Abstract
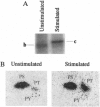
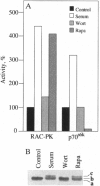
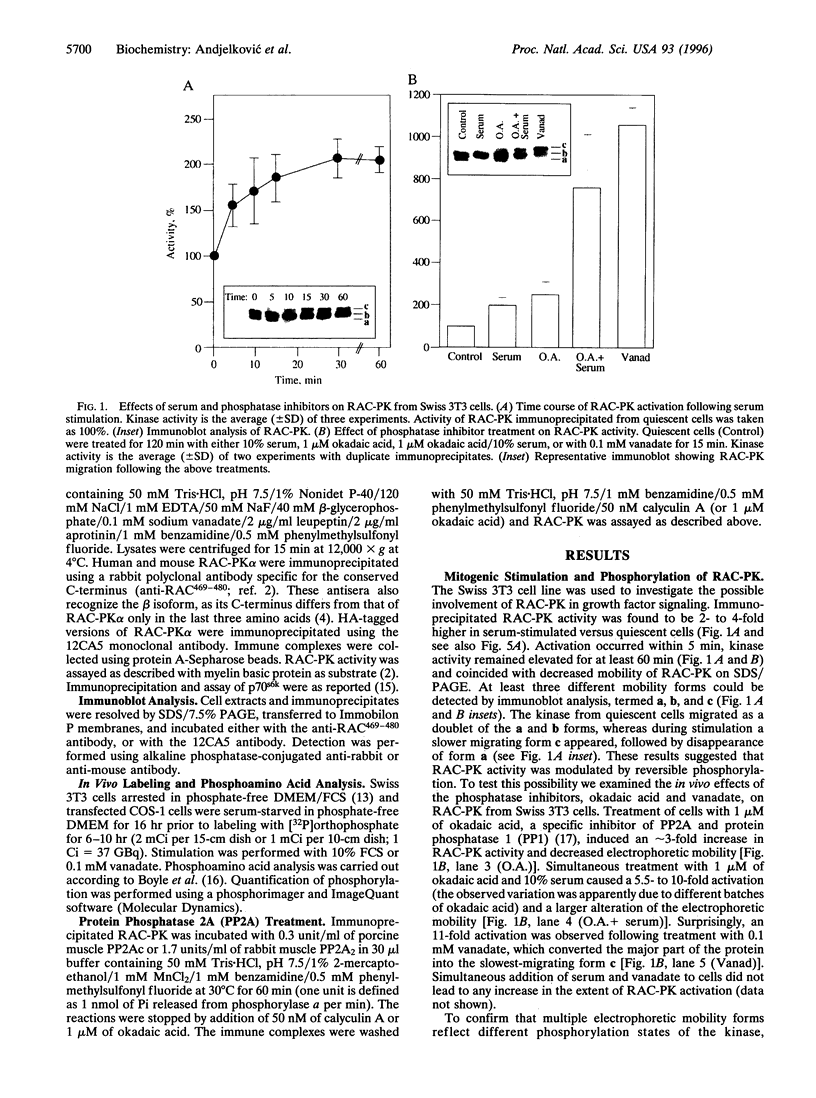
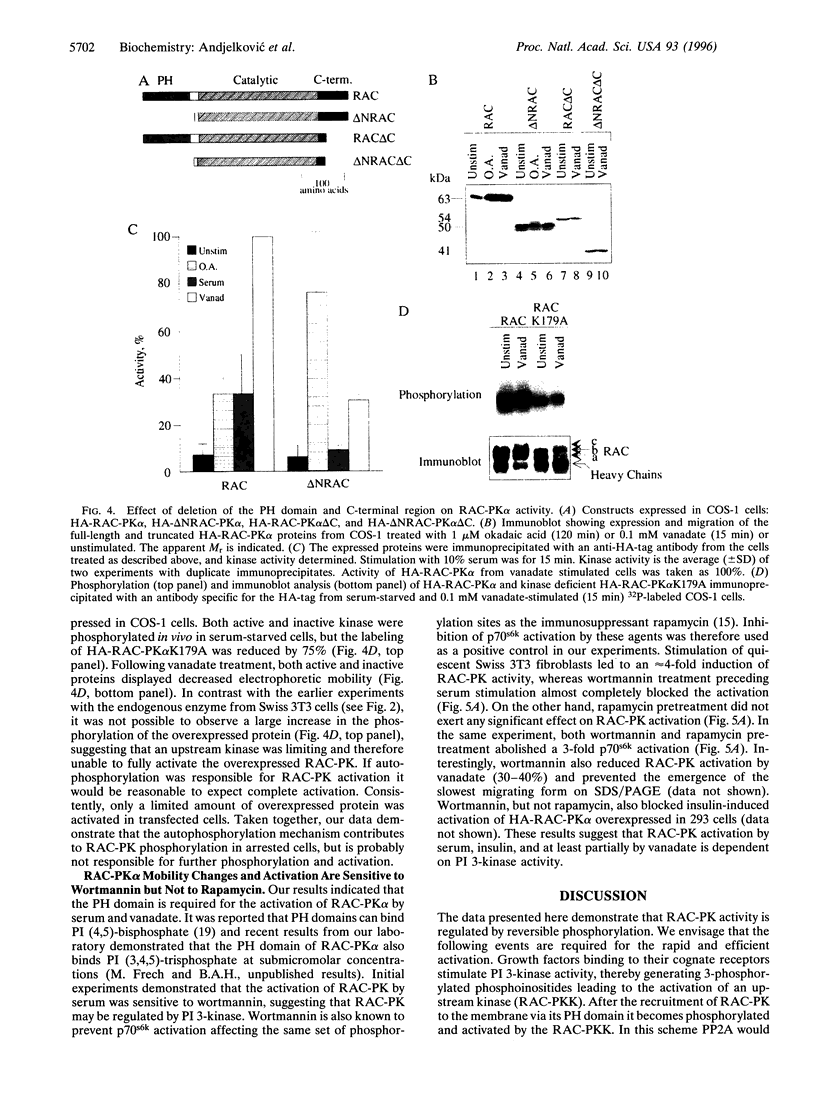
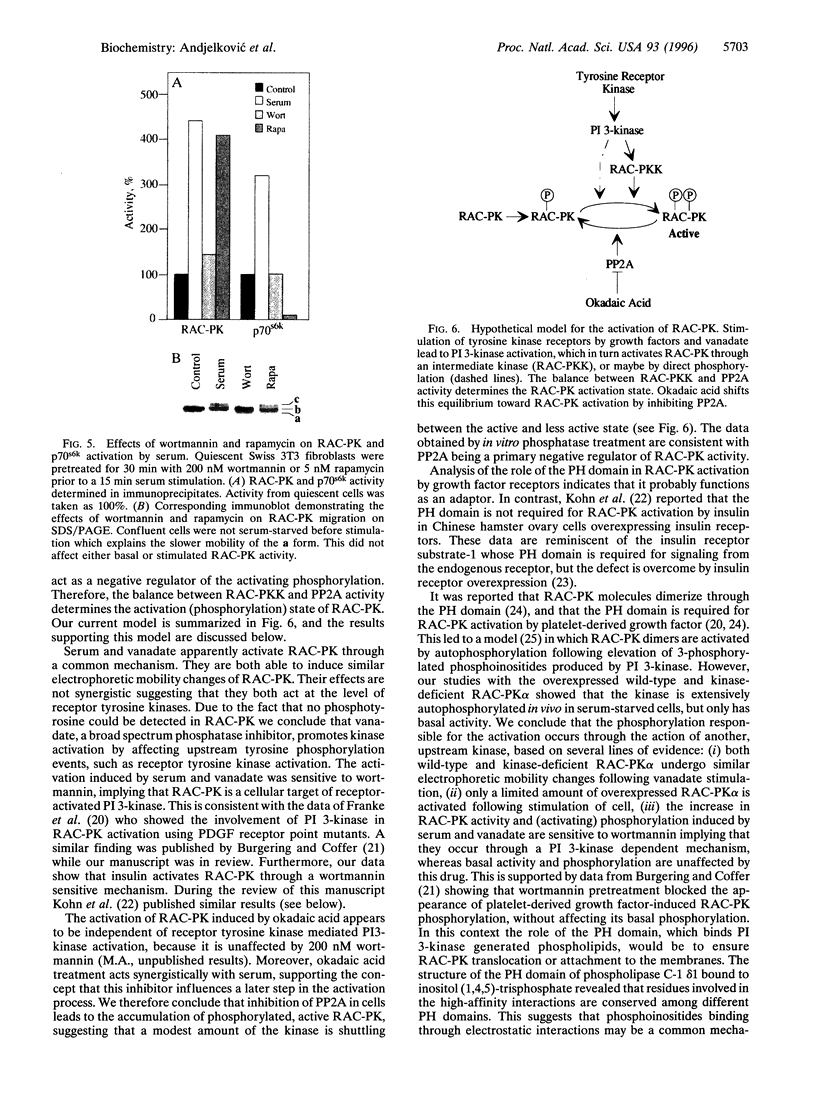
Treatment of quiescent Swiss 3T3 fibroblasts with serum, or with the phosphatase inhibitors okadaic acid and vanadate, induced a 2- to 11-fold activation of the serine/ threonine RAC protein kinase (RAC-PK). Kinase activation was accompanied by decreased mobility of RAC-PK on SDS/PAGE such that three electrophoretic species (a to c) of the kinase were detected by immunoblot analysis, indicative of differentially phosphorylated forms. Addition of vanadate to arrested cells increased the RAC-PK phosphorylation level 3-to 4-fold. Unstimulated RAC-PK was phosphorylated predominantly on serine, whereas the activated kinase was phosphorylated on both serine and threonine residues. Treatment of RAC-PK in vitro with protein phosphatase 2A led to kinase inactivation and an increase in electrophoretic mobility. Deletion of the N-terminal region containing the pleckstrin homology domain did not affect RAC-PK activation by okadaic acid, but it reduced vanadate-stimulated activity and also blocked the serum-induced activation. Deletion of the serine/threonine rich C-terminal region impaired both RAC-PKalpha basal and vanadate-stimulated activity. Studies using a kinase-deficient mutant indicated that autophosphorylation is not involved in RAC-PKalpha activation. Stimulation of RAC-PK activity and electrophoretic mobility changes induced by serum were sensitive to wortmannin. Taken together the results suggest that RAC-PK is a component of a signaling pathway regulated by phosphatidylinositol (PI) 3-kinase, whose action is required for RAC-PK activation by phosphorylation.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahmed N. N., Franke T. F., Bellacosa A., Datta K., Gonzalez-Portal M. E., Taguchi T., Testa J. R., Tsichlis P. N. The proteins encoded by c-akt and v-akt differ in post-translational modification, subcellular localization and oncogenic potential. Oncogene. 1993 Jul;8(7):1957–1963. [PubMed] [Google Scholar]
- Andjelković M., Jones P. F., Grossniklaus U., Cron P., Schier A. F., Dick M., Bilbe G., Hemmings B. A. Developmental regulation of expression and activity of multiple forms of the Drosophila RAC protein kinase. J Biol Chem. 1995 Feb 24;270(8):4066–4075. doi: 10.1074/jbc.270.8.4066. [DOI] [PubMed] [Google Scholar]
- Bellacosa A., Testa J. R., Staal S. P., Tsichlis P. N. A retroviral oncogene, akt, encoding a serine-threonine kinase containing an SH2-like region. Science. 1991 Oct 11;254(5029):274–277. doi: 10.1126/science.254.5029.274. [DOI] [PubMed] [Google Scholar]
- Boyle W. J., van der Geer P., Hunter T. Phosphopeptide mapping and phosphoamino acid analysis by two-dimensional separation on thin-layer cellulose plates. Methods Enzymol. 1991;201:110–149. doi: 10.1016/0076-6879(91)01013-r. [DOI] [PubMed] [Google Scholar]
- Burgering B. M., Coffer P. J. Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature. 1995 Aug 17;376(6541):599–602. doi: 10.1038/376599a0. [DOI] [PubMed] [Google Scholar]
- Cheng J. Q., Godwin A. K., Bellacosa A., Taguchi T., Franke T. F., Hamilton T. C., Tsichlis P. N., Testa J. R. AKT2, a putative oncogene encoding a member of a subfamily of protein-serine/threonine kinases, is amplified in human ovarian carcinomas. Proc Natl Acad Sci U S A. 1992 Oct 1;89(19):9267–9271. doi: 10.1073/pnas.89.19.9267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cross D. A., Alessi D. R., Cohen P., Andjelkovich M., Hemmings B. A. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature. 1995 Dec 21;378(6559):785–789. doi: 10.1038/378785a0. [DOI] [PubMed] [Google Scholar]
- Datta K., Franke T. F., Chan T. O., Makris A., Yang S. I., Kaplan D. R., Morrison D. K., Golemis E. A., Tsichlis P. N. AH/PH domain-mediated interaction between Akt molecules and its potential role in Akt regulation. Mol Cell Biol. 1995 Apr;15(4):2304–2310. doi: 10.1128/mcb.15.4.2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Downward J. Signal transduction. A target for PI(3) kinase. Nature. 1995 Aug 17;376(6541):553–554. doi: 10.1038/376553a0. [DOI] [PubMed] [Google Scholar]
- Ellis L., Clauser E., Morgan D. O., Edery M., Roth R. A., Rutter W. J. Replacement of insulin receptor tyrosine residues 1162 and 1163 compromises insulin-stimulated kinase activity and uptake of 2-deoxyglucose. Cell. 1986 Jun 6;45(5):721–732. doi: 10.1016/0092-8674(86)90786-5. [DOI] [PubMed] [Google Scholar]
- Ferguson K. M., Lemmon M. A., Schlessinger J., Sigler P. B. Structure of the high affinity complex of inositol trisphosphate with a phospholipase C pleckstrin homology domain. Cell. 1995 Dec 15;83(6):1037–1046. doi: 10.1016/0092-8674(95)90219-8. [DOI] [PubMed] [Google Scholar]
- Franke T. F., Tartof K. D., Tsichlis P. N. The SH2-like Akt homology (AH) domain of c-akt is present in multiple copies in the genome of vertebrate and invertebrate eucaryotes. Cloning and characterization of the Drosophila melanogaster c-akt homolog Dakt1. Oncogene. 1994 Jan;9(1):141–148. [PubMed] [Google Scholar]
- Franke T. F., Yang S. I., Chan T. O., Datta K., Kazlauskas A., Morrison D. K., Kaplan D. R., Tsichlis P. N. The protein kinase encoded by the Akt proto-oncogene is a target of the PDGF-activated phosphatidylinositol 3-kinase. Cell. 1995 Jun 2;81(5):727–736. doi: 10.1016/0092-8674(95)90534-0. [DOI] [PubMed] [Google Scholar]
- Han J. W., Pearson R. B., Dennis P. B., Thomas G. Rapamycin, wortmannin, and the methylxanthine SQ20006 inactivate p70s6k by inducing dephosphorylation of the same subset of sites. J Biol Chem. 1995 Sep 8;270(36):21396–21403. doi: 10.1074/jbc.270.36.21396. [DOI] [PubMed] [Google Scholar]
- Harlan J. E., Hajduk P. J., Yoon H. S., Fesik S. W. Pleckstrin homology domains bind to phosphatidylinositol-4,5-bisphosphate. Nature. 1994 Sep 8;371(6493):168–170. doi: 10.1038/371168a0. [DOI] [PubMed] [Google Scholar]
- Haslam R. J., Koide H. B., Hemmings B. A. Pleckstrin domain homology. Nature. 1993 May 27;363(6427):309–310. doi: 10.1038/363309b0. [DOI] [PubMed] [Google Scholar]
- Hunter T. When is a lipid kinase not a lipid kinase? When it is a protein kinase. Cell. 1995 Oct 6;83(1):1–4. doi: 10.1016/0092-8674(95)90225-2. [DOI] [PubMed] [Google Scholar]
- Ingley E., Hemmings B. A. Pleckstrin homology (PH) domains in signal transduction. J Cell Biochem. 1994 Dec;56(4):436–443. doi: 10.1002/jcb.240560403. [DOI] [PubMed] [Google Scholar]
- Jones P. F., Jakubowicz T., Hemmings B. A. Molecular cloning of a second form of rac protein kinase. Cell Regul. 1991 Dec;2(12):1001–1009. doi: 10.1091/mbc.2.12.1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones P. F., Jakubowicz T., Pitossi F. J., Maurer F., Hemmings B. A. Molecular cloning and identification of a serine/threonine protein kinase of the second-messenger subfamily. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4171–4175. doi: 10.1073/pnas.88.10.4171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohn A. D., Kovacina K. S., Roth R. A. Insulin stimulates the kinase activity of RAC-PK, a pleckstrin homology domain containing ser/thr kinase. EMBO J. 1995 Sep 1;14(17):4288–4295. doi: 10.1002/j.1460-2075.1995.tb00103.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKintosh C., MacKintosh R. W. Inhibitors of protein kinases and phosphatases. Trends Biochem Sci. 1994 Nov;19(11):444–448. doi: 10.1016/0968-0004(94)90127-9. [DOI] [PubMed] [Google Scholar]
- Morgan D. O., De Bondt H. L. Protein kinase regulation: insights from crystal structure analysis. Curr Opin Cell Biol. 1994 Apr;6(2):239–246. doi: 10.1016/0955-0674(94)90142-2. [DOI] [PubMed] [Google Scholar]
- Myers M. G., Jr, Grammer T. C., Brooks J., Glasheen E. M., Wang L. M., Sun X. J., Blenis J., Pierce J. H., White M. F. The pleckstrin homology domain in insulin receptor substrate-1 sensitizes insulin signaling. J Biol Chem. 1995 May 19;270(20):11715–11718. doi: 10.1074/jbc.270.20.11715. [DOI] [PubMed] [Google Scholar]
- Pearson R. B., Dennis P. B., Han J. W., Williamson N. A., Kozma S. C., Wettenhall R. E., Thomas G. The principal target of rapamycin-induced p70s6k inactivation is a novel phosphorylation site within a conserved hydrophobic domain. EMBO J. 1995 Nov 1;14(21):5279–5287. doi: 10.1002/j.1460-2075.1995.tb00212.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Posner B. I., Faure R., Burgess J. W., Bevan A. P., Lachance D., Zhang-Sun G., Fantus I. G., Ng J. B., Hall D. A., Lum B. S. Peroxovanadium compounds. A new class of potent phosphotyrosine phosphatase inhibitors which are insulin mimetics. J Biol Chem. 1994 Feb 11;269(6):4596–4604. [PubMed] [Google Scholar]
- Saraste M., Hyvönen M. Pleckstrin homology domains: a fact file. Curr Opin Struct Biol. 1995 Jun;5(3):403–408. doi: 10.1016/0959-440x(95)80104-9. [DOI] [PubMed] [Google Scholar]
- Susa M., Thomas G. Identical Mr 70,000 S6 kinase is activated biphasically by epidermal growth factor: a phosphopeptide that characterizes the late phase. Proc Natl Acad Sci U S A. 1990 Sep;87(18):7040–7044. doi: 10.1073/pnas.87.18.7040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J., Wang L., Schwartz J., Bond R. W., Bishop W. R. Phosphorylation of Thr642 is an early event in the processing of newly synthesized protein kinase C beta 1 and is essential for its activation. J Biol Chem. 1994 Jul 29;269(30):19578–19584. [PubMed] [Google Scholar]