Abstract
OBJECTIVE:
To evaluate whether the pathophysiology of shock syndromes can be better understood by comparing central hemodynamics with kinetic data on fluid and electrolyte shifts.
METHODS:
We studied the dilutional hyponatremic shock that developed in response to overhydration with electrolyte-free irrigating fluid – the so-called ‘transurethral resection syndrome' – by comparing cardiac output, arterial pressures, and volume kinetic parameters in 17 pigs that were administered 150 ml/kg of either 1.5% glycine or 5% mannitol by intravenous infusion over 90 minutes.
RESULTS:
Natriuresis appeared to be the key factor promoting hypovolemic hypotension 15–20 minutes after fluid administration ended. Excessive sodium excretion, due to osmotic diuresis caused by the irrigant solutes, was associated with high estimates of the elimination rate constant (k10) and low or negative estimates of the rate constant describing re-distribution of fluid to the plasma after translocation to the interstitium (k21). These characteristics indicated a high urinary flow rate and the development of peripheral edema at the expense of plasma volume and were correlated with reductions in cardiac output. The same general effects of natriuresis were observed for both irrigating solutions, although the volume of infused 1.5% glycine had a higher tendency to enter the intracellular fluid space.
CONCLUSION:
Comparisons between hemodynamics and fluid turnover showed a likely sequence of events that led to hypovolemia despite intravenous administration of large amounts of fluid.
Keywords: Kinetic Model, Shock Syndrome, Electrolyte, Hypovolemia, Hyponatremia
INTRODUCTION
A bewildering volume overload syndrome, called “transurethral resection (TUR) syndrome,” is caused by the absorption of >3 liters of electrolyte-free irrigating fluid during transurethral operations, usually transurethral resection of the prostate (TURP) ,1-3. In 1956, the clinical picture was summarized as “dilutional hyponatremic shock,” due to the severe arterial hypotension and even cardiovascular collapse that occurs postoperatively ,4-7. Although the incidence of this syndrome is decreasing, complete or incomplete forms of TUR syndrome occur in up to 5% percent of patients in some case series of TURPs performed with monopolar electrocautery ,8-10.
Bradycardia, hypotension, and nausea are the most typical early signs of mild and moderately severe forms of TUR syndrome ,11-13. The hypotension is counterintuitive, as fluid overload is the cause of the syndrome. Medical textbooks typically suggest that infusion fluids be withheld and diuretics given, while others have claimed that colloid volume loading is the appropriate remedy 5,14. The majority of authorities agree that slow administration of hypertonic saline is indicated in severe cases, at least when the cardiovascular situation has stabilized.
The aim of the present evaluation was to provide a better explanation of the pathophysiology of this shock-like hypotension syndrome. We hoped to find an approach that can also be used in the study of other syndromes, such as diabetic ketoacidosis and sepsis. For this purpose, the relationship between central hemodynamics and the kinetics of a large volume of irrigating fluid, consisting of either 1.5% glycine or 5% mannitol, was examined in a series of infusion experiments in pigs. Studies of volume kinetics use pharmacokinetic compartment models to describe the distribution and elimination of infusion fluids 15,16. The version of the model used here allows the description of an uneven distribution of fluid by having two rate constants instead of a single one to describe the distribution of fluid between the plasma and interstitial fluid 17,18.
MATERIALS AND METHODS
After obtaining approval from the local Ethics Committee for Experiments on Animals, 17 pigs between 8 and 10 weeks of age, with a mean body weight of 22 (range 20–24) kg, were studied under general anesthesia using invasive measurements of hemodynamic parameters, blood samples, and urine output.
Following a minimum acclimatization period of 3 days in an in-laboratory sty, each pig was premedicated with an intramuscular injection of 20-30 mg of diazepam, 400-600 mg of ketamine, and 1 mg of atropine. Subsequent surgical anesthesia, monitored by repeated pinching of the skin between the hoofs, was then maintained by thiopental, ketamine, and diazepam. The key anesthetic was ketamine, as this drug does not depress the hemodynamic responses, which are very similar in pig and man. The animals were endotracheally intubated and ventilated to normocapnia using a Servo 900C (Siemens-Elema, Solna, Sweden).
Surgical preparation and measurements
Surgical preparation included dissection of the jugular vein and insertion of a 5.5 F pulmonary artery catheter (Abbott Critical Care Systems, Mountain View, CA) and a central venous catheter. A catheter was also introduced into the femoral artery via an incision in the left groin to monitor the pressure and to sample blood, while a urinary bladder cystostomy was established to monitor urinary excretion. After a 30-min rest to reach a hemodynamic steady state, the animals were randomly allocated to receive an intravenous infusion of 150 ml/kg of either glycine 1.5% (n = 9) or mannitol 5% (n = 8; Baxter Healthcare, Thetford, UK) over 90 min with the aid of infusion pumps.
The measurements comprised cardiac output (CO) and arterial pressures, which were taken at 0, 5, 10, 15, 20, 30, 40, 50, 60, 70, 80, 90, 100, 110, and 120 min from the start of the infusion. CO was taken as the mean of three successive injections of ice-cold saline. The blood hemoglobin (B-Hb) concentration was measured at the same times, as well as every 2.5 min during the first 20 min of the study, and then every 5 min up to 80 min. The urine volume was measured, and samples were taken for assessment of the serum and urinary sodium concentrations every 10th minute throughout the study.
B-Hb and the serum and urine sodium concentrations were measured using Multi-Species software on a Technicon H1 (Bayer Diagnostica, Tarrytown, NY). At 120 min, the pigs were sacrificed using an intracardiac injection of pentothal and ethanol. Crude data from the experiments, as well as further details about the anesthesia and the surgical methods, have been published by Sandfeldt et al. 19.
Volume kinetics
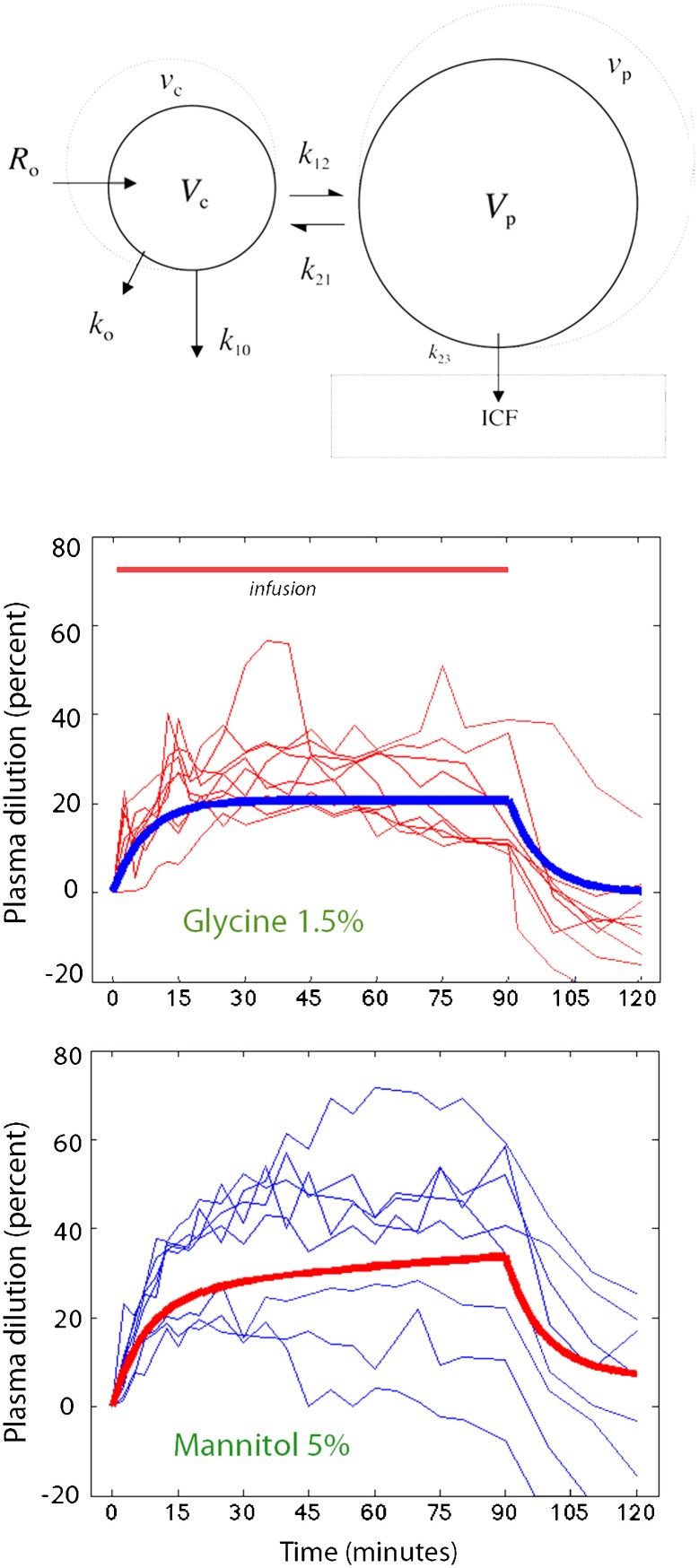
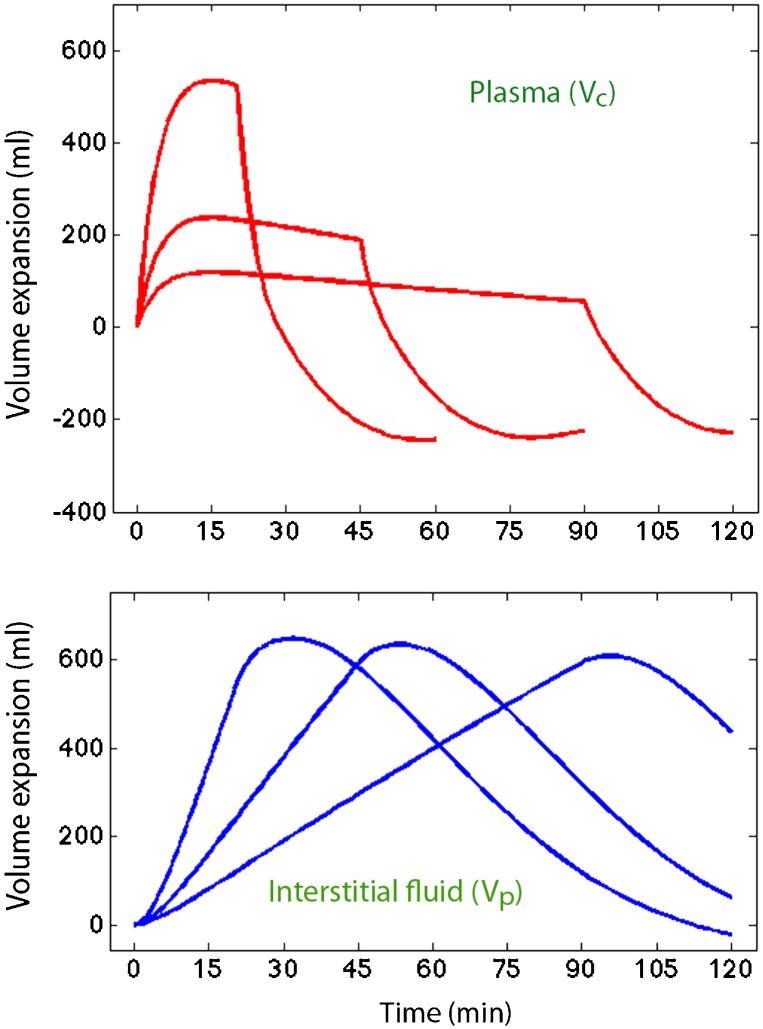
The distribution and elimination of the infused irrigating fluid were analyzed by a two-volume kinetic model (Figure 1, top). Fluid was infused at rate Ro to increase the volume of the central body fluid space Vc to vc (plasma compartment). The rate of elimination was given as the product of the volume expansion of Vc and the elimination rate constant k10 (unit: min-1). “Perspiratio insensibilis” was accounted for by constant ko, which was pre-set to 0.1 ml/min. The distribution of fluid to a peripheral body fluid space Vp (interstitial compartment) was governed by k12, and its return from vp to vc was governed by the rate constant k21. The differential equations are 17,18:
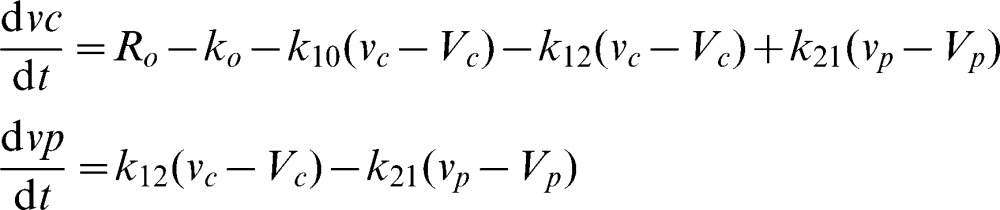 |
Figure 1.
Schematic drawing of the kinetic model used to analyze the distribution and elimination of irrigating fluid (top) and the dilution of arterial plasma during and after infusion of 1.5% glycine and 5% mannitol over 90 min (bottom). Thin blue lines represent individual experiments, and the thick red line is a computer simulation based on the group median value of each parameter in the kinetic model.
The Hb-derived fractional plasma dilution was used to indicate the volume expansion of Vc resulting from the infusion:
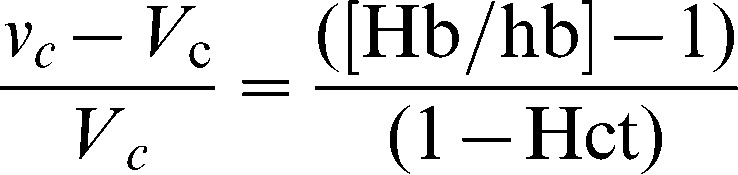 |
The symbols in capital letters denote the baseline values. A correction for the effect of blood sampling and blood loss was made in the plasma dilution 16.
The main parameters in the model (Vc, k12, and k21) were estimated by the application of a non-linear, least-squares regression routine (fminsearch) within Matlab R2010a software (Math Works Inc., Natick, MA), while k10 was calculated as follows:
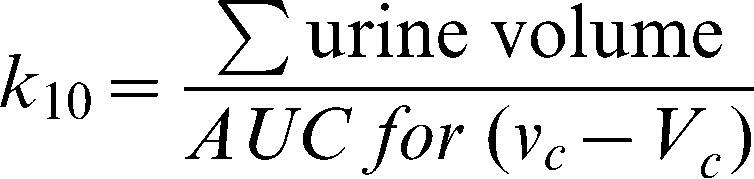 |
where AUC is the area under the curve.
The conventional volume kinetic model assumes that a single clearance constant governs the rate of distribution of infused fluid between the plasma and interstitial fluid spaces in proportion to the extent that they have become diluted 15,16. As this assumption may not be valid in shock states, the present study describes the rate of fluid distribution as being governed by two different rate constants (k12 and k21) in proportion to the extent that the plasma and interstitial fluid spaces have become expanded 17,18. Stability is maintained by having the model apply volume shifts instead of dilution in the non-linear regression analysis, which makes it possible to analyze the fluid distribution without estimating Vc or Vp. Their sizes can still be calculated as secondary parameters (this was done with Vc), but they are not needed for simulation of the distribution and elimination of infused fluid.
Intracellular distribution of fluid
Distribution of the infused fluid into the intracellular fluid (ICF) space was calculated using sodium as an endogenous tracer. All infused and voided amounts of fluid and sodium were known, so the change in the ICF volume could be estimated for each 10-min period, based on the assumption that sodium was evenly distributed in the extracellular fluid (ECF) volume, which makes up 20% of the body weight 19. For each 10-min time interval, we obtain 15,16,20,21:
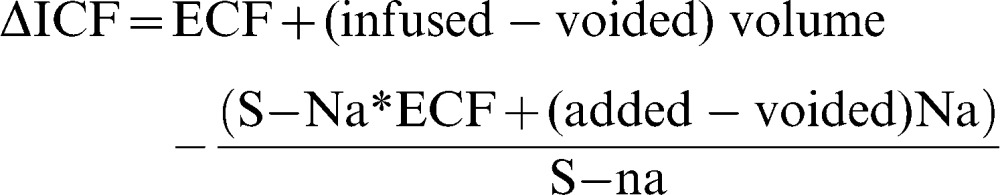 |
where S-Na and S-na are the serum sodium concentrations measured at the beginning and end of each assessed time period, respectively.
As ΔICF is derived from the peripheral (interstitial) space, ΔICF was subtracted from vp at each time point of the curve-fitting procedure to arrive at the correct distribution of fluid between vc and vp (if not, ΔICF would be included in vp).
For simulation purposes, we calculated a rate parameter k23 to express the rate of transfer of irrigating fluid to the ICF space from vp:
 |
Simulation of the distribution and elimination of the irrigating fluid following infusion times other than the 90 min used in the study was performed by inserting parameter values into the same kinetic model as used for the evaluation. Using k23 allowed us to simulate the distribution of fluid without involving serum sodium.
Statistics
The results are reported as the median and 25th–75th percentiles. Changes in parameter values were studied by the Wilcoxon matched-pair test, and differences between the groups were evaluated by the Mann-Whitney U test. Correlations were evaluated by simple and multiple linear regression analyses. P<0.05 was considered statistically significant.
RESULTS
The kinetic model could be applied in all experiments (Table 1). A plot of the measured dilution of arterial plasma over time revealed a marked variability in the response to the administration of irrigating fluid (Figure 1, bottom).
Table 1.
Kinetic parameters for infusion experiments with irrigating fluids in the pig. Group data are the median (25th-75th percentiles) of the optimal estimate with regard to the type of infused irrigating fluid (top rows) and whether cardiac output (CO) had decreased or increased at the end of the infusions compared to baseline (bottom rows).
| N | Vc | k12 | k21 | k10 | k23 | |
| ml/kg b.w. | 10-3; min-1 | 10-3; min-1 | 10-3; min-1 | 10-3; min-1 | ||
| Glycine 1.5% | 9 | 60 (51-97) | 57 (24-92) | -0.1 ((-25.3)- 6.9) | 35 (25-67) | 33 (18-47)* |
| Mannitol 5% | 8 | 45 (32-91) | 67 (36-135) | 9.6 ((-8.0) -16.6) | 42 (29-56) | 16 (9-26) |
| CO decrease | 7 | 88 (53-03) | 39 (22-93)** | -11 ((-23)-5)*** | 56 (40-119) | 47 (21-82) |
| CO increase | 9 | 43 (29-60) | 93 (53-148) | 12 (5-19) | 39 (31-56) | 27 (13-40) |
*p<0.003 versus mannitol 5%. **p<0.04 versus CO increase; ***p<0.02 versus CO increase.
Hemodynamics
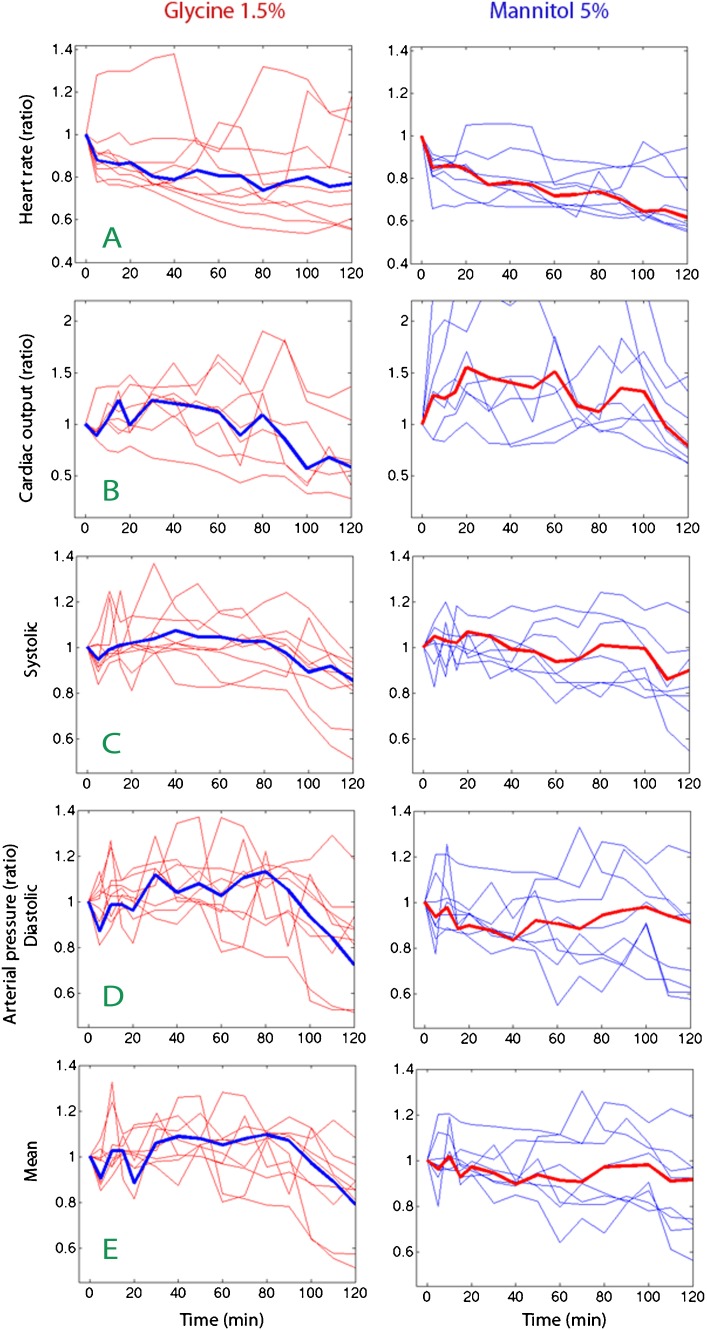
Bradycardia developed early during the infusions (Figure 2A). CO increased, particularly in response to mannitol, but when the infusions were stopped, CO ultimately decreased to 67% of baseline (median) at 120 min (p<0.03; Figure 2B).
Figure 2.
Heart rate (A), cardiac out (B), and arterial pressures (C, D, E) over time. Each thin line represents one infusion experiment, and the thick lines are the median values.
The systolic arterial pressure was lower at 110 and 120 min compared to baseline (Wilcoxon's matched-pair test p<0.01, Figure 2C). The diastolic pressure increased in response to glycine but dropped promptly when the infusion was stopped (Figure 2D).
The mean arterial pressure (MAP) decreased to 85% of baseline, from 83 mmHg (median; 25th–75th percentile 76–87) before the infusions to 67 (52–81) mmHg at 120 min (pooled data, p<0.0042, Figure 2E).
Natriuresis versus volume kinetics
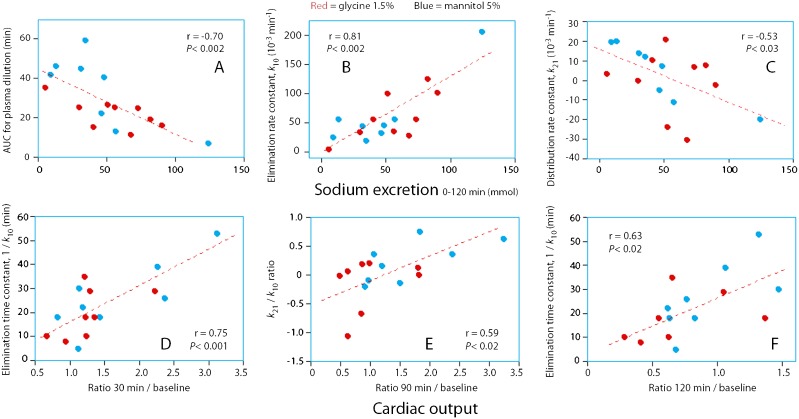
Sodium excretion appeared to have a marked effect on the fluid volume kinetics. A strong natriuresis promoted a smaller plasma dilution over time (Figure 3A), a higher elimination rate constant (Figure 3B), and a negative value of the rate constant for the re-distribution of fluid from peripheral tissues to the plasma (Figure 3C).
Figure 3.
Relationship between selected volume kinetic parameters and the total sodium excretion (top row) and the change in cardiac output (bottom row) during infusion experiments with urological irrigating fluids. Each point represents one experiment.
Multiple regression analysis showed that both k10 (p<0.001) and 1/k21 (p<0.0017) were independently associated with increasing natriuresis (r = 0.91).
Hemodynamics versus volume kinetics
Multiple regression analysis showed that higher values of both 1/k10 (p<0.004) and k21 (p<0.04) were independently correlated with the increase in CO during the infusion (r = 0.80). Therefore, the marked increase in CO expected to result from the rapid, massive infusion of fluid was counteracted by a high k10 and a low or negative k21 (Figure 3D, E and F).
These relationships were further illustrated by separating the data for the animals with CO below and above baseline at the end of the infusions (90 min); the former group had a higher total urinary excretion compared to the others (1729 vs. 1290 ml; ANOVA p<0.05). Those with a decrease in CO also had a low or negative k21, which was not the case for animals showing an increase in CO (Table 1, bottom). A similar trend was also noted for changes in stroke volume, but this was less apparent than for CO.
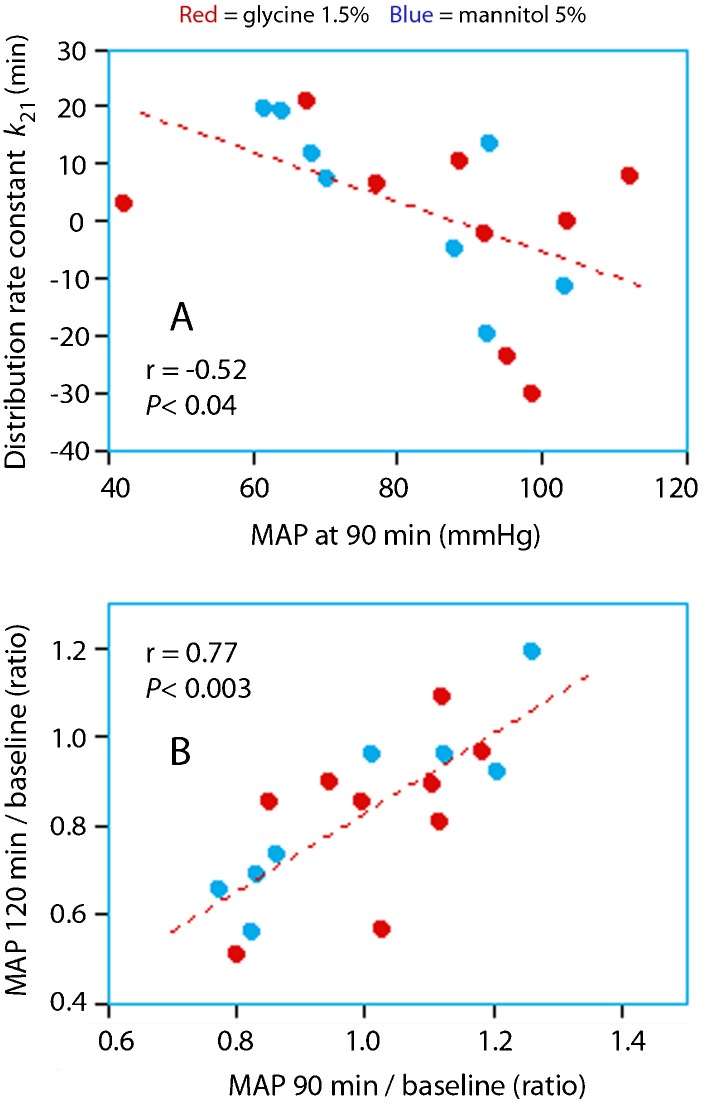
A low MAP at the end of the infusion was the only factor associated with a positive value of k21, which was otherwise negative (Figure 4A).
Figure 4.
Relationship between the mean arterial pressure at 90 min and k21 (A) and the changes in mean arterial pressure at 90 min and 120 min compared to baseline (B).
Completion of the infusion resulted in a consistent decrease in MAP over the subsequent 30 min, which amounted to almost 20% (Figure 4B). This means that an increase in MAP during the infusion prevented, rather than promoted, serious hypotension during the postinfusion period.
Computer simulation
Simulations of the distribution of 150 ml/kg (3.3 L) of irrigating fluid were performed for administrations taking place over three different periods of time. Values of k10 and k21 were applied that are likely to ensue when natriuresis occurs quickly (as suggested by Figure 3). With respect to k12 and k23, which are independent of natriuresis, we used the median optimal estimates obtained when 1.5% glycine was infused (Table 1).
These curves showed that the volume expansion of the central compartment (Vc) was quite modest, except for a peak when the irrigating fluid was infused rapidly. However, the volume expansion invariably turned into a deficit within 15 min after the infusion was stopped (Figure 5A). Volume expansion of the peripheral fluid space (Vp) was more consistent for the three infusion rates, but it did not last for more than 1 hour (Figure 5B).
Figure 5.
– Computer simulation of the fluid distribution when 150 ml/kg of electrolyte-free irrigating solution is infused over 20, 45, and 90 minutes in a pig weighing 20 kg (all infusions amounted to 3.3 L). Parameter settings: k10 = 150, k12 = 57, k21 = -30, k23 = 33; all have the unit 10-3 min-1.
The urinary excretion amounted to 1550, 1400, and 1200 ml for the 20-min, 45-min, and 90-min infusions, respectively, but according to the model, further diuresis was arrested when vc<0. The final intracellular accumulation of fluid amounted to 1300 ml for all three infusions (data not shown).
DISCUSSION
Natriuresis
Infusion of electrolyte-free irrigating fluid decreases the serum sodium concentration. In the present study, the lowest serum sodium occurred at the end of the infusions and averaged 105 mmol/l for 1.5% glycine and 100 mmol/l for 5% mannitol 20. Dilution caused by the lack of sodium in the infused fluid is mainly responsible for the hyponatremia, but interestingly, the influence of dilution is gradually overshadowed by natriuresis-induced hyponatremia 22.
The results of our present evaluation suggest that natriuresis is the key element promoting dilutional hyponatremic shock when the body is loaded with an electrolyte-free irrigating fluid. The cause of the sodium loss is the osmotic diuresis that occurs when the renal reabsorption mechanisms are either overwhelmed (glycine) or absent (mannitol). The capacity of the kidneys to control the urine composition is then undermined, and various small solutes, including amino acids and sodium, are ultimately lost from the body. The largest amounts of solutes are excreted at the end of the fluid administration and during the subsequent hour 21,23.
High natriuresis acted to reduce the plasma dilution, which was a sign of modest plasma volume expansion, despite vigorous fluid administration (Figure 3A). Factors decreasing the volume expansion were a high urinary flow (higher k10) and the surprising finding that k21 became low or negative (Figure 3B, C). The latter phenomenon is particularly important in the development of hypovolemic shock, as it means and that, once distributed from the plasma to the interstitial space, the fluid does not return to the plasma.
A negative k21 implies that fluid is driven from the plasma to the interstitium, probably due to a sodium gradient between these fluid compartments. This type of a gradient likely exists because natriuresis and the infusion of irrigating fluid both reduce the plasma sodium concentration, while translocation of fluid to the ICF raises the sodium concentration in the interstitium. The only factor that occasionally keeps k21 positive is a marked decrease in MAP, which is a compensatory mechanism that can be understood from the Starling equilibrium (Figure 4A).
The end result of these opposing mechanisms (i.e., that sodium is diluted in the plasma and concentrated in the interstitium) is that the plasma volume rapidly decreases during a 15–20 min period after the infusion ends (Figure 5). As expected, the hypovolemia reduces CO and a hypokinetic state ensues. During an operation, the hypovolemia would be even more pronounced because fluid absorption is typically associated with a temporary increase in blood loss 24. The latter also promotes further loss of sodium, as the majority of the sodium in the bled plasma is derived from the interstitium 25.
The fluid shifts in TUR syndrome have similarities to those occurring in “hypotonic dehydration”. Cellular edema and a low plasma volume occur in both entities. However, the sodium gradient between the plasma and the interstitial fluid must be much greater in TUR syndrome due to the fact that electrolyte-free irrigating fluid is continuously infused into the plasma (until the operation ends). Thus, the tendency of volume to remain translocated to the interstitium at the expense of the plasma volume becomes exceptionally strong.
Many specific factors other than natriuresis have been proposed as explanations for dilutional hyponatremic shock, but with scant evidence of the pathophysiological importance 26. These include hyperkalemia 27, hypocalcemia 28, and the release of prostatic tissue substances 29,30 and endotoxins 31 from damaged cells in the operating area. Although these factors may be important in certain patients, the present study supports the idea that natriuresis is the key factor explaining why hypovolemic hypotension develops despite administration of large amounts of fluid.
Implications for treatment
The treatment of TUR syndrome has long been controversial. The primary challenge is to prevent cardiovascular collapse and brain edema, which are the main causes of death 32. Depressed consciousness and renal failure are easier to manage and therefore become secondary issues.
The majority of textbooks suggest that furosemide be used to combat the fluid overload, but this treatment is based on logic and not on evidence. Furosemide operates by increasing natriuresis, and the present results suggest that this treatment would aggravate the hemodynamic problems by further reducing the plasma volume and increasing the cellular edema. Clinical observations support this view 33.
A less popular interpretation is that dilutional hyponatremic shock represents a hypovolemic condition that should be treated with plasma volume expanders 5,14. A step-by-step evaluation of 12 prostatectomy operations over 10-min periods indeed has shown that only a modest rise in blood volume occurs in response to fluid absorption, which is soon turned into hypovolemia and hypotension after the absorption has slowed or stopped 34. Other authors also report a low CO postoperatively 35.
The results of the present study support the view that TUR syndrome is a hypovolemic condition and should be treated with plasma volume expansion to prevent cardiovascular collapse. Treatment should be initiated within 15 min after the fluid absorption, or the operation, has ended. The presence of marked interstitial edema suggests that plasma volume expansion is best performed using a colloid fluid. From a modeling perspective, the marked disequilibrium of fluid, with hypovolemia occurring despite marked volume expansion of the interstitium (Figure 5), could not have been discovered by conventional volume kinetics measurements that use a single clearance constant to describe the rate of distribution.
Greater agreement exists for the use of hypertonic saline to combat severe TUR syndrome, although warnings of excessive fluid overload and cerebral damage from rapid administration have been given. Animal experiments show that hypertonic saline increases the plasma volume, dehydrates the cells 21, and improves survival 36. Clinical evaluations have also demonstrated the benefit of hypertonic saline 37,38. The administration does not need to be slow, but it should extend over the period of the osmotic diuresis, at least 1–2 hours, and be guided by measurements of serum sodium. Full compensation does not need to be given.
The electrolyte-free irrigating fluids used in the present study are the most commonly used worldwide. They differ from each other in that 1.5% glycine more easily enters the cells (i.e., its k23 is higher) by virtue of its lower osmolality (200 mOsmol/kg) than 5% mannitol (295 mOsmol/kg). Glycine is also actively pumped into the cells, and this exerts an effect on fluid distribution. Besides the k23, no differences in kinetic parameters between the two fluids could be established statistically, although k21 tended to be lower for glycine. Therefore, there is no clear explanation for why different treatments should be suggested depending on whether hyponatremic shock is induced by absorption of 1.5% glycine or 5% mannitol. Clinically, the incidence of hypotensive events after absorption of 1.5% glycine and 3% mannitol during prostate surgery appears to be identical 13.
Limitations
Limitations of this study include the fact that the data were obtained from animals and may therefore not be uncritically extrapolated to humans. Hemodynamic responses in the pig are still believed to be quite similar to those observed in humans. Ketamine was used for anesthesia, and this is known to alter hemodynamics to only a small degree, while both spinal anesthesia and surgical blood loss, which are inherent components of surgical operations in which irrigating fluid might be absorbed, may aggravate hypovolemic hypotension. Senior citizens subjected to TURP surgery also have a compromised cardiovascular baroreflex and weak reactivity to catecholamines, which makes them dependent on preload. Hence, they are more poorly suited than are young pigs to maintain normal hemodynamics when challenged with the fluid volume shifts associated with TUR syndrome.
The amount of administered fluid is also worth mentioning. Infused volumes per body weight cannot be extrapolated directly between animals and humans. As differences in toxicokinetics are related to the 2/3 power of body weight 39, the toxic effects of the infused fluid correspond to 60 ml/kg (≈4.5 liters) in an adult male. Absorption of this volume occurs over 20-35 min during prostatectomy operations 40, and assuming that this would also take place in simulations is not unreasonable. Finally, the hemodynamic data presented here are selective and are limited to those that are essential for providing a link to the fluid distribution. For more complete data, we refer readers to our previous report 20.
Comparisons between hemodynamic responses and volume kinetic parameters can be a useful approach to better understand the pathophysiology of shock syndromes. The present results identify natriuresis as the key factor promoting hypovolemic hypotension after administration of 3.3 L of electrolyte-free irrigating fluid in pigs. Excessive sodium loss, which is the result of osmotic diuresis, was associated with a higher urinary flow rate and the uptake of fluid to the ICF space at the expense of the plasma volume alone. Treatment should consist of plasma volume expansion, preferably by a colloid, initiated within 15 min after fluid absorption (or the operation) ends. Thereafter, hypertonic saline should be provided to replace the excreted sodium. The administration should be guided by measurements of serum sodium.
ACKNOWLEDGMENTS
The authors are indebted to Drs. Lars Sandfeldt, Louis Riddez, Carl-Arne Ewaldsson, and David Piros for their assistance in performing the animal experiments.
Footnotes
No potential conflict of interest was reported.
REFERENCES
- 1.Demirel I, Ozer AB, Bayar M, Erhan OL. TURP syndrome and severe hyponatremia under general anesthesia. BMJ Case Rep. 2012;2012 doi: 10.1136/bcr-2012-006899. doi:pii:bcr-2012-006899. 10.1136/bcr-2012-006899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cavallini M, Sarocco MG, Aguggia M. Postoperative delirium with hyponatremia after transurethral resection of theprostate. Acta Neurol Belg. 2012;111(2):152–4. [PubMed] [Google Scholar]
- 3.Tuccori M, Guidi B, Montagnani S, Fornai M, Antonioli L, Brandizzi C, et al. Transient acute liver failure complicating transurethral resection syndrome. Scand J Urol Nephrol. 2010;44(4):269–72. doi: 10.3109/00365591003636612. [DOI] [PubMed] [Google Scholar]
- 4.Harrison RH, Boren JS, Robison JR. Dilutional hyponatremic shock: another concept of the transurethral prostatic resection reaction. J Urol. 1956;75(1):95–110. doi: 10.1016/S0022-5347(17)66783-1. [DOI] [PubMed] [Google Scholar]
- 5.Hahn RG. Hallucination and visual disturbances during transurethral prostatic resection. Intensive Care Med. 1988;14(6):668–71. doi: 10.1007/BF00256777. [DOI] [PubMed] [Google Scholar]
- 6.Wang JH, He Q, Liu YL, Hahn RG. Pulmonary edemain the transurethral resection syndrome induced with mannitol 5% Acta Anaesthesiol Scand. 2009;53(8):1094–6. doi: 10.1111/j.1399-6576.2009.01983.x. [DOI] [PubMed] [Google Scholar]
- 7.Boukatta B, Sbai H, Messaoudi F, Lafrayiji Z, El Bouazzaoui A, Kanjaa N. Transurethral resection of prostate syndrome: report of a case. Pan Afr Med J. 2013;14:14. doi: 10.11604/pamj.2013.14.14.1906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Collins JW, MacDermott S, Bradbrook RA, Drake B, Keeley FX, Timoney AG. The effect of the choice of irrigation fluid on cardiac stress during transurethral resectionof the prostate: a comparison between 1.5% glycine and 5% glucose. J Urol. 2007;177(4):1369–73. doi: 10.1016/j.juro.2006.11.042. [DOI] [PubMed] [Google Scholar]
- 9.Yousef AA, Suliman GA, Elashry OM, Elsharaby MD, Elgamasy AEK. A randomized comparison between three types of irrigating fluids during transurethral resection in benign prostatic hyperplasia. BMC Anesthesiol. 2010;10:7. doi: 10.1186/1471-2253-10-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shrestha B, Baidhya JL. Morbidity and early outcome of transurethral resection of prostate: a prospective single-institute evaluation of 100 patients. Kathmandu Univ Med J. 2010;830):233–7. doi: 10.3126/kumj.v8i2.3559. [DOI] [PubMed] [Google Scholar]
- 11.Olsson J, Nilsson A, Hahn RG. Symptoms of the transurethral resection syndrome using glycine as the irrigant. J Urol. 1995;154(1):123–8. [PubMed] [Google Scholar]
- 12.Hahn RG, Shemais H, Essén P. Glycine 1.0% versus glycine 1.5% as irrigating fluid during transurethral resection of the prostate. Br J Urol. 1997;79(3):394–400. doi: 10.1046/j.1464-410x.1997.00016.x. [DOI] [PubMed] [Google Scholar]
- 13.Hahn RG, Sandfeldt L, Nyman CR. Double-blind randomized study of symptoms associated with absorption of glycine 1.5% or mannitol 3% during transurethral resection of the prostate. J Urol. 1998;160(2):397–401. [PubMed] [Google Scholar]
- 14.Hahn RG. The transurethral resection syndrome. Acta Anaesthesiol Scand. 1991;35(7):557–67. doi: 10.1111/j.1399-6576.1991.tb03348.x. [DOI] [PubMed] [Google Scholar]
- 15.Hahn RG. Volume kinetics for infusion fluids. Anesthesiology. 2010;113(2):470–81. doi: 10.1097/ALN.0b013e3181dcd88f. [DOI] [PubMed] [Google Scholar]
- 16.Ewaldsson C-A, Hahn RG. Kinetics and extravascular retention of acetated Ringer's solution during isoflurane and propofol anesthesia for thyroid surgery. Anesthesiology. 2005;103(3):460–9. doi: 10.1097/00000542-200509000-00006. [DOI] [PubMed] [Google Scholar]
- 17.Li Y, Zhu HB, Zheng X, Chen HJ, Shao L, Hahn RG. Low doses of esmolol and phenylephrine act as diuretics during intravenous anesthesia. Crit Care. 2012;16(1):R18. doi: 10.1186/cc11175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hahn RG, Bergek C, Gebäck T, Zdolsek J. Interactions between the volume effects of hydroxyethyl starch 130/0.4 and Ringer's acetate. 2013;17(3):R104. doi: 10.1186/cc12749. Crit Care. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guyton AC, Hall JE. Textbook of medical physiology, 9th Edition. Philadelphia: W.B. Saunders Company. 1996;185-6:298–302. [Google Scholar]
- 20.Sandfeldt L, Riddez L, Rajs J, Ewaldsson C, Piros D, Hahn RG. High-dose intravenous infusion of irrigating fluids containing glycine and mannitol in the pig. J Surg Res. 2001;95(2):114–25. doi: 10.1006/jsre.2000.6028. [DOI] [PubMed] [Google Scholar]
- 21.Hahn RG, Nilsson A, Hjelmqvist H, Zhang W, Rundgren M. Renal function during intravenous infusion of urological irrigating fluids in the sheep. Acta Anaesthesiol Scand. 1996;40(6):671–8. doi: 10.1111/j.1399-6576.1996.tb04508.x. [DOI] [PubMed] [Google Scholar]
- 22.Hahn RG. Natriuresis and “dilutional” hyponatremia after infusion of glycine 1.5% J Clin Anesth. 2001;13(3):167–74. doi: 10.1016/s0952-8180(01)00228-8. [DOI] [PubMed] [Google Scholar]
- 23.Hahn RG. Aminoacid concentrations in serum and urine after intravenous infusion of 1.5% glycine in prostatectomy patients. Prostate. 1992;21(3):173–81. doi: 10.1002/pros.2990210302. [DOI] [PubMed] [Google Scholar]
- 24.Hahn RG, Berlin T, Lewenhaupt A. Irrigating fluid absorption and blood loss during transurethral resection of the prostate studied by a regular interval monitoring (RIM) method. Scand J Urol Nephrol. 1988;22(1):23–30. doi: 10.1080/00365599.1988.11690379. [DOI] [PubMed] [Google Scholar]
- 25.Hahn RG. Trapping of electrolytes during fluid absorption in transurethral resection of the prostate. Scand J Urol Nephrol. 1997;31(3):259–63. doi: 10.3109/00365599709070344. [DOI] [PubMed] [Google Scholar]
- 26.Hoyt HS, Goebel JL, Lee HI, Choenbrod J. Types of shock-like reaction during transurethral resection and relation to acute renal failure. J Urol. 1958;79(3):500–6. doi: 10.1016/S0022-5347(17)66300-6. [DOI] [PubMed] [Google Scholar]
- 27.Browne GF, Walsh A. Transient hyperkalaemia due to systemic absorption of glycine irrigating fluid during transurethral resection of the prostate: a possible cause of cardiovascular collapse. Eur J Anaesth. 1983;1:150P–1P. [Google Scholar]
- 28.Krohn JS. Dilutional hypocalcemia in association with dilutional hyponatremia. Anesthesiology. 1993;79(5):1136–8. doi: 10.1097/00000542-199311000-00035. [DOI] [PubMed] [Google Scholar]
- 29.Verilli RA, Uhlman RC, Viek NF, Hunsicker WC. The hypotensive effect of a prostatic extract. J Urol. 1962;87(2):184–6. doi: 10.1016/S0022-5347(17)64935-8. [DOI] [PubMed] [Google Scholar]
- 30.O′Donnell P. Effects of intravenous infusion of human prostate tissue substances in dogs. The Prostate. 1990;16(1):49–56. doi: 10.1002/pros.2990160106. [DOI] [PubMed] [Google Scholar]
- 31.Sohn MH, Vogt C, Heinen G, Erkens M, Nordmeyer N, Jakse G. Fluid absorption and circulating endotoxins during transurethral resection of the prostate. Br J Urol. 1993;72(5):605–10. doi: 10.1111/j.1464-410x.1993.tb16218.x. [DOI] [PubMed] [Google Scholar]
- 32.Radal M, Jonville-Béra AP, Leisner C, Haillot O, Autret-Leca E. Effects indésirables des solutions d′irrigation glycollées. Thérapie. 1999;54(2):233–6. [PubMed] [Google Scholar]
- 33.Crowley K, Clarkson K, Hannon V, McShane A, Kelly DG. Diuretics after transurethral prostatectomy: a double-blind controlled trial comparing frusemide and mannitol. Br J Anaesth. 1990;65(3):337–41. doi: 10.1093/bja/65.3.337. [DOI] [PubMed] [Google Scholar]
- 34.Hahn RG. Fluidand electrolyte dynamics during development of the TURP syndrome. Br J Urol. 1990;66(1):79–84. doi: 10.1111/j.1464-410x.1990.tb14869.x. [DOI] [PubMed] [Google Scholar]
- 35.Singer M, Patel M, Webb A, Bullen C. Management of the transurethral prostate resection syndrome: time for reappraisal. Crit Care Med. 1990;18(12):1479–80. doi: 10.1097/00003246-199012000-00031. [DOI] [PubMed] [Google Scholar]
- 36.Bernstein GT, Loughlin KR, Gittes RF. The physiologic basis of the TUR syndrome. J Surg Res. 1989;46(2):135–41. doi: 10.1016/0022-4804(89)90216-3. [DOI] [PubMed] [Google Scholar]
- 37.Henderson DJ, Middleton RG. Coma from hyponatraemia following transurethral resection of the prostate. Urology. 1980;XV(3):267–71. doi: 10.1016/0090-4295(80)90441-0. [DOI] [PubMed] [Google Scholar]
- 38.Ayus JC, Krothapalli RK, Arieff AI. Treatment of symptomatic hyponatraemia and its relation to brain damage. A prospective study. New Engl J Med. 1987;317(19):1190–5. doi: 10.1056/NEJM198711053171905. [DOI] [PubMed] [Google Scholar]
- 39.Boxenbaum H. Interspecies scaling, allometry, physiological time and the ground plan for pharmacokinetics. J Pharmacokin et Biopharm. 1982;10(2):201–27. doi: 10.1007/BF01062336. [DOI] [PubMed] [Google Scholar]
- 40.Hahn RG. Early detection of the TUR syndrome by marking the irrigating fluid with 1% ethanol. Acta Anaesthesiol Scand. 1989;33(2):146–51. doi: 10.1111/j.1399-6576.1989.tb02878.x. [DOI] [PubMed] [Google Scholar]