Abstract
Purpose
This pilot trial sought to investigate whether any of three doses of American ginseng (Panax quinquefolius) might help cancer-related fatigue. A secondary aim was to evaluate toxicity.
Methods
Eligible adults with cancer were randomized in a double-blind manner, to receive American ginseng in doses of 750, 1,000, or 2,000 mg/day or placebo given in twice daily dosing over 8 weeks. Outcome measures included the Brief Fatigue Inventory, vitality subscale of the Medical Outcome Scale Short Form-36 (SF-36), and the Global Impression of Benefit Scale at 4 and 8 weeks.
Results
Two hundred ninety patients were accrued to this trial. Nonsignificant trends for all outcomes were seen in favor of the 1,000- and 2,000-mg/day doses of American ginseng. Area under the curve analysis of activity interference from the Brief Fatigue Inventory was 460–467 in the placebo group and 750 mg/day group versus 480–551 in the 1,000- and 2,000-mg/day arms, respectively. Change from baseline in the vitality subscale of the SF-36 was 7.3– 7.8 in the placebo and the 750-mg/day arm, versus 10.5– 14.6 in the 1,000- and 2,000-mg/day arms. Over twice as many patients on ginseng perceived a benefit and were satisfied with treatment over those on placebo. There were no significant differences in any measured toxicities between any of the arms.
Conclusion
There appears to be some activity and tolerable toxicity at 1,000–2,000 mg/day doses of American ginseng with regard to cancer-related fatigue. Thus, further study of American ginseng is warranted.
Keywords: Panax quinquefolius, American ginseng, Cancer-related fatigue, Botanicals
Background and significance
Definition and prevalence of cancer-related fatigue
Cancer-related fatigue has been defined by a panel of the National Comprehensive Cancer Network as “a persistent, subjective sense of tiredness related to cancer or cancer treatment that interferes with usual functioning” [1]. One of the distinguishing characteristics of cancer-related fatigue, compared to other fatigue, is that it is not relieved by sleep or rest [1–3] and patients report being “unusually” or overwhelmingly tired [1–4]. Cancer-related fatigue is out of proportion to the amount of physical activity and hence is often accompanied by negative affect or distress [1–4].
Fatigue is one of the most common unmanaged symptoms in people diagnosed with cancer and exists in those receiving chemotherapy, radiation therapy, biologic therapies, and in those having completed treatment [1, 2, 5, 6]. The prevalence of fatigue is reported to be between 59–96% in patients undergoing chemotherapy, 65–100% in patients receiving radiation therapy, and 30% in long term survivors [7–9].
Evidence-based interventions
Few randomized controlled trials of nonpharmacologic or pharmacologic interventions for improving fatigue in cancer patients have been performed. Of the nonpharmacologic interventions, exercise has the strongest evidence base for treating fatigue [10, 11]. Most studies have used home-based walking, three to five times per week for 30 min each time [2, 12, 13]. Other interventions with some evidence of efficacy include restorative therapy such as communing with nature, dietary management, sleep therapy, and activity management [10–13]. However, though preliminary evidence is positive, large randomized trials are needed to properly define the role and effect size of many of these interventions in clinical practice.
Several pharmacologic agents have been explored, such as methylphenidate [14–18], corticosteroids, anabolic steroids, antidepressants, donepezil [19], L-carnitine, modafanil [20], and amantadine, a drug which as shown some efficacy in relieving fatigue related to multiple sclerosis [11]. However, few of these agents have been studied in large placebocontrolled trials and, so far, of those that have been studied, none have been proven to be helpful and ready for general clinical practice.
The evidence for ginseng
Within the context of traditional Chinese medicine, ginseng is generally viewed as an “adaptogen”, a substance which can help reduce the impact of environmental stress. There are different species of ginseng, the two most common being Asian (Panax ginseng) and American (Panax quinquefolius), both from the genus Panax of the Araliaceae family of plants [21, 22]. Both Asian and American ginseng have a common mixture of active ingredients, the most important being ginsenosides, in varying amounts, strengths, and ratios and both ginseng preparations are thought to have broad and similar activity [21–23].
Studies with ginseng often do not denote the specific species of ginseng nor the ginsenoside constituents, which makes it very difficult to accurately summarize the literature with respect to scientific thoughts. Despite this weakness, there are several studies looking at ginseng for fatigue.
Most of the preliminary data, both in animals and in pilot studies with humans, have been with Asian ginseng. Studies in mice have shown that Asian ginseng has improved fatigue related to stress using the forced swimming test as well as electroshock stress [24, 25]. One large study evaluating Asian ginseng in combination with vitamins and minerals, in 232 patients who had functional fatigue for over 10 years, concluded that the ginseng formula improved a fatigue symptom score statistically significantly more than did a vitamin placebo [26]. Furthermore, an abstract presented at the 2003 American Society of Clinical Oncology meeting evaluated Asian ginseng in chemonäive people with cancer. It reported that ginseng significantly improved total and average fatigue levels, as measured by the Brief Fatigue Inventory, even though the sample size was quite small (N=20) [27].
Though much of the published research evaluating properties of ginseng with respect to fatigue has been done with Asian ginseng, there are some important considerations in favor of studying American ginseng for cancer-related fatigue. These issues include the fact that both species have similar ginsenosides [28, 29], the positive experience of American ginseng in research with respect to toxicities [30], and the quality assurance available with respect to the product.
As noted above, both species of ginseng (Asian and American) have many of the same ginsenosides but in different ratios. With respect to fatigue, one study looked at the specific ginsenosides attributable to the ergogenic properties of ginseng [31]. These investigators found that either the Rb1 or Rg1 ginsenoside was necessary to enhance activity performance in mice. Without either of these ginsenosides, the ginseng was not effective [31]. Both American and Asian ginseng have both of these ginsenosides; American has more Rb1, while Asian has more Rg1 [28, 29]. Therefore, operating under the hypothesis that either Rb1 or Rg1 was the active ginsenosides related to fatigue, either Asian or American ginseng would have the potential to impact fatigue.
American ginseng is currently being studied fervently with respect to diabetes [32]. Doses up to 3,000 mg have been studied with no significant toxicity being reported [22]. The only reported side effect in the literature attributed to American ginseng has been insomnia [32, 33]. In addition, American ginseng has been shown to have antiproliferative effects in breast tissue and is beginning to be studied in colon cancer [34–36].
Finally, the availability and quality control of American ginseng may be better, as Asian ginseng is difficult to find and many products studied have been found to be inadequate [37]. American ginseng grown in some areas of the USA is subject to strict pesticide use standards for agricultural products. Specifically, American ginseng from Wisconsin, which was used in this pilot trial, has been tested by the US Environmental Protection Agency and is governed by the Ginseng Board of Wisconsin which has been working with the US Department of Agriculture to develop grading standards to ensure consistency of ginseng marketed as Wisconsin ginseng in the USA [38].
Due to the high prevalence of fatigue and the limited number of effective interventions, the purpose of this pilot trial was to investigate three doses of American ginseng (P. quinquefolius), to evaluate the potential for alleviating cancer-related fatigue and to examine toxicity. This pilot trial was designed to delineate an appropriate dose of American ginseng to study in a more definitive phase III placebo-controlled trial.
Materials and methods
Eligibility
To be eligible for this trial, adult patients had to have a history of cancer-related fatigue as defined by a score of 4 or more on a screening question about fatigue level, that went from 0 (no fatigue) to 10 (fatigue as bad as you can imagine). Other causes of fatigue, such as uncontrolled pain, insomnia, and hypothyroidism, were to be ruled out. Participants had to be experiencing fatigue for at least 1 month and had to have a life expectancy of 6 months or more.
Randomization
Participants were stratified according to stage of disease (stage I/II versus III/IV versus unknown), gender, baseline fatigue score (4–7 versus 8–10), and current treatment (chemotherapy versus not and radiation therapy versus not). Randomization was computer generated and assignments were made using a dynamic allocation procedure balancing marginal distribution of the stratification factors. Participants were randomized to receive a placebo versus 750, 1,000, or 2,000 mg per day of P. quinquefolius. The total milligrams were divided into twice daily dosing to be taken in the morning and midafternoon with food. All participants and treating study personnel were blinded to treatment assignments. Informed consent was obtained through all local Institutional Review Boards according to federal regulations.
Intervention
American ginseng is also known by a number of names (North American ginseng, Ontario ginseng, Canadian ginseng, western ginseng, and Wisconsin ginseng, for example), depending on where it was grown. The ginseng used in this study was Wisconsin ginseng. A 4-year-old root was used and the product was fingerprinted by an independent company, Covance, in Madison, WI, USA. The ginseng met with quality control for pesticides and contaminants. It also contained 5% ginsenosides, with Rb1 being the most prevalent ginsenosides, with 24,000 ppm, compared to 3,230 ppm of Rg1. Other ginsenosides present in this product included (in declining amounts) Re (22,800 ppm), Rc (8,130 ppm), Rd (7,080 ppm), Rb2 (1,160 ppm), and Rf (<850 ppm). The root was made into capsules via Good Manufacturing Practices by Beehive Botanicals in Hayward, WI, USA. Matching placebo was also made by Beehive Botanicals and consisted of long grain white rice flour. Both placebo and Wisconsin ginseng were supplied for this trial by the Ginseng Board of Wisconsin and the Ginseng Research Institute of America (Wausau, WI, USA).
Outcome measures
The primary outcome measure was the Brief Fatigue Inventory (BFI) developed by Mendoza and colleagues [39]. As this was a pilot trial, efficacy trends for the three dose levels were examined. Secondary outcome measures included the vitality subscale of the Medical Outcome Scale Short Form-36 (SF-36) [40], the Pittsburgh Sleep Quality Index (PSQI) [41], the Global Impression of Change [42], and the Linear Analogue Self Assessment Scale [43–46], which has been validated for application in cancer patient clinical trials by the North Central Cancer Treatment Group.
Toxicities were evaluated with a symptom experience diary, which is a self-report diary of potential side effects of ginseng. It measured the severity of the side effects experienced on a 0 to 10 scale with 0 being not a problem to 10 being “as bad as it can be”. Toxicities graded per the National Cancer Institute Common Terminology Criteria for Adverse Events (Version 3.0) every other week during treatment included agitation, anxiety, insomnia, thrombosis, nausea, and vomiting.
All primary and secondary outcome measures were completed at baseline and at 4 and 8 weeks after starting study treatment. Self-reported toxicities were completed weekly and provider-graded toxicities were evaluated every other week.
Statistical analysis
The primary endpoint was the total activity interference as measured by the area under the curve (AUC) of the summated BFI activities of daily living items. The average AUC for the placebo arm was compared to the average AUC for the collective ginseng treatment arms, using a single two sample t test with a two-sided alternative. Subsequent testing for differences in efficacy among the three ginseng dose levels was carried out by paired comparison procedures. Confidence intervals were constructed for the mean reduction in total AUC fatigue score for each treatment group. Understanding that this was designed as a pilot study, the trial was powered for a total of 256 patients with 192 patients in the ginseng arms and 64 patients in the placebo arm providing 80% power to detect a difference of 41% times the standard deviation, using a two sample t test. This is considered a moderate effect size [47]. The study was powered to look at the three ginseng arms combined and not look at individual differences in the doses. To examine differences between specific arms, there was 80% power to detect 61% times the standard deviation, which is a large effect.
The analytical procedures detailed for the primary endpoint were also applied to the secondary endpoints, including the usual fatigue question from the BFI. Supplementary analyses involved a t test and Wilcoxon procedures at each time point as well as a repeated measure of analysis of variance and generalized estimating equations linear model, using data from all time points for the average fatigue score, sleep quality score, sleep latency, sleep duration, overall quality of life, and various domains, and the vitality subscale of the SF-36 [48].
All scales were transformed to 0 to 100 point scales for improved ability to compare effects and determine clinical significance. Toxicities were compared across treatment groups using chi-square testing.
Results
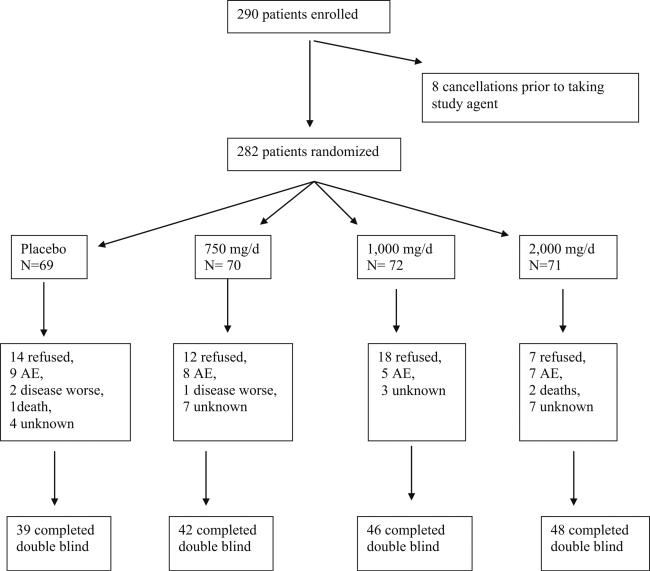
A total of 290 patients were randomized on this trial from October 21, 2005 to July 5, 2006. Patient flow is illustrated in the CONSORT diagram in Fig. 1. One hundred seventy-five completed the double blind 8-week phase of the study, with 39 to 48 patients per arm. Baseline characteristics, shown in Table 1, were well balanced with the randomization and stratification process. There were no statistically significant differences between the arms in these variables at baseline.
Fig. 1.
CONSORT diagram
Table 1.
Demographics
| Placebo (n=69) | 750mg (n=70) | 1,000mg (n=72) | 2,000mg (n=71) | Total (n=282) | |
|---|---|---|---|---|---|
| Age | |||||
| n | 69 | 70 | 72 | 71 | 282 |
| Mean (SD) | 62 (13) | 58 (11) | 60 (12) | 62 (11) | 60 (12) |
| Race | |||||
| White | 66 (96%) | 64 (91%) | 70 (97%) | 67 (94%) | 267 (95%) |
| Black or African American | 2 (3%) | 3 (4%) | 0 (0%) | 3 (4%) | 8 (3%) |
| Native Hawaiian or other Pacific Islander | 0 (0%) | 1 (1%) | 0 (0%) | 0 (0%) | 1 (0.4%) |
| Asian | 0 (0%) | 1 (1%) | 1 (1%) | 1 (1%) | 3 (1%) |
| American Indian or Alaska Native | 1 (1%) | 1 (1%) | 0 (0%) | 0 (0%) | 2 (1%) |
| Gender | |||||
| Female | 45 (65%) | 46 (66%) | 47 (65%) | 48 (68%) | 186 (66%) |
| Disease stage | |||||
| I/II | 22 (32%) | 21 (30%) | 24 (33%) | 23 (32%) | 90 (32%) |
| III/IV | 44 (64%) | 45 (64%) | 44 (61%) | 42 (59%) | 175 (62%) |
| Unknown | 3 (4%) | 4 (6%) | 4 (6%) | 6 (9%) | 17 (6%) |
| Baseline Fatigue Score | |||||
| 4-7 | 50 (73%) | 50 (71%) | 50 (70%) | 51 (72%) | 201 (71%) |
| 8-10 | 19 (28%) | 20 (29%) | 22 (31%) | 20 (28%) | 81 (29%) |
| Current chemotherapy | |||||
| Yes | 39 (57%) | 39 (56%) | 42 (58%) | 40 (56%) | 160 (57%) |
| Current radiation therapy | |||||
| Yes | 12 (17%) | 12 (17%) | 14 (19%) | 13 (18%) | 51 (18%) |
| Type of cancer | |||||
| Breast | 24 (35%) | 29 (41%) | 26 (36%) | 30 (42%) | 109 (39%) |
| Colon | 7 (10%) | 7 (10%) | 5 (7%) | 10 (14%) | 29 (10%) |
| Lung | 8 (12%) | 8 (11%) | 10 (14%) | 9 (13%) | 35 (12%) |
| Combination/unknown/other | 30 (44%) | 26 (37%) | 31 (43%) | 22 (31%) | 109 (39%) |
| Menopausal status | |||||
| Premenopausal | 7 (10%) | 7 (10%) | 7 (10%) | 6 (9%) | 27 (10%) |
| Postmenopausal | 34 (49%) | 35 (50%) | 37 (51%) | 39 (55%) | 145 (51%) |
| Hysterectomy without oophorectomy | 4 (6%) | 4 (6%) | 3 (4%) | 3 (4%) | 14 (5%) |
| Previous radiation therapy | |||||
| Yes | 23 (50%) | 16 (33%) | 19 (39%) | 14 (30%) | 72 (38%) |
| Previous chemotherapy | |||||
| Yes | 30 (65%) | 32 (67%) | 34 (69%) | 28 (60%) | 124 (65%) |
The primary endpoint of AUC activity interference (p=0.21) and secondary endpoint of usual fatigue (p=0.08) were not statistically significantly different between the collective ginseng arms versus placebo. However, planned subset analyses revealed a trend for greater positive effects with the highest dose of ginseng, 2,000 mg/day, over placebo, with the 2,000-mg arm having an effect size of over 50% times the standard deviation (Table 2).
Table 2.
Efficacy results: mean BFI AUC with 95% confidence intervals at 8 weeks (higher numbers are better)
| Endpoints | Placebo (95% CI) | 750mg ginseng (95% CI) | ES (%) | 1,000mg ginseng (95% CI) | ES (%) | 2,000mg ginseng (95% CI) | ES (%) | P value, SD |
|---|---|---|---|---|---|---|---|---|
| Activity interference | 460 (405-515) | 467 (409-524) | 4.2 | 480 (429-531) | 12 | 551 (506-597) | 56 | 0.08, 163 |
| Usual fatigue | 410 (360-460) | 425 (373-177) | 10 | 448 (408-488) | 27 | 491 (449-532) | 57 | 0.08, 143 |
95% CI 95% confidence interval, ES effect size, SD standard deviation
The mean change from baseline for all secondary endpoints at weeks 4 and 8 are shown in Tables 3 and 4, respectively. The mean scores for the vitality subscale of the SF-36 demonstrate that there was a greater effect on fatigue in the highest two doses (1,000 and 2,000 mg/day) of ginseng. An increase in quality of life as measured by the overall physical, emotional, mental, and spiritual well-being scales were also evident in the higher dose levels. This trend is not evident for improvement in sleep as measured by the PSQI, where the placebo arm showed the most improvement in sleep.
Table 3.
Change from baseline at week 4 (higher numbers are better)
| Endpoint | Placebo | 750mg ginseng | ES (%) | 1,000mg ginseng | ES (%) | 2,000mg ginseng | ES (%) | P value, SD |
|---|---|---|---|---|---|---|---|---|
| Vitality subscale | 4.1 | 3.5 | −3 | 13.6 | 50 | 10.4 | 33 | 0.06, 19 |
| Physical well being | −1.0 | −0.5 | 2 | 2.2 | 13 | 7.3 | 33 | 0.23, 25 |
| Emotional well being | −0.5 | −0.5 | 0 | 0.0 | 2 | 6.9 | 34 | 0.12, 22 |
| Mental well being | 2.4 | −1.0 | −15 | −6.1 | −37 | 4.7 | 10 | 0.06, 23 |
| Spiritual well being | 0.0 | −3.6 | −17 | −0.8 | −2 | 5.1 | 24 | 0.05, 21 |
| Social activity | 4.6 | −0.2 | −20 | 8.2 | 15 | 2.2 | −10 | 0.10, 24 |
| Sleep | 7.4 | 0.7 | −39 | 5.3 | −12 | 1.4 | −51 | 0.61, 17 |
ES effect size, SD standard deviation of change from baseline
Table 4.
Change from baseline at week 8 (higher numbers are better)
| Endpoint | Placebo | 750mg ginseng | ES (%) | 1,000mg ginseng | ES (%) | 2,000mg ginseng | ES (%) | P value, SD |
|---|---|---|---|---|---|---|---|---|
| Vitality subscale | 7.3 | 7.8 | 3 | 14.6 | 37 | 10.5 | 17 | 0.39, 19 |
| Physical well being | 5.6 | 5.3 | −1 | 12.0 | 26 | 6.5 | 4 | 0.65, 25 |
| Emotional well being | 3.6 | 3.0 | −3 | 6.1 | 11 | 9.4 | 26 | 0.47, 22 |
| Mental well being | 4.4 | 3.3 | −5 | 0.7 | −16 | 7.5 | 13 | 0.55, 23 |
| Spiritual well being | 2.9 | 3.9 | 5 | 2.9 | 0 | 6.3 | 16 | 0.47, 21 |
| Social activity | 10.3 | 9.2 | −4 | 16.1 | 26 | 9.4 | −4 | 0.51, 24 |
| Sleep | 12.9 | −1.4 | −8.4 | 10.1 | −16 | 2.9 | −59 | 0.10, 17 |
ES effect size, SD standard deviation of change from baseline
There were no statistically significant differences between the study arms, in the numbers of patients who withdrew from the study (Fig. 1). Investigator-reported toxicities were spread fairly evenly throughout the treatment arms, including placebo, as is shown in Table 5. In addition, there were no statistically significant differences by arm for the following self-reported side effects evaluated per the symptom experience diary: nausea, dizziness, nervousness, headache, trouble falling asleep, and trouble staying asleep.
Table 5.
CTC graded toxicities—numbers of patients
| Toxicity | Grade | Placebo | 750mg | 1,000mg | 2,000mg | P value |
|---|---|---|---|---|---|---|
| Agitation | Grade 1 (N) | 7 | 6 | 6 | 6 | 0.38 |
| Grade 2 (N) | 0 | 1 | 0 | 0 | ||
| Grade 3 (N) | 0 | 0 | 0 | 1 | ||
| Anxiety | Grade 1 (N) | 7 | 6 | 6 | 10 | 0.38 |
| Grade 2 (N) | 2 | 5 | 3 | 1 | ||
| Grade 3 (N) | 0 | 0 | 0 | 1 | ||
| Insomnia | Grade1 (N) | 28 | 22 | 19 | 19 | 0.57 |
| Grade 2 (N) | 2 | 5 | 5 | 3 | ||
| Grade 3 (N) | 2 | 4 | 1 | 1 | ||
| Nausea | Grade1 (N) | 13 | 14 | 17 | 16 | 0.64 |
| Grade 2 (N) | 2 | 8 | 8 | 5 | ||
| Grade 3 (N) | 1 | 2 | 1 | 1 | ||
| Vomiting | Grade1 (N) | 3 | 5 | 7 | 5 | 0.29 |
| Grade 2 (N) | 0 | 5 | 5 | 4 | ||
| Grade 3 (N) | 2 | 2 | 1 | 1 |
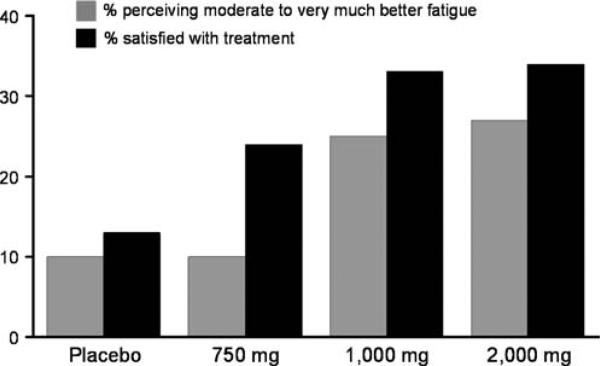
More than twice as many participants on the 1,000- and 2,000-mg/day doses of ginseng perceived a moderate to very much better improvement in fatigue at the end of the 8-week study period compared to those on placebo as shown in Fig. 2. In addition, more participants on the higher doses of ginseng were satisfied with their treatment for fatigue than those on placebo. These data were obtained while patients were still blinded to their treatment assignment.
Fig. 2.
Perception of benefit
Discussion
This pilot trial evaluated Wisconsin ginseng in a heterogeneous group of patients with cancer, ranging from those with localized resected disease to those with advanced incurable cancer. Overall, this study suggested that Wisconsin ginseng, at a dose of 750 mg/day, did not provide any benefit over that seen with a placebo. However, the two highest doses of Wisconsin ginseng (1,000 and 2,000 mg/day) did appear to decrease fatigue more than did a placebo, as measured by various scales of fatigue, vitality, and well being.
Perhaps the most compelling preliminary evidence from this current trial is that more than twice the patients who were on the higher doses of ginseng perceived a benefit for their fatigue, with 40% of patients who actually completed treatment with the 1,000- and 2,000-mg doses perceiving a moderate to very much better benefit as compared to 17% of the participants who were on the placebo arm. If the magnitude of benefit suggested by this pilot trial experience is observed in a larger more definitive trial, Wisconsin ginseng has the potential to help over 220,000 survivors based on conservative estimates from the literature that 30% to 60% of cancer survivors experience fatigue through at least 1 year past diagnosis and the fact that there was an estimated 1.4 million people diagnosed with cancer in 2006.
In this study, there were no significant toxicities apparent in the active arms compared to the incidences seen in those taking the placebo. This was true despite the fact that many of these participants had advanced disease and were taking cytotoxic treatment for their disease.
It is curious that there was not a clear trend for a linear dose effect on all endpoints. Further study is needed to clarify this issue. The reason for the variability in this may be due to the challenges in measuring fatigue or may be due to the heterogeneous sample. However, the effect sizes seen are consistent with the potential for meaningful clinical activity and, coupled with low toxicity, suggest that this herb be studied further.
One of the difficulties in studying herbal or dietary supplements is the lack of regulation and hence standardization of product. Plant products are affected by growing conditions including climate, soil, fertilizers, and pesticides [21, 37]. In addition, the harvesting and processing of plants can alter their biologic viability. In any one crop of ginseng, the percentage of ginsenosides can vary and can be further influenced by the manufacturing process. There is evidence to suggest that the strength of effect of ginseng may be related to the percentage of ginsenosides [49]. This principal was supported in this study in that there was suggestion of a dose response that is that with more ginsenosides, a greater effect was observed.
The precise pharmacokinetic activity of Wisconsin ginseng is not yet known [21]. Ginsenosides, the active constituents of ginseng, are steroidal saponins. Therefore, each ginsenoside, as well as other nonginsenoside constituents of ginseng such as polysaccharides and peptides, may have more than one effect on any given tissue [21]. There are animal model studies suggesting that Wisconsin ginseng can directly affect the brain through activating intracellular steroid receptors [21], regulating gamma-aminobutyric acid (GABAergic) neurotransmission [50] and/or providing neuroprotection in various brain regions [51]. The ability to modulate important neurotransmitters, such as dopamine, noradrenalin, serotonin, and GABA may be one explanation for how ginseng may help cancer-related fatigue.
Thus, given the multiple potential effects of Wisconsin ginseng and the present, very preliminary, data suggesting that the higher doses studied may be helpful in cancer-related fatigue, it appears worthwhile to more clearly and definitively evaluate the benefits of the effects of Wisconsin ginseng in a larger, more definitive, placebo-controlled clinical trial. Such a trial is actively recruiting patients in the North Central Cancer Treatment Group.
Acknowledgments
This study was conducted as a collaborative trial of the North Central Cancer Treatment Group and Mayo Clinic and was supported in part by Public Health Service grants CA-25224, CA-37404, CA-63849, CA-63848, CA-35267, CA-35431, CA-35269, CA-52352, CA-37417, CA-35415, CA-35103, CA-35119, CA-35195, CA-35448, CA-60276, CA-35101, CA-35113, CA-35103, and CA-35090.
Footnotes
Additional participating institutions include Iowa Oncology Research Association CCOP, Des Moines, IA 50314 (Roscoe F. Morton, M.D.); CentraCare Clinic, St. Cloud, MN 56301 (Harold E. Windschitl, M. D.); Sioux Community Cancer Consortium, Sioux Falls, SD 57105 (Loren K. Tschetter, M.D.); Siouxland Hematology–Oncology Associates, Sioux City, IA 51105 (Donald B. Wender, M.D.); Illinois Oncology Research Assn. CCOP, Peoria, IL 61615-7828 (John W. Kugler, M.D.); Montana Cancer Consortium, Billings, MT 59101 (Benjamin T. Marchello, M.D.); Meritcare Hospital CCOP, Fargo, ND 58122 (Preston D. Steen, M.D.); Toledo Community Hospital Oncology Program CCOP, Toledo, OH 43623 (Paul L. Schaefer, M.D.); Cancer Care Associates, Tulsa, OK 74136 (Mark R. Olsen, M.D.); Upstate Carolina CCOP, Spartanburg, SC 29303 (James D. Bearden, III, M.D.); Medical College of Georgia, Augusta, GA 30912 (Anand P. Jillella, M.D.); Geisinger Clinic & Medical Center CCOP, Danville, PA 17822 (Albert M. Bernath, Jr, M.D.); Mayo Clinic Jacksonville, Jacksonville, FL 32224 (Edith A. Perez, M.D.); Mayo Clinic Scottsdale, Scottsdale, AZ 85259-5404 (Tom R. Fitch, M.D.); Lehigh Valley Hospital, Allentown, PA 18103 (Suresh Nair, M.D.); and Hematology & Oncology of Dayton, Inc., Dayton, OH 45415 (Howard M. Gross, M.D.)
Contributor Information
Debra L. Barton, Mayo Clinic and Mayo Foundation, 200 First Street, SW, Rochester, MN 55905, USA
Gamini S. Soori, Missouri Valley Cancer Consortium, Omaha, NE 68106, USA
Brent A. Bauer, Mayo Clinic and Mayo Foundation, 200 First Street, SW, Rochester, MN 55905, USA
Jeff A. Sloan, Mayo Clinic and Mayo Foundation, 200 First Street, SW, Rochester, MN 55905, USA
Patricia A. Johnson, Carle Cancer Center CCOP, Urbana, IL 61801, USA
Cesar Figueras, Michigan Cancer Research Consortium, Ann Arbor, MI 48106, USA.
Steven Duane, Metro-Minnesota Community Clinical Oncology Program, St. Louis Park, MN 55416, USA.
Bassam Mattar, Wichita Community Clinical Oncology Program, Wichita, KS 67214-3882, USA.
Heshan Liu, Mayo Clinic and Mayo Foundation, 200 First Street, SW, Rochester, MN 55905, USA.
Pamela J. Atherton, Mayo Clinic and Mayo Foundation, 200 First Street, SW, Rochester, MN 55905, USA
Bradley Christensen, Mayo Clinic and Mayo Foundation, 200 First Street, SW, Rochester, MN 55905, USA.
Charles L. Loprinzi, Mayo Clinic and Mayo Foundation, 200 First Street, SW, Rochester, MN 55905, USA
References
- 1.NCCN Cancer Related Fatigue Clinical Practice Guidelines in Oncology V.1. 2003 doi: 10.6004/jnccn.2003.0029. http://www.nccn.org/ [DOI] [PubMed]
- 2.Mock V. Fatigue management, evidence and guidelines for practice. Cancer. 2001;92(6):1699–1707. doi: 10.1002/1097-0142(20010915)92:6+<1699::aid-cncr1500>3.0.co;2-9. doi:10.1002/1097-0142(20010915)92:6+<1699::AID-CNCR1500>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 3.Fulton C, Knowles G. Cancer fatigue. Eur J Cancer Care (Engl) 2000;9(3):167–171. [PubMed] [Google Scholar]
- 4.Gutstein HB. The biological basis of fatigue. Cancer. 2001;92(6):1678–1683. doi: 10.1002/1097-0142(20010915)92:6+<1678::aid-cncr1496>3.0.co;2-r. doi:10.1002/1097-0142(20010915)92:6+<1678::AID-CNCR1496>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 5.Berger AM, Farr LA, Kuhn BR, Fischer P, Agrawal S. Values of sleep/wake, activity/rest, circadian rhythms, and fatigue prior to adjuvant breast cancer chemotherapy. J Pain Symptom Manage. 2007;33(4):398–409. doi: 10.1016/j.jpainsymman.2006.09.022. doi:10.1016/j.jpainsymman.2006.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lawrence DP, Kupelnick B, Miller K, Devine D, Lau J. Evidence report on the occurrence, assessment, and treatment of fatigue in cancer patients. J Natl Cancer Inst Monogr. 2004;32:40–50. doi: 10.1093/jncimonographs/lgh027. doi:10.1093/jncimonographs/lgh027. [DOI] [PubMed] [Google Scholar]
- 7.Bower JE, Ganz PA, Desmond KA, Rowland KA, Meyerowitz JH, Belin BE, Thomas R. Fatigue in breast cancer survivors: occurrences, correlates, and impact on quality of life. J Clin Oncol. 2000;18:743–753. doi: 10.1200/JCO.2000.18.4.743. [DOI] [PubMed] [Google Scholar]
- 8.Bower JE, Ganz PA, Desmond KA, Bernaards C, Rowland JH, Meyerowitz BE, Belin TR. Fatigue in long-term breast carcinoma survivors. A longitudinal investigation. Cancer. 2006;106:751–758. doi: 10.1002/cncr.21671. doi:10.1002/cncr.21671. [DOI] [PubMed] [Google Scholar]
- 9.Baker F, Denniston M, Smith T, West MM. Adult cancer survivors: how are they faring. Cancer. 2005;104(Suppl 11):2565–2576. doi: 10.1002/cncr.21488. doi:10.1002/cncr.21488. [DOI] [PubMed] [Google Scholar]
- 10.Mitchell SA, Beck SL, Edward Hood L, Moore K, Tanner ER. Putting evidence into practice: evidence-based interventions of fatigue during and following cancer and its treatment. Clin J Oncol Nurs. 2007;11(1):99–113. doi: 10.1188/07.CJON.99-113. doi:10.1188/07.CJON.99-113. [DOI] [PubMed] [Google Scholar]
- 11.Mitchell SA, Berger AM. Cancer-related fatigue: the evidence base for assessment and management. Cancer J. 2006;12:374–387. doi: 10.1097/00130404-200609000-00007. doi:10.1097/00130404-200609000-00007. [DOI] [PubMed] [Google Scholar]
- 12.Mock V, Pickett M, Ropka M, Lin E, Grimm P, Rhodes V, McDaniel R, Grimm PM, Krumm S, McCorkle R. Fatigue, physical functioning, emotional distress, and quality of life outcomes of a walking intervention during breast cancer treatment. Cancer Pract. 2001;9:119–127. doi: 10.1046/j.1523-5394.2001.009003119.x. doi:10.1046/j.1523-5394.2001.009003119.x. [DOI] [PubMed] [Google Scholar]
- 13.Schwartz AL, Mori M, Gao R, Nail R, King LM, Marjorie E. Exercise reduces daily fatigue in women with breast cancer receiving chemotherapy. Med Sci Sports Exerc. 2001;33:718–723. doi: 10.1097/00005768-200105000-00006. doi:10.1097/00005768-200105000-00006. [DOI] [PubMed] [Google Scholar]
- 14.Stone P, Richards M, Hardy J. Fatigue in patients with cancer. Eur J Cancer. 1998;34(11):1670–1676. doi: 10.1016/s0959-8049(98)00167-1. doi:10.1016/S0959-8049(98)00167-1. [DOI] [PubMed] [Google Scholar]
- 15.Portenoy RK, Itri LM. Cancer-related fatigue: guidelines for evaluation and management. Oncologist. 1999;4:1–10. [PubMed] [Google Scholar]
- 16.Burks TF. New agents for the treatment of cancer-related fatigue. Cancer. 2001;92(Suppl 6):1714–1718. doi: 10.1002/1097-0142(20010915)92:6+<1714::aid-cncr1502>3.0.co;2-n. doi:10.1002/1097-0142(20010915)92:6+<1714::AID-CNCR1502>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- 17.Bruera E, Driver L, Barnes EA, Willey J, Shen L, Palmer JL, Escalante C. Patient-controlled methylphenidate for the management of fatigue in patients with advanced cancer: a preliminary report. J Clin Oncol. 2003;21(23):4439–4443. doi: 10.1200/JCO.2003.06.156. doi:10.1200/JCO.2003.06.156. [DOI] [PubMed] [Google Scholar]
- 18.Bruera E, Valero V, Driver L, Shen L, Willey J, Zhang T, Palmer JL. Patient-controlled methylphenidate for cancer fatigue: a double-blind, randomized, placebo-controlled trial. J Clin Oncol. 2006;24(13):2073–2078. doi: 10.1200/JCO.2005.02.8506. doi:10.1200/JCO.2005.02.8506. [DOI] [PubMed] [Google Scholar]
- 19.Bruera E, Osta E, Vlaero V, Driver L, Palmer J, Pei B, Shen L, Poulter V. Donepezil for cancer-related fatigue: a double-blind, randomized, placebo-controlled study. J Clin Oncol. 2007;25(23):3475–3481. doi: 10.1200/JCO.2007.10.9231. Part I II 493s(9003) [DOI] [PubMed] [Google Scholar]
- 20.Stankoff B, Waubant E, Confavreau C, Edan G, Debouverie M, Rumback L, Moreau Tl, Pelletier J, Lubetzki C, Clanet M, French Modafinil Study Group Modafinil for fatigue in MS: a randomized placebo-controlled double blind study. Neurology. 2005;64:1139–1143. doi: 10.1212/01.WNL.0000158272.27070.6A. [DOI] [PubMed] [Google Scholar]
- 21.Attele AS, Wu JA, Yuan C. Ginseng pharmacology. Biochem Pharmacol. 1999;58:1685–1693. doi: 10.1016/s0006-2952(99)00212-9. doi:10.1016/S0006-2952(99)00212-9. [DOI] [PubMed] [Google Scholar]
- 22.Ginseng (American ginseng, Asian ginseng, Chinese ginseng, Korean red ginseng, Panax ginseng: Panax spp. Including P. ginseng C.C. Meyer and P. quinquefolius L., excluding Eleutherococcus senticosus). [4 April 2007]; http://natuaralstandard.com/monographys/herssupplements/ginseng.asp.
- 23.Cui J. Identification and quantification of ginsenosides in various commercial ginseng preparations. Eur J Pharm Sci. 1995;3:77–85. doi:10.1016/0928-0987(94)00077-D. [Google Scholar]
- 24.Tadano T, Nakagawasai O, Niijima F, Tan-No K, Kisara K. The effects of traditional tonics on fatigue in mice differ from those of the antidepressant imipramine: a pharmacological and behavioral study. Am J Chin Med. 2000;28(1):97–104. doi: 10.1142/S0192415X0000012X. doi:10.1142/S0192415X0000012X. [DOI] [PubMed] [Google Scholar]
- 25.Banerjee U, Izquierdo JA. Antistress and antifatigue properties of Panax ginseng: comparison with piracetam. Acta Physiol Lat Am. 1982;32(4):277–285. [PubMed] [Google Scholar]
- 26.Marasco CA, Ruiz VR, Villagomex SA, Infante BC. Double-blind study of a multivitamin complex supplemented with ginseng extract. Drugs Exp Clin Res XXII. 1996;6:323–329. [PubMed] [Google Scholar]
- 27.Younus J, Collins A, Wang X, Saunders M, Manuel J, Freake C, Defen P. A double blind placebo controlled pilot study to evaluate the effect of ginseng on fatigue and quality of life in adult chemo-naïve cancer patients. J Clin Oncol. 2003;22:733. abstract # 2947. [Google Scholar]
- 28.Asafu-Adjaye EB, Wong SK. Determination of ginsenosides (ginseng saponins) in dry root powder from Panax ginseng, Panax quinquefolius, and selected commercial products by liquid chromatography: interlaboratory study. J AOAC Int. 2003;86(6):1112–1123. [PubMed] [Google Scholar]
- 29.Wang X, Sakuma T, Asafu-Adjaye E, Shiu GK. Determination of ginsenosides in plant extracts from Panax ginseng and Panax quinquefolius L. by LC/MS/MS. Anal Chem. 1999;8:1579–1584. doi: 10.1021/ac980890p. doi:10.1021/ac980890p. [DOI] [PubMed] [Google Scholar]
- 30.Xie JT, Maleckar SA, Yuan CS. Is ginseng free from adverse effects? In: Yuan CS, Bieber E, editors. Complementary and alternative medicine. CRC; Boca Raton: 2003. chapter 17. [Google Scholar]
- 31.Wang LC, Lee TF. Effect of ginseng saponins on exercise performance in non-trained rats. Planta Med. 1998;64(2):130–133. doi: 10.1055/s-2006-957389. doi:10.1055/s-2006-957389. [DOI] [PubMed] [Google Scholar]
- 32.Vuksan V, Sievenpiper JL, Koo VY, Francis T, Beljan-Zdravkovic U, Xu Z, Vidgen E. American ginseng (Panax quinquefolius L) reduces postprandial glycemia in nondiabetic subjects and subjects with type 2 diabetes mellitus. Arch Intern Med. 2000;160(7):1009–1013. doi: 10.1001/archinte.160.7.1009. doi:10.1001/archinte.160.7.1009. [DOI] [PubMed] [Google Scholar]
- 33.Sotaniemi EA, Haapakoski E, Rautio A. Ginseng therapy in non-insulin-dependent diabetic patients. Diabetes Care. 1995;18(10):1373–1375. doi: 10.2337/diacare.18.10.1373. doi:10.2337/diacare.18.10.1373. [DOI] [PubMed] [Google Scholar]
- 34.King ML, Adler SR, Murphy LL. Extraction-dependent effects of American ginseng (Panax quinquefolium) on human breast cancer cell proliferation and estrogen receptor activation. Integr Cancer Ther. 2006;5(3):236–243. doi: 10.1177/1534735406291341. doi:10.1177/1534735406291341. [DOI] [PubMed] [Google Scholar]
- 35.King ML, Murphy LL. American ginseng (Panax quinquefolius L.) extract alters mitogen-activated protein kinase cell signaling and inhibits proliferation of MCF-7 cells. J Exp Ther Oncol. 2007;6:147–155. [PubMed] [Google Scholar]
- 36.Duda RB, Zhong Y, Navas V, Li MZ, Toy BR, Alavarez JG. American ginseng and breast cancer therapeutic agents synergistically inhibit MCF-7 breast cancer cell growth. J Surg Oncol. 1999;72(4):230–239. doi: 10.1002/(sici)1096-9098(199912)72:4<230::aid-jso9>3.0.co;2-2. doi:10.1002/(SICI)1096-9098(199912)72:4<230::AID-JSO9>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 37.Bahrke MS, Morgan WR. Evaluation of the ergogenic properties of ginseng: an update. Sports Med. 2000;29(2):113–133. doi: 10.2165/00007256-200029020-00004. doi:10.2165/00007256-200029020-00004. [DOI] [PubMed] [Google Scholar]
- 38. [November 2007];Ginseng Board of Wisconsin. http://www.ginsengboard.com.
- 39.Mendoza TR, Wang XS, Cleeland CS, Morrissey M, Johnson BA, Wendt JK, Huber SL. The rapid assessment of fatigue severity in cancer patient's use of the Brief Fatigue Inventory. Cancer. 1999;85:1186–1196. doi: 10.1002/(sici)1097-0142(19990301)85:5<1186::aid-cncr24>3.0.co;2-n. doi:10.1002/(SICI)1097-0142(19990301)85:5<1186::AID-CNCR24>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- 40.Ware JE, Sherbourne CD. The MOS 36-Item Short-Form Health Survey (SF-36). Conceptual framework and item selection. Med Care. 1992;30(6):473–481. doi:10.1097/00005650-199206000-00002. [PubMed] [Google Scholar]
- 41.Buysse D, Reynolds C, Monk T, Berman S, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193–213. doi: 10.1016/0165-1781(89)90047-4. doi:10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 42.Guyatt GH, Osoba D, Wu AW, Wyrwich KW, Norman GR, Sloan JA, Clinical Significance Consensus Meeting Group Methods to explain the clinical significance of health status measures. Mayo Clin Proc. 2002;77:371–383. doi: 10.4065/77.4.371. doi:10.4065/77.4.371. [DOI] [PubMed] [Google Scholar]
- 43.Hyland ME, Sodergren SC. Development of a new type of global quality of life scale and comparison and preference for 12 global scales. Qual Life Res. 1996;5(5):469–480. doi: 10.1007/BF00540019. doi:10.1007/BF00540019. [DOI] [PubMed] [Google Scholar]
- 44.Sloan A, O'Fallon JR, Suman VJ. Incorporating quality of life measurement into oncology clinical trials. Proc Am Stat Assoc. 1998:282–287. [Google Scholar]
- 45.Wewers ME, Lowe NK. A critical review of visual analogue scales in the measurement of clinical phenomena. Res Nurs Health. 1990;13:227–236. doi: 10.1002/nur.4770130405. doi:10.1002/nur.4770130405. [DOI] [PubMed] [Google Scholar]
- 46.Grunberg SM, Groshen S, Steingass S, Zaretsky S, Meyerowitz B. Comparison of conditional quality of life terminology and visual analogue scale measurements. Qual Life Res. 1996;5:65–72. doi: 10.1007/BF00435970. doi:10.1007/BF00435970. [DOI] [PubMed] [Google Scholar]
- 47.Cohen J. Statistical power analysis for the behavioral sciences. Erlbaum; Hillsdale, NJ: 1988. [Google Scholar]
- 48.Lipsitz SR, Fitzmaurice GM, Orav EJ, Laird NM. Performance of generalized estimating equations in practical situations. Biometrics. 1994;50:270–278. doi:10.2307/2533218. [PubMed] [Google Scholar]
- 49.Sievenpiper JL, Arnason JT, Leiter LA, Vuksan V. Variable effects of American ginseng: a batch of American ginseng (Panax quinquefolius L.) with a depressed ginsenoside profile does not affect postprandial glycemia. Eur J Clin Nutr. 2003;57(2):243–248. doi: 10.1038/sj.ejcn.1601550. doi:10.1038/sj.ejcn.1601550. [DOI] [PubMed] [Google Scholar]
- 50.Yuan C, Attele AS, Wu JA, Liu D. Modulation of American ginseng on brainstem GABAergic effects in rats. J Ethnopharmacol. 1998;62:215–222. doi: 10.1016/s0378-8741(98)00066-x. doi:10.1016/S0378-8741(98)00066-X. [DOI] [PubMed] [Google Scholar]
- 51.Lian XY, Zhang Z, Stringer JL. Protective effects of ginseng components in a rodent model of neurodegeneration. Ann Neurol. 2005;57(5):642–648. doi: 10.1002/ana.20450. doi:10.1002/ana.20450. [DOI] [PubMed] [Google Scholar]