MRSA infections (4, 5). In this study, the VAN MICs of ≥1.5 μg/ml from the VAN-treated group and the TEC MICs of ≥4.0 μg/ml from TEC-treated group were more common in the nonsurvivors than the survivors but were not a significant factor for clinical failure. These findings indicate that the threshold VAN or TEC MICs for clinical outcomes might be different among the study populations.
This study has some limitations. First, this prospective study was not designed to include the detailed complications associated with MRSA bacteremia. This may have resulted in a falsely low complication rate. However, catheter-related infections accounted for 47.9% of the HA-MRSA bacteremia cases in this study, which were easily controlled with catheter removal and antibiotic therapy. Thus, the related confounding factors might be minimal. Second, this was not a randomized clinical trial: the doctors chose VAN to initiate treatment due to the reimbursement system in Korea. Therefore, the patients who received TEC therapy may not be representative of the larger population with HA-MRSA bacteremia. Third, individualized dosing regimens of VAN or TEC relative to the MICs were not undertaken in this study. Implementation of the individualized VAN dosing approach targeting an AUC/MIC ratio of 400 μg · h/ml or greater, rather than a trough serum concentration, may lead to improved clinical outcomes in critically ill patients (38). Lastly, other antibiotics switched from VAN or TEC or concomitant antibiotics with VAN or TEC might have influenced the treatment outcome. The clinical effect of these antibiotics were not evaluated owing to the small number of study cases.
In conclusion, this multicenter prospective study indicates that TEC is an effective and safe alternative to VAN for the treatment of HA-MRSA bacteremia. Further studies that take the AUC/MIC ratio of VAN and TEC into account may be required for better clinical outcomes in treating patients with HA-MRSA bacteremia.
ACKNOWLEDGMENTS
This work was supported by a grant (A102065) from the Korean Health 21 R&D project of the Ministry for Health, Welfare and Family Affairs, Republic of Korea.
We have no conflicts of interest.
Footnotes
Published ahead of print 28 October 2013
REFERENCES
- 1.Primo MG, Guilarde AO, Martelli CM, Batista LJ, Turchi MD. 2012. Healthcare-associated Staphylococcus aureus bloodstream infection: length of stay, attributable mortality, and additional direct costs. Braz. J. Infect. Dis. 16:503–509. 10.1016/j.bjid.2012.10.001 [DOI] [PubMed] [Google Scholar]
- 2.Park SY, Son JS, Oh IH, Choi JM, Lee MS. 2011. Clinical impact of methicillin-resistant Staphylococcus aureus bacteremia based on propensity scores. Infection 39:141–147. 10.1007/s15010-011-0100-1 [DOI] [PubMed] [Google Scholar]
- 3.Moise-Broder PA, Sakoulas G, Eliopoulos GM, Schentag JJ, Forrest A, Moellering RC., Jr 2004. Accessory gene regulator group II polymorphism in methicillin-resistant Staphylococcus aureus is predictive of failure of vancomycin therapy. Clin. Infect. Dis. 38:1700–1705. 10.1086/421092 [DOI] [PubMed] [Google Scholar]
- 4.Jacob JT, Diazgranados CA. 2013. High vancomycin minimum inhibitory concentration and clinical outcomes in adults with methicillin-resistant Staphylococcus aureus infections: a meta-analysis. Int. J. Infect. Dis. 17:e93–e100. 10.1016/j.ijid.2012.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van Hal SJ, Lodise TP, Paterson DL. 2012. The clinical significance of vancomycin minimum inhibitory concentration in Staphylococcus aureus infections: a systematic review and meta-analysis. Clin. Infect. Dis. 54:755–771. 10.1093/cid/cir935 [DOI] [PubMed] [Google Scholar]
- 6.Rybak MJ, Lomaestro BM, Rotschafer JC, Moellering RC, Craig WA, Billeter M, Dalovisio JR, Levine DP. 2009. Vancomycin therapeutic guidelines: a summary of consensus recommendations from the infectious diseases Society of America, the American Society of Health-System Pharmacists, and the Society of Infectious Diseases Pharmacists. Clin. Infect. Dis. 49:325–327. 10.1086/600877 [DOI] [PubMed] [Google Scholar]
- 7.Brown J, Brown K, Forrest A. 2012. Vancomycin AUC24/MIC ratio in patients with complicated bacteremia and infective endocarditis due to methicillin-resistant Staphylococcus aureus and its association with attributable mortality during hospitalization. Antimicrob. Agents Chemother. 56:634–638. 10.1128/AAC.05609-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lodise TP, Patel N, Lomaestro BM, Rodvold KA, Drusano GL. 2009. Relationship between initial vancomycin concentration-time profile and nephrotoxicity among hospitalized patients. Clin. Infect. Dis. 49:507–514. 10.1086/600884 [DOI] [PubMed] [Google Scholar]
- 9.Verbist L, Tjandramaga B, Hendrickx B, Van Hecken A, Van Melle P, Verbesselt R, Verhaegen J, De Schepper PJ. 1984. In vitro activity and human pharmacokinetics of teicoplanin. Antimicrob. Agents Chemother. 26:881–886. 10.1128/AAC.26.6.881 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Svetitsky S, Leibovici L, Paul M. 2009. Comparative efficacy and safety of vancomycin versus teicoplanin: systematic review and meta-analysis. Antimicrob. Agents Chemother. 53:4069–4079. 10.1128/AAC.00341-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Williams AH, Gruneberg RN. 1988. Teicoplanin revisited. J. Antimicrob. Chemother. 22:397–401. 10.1093/jac/22.4.397 [DOI] [PubMed] [Google Scholar]
- 12.Pedrera MI, Barriga C, Rodriguez AB. 1995. Intracellular activity of both teicoplanin and vancomycin against Staphylococcus aureus in human neutrophils. Comp. Immunol. Microbiol. Infect. Dis. 18:123–128. 10.1016/0147-9571(95)98853-A [DOI] [PubMed] [Google Scholar]
- 13.Lin SH, Lai CC, Tan CK, Liao WH, Hsueh PR. 2011. Comparative efficacy of vancomycin and teicoplanin in the treatment of hospitalised elderly patients with persistent meticillin-resistant Staphylococcus aureus (MRSA) bacteraemia. Int. J. Antimicrob. Agents 37:179–181. 10.1016/j.ijantimicag.2010.10.018 [DOI] [PubMed] [Google Scholar]
- 14.Chang HJ, Hsu PC, Yang CC, Siu LK, Kuo AJ, Chia JH, Wu TL, Huang CT, Lee MH. 2012. Influence of teicoplanin MICs on treatment outcomes among patients with teicoplanin-treated methicillin-resistant Staphylococcus aureus bacteraemia: a hospital-based retrospective study. J. Antimicrob. Chemother. 67:736–741. 10.1093/jac/dkr531 [DOI] [PubMed] [Google Scholar]
- 15.Gilbert DN, Moellering RC, Jr, Eliopoulos GM. (ed). 2010. The Sanford guide to antimicrobial therapy 2010, 40th ed. Antimicrobial Therapy, Sperryville, VA [Google Scholar]
- 16.Horan TC, Andrus M, Dudeck MA. 2008. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am. J. Infect. Control 36:309–332. 10.1016/j.ajic.2008.03.002 [DOI] [PubMed] [Google Scholar]
- 17.Longo DL, Fauci A, Kasper D, Hauser S, Jameson J, Loscalzo J. 2012. Harrison's principles of internal medicine, 18th ed. McGraw-Hill, New York, NY [Google Scholar]
- 18.O'Brien FG, Lim TT, Chong FN, Coombs GW, Enright MC, Robinson DA, Monk A, Said-Salim B, Kreiswirth BN, Grubb WB. 2004. Diversity among community isolates of methicillin-resistant Staphylococcus aureus in Australia. J. Clin. Microbiol. 42:3185–3190. 10.1128/JCM.42.7.3185-3190.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.David MZ, Glikman D, Crawford SE, Peng J, King KJ, Hostetler MA, Boyle-Vavra S, Daum RS. 2008. What is community-associated methicillin-resistant Staphylococcus aureus? J. Infect. Dis. 197:1235–1243. 10.1086/533502 [DOI] [PubMed] [Google Scholar]
- 20.Charlson M, Szatrowski TP, Peterson J, Gold J. 1994. Validation of a combined comorbidity index. J. Clin. Epidemiol. 47:1245–1251. 10.1016/0895-4356(94)90129-5 [DOI] [PubMed] [Google Scholar]
- 21.Knaus WA, Draper EA, Wagner DP, Zimmerman JE. 1985. APACHE II: a severity of disease classification system. Crit. Care Med. 13:818–829. 10.1097/00003246-198510000-00009 [DOI] [PubMed] [Google Scholar]
- 22.Chow JW, Fine MJ, Shlaes DM, Quinn JP, Hooper DC, Johnson MP, Ramphal R, Wagener MM, Miyashiro DK, Yu VL. 1991. Enterobacter bacteremia: clinical features and emergence of antibiotic resistance during therapy. Ann. Intern. Med. 115:585–590. 10.7326/0003-4819-115-8-585 [DOI] [PubMed] [Google Scholar]
- 23.Lee K, Kim MN, Kim JS, Hong HL, Kang JO, Shin JH, Park YJ, Yong D, Jeong SH, Chong Y. 2011. Further increases in carbapenem-, amikacin-, and fluoroquinolone-resistant isolates of Acinetobacter spp. and P. aeruginosa in Korea: KONSAR study 2009. Yonsei Med. J. 52:793–802. 10.3349/ymj.2011.52.5.793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cosgrove SE, Sakoulas G, Perencevich EN, Schwaber MJ, Karchmer AW, Carmeli Y. 2003. Comparison of mortality associated with methicillin-resistant and methicillin-susceptible Staphylococcus aureus bacteremia: a meta-analysis. Clin. Infect. Dis. 36:53–59. 10.1086/345476 [DOI] [PubMed] [Google Scholar]
- 25.Moore CL, Lu M, Cheema F, Osaki-Kiyan P, Perri MB, Donabedian S, Haque NZ, Zervos MJ. 2011. Prediction of failure in vancomycin-treated methicillin-resistant Staphylococcus aureus bloodstream infection: a clinically useful risk stratification tool. Antimicrob. Agents Chemother. 55:4581–4588. 10.1128/AAC.00115-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hall RG, II, Giuliano CA, Haase KK, Hazlewood KA, Frei CR, Forcade NA, Brouse SD, Bell T, Bedimo RJ, Alvarez CA. 2012. Empiric guideline-recommended weight-based vancomycin dosing and mortality in methicillin-resistant Staphylococcus aureus bacteremia: a retrospective cohort study. BMC Infect. Dis. 12:104. 10.1186/1471-2334-12-104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lin SH, Liao WH, Lai CC, Liao CH, Tan CK, Wang CY, Huang YT, Hsueh PR. 2010. Risk factors for mortality in patients with persistent methicillin-resistant Staphylococcus aureus bacteraemia in a tertiary care hospital in Taiwan. J. Antimicrob. Chemother. 65:1792–1798. 10.1093/jac/dkq188 [DOI] [PubMed] [Google Scholar]
- 28.Liao CH, Chen SY, Huang YT, Hsueh PR. 2008. Outcome of patients with meticillin-resistant Staphylococcus aureus bacteraemia at an Emergency Department of a medical centre in Taiwan. Int. J. Antimicrob. Agents 32:326–332. 10.1016/j.ijantimicag.2008.04.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang JL, Wang JT, Sheng WH, Chen YC, Chang SC. 2010. Nosocomial methicillin-resistant Staphylococcus aureus (MRSA) bacteremia in Taiwan: mortality analyses and the impact of vancomycin, MIC = 2 mg/L, by the broth microdilution method. BMC Infect. Dis. 10:159. 10.1186/1471-2334-10-159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cavalcanti AB, Goncalves AR, Almeida CS, Bugano DD, Silva E. 2010. Teicoplanin versus vancomycin for proven or suspected infection. Cochrane Database Syst. Rev. 2010:CD007022. 10.1002/14651858.CD007022.pub2 [DOI] [PubMed] [Google Scholar]
- 31.Wood MJ. 1996. The comparative efficacy and safety of teicoplanin and vancomycin. J. Antimicrob. Chemother. 37:209–222. 10.1093/jac/37.2.209 [DOI] [PubMed] [Google Scholar]
- 32.Huang JH, Hsu RB. 2008. Treatment of infective endocarditis caused by methicillin-resistant Staphylococcus aureus: teicoplanin versus vancomycin in a retrospective study. Scand. J. Infect. Dis. 40:462–467. 10.1080/00365540701837126 [DOI] [PubMed] [Google Scholar]
- 33.Liu C-Y, Lee W-S, Fung C-P, Cheng N-C, Liu C-L, Yang S-P, Chen S-L. 1996. Comparative study of teicoplanin vs vancomycin for the treatment of methicillin-resistant Staphylococcus aureus bacteraemia. Clin. Drug Invest. 12:80–87. 10.2165/00044011-199612020-00003 [DOI] [PubMed] [Google Scholar]
- 34.Liu C, Bayer A, Cosgrove SE, Daum RS, Fridkin SK, Gorwitz RJ, Kaplan SL, Karchmer AW, Levine DP, Murray BE, M JR, Talan DA, Chambers HF. 2011. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin. Infect. Dis. 52:285–292. 10.1093/cid/cir034 [DOI] [PubMed] [Google Scholar]
- 35.Park HJ, Kim SH, Kim MJ, Lee YM, Park SY, Moon SM, Park KH, Chong YP, Lee SO, Choi SH, Woo JH, Kim YS. 2012. Efficacy of linezolid-based salvage therapy compared with glycopeptide-based therapy in patients with persistent methicillin-resistant Staphylococcus aureus bacteremia. J. Infect. 65:505–512. 10.1016/j.jinf.2012.08.007 [DOI] [PubMed] [Google Scholar]
- 36.Hsiao SH, Chou CH, Lin WL, Lee EJ, Liao LH, Chang HJ, Yeh PY, Lin CY, Wu TJ. 2012. High risk of cross-reactivity between vancomycin and sequential teicoplanin therapy. J. Clin. Pharm. Ther. 37:296–300. 10.1111/j.1365-2710.2011.01291.x [DOI] [PubMed] [Google Scholar]
- 37.Hung YP, Lee NY, Chang CM, Lee HC, Wu CJ, Chen PL, Lee CC, Chung CH, Ko WC. 2009. Tolerability of teicoplanin in 117 hospitalized adults with previous vancomycin-induced fever, rash, or neutropenia: a retrospective chart review. Clin. Ther. 31:1977–1986. 10.1016/j.clinthera.2009.09.010 [DOI] [PubMed] [Google Scholar]
- 38.DeRyke CA, Alexander DP. 2009. Optimizing vancomycin dosing through pharmacodynamic assessment targeting area under the concentration-time curve/minimum inhibitory concentration. Hosp. Pharm. 44:751–765. 10.1310/hpj4409-751 [DOI] [Google Scholar]


