Abstract
Despite the growing use of poxvirus vectors as vaccine candidates for multiple pathogens and cancers, their innate stimulatory properties remain poorly characterized. Here we show that the canarypox virus-based vector ALVAC induced distinct systemic proinflammatory and antiviral cytokine and chemokine levels following the vaccination of rhesus monkeys compared to the vaccinia virus-based vectors MVA and NYVAC. These data suggest that there are substantial biological differences among leading poxvirus vaccine vectors that may influence resultant adaptive immune responses following vaccination.
TEXT
Poxvirus vectors are used for vaccination against multiple pathogens, including human immunodeficiency virus type 1 (HIV-1) and cancer (1–3). A recent study that used the canarypox virus-based vector ALVAC with a HIV-1 gp120 protein boost showed 31.2% efficacy against HIV-1 infection in a phase III trial in Thailand (RV144) (4). While analysis of data from the RV144 trial has indicated several possible correlates of vaccine-induced protection (5), the innate immune stimulatory properties of ALVAC vectors and how they might influence adaptive immune responses remain poorly described. In this study, we compared innate immune stimulation as measured by serum cytokine and chemokine levels following vaccination of rhesus monkeys with ALVAC and two vaccinia virus-based vectors, modified vaccinia virus Ankara (MVA) (6) and New York vaccinia virus (NYVAC) (7).
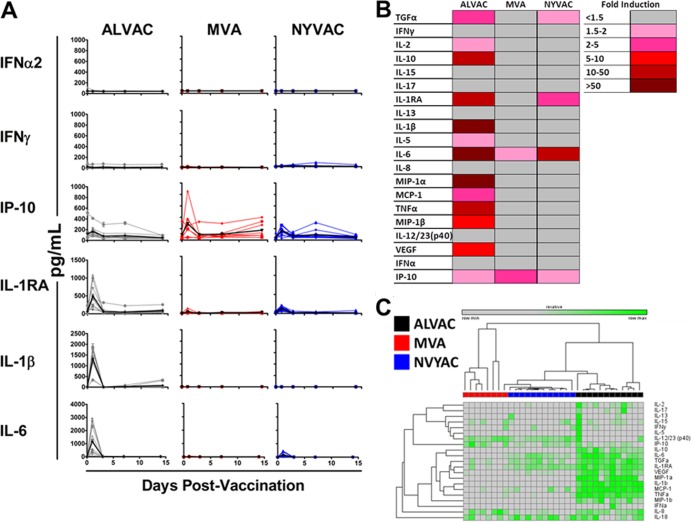
We initiated studies by assessing serum cytokine and chemokine levels following vaccination with the replication-incompetent ALVAC, MVA, or NYVAC vector in 32 rhesus monkeys (Macaca mulatta). All studies with animals and human cells were approved by the relevant Institutional Animal Care and Use Committee (IACUC) and Institutional Review Board (IRB). Monkeys (n = 8 to 12/group) were injected intramuscularly (i.m.) with 1 × 108 PFU of the ALVAC, MVA, or NYVAC vector expressing simian immunodeficiency virus (SIV) Gag, Pol, and Env (8–10). All of the vectors used in these studies were similar in purity and infectivity. Sera were collected on days 0, 1, 3, 7, and 14 following vaccination. Systemic cytokine and chemokine levels were assessed by Luminex assays and enzyme-linked immunosorbent assays (ELISAs) as previously described (11). Longitudinal analysis of systemic cytokine and chemokine levels following ALVAC vaccination revealed potent but transient induction of proinflammatory cytokines and chemokines on day 1 postimmunization (Fig. 1A and B). In contrast, lower induction of proinflammatory cytokines and chemokines was observed in monkeys vaccinated with MVA or NYVAC (Fig. 1A and B). Animals that received ALVAC displayed greater fold induction over the averaged group baseline than those that received MVA or NYVAC for interleukin-1β (IL-1β) (411- and 408-fold greater induction, respectively; P = 0.0002 and P < 0.0001, respectively; Mann-Whitney U Test), IL-6 (83- and 6.3-fold greater induction, respectively; P = 0.0003 and P = 0.0043, respectively), tumor necrosis factor alpha (TNF-α) (48- and 29-fold greater induction, respectively; P = 0.0002 and P < 0.0001, respectively), monocyte chemotactic protein 1 (MCP-1) (4- and 3-fold greater induction, respectively; P = 0.0002 and P < 0.0001, respectively), macrophage inflammatory protein alpha (MIP-1α) (37- and 79-fold greater induction, respectively; P = 0.003 and P = 0.0004, respectively), and MIP-1β (9- and 10-fold greater induction, respectively; P = 0.0002 and P < 0.0001) at day 1 following vaccination. These data show that ALVAC induced a distinct proinflammatory response compared to MVA and NYVAC in rhesus monkeys.
FIG 1.
Serum cytokine and chemokine concentrations of rhesus monkeys following poxvirus vector vaccination. Rhesus monkeys (n = 8 to 12/group) were inoculated i.m. with 1 × 108 PFU of ALVAC, MVA, or NYVAC expressing SIV Gag, Pol, and Env. Sera were collected on days 0, 1, 3, 7, and 14 following vaccination, and systemic levels of cytokines and chemokines were analyzed by Luminex assays and ELISAs. (A) Systemic levels of selected cytokines and chemokines following vaccination with the various poxvirus vectors, with individual levels shown as colored lines and group means over time shown as black lines. (B) Mean fold induction of all cytokines and chemokines at day 1 postvaccination of rhesus monkeys with poxvirus vectors. The values shown are group mean fold induction over the averaged group baseline. (C) Fold cytokine induction over the grouped baseline of rhesus monkeys following vaccination with ALVAC, MVA, and NYVAC. Monkeys are clustered in an unsupervised hierarchical analysis based on individual fold induction over the baseline values of all of the cytokines. Clustering analysis was performed in Gene-E. Values are log individual fold induction of the indicated cytokines and chemokines over the grouped baseline at day 1 following vaccination with the various poxvirus vectors.
Unsupervised hierarchical clustering of animals based upon fold induction at day 1 postvaccination over the individual baselines of all of the cytokines and chemokines analyzed indicated further that the responses elicited by ALVAC, MVA, and NYVAC are all distinct (Fig. 1C). ALVAC induced a distinct proinflammatory cytokine milieu, but differences were also observed between MVA and NYVAC for several cytokines, such as IL-6, transforming growth factor alpha, and IL-1RA (Fig. 1C). Together, these data indicate that ALVAC, MVA, and NYVAC all induce qualitatively distinct cytokine profiles following vaccination.
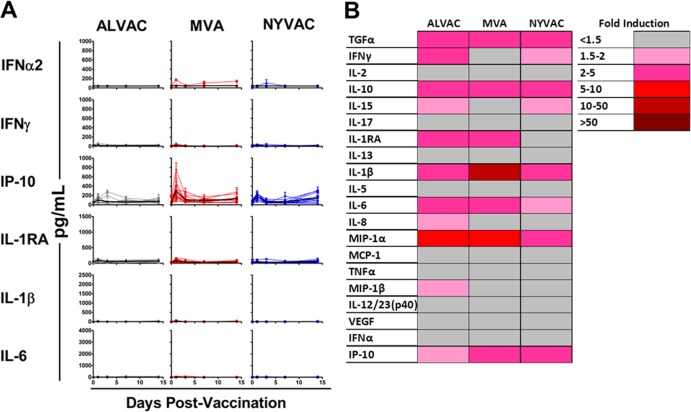
We next analyzed the impact of priming with a heterologous vector on elicited cytokines following boosting with ALVAC, MVA, or NVYAC. Rhesus monkeys (n = 12/group) were injected i.m. with 3 × 1010 viral particles of an adenovirus serotype 26 (Ad26) vector expressing SIV Gag, Pol, and Env. Twenty-four weeks following priming vaccination, animals were boosted i.m. with 1 × 108 PFU of ALVAC, MVA, or NYVAC expressing SIV Gag, Pol, and Env. Longitudinal analysis of serum cytokines and chemokines following the boost immunization with ALVAC, MVA, or NYVAC revealed clear induction of proinflammatory and antiviral cytokines such as IL-1β, IL-6, gamma interferon (IFN-γ), and IFN-γ-induced protein 10 (IP-10), on day 1 following the boost (Fig. 2A). However, the cytokine levels elicited following boosting were lower than those observed following priming for all of the vectors, and particularly for ALVAC (Fig. 1A and 2A). Analysis of the average fold induction over the group average baseline indicated that boosting with ALVAC, MVA, or NYVAC induced comparable cytokine profiles, characterized by IL-1β, IL-6, IL-1RA, IFN-γ, and IP-10 (Fig. 2B). These data suggest that Ad26 priming attenuated innate cytokine induction as measured by serum cytokine profiles of ALVAC-, MVA-, or NYVAC-boosted animals. We suspect that this may be related to enhanced vector clearance as a result of the adaptive immune responses elicited by the priming immunization, although other factors may play a role.
FIG 2.
Serum cytokine and chemokine concentrations of rhesus monkeys following poxvirus vector boosting. Rhesus monkeys (n = 12/group) were primed i.m. with 3 × 1010 viral particles of Ad26 expressing SIV Gag, Pol, and Env and then boosted i.m. with 1 × 108 PFU of the ALVAC, MVA, or NYVAC vector expressing transgenes. Sera were collected on days 0, 1, 3, 7, and 14 following vaccination, and systemic levels of cytokines and chemokines were analyzed by Luminex assays and ELISAs. (A) Systemic levels of selected cytokines and chemokines following boosting with the various poxvirus vectors, with individual levels shown as colored lines and group means over time shown as black lines. (B) Mean fold induction of all cytokines and chemokines in rhesus monkeys at day 1 postboosting with poxvirus vectors. The values shown are group mean fold induction over the averaged group baseline prior to boosting with the various poxvirus vectors.
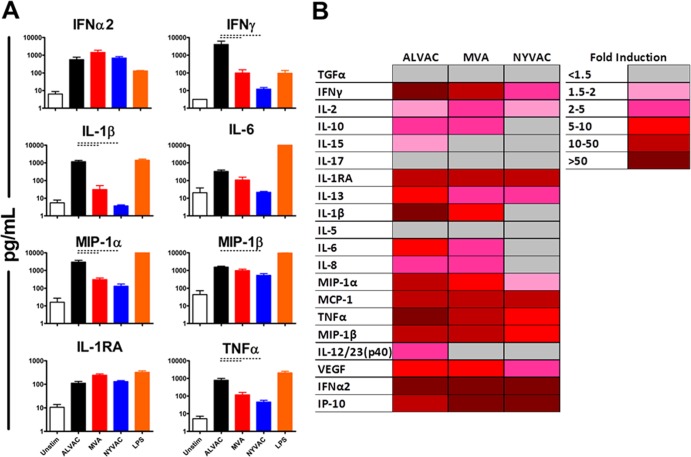
We next sought to assess cytokine and chemokine induction by ALVAC, MVA, and NYVAC in vitro in freshly isolated human peripheral blood mononuclear cells (PBMC). Fresh human PBMC (n = 4/group) were isolated by Ficoll-Hypaque density gradient centrifugation (12). In order to include all of the possible cell types shown to be infectible with poxvirus vectors, whole PBMC were infected with the ALVAC, MVA, or NYVAC vector at a multiplicity of infection (MOI) of 10 PFU/cell (13). Culture supernatant was analyzed for cytokine and chemokine levels 24 h postinfection by Luminex assays. Similar to our in vivo results, stimulation of human PBMC with ALVAC resulted in higher levels of proinflammatory and IFN-related antiviral cytokines and chemokines than did stimulation with MVA or NYVAC (Fig. 3A). Stimulation of human PBMC with ALVAC elicited higher levels than that with MVA or NYVAC for IFN-γ (41- and 334-fold greater levels, respectively; both P = 0.0286, Mann-Whitney U test), IL-1β (37- and 315-fold greater levels, respectively; both P = 0.0286), IL-6 (3- and 15-fold greater levels, respectively; P = 0.0571 and P = 0.0286, respectively), and TNF-α (7- and 17-fold greater levels, respectively; both P = 0.0286). These in vitro results are comparable to our in vivo findings, with a few notable exceptions, such as a lack of difference observed in the elicitation of IFN-α2 in vitro.
FIG 3.
Induction of cytokines and chemokines by poxvirus vector stimulation of human PBMC. (A) Fresh whole human PBMC (n = 4/group) were stimulated at an MOI of 10 PFU/cell with the ALVAC, MVA, or NYVAC vector expressing no transgene. Cytokine and chemokines were measured 24 h postinfection by Luminex assays. Lipopolysaccharide (LPS; 1 ng/ml) was included as the positive control. The data are means and standard errors of the means. Bars indicate P values of <0.05 by Kruskal-Wallis tests. (B) Mean fold induction of all cytokines and chemokines at 24 h postinfection of human PBMC with poxvirus vectors. The values shown are group mean fold induction over the averaged group baseline of the unstimulated PBMC control.
We sought to assess the contributions of various PBMC subsets, some of which were previously shown to be readily transduced by vaccinia virus, to the production of cytokines and chemokines elicited by the ALVAC, MVA, and NYVAC vectors (13). Human PBMC (n = 2 to 6/group) were isolated as described above and depleted of T cells, monocytes and macrophages (MonoMac), myeloid dendritic cells, or plasmacytoid dendritic cells (pDC) by magnetic separation as previously described (11). Depletion was confirmed by flow cytometry (data not shown). Cells were then infected with ALVAC, MVA, or NYVAC at an MOI of 10 PFU/cell. Supernatants were analyzed 24 h postinfection for elicited cytokines and chemokines by Luminex analyses. Depletion of pDC led to a marked reduction in the levels of IFN-α2 (16-fold lower induction; P = 0.0022) elicited in response to ALVAC stimulation relative to those in unseparated cells (Fig. 4). Depletion of MonoMac resulted in reduced induction of IL-1β and TNF-α (117- and 37-fold lower induction; both P = 0.0043) in response to ALVAC stimulation. Depletion of T cells markedly decreased the levels of elicited IFN-γ (15-fold lower induction; P = 0.0087) following ALVAC stimulation. A similar trend was observed with lowered levels of elicited IL-1β and TNF-α in response to MVA or NYVAC stimulation following depletion of MonoMac. Interestingly, pDC depletion had a more modest impact on IFN-α2 secretion following MVA or NYVAC stimulation than did ALVAC stimulation. Taken together, these results indicate that multiple PBMC subsets likely contribute to the overall cytokine milieu elicited by the ALVAC, MVA, and NYVAC vectors.
FIG 4.
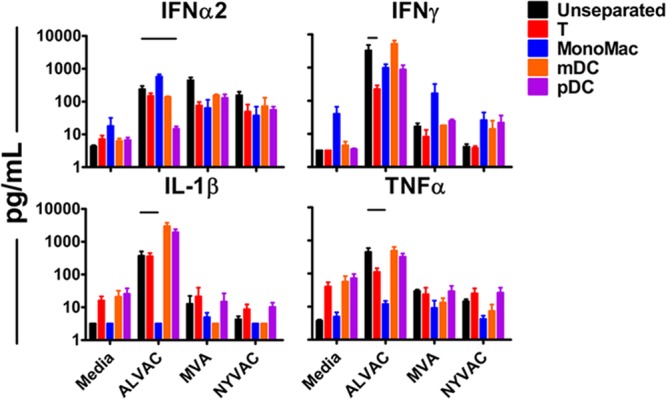
Cytokine and chemokine responses elicited by ALVAC in human PBMC depleted of various cell populations. Fresh whole human PBMC (n = 2 to 6/group) were depleted of the indicated PBMC populations, and depletion was confirmed by flow cytometry (data not shown). Unseparated and depleted cell populations were stimulated at an MOI of 10 PFU/cell with ALVAC, MVA, or NYVAC, and cytokine and chemokine responses were measured by Luminex assays at 24 h postinfection. The data shown are means and standard errors of the means. Lines indicate P < 0.01 by Kruskal-Wallis tests.
In this study, we show that the innate immune profiles elicited by ALVAC, MVA, and NYVAC are all distinct. ALVAC elicited an innate immune response qualitatively and quantitatively different from that elicited by MVA and NYVAC both in vivo in rhesus monkeys and in vitro in human PBMC, characterized by a higher induction of proinflammatory and IFN-related antiviral cytokines and chemokines on day 1 following immunization. Moreover, MVA and NYVAC also proved different, although these differences were less striking. The stimulatory phenotypes observed following priming with ALVAC, MVA, or NYVAC were all reduced when these poxvirus vectors were used as a boost. Furthermore, ALVAC's stimulatory phenotype was influenced by several PBMC subsets such as T cells, MonoMac, and pDC. These data suggest potentially important biological differences among these three clinically relevant poxvirus vectors. Additional studies are required to evaluate the correlation between these different innate signatures and subsequent adaptive immune responses and protective efficacy.
A possible source of the observed differences in innate stimulatory phenotypes among ALVAC, MVA, and NYVAC involves their different arsenals of immune regulatory genes. Poxviruses possess a wide array of proteins that serve to block host antiviral immune responses (14–18). Importantly, ALVAC differs significantly from both vaccinia virus-derived vectors MVA and NYVAC in terms of extensive phylogenetic divergence, as well as genome size (approximately 365 kbp versus approximately 178 kbp, respectively) (14, 19) and the number of open reading frame (ORFs) (7, 19, 20). As stated above, the ALVAC, MVA, and NYVAC vectors are replication incompetent when used in vivo and many of ALVAC's ORFs may not be functional in mammalian cells, as evidenced by the inability of avipoxviruses to replicate in mammalian cells (21–23). Previous reports have indicated increased innate immune gene upregulation following MVA and NYVAC infection relative to vaccinia virus Western Reserve infection in HeLa cells (24–26), highlighting that attenuation of these vectors by the removal of viral immune regulatory genes confers a stronger innate stimulatory phenotype. These studies reported that MVA elicited a stronger IFN-stimulatory phenotype and NYVAC induced a more proinflammatory phenotype, congruent with our results in the present study. Differences in immune gene upregulation and induced immune phenotypes were further seen following MVA and NYVAC infections in vitro and in vivo (27–30). Further studies showed increased innate stimulation by these vectors following expanded deletion of their immune regulatory gene repertoire, highlighting the role of the immune gene repertoire of poxvirus vectors in their innate stimulatory phenotypes (31–34). However, far less is known about the innate immune profile of ALVAC and its immune regulatory genes (35). Our findings suggest that ALVAC stimulation of innate immunity requires several PBMC cellular subsets, which is consistent with prior studies that have suggested that monocyte tropism is a key difference in innate triggering by ALVAC, MVA, and NVYAC (36). The unique innate immune profile of ALVAC may also relate to its avian origin, as opposed to MVA and NYVAC. The molecular basis of this innate stimulatory phenotype and whether it is unique to ALVAC or extends to other avipoxviruses warrant further study.
Our results indicate that the innate immune profiles elicited by the leading poxvirus vaccine vectors are different, suggesting potentially important biological differences. In particular, ALVAC induced a unique proinflammatory cytokine and chemokine response following the vaccination of rhesus monkeys and infection of human PBMC. The extent to which these properties are advantageous to vaccines, however, remains to be determined. We previously reported substantial differences in innate immune profiles among various serotypes of adenovirus vectors (11). Taken together, these data suggest that vectors from the same family can differ markedly in their biological and innate stimulatory properties, which may potentially impact the resultant adaptive immune responses and protective efficacy of vector-based vaccines.
ACKNOWLEDGMENTS
We thank S. Blackmore, E. Borducci, A. McNally, L. Parenteau, L. Peter, N. Provine K. Stanley, and J. Smith for assistance. We thank Jim Tartaglia (Sanofi-Pasteur) for ALVAC vectors, as well as technical and scientific advice.
We acknowledge support from the National Institutes of Health (AI078526, AI096040, AI095985); the U.S. Military HIV Research Program (W81XWH-07-2-0067); and the Ragon Institute of MGH, MIT, and Harvard (D.H.B.); as well as a National Defense Science & Engineering Graduate Fellowship and the Herchel Smith Graduate Fellowship from Harvard University (J.E.T.).
Footnotes
Published ahead of print 20 November 2013
REFERENCES
- 1.Perkus M, Tartaglia J, Paoletti E. 1995. Poxvirus-based infectious vaccine for cancer, AIDS, and other diseases. J. Leukoc. Biol. 58:1–13 [DOI] [PubMed] [Google Scholar]
- 2.Walsh SR, Dolin R. 2011. Vaccinia viruses: vaccines against smallpox and vectors against infectious diseases and tumors. Expert Rev. Vaccines 10:1221–1240. 10.1586/erv.11.79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Franchini G, Gurunathan S, Bagylos L, Plotkin S, Tartaglia J. 2004. Poxvirus-based vaccine candidates for HIV: two decades of experience with special emphasis on canarypox vectors. Expert Rev. Vaccines 3:S75–S88. 10.1586/14760584.3.4.S75 [DOI] [PubMed] [Google Scholar]
- 4.Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, Paris R, Premsri N, Namwat C, de Souza M, Adams E, Benenson M, Gurunathan S, Tartaglia J, McNeil JG, Francis DP, Stablein D, Birx DL, Chunsuttiwat S, Khamboonruang C, Thongcharoen P, Robb ML, Michael NL, Kunasol P, Kim JH. 2009. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 361:2209–2220. 10.1056/NEJMoa0908492 [DOI] [PubMed] [Google Scholar]
- 5.Haynes B, Tomaras GD, Alam SM, Evans D, Montefiori D, Decamp A, Huang Y, Rao M, Billings E, Karasavvas N, Bailer RT, Soderberg KA, Andrews C, Berman PW, Frahm N, Rosa De SC, Alpert MD, Yates NL, Shen X, Koup RA. 2012. Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N. Engl. J. Med. 366:1275–1286. 10.1056/NEJMoa1113425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mayr A, Stickl H, Muller H, Danner K, Singer H. 1978. The smallpox vaccination strain MVA: marker, genetic structure, experience gained with the parenteral vaccination and behavior in organisms with a debilitated defence mechanism. Zentralbl. Bakteriol. B 167:375–390 [PubMed] [Google Scholar]
- 7.Tartaglia J, Perkus M, Taylor J, Norton E, Audonnet J, Cox W, Davis S, van der Hoeven J, Meignier B, Riviere M. 1992. NYVAC: a highly attenuated strain of vaccinia virus. Virology 188:217–232. 10.1016/0042-6822(92)90752-B [DOI] [PubMed] [Google Scholar]
- 8.Pal R, Venzon D, Letvin N, Santra S, Montefiori D, Miller N, Tryniszewska E, Lewis M, Vancott T, Hirsch V, Woodward R, Gibson A, Grace M, Dobratz E, Markham P, Hel Z, Nacsa J, Klein M, Tartaglia J, Franchini G. 2002. ALVAC-SIV-gag-pol-env-based vaccination and macaque major histocompatibility complex class I (A*01) delay simian immunodeficiency virus SIVmac-induced immunodeficiency. J. Virol. 76:292–302. 10.1128/JVI.76.1.292-302.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ourmanov I, Brown CR, Moss B, Carroll M, Wyatt L, Pletneva L, Goldstein S, Venzon D, Hirsch VM. 2000. Comparative efficacy of recombinant modified vaccinia virus Ankara expressing simian immunodeficiency virus (SIV) Gag-Pol and/or Env in macaques challenged with pathogenic SIV. J. Virol. 74:2740–2751. 10.1128/JVI.74.6.2740-2751.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Benson J, Chougnet C, Robert-Guroff M, Montefiori D, Markham P, Shearer G, Gallo R, Cranage M, Paoletti E, Limbach K, Venzon D, Tartaglia J, Franchini G. 1998. Recombinant vaccine-induced protection against the highly pathogenic simian immunodeficiency virus SIV(mac251): dependence on route of challenge exposure. J. Virol. 72:4170–4182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Teigler JE, Iampietro MJ, Barouch DH. 2012. Vaccination with adenovirus serotypes 35, 26, and 48 elicits higher levels of innate cytokine responses than adenovirus serotype 5 in rhesus monkeys. J. Virol. 86:9590–9598. 10.1128/JVI.00740-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bennett S, Breit SN. 1994. Variables in the isolation and culture of human monocytes that are of particular relevance to studies of HIV. J. Leukoc. Biol. 56:236–240 [DOI] [PubMed] [Google Scholar]
- 13.Byrd D, Amet T, Hu N, Lan J, Hu S, Yu Q. 2013. Primary human leukocyte subsets differentially express vaccinia virus receptors enriched in lipid rafts. J. Virol. 87:9301–9312. 10.1128/JVI.01545-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gubser C. 2004. Poxvirus genomes: a phylogenetic analysis. J. Gen. Virol. 85:105–117. 10.1099/vir.0.19565-0 [DOI] [PubMed] [Google Scholar]
- 15.Cuff S, Ruby J. 1996. Evasion of apoptosis by DNA viruses. Immunol. Cell Biol. 74:527–537. 10.1038/icb.1996.86 [DOI] [PubMed] [Google Scholar]
- 16.McFadden G. 2005. Poxvirus tropism. Nat. Rev. Microbiol. 3:201–213. 10.1038/nrmicro1099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Perdiguero B, Esteban M. 2009. The interferon system and vaccinia virus evasion mechanisms. J. Interferon Cytokine Res. 29:581–598. 10.1089/jir.2009.0073 [DOI] [PubMed] [Google Scholar]
- 18.Seet BT, Johnston JB, Brunetti CR, Barrett JW, Everett H, Cameron C, Sypula J, Nazarian SH, Lucas A, McFadden G. 2003. Poxviruses and immune evasion. Annu. Rev. Immunol. 21:377–423. 10.1146/annurev.immunol.21.120601.141049 [DOI] [PubMed] [Google Scholar]
- 19.Tulman E, Afonso C, Lu Z, Zsak L, Kutish G, Rock D. 2004. The genome of canarypox virus. J. Virol. 78:353–366. 10.1128/JVI.78.1.353-366.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Antoine G, Scheiflinger F, Dorner F, Falkner F. 1998. The complete genomic sequence of the modified vaccinia Ankara strain: comparison with other orthopoxviruses. Virology 244:365–396. 10.1006/viro.1998.9123 [DOI] [PubMed] [Google Scholar]
- 21.Taylor J, Weinberg R, Languet B, Desmettre P, Paoletti E. 1988. Recombinant fowlpox virus inducing protective immunity in non-avian species. Vaccine 6:497–503. 10.1016/0264-410X(88)90100-4 [DOI] [PubMed] [Google Scholar]
- 22.Baxby D, Paoletti E. 1992. Potential use of non-replicating vectors as recombinant vaccines. Vaccine 10:8–9. 10.1016/0264-410X(92)90411-C [DOI] [PubMed] [Google Scholar]
- 23.Meyer H, Sutter G, Mayr A. 1991. Mapping of deletions in the genome of the highly attenuated vaccinia virus MVA and their influence on virulence. J. Gen. Virol. 72:1031–1038. 10.1099/0022-1317-72-5-1031 [DOI] [PubMed] [Google Scholar]
- 24.Guerra S, López-Fernández LA, Pascual-Montano A, Najera J, Zaballos A, Esteban M. 2006. Host response to the attenuated poxvirus vector NYVAC: upregulation of apoptotic genes and NF-kappaB-responsive genes in infected HeLa cells. J. Virol. 80:985–998. 10.1128/JVI.80.2.985-998.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guerra S, Lopez-Fernandez L, Pascual-Montano A, Munoz M, Harshman K, Esteban M. 2003. Cellular gene expression survey of vaccinia virus infection of human HeLa cells. J. Virol. 77:6493–6506. 10.1128/JVI.77.11.6493-6506.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guerra S, López-Fernández, Conde LAR, Pascual-Montano A, Harshman K, Esteban M. 2004. Microarray analysis reveals characteristic changes of host cell gene expression in response to attenuated modified vaccinia virus Ankara infection of human HeLa cells. J. Virol. 78:5280–5834. 10.1128/JVI.78.11.5820-5834.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Guerra S, Nájera JL, González JM, López-Fernández L, Climent N, Gatell J, Gallart T, Esteban M. 2007. Distinct gene expression profiling after infection of immature human monocyte-derived dendritic cells by the attenuated poxvirus vectors MVA and NYVAC. J. Virol. 81:8707–8721. 10.1128/JVI.00444-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gómez CE, Perdiguero B, Jiménez V, Filali-Mouhim A, Ghneim K, Haddad EK, Quakkelaar ED, Quakkerlaar ED, Delaloye J, Harari A, Roger T, Duhen T, Dunhen T, Sékaly RP, Melief CJM, Calandra T, Sallusto F, Lanzavecchia A, Wagner R, Pantaleo G, Esteban M. 2012. Systems analysis of MVA-C induced immune response reveals its significance as a vaccine candidate against HIV/AIDS of clade C. PLoS One 7:e35485. 10.1371/journal.pone.0035485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mooij P, Balla-Jhagjhoorsingh SS, Koopman G, Beenhakker N, van Haaften P, Baak I, Nieuwenhuis IG, Kondova I, Wagner R, Wolf H, Gómez CE, Nájera JL, Jiménez V, Esteban M, Heeney JL. 2008. Differential CD4+ versus CD8+ T-cell responses elicited by different poxvirus-based human immunodeficiency virus type 1 vaccine candidates provide comparable efficacies in primates. J. Virol. 82:2975–2988. 10.1128/JVI.02216-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ferrier-Rembert A, Drillien R, Tournier J-N, Garin D, Crance J-M. 2008. Short- and long-term immunogenicity and protection induced by non-replicating smallpox vaccine candidates in mice and comparison with the traditional 1st generation vaccine. Vaccine 26:1794–1804. 10.1016/j.vaccine.2007.12.059 [DOI] [PubMed] [Google Scholar]
- 31.Gómez CE, Perdiguero B, Nájera JL, Sorzano CO, Jiménez V, González-Sanz R, Esteban M. 2012. Removal of vaccinia virus genes that block interferon type I and II pathways improves adaptive and memory responses of the HIV/AIDS vaccine candidate NYVAC-C in mice. J. Virol. 86:5026–5038. 10.1128/JVI.06684-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.García-Arriaza J, Nájera JL, Gómez CE, Tewabe N, Sorzano CO, Calandra T, Roger T, Esteban M. 2011. A candidate HIV/AIDS vaccine (MVA-B) lacking vaccinia virus gene C6L enhances memory HIV-1-specific T-cell responses. PLoS One 6:e24244. 10.1371/journal.pone.0024244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Quakkelaar ED, Redeker A, Haddad EK, Harari A, McCaughey SM, Duhen T, Filali-Mouhim A, Goulet J-P, Loof NM, Ossendorp F, Perdiguero B, Heinen P, Gomez CE, Kibler KV, Koelle DM, Sékaly RP, Sallusto F, Lanzavecchia A, Pantaleo G, Esteban M, Tartaglia J, Jacobs BL, Melief CJ. 2011. Improved innate and adaptive immunostimulation by genetically modified HIV-1 protein expressing NYVAC vectors. PLoS One 6:e16819. 10.1371/journal.pone.0016819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Falivene J, Del Médico Zajac MP, Pascutti MF, Rodríguez AM, Maeto C, Perdiguero B, Gómez CE, Esteban M, Calamante G, Gherardi MM. 2012. Improving the MVA vaccine potential by deleting the viral gene coding for the IL-18 binding protein. PLoS One 7:e32220. 10.1371/journal.pone.0032220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zanotto C, Pozzi E, Pacchioni S, Volonté L, De Giuli Morghen C, Radaelli A. 2010. Canarypox and fowlpox viruses as recombinant vaccine vectors: a biological and immunological comparison. Antiviral Res. 88:53–63. 10.1016/j.antiviral.2010.07.005 [DOI] [PubMed] [Google Scholar]
- 36.Yu Q, Jones B, Hu N, Chang H, Ahmad S, Liu J, Parrington M, Ostrowski M. 2006. Comparative analysis of tropism between canarypox (ALVAC) and vaccinia viruses reveals a more restricted and preferential tropism of ALVAC for human cells of the monocytic lineage. Vaccine 24:6376–6391. 10.1016/j.vaccine.2006.06.011 [DOI] [PubMed] [Google Scholar]