Abstract
Blood vessels and neurons grow often side by side. However, the molecular and cellular mechanisms underlying their parallel development remain unclear. Here, we report that a subpopulation of secondary motoneurons extends axons ventrally outside of the neural tubes and rostrocaudally as a fascicle beneath the dorsal aorta (DA) in zebrafish. We tried to clarify the mechanism by which these motoneuron axons grow beneath the DA and found that Vegfc in the DA and Vegfr3 in the motoneurons were essential for the axon growth. Forced expression of either Vegfc in arteries or Vegfr3 in motoneurons resulted in enhanced axon growth of motoneurons over the DA. Both vegfr3 morphants and vegfc morphants lost the alignment of motoneuron axons with DA. In addition, forced expression of two mutant forms of Vegfr3 in motoneurons, potentially trapping endogenous Vegfc, resulted in failure of growth of motoneuron axons beneath the DA. Finally, a vegfr3 mutant fish lacked the motoneuron axons beneath the DA. Collectively, Vegfc from the preformed DA guides the axon growth of secondary motoneurons.
Keywords: Vegfc, Vegfr3, Motoneuron, Wiring, Guidance
INTRODUCTION
Blood vessels exhibit hierarchical branching from the central vessel (aorta) to the periphery (capillaries in the organs). Similarly, the neural network is formed according to the hierarchical connection originating from the central nervous system to the peripheral nervous system. Blood vessels and neurons are found side by side in the vertebrates (Bearden and Segal, 2005; Martin and Lewis, 1989), suggesting that both mutually affect the growth direction or that both develop following the guidance cues from the tissue surrounding them. Indeed, neurons extend or make contact according to repulsive or attractive cues, including Ephrin/Eph, Netrin/Unc5, Semaphorin/Plexin and Neuropilin (NRP), and Slit/Robo signaling, to form the network in the whole body (Carmeliet and Tessier-Lavigne, 2005; Guan and Rao, 2003). During blood vessel formation, migration and retraction of endothelial cells also follow these guidance cues (Adams and Eichmann, 2010; Eichmann et al., 2005; Larrivée et al., 2009).
There are three possible patterns for parallel growth of neurons and blood vessels: simultaneous growth, one preceding the other and vice versa. In the latter, the pre-formed network might provide the guiding cues to the following network. Artemin is primarily expressed in vascular smooth muscle cells and functions as a guidance molecule for axon growth of sympathetic nerves (Honma et al., 2002). Endothelin 3 released from smooth muscle cells of the carotid artery contributes to the alignment of sympathetic neurons with the carotid artery (Makita et al., 2008). The essential role of vascular endothelial growth factor A (VEGFA) from the preformed sensory neurons for vascular patterning has been reported in mice; loss of nerve-derived VEGFA results in impaired arterial differentiation in the skin of mice (Mukouyama et al., 2002). The congruence of neurons and blood vessels is also found in adult mouse skin, suggesting that VEGFA might not only regulate branching pattern of blood vessels but might also promote co-alignment of both nerves and vessels (James and Mukouyama, 2011). Recently, peripheral nerve-derived VEGFA and CXCL12 have been reported to regulate the patterning of arteries (Li et al., 2013). In addition, VEGFA not only functions as an axonal guidance cue but also regulates neuronal migration and plays a trophic factor for neurons (Mackenzie and Ruhrberg, 2012).
The VEGF family consists of VEGFA, VEGFB, VEGFC, VEGFD (FIGF) and PLGF (PGF). Whereas VEGFA mainly activates VEGFR1 (FLT1) and VEGFR2 (FLK1; KDR) expressed on the vascular endothelial cells (Olsson et al., 2006), VEGFB and PLGF activate VEGFR1. VEGFC and VEGFD activate VEGFR3 (FLT4), which is mainly expressed on lymphatic endothelial cells (Karkkainen et al., 2004). During developmental angiogenesis of zebrafish, Vegfa from somites determines the fate of angioblasts and common precursor vessels expressing Vegfr2 (Kdrl) in the main trunk (Lawson et al., 2002). Posterior cardinal vein (PCV) segregated from the dorsal aorta (DA) expresses Vegfr3 (Flt4), although the DA does not express Vegfr3 but rather Vegfc (Covassin et al., 2006; Lawson et al., 2001). In the central nervous system, VEGFR3 is expressed in the neural progenitor cells in Xenopus laevis and mouse embryos (Le Bras et al., 2006). VEGFC is able to stimulate VEGFR3-expressing neural stem cells in mice (Calvo et al., 2011). The proliferation of neural progenitor cells depends on the VEGFC/VEGFR3-mediated signal. In addition, VEGFC acts as a neurotrophic factor for dopamine neurons (Piltonen et al., 2011). These reports indicate that the signal mediated by VEGFC/VEGFR3 is not restricted to within the mesoderm-derived cells but is also used outside of mesodermal tissues. Consistent with this, in zebrafish, Vegfc is required for coalescence of endodermal cells in the anterior midline and for the initial formation of dorsal endoderm (Ober et al., 2004).
Among the primary motoneurons of zebrafish [rostral primary (RoP), middle primary (MiP) and caudal primary (CaP) motoneurons] and CaP-like secondary motoneurons, RoP, CaP and CaP-like motoneurons exit the neural tube and extend their axons ventrally towards the axial vessels (Lewis and Eisen, 2003). In addition to these motoneurons, dorsoventrally projecting secondary motoneurons, ventrally projecting secondary motoneurons and intermyotomal secondary motoneurons extend axons ventrally (Asakawa et al., 2013; Menelaou and McLean, 2012). In contrast to the initial neural axon growth of these motoneurons, intersegmental vessels sprout from the DA and extend dorsally towards the neural tube (Isogai et al., 2001). However, once the former and the latter reach the ventral-most and dorsal-most points, respectively, both extend rostrally and caudally along the anterior-posterior axis. These neural and vascular networks during embryogenesis can be spatiotemporally monitored in transgenic fish in which fluorescence proteins are produced under the control of neuron-specific or endothelial cell-specific promoters.
Here, we demonstrate the growth of secondary motoneuron axons descending ventrally and extending both rostrally and caudally as a fascicle beneath the DA using transgenic fish expressing fluorescent proteins: monomeric Cherry (mCherry) in endothelial cells and green fluorescent protein (GFP) in motoneurons. We show that the parallel growth of secondary motoneuron axons with the preformed DA is regulated by Vegfc/Vegfr3 signaling.
MATERIALS AND METHODS
Zebrafish and transgenesis
The experiments using zebrafish were approved by the institutional animal committee of National Cerebral and Cardiovascular Center and performed according to the guidelines of the Institute.
Zebrafish (Danio rerio) embryos were obtained from natural spawning of laboratory lines. Tg(fli1a:egfp)y1 fish were kindly provided by Nathan Lawson (University of Massachusetts Medical School, MA, USA). Tg(mnx1:gfp)ml2 fish were obtained from the Zebrafish International Resource Center (University of Oregon, OR, USA). Tg(mnx2b:gff) fish in which Gal4FF was expressed under the BAC-derived mnx2b promoter were established (Asakawa et al., 2008). Mutant vegfr3hu4602/wt (expando) in double-transgenic Tg(fli1a:egfp)y1;(kdrl:mcherry) was previously reported (Hogan et al., 2009). Zebrafish were raised, injected and maintained under standard laboratory conditions (Westerfield, 2000). We used wild-type (AB), Tg(fli1a:myr-mcherry), Tg(huc:gfp), Tg(mnx1:gfp)ml2, Tg(mnx2b:gff) and Tg(fli1a:egfp)y1 embryos of either sex. Tg(fli1a:myr-mcherry) fish were developed by injecting the Tol2-based plasmid containing fli1a promoter followed by cDNA coding myristoylated (Myr) mCherry (pTol fli1a:myr-mcherry; 25 ng) with Tol2 transposase mRNA (25 ng) into one-cell-stage embryos of AB fish. Embryos were selected at 2 days post-fertilization (dpf) for high expression and grown to adults, among which germline founders were identified by specific expression of Myr-mCherry in the blood vessels.
Plasmids
pTol fli1a vector was constructed by modifying pTol2 vector and inserting the fli1a promoter as a driver of expression of the target molecule (Kawakami et al., 2004; Lawson and Weinstein, 2002). pTol mnx2b vector was similarly constructed by inserting the mnx2b promoter (Asakawa et al., 2012). The pTol flt1 vector was constructed by inserting the flt1 (vegfr1) promoter/enhancer corresponding to conserved non-coding elements (9a and 11) and -181 bp of the flt1 gene (Bussmann et al., 2010). An oligonucleotide encoding the myristoylation (Myr) signal derived from Lyn kinase was subcloned into pmCherry-N1 vectors (Takara) to construct the plasmid expressing Myr signal-tagged mCherry. pTol fli1a:myr-mcherry was constructed by inserting Myr-mCherry cDNA into pTol fli1a vector. The DNA encoding zebrafish (z)Vegfr3 tagged with Flag followed by 2A peptide and mCherry was subcloned into pcDNA3.1 (Invitrogen), pPBbsr2 (provided by Michiyuki Matsuda, Kyoto University, Kyoto, Japan) for piggyBac transposon-mediated gene transfer (provided by Allan Bradley, Wellcome Trust Sanger Institute, Cambridge, UK), and pTol mnx2b for Tol2 transposon-mediated gene transfer. These plasmids were named as follows; pcDNA3.1(z)vegfr3-f2amcherry, pPBbsr2(z)vegfr3-f2amcherry and pTol mnx2b:(z)vegfr3-f2amcherry. The DNA encoding (z)Vegfr3 lacking tyrosine kinase domain and tagged with Flag followed by 2A peptide and mCherry were inserted into pcDNA3.1 [designated as pcDNA3.1(z)vegfr3delta RTK-f2amcherry] and pTol mnx2b [pTol2mnx2b:(z)vegfr3deltaRTK-f2amcherry]. The DNA encoding partial extracellular domain of human VEGFR3 fused with human Ig Fc fragment was subcloned into pcDNA3.1 and pTol mnx2 plasmids and named as follows; pcDNA3.1(h)VEGFR3-Fc and pTol mnx2b:(h)VEGFR3-Fc. The DNA encoding (z)Vegfc was subcloned into pCS2+ and pTol flt1 plasmids and named as follows: pCS2(z)vegfc and pTol flt1:(z)vegfc. mCherry cDNA was inserted into pTol flt1 (pTol flt1:mcherry). pTol uas(Gal4-specific recognition sequence; for upstream activating sequence):egfp was used to identify motoneurons by being injected into one-cell-stage Tg(mnx2b:gff) embryos.
FACS, RT-PCR and quantitative RT-PCR
Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos at 72 hours post-fertilization (hpf) were digested with 5 mg/ml trypsin in PBS. The separated cells were sorted by a FACS Aria III Cell Sorter (BD Biosciences) according to GFP and mCherry fluorescence. The cells sorted by FACS were suspended in 0.1% fetal bovine serum (FBS) in PBS. RNAs were isolated using the RNeasy Micro Kit (Qiagen). For RT-PCR, RNAs were reverse transcribed by random hexamer primers using Superscript III (Invitrogen) according to the manufacturer’s instructions. PCR was performed using SpeedSTAR HS DNA polymerase (Takara). Real-time quantitative (q) RT-PCR was performed using the QuantiFast SYBR RT-PCR Kit (Qiagen). Primer sequences for the target mRNAs used for PCR and qRT-PCR amplification are described in supplementary material Table S1 and Fig. S3.
Microinjections of plasmids, morpholinos and quantum dot
Transient expression of fluorescent proteins, (z)Vegfc, (z)Vegfr3 tagged with Flag followed by 2A peptide and mCherry [(z)Vegfr3-F2AmCherry], (h)VEGFR3-Fc, and (z)Vegfr3deltaRTK tagged with Flag followed by 2A peptide and mCherry [(z)Vegfr3deltaRTK2AmCherry] was carried out using the Tol2 system (Kawakami et al., 2004). Capped Tol2 transposase mRNA (25 pg) and the Tol2 plasmids (25 ng) containing either the artery-specific flt1 promoter or the motoneuron-specific mnx2b promoter were co-injected into one-cell-stage transgenic embryos. Quantum (Q) dot 655 (Molecular Probes) was injected into the axial blood vessels at 4 dpf. Three nanograms of morpholino (MO) (Gene Tools) was injected into yolk of one- to two-cell-stage embryos of transgenic fish. Details of Tol2 plasmids, MOs and primers for the validation of inhibition of splicing by MOs are described in supplementary material Table S1 and Fig. S3.
Ex vivo assay
Tg(mnx1:gfp)ml2 embryos at 36 hpf were manually dechorionated with fine forceps and were rinsed in sterile 0.5× E3 medium. Embryos were cut at the dorsoventral line between yolk and caudally extended yolk using operating scissors to expose the neural tissues of the trunk. Embryos, from which heads had been removed, were embedded in matrigel (BD Biosciences) with or without recombinant human (h)VEGFC (R&D Systems). Embedded embryos were immersed in culture media consisting of CO2-independent media (Gibco) supplemented with 1× penicillin/streptomycin (Gibco) in 5% CO2 at 28°C for 12 hours.
Vegfr inhibitors
Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos were incubated from 60 to 96 hpf in E3 medium containing 25 μM maz51 (Merck) or 0.5 μM ki8751 (Tocris) (Kubo et al., 2005; Ny et al., 2008). For the ex vivo assay, zebrafish embryos, from which the heads and yolk had been dissected from the trunks, were incubated at 28°C for 12 hours in the medium containing 25 μM maz51 or 0.5 μM ki8751.
Cell culture and preparation of conditioned medium
Human umbilical vein endothelial cells (HUVECs) were cultured in EBM2 medium with essential growth media (Lonza). HEK293 cells and 293T cells were cultured in DMEM (Invitrogen) containing 10% FBS supplemented with 1× penicillin/streptomycin (Gibco). Cells were transfected using Lipofectamine2000 according to the manufacturer’s instruction (Invitrogen). Conditioned media (CM) containing (z)Vegfc and (h)VEGFR3-Fc were prepared by the culture media of the 293T cells transfected with pCS2 (z)vegfc and pcDNA3.1(h)VEGFR3-Fc plasmids, respectively. Media were replaced with FBS-free medium 24 hours after transfection. FBS-free media from transfected cells were collected after 24 hours incubation and concentrated through an Amicon Ultra-4 filter device (10 kDa) (Millipore). The HEK293 cells stably expressing (z)Vegfr3 were established by transfecting the cells with pPBbsr2(z)vegfr3-f2amcherry and pCMV-mPBase (neo-) by using Lipofectamine2000 (Invitrogen) according to the piggyBac system (Komatsu et al., 2011; Yusa et al., 2009). The cells were cultured for 2 weeks in DMEM media containing blasticidin (6 μg/ml). Those surviving under the medium containing blasticidin were used as the cells stably expressing (z)Vegfr3. Expression of (z)Vegfr3 was confirmed by immunoblot analysis using anti-Flag antibody (Sigma-Aldrich).
Immunoblot analyses and precipitation
Cells were lysed in a lysis buffer containing 10 mM HEPES (pH 7.9), 400 mM NaCl, 0.1 mM EDTA, 5% glycerol, 1 mM dl-dithiothreitol (DTT) and a protease inhibitor cocktail (Roche). Whole-cell lysates were resolved by SDS-PAGE followed by the electrophoretic transfer onto a polyvinyl difluoride membrane (Millipore). The antibodies in this study for immunoblot analyses were: anti-phospho Erk1/2 (Santa Cruz), anti-Erk1/2 (Cell Signaling Technology), anti-Flag, anti-β-actin (Sigma-Aldrich), horseradish peroxidase-coupled goat anti-mouse IgG, horseradish peroxidase-coupled goat anti-rabbit IgG (GE Healthcare) and horseradish peroxidase-coupled goat anti-human IgG. (h)VEGFR3-Fc was precipitated on Protein G-agarose (GE Healthcare).
Image acquisition, processing and quantitative analyses of neurite outgrowth and of parallel growth
Pigmentation of embryos was inhibited by 1-phenyl-2-thiourea (Sigma). The embryos were treated with 100 μg/ml tricaine (Sigma), mounted in a drop of 1.5% low melting agarose in the E3 medium, and placed onto a glass-based dish. Fluorescence images were obtained using a FluoView FV1000 confocal microscope (Olympus) with either a ×40 or a ×60 water objective lens (LUMPlanFL N, Olympus). z-stack images were 3D volume rendered with fluorescence mode using Volocity 3D Image Analysis Software (PerkinElmer). In the ex vivo assay, the number of GFP-positive outgrown neurites from the embryos was counted after 12 hours incubation at 28°C.
Impairment of parallel growth was defined as shortened or absent axons (defined as the length of axons becoming >90% shorter than those of either untreated or uninjected controls). For quantitative analyses of the parallel growth of motoneuron axons with the DA, ‘complete’ indicates the no defects in the motoneuron axons beneath the DA above yolk tubes. For that of increased parallel growth, ‘increase’ denotes the branching over the DA in addition to the parallel growth. The number of the embryos showing ‘impaired’ parallel growth divided by the total number of the embryos we observed is indicated as the percentage of the impaired parallel growth of the embryos.
Statistical analysis
Data are expressed as mean±s.d., as indicated in figure legends. Statistical significance for paired samples and for multiple comparisons was determined by Student’s t-test and by one-way analysis of variance with Tukey’s test, respectively. Data were considered statistically significant if P value was less than 0.05.
RESULTS
Parallel growth of motoneuron axons and dorsal aorta
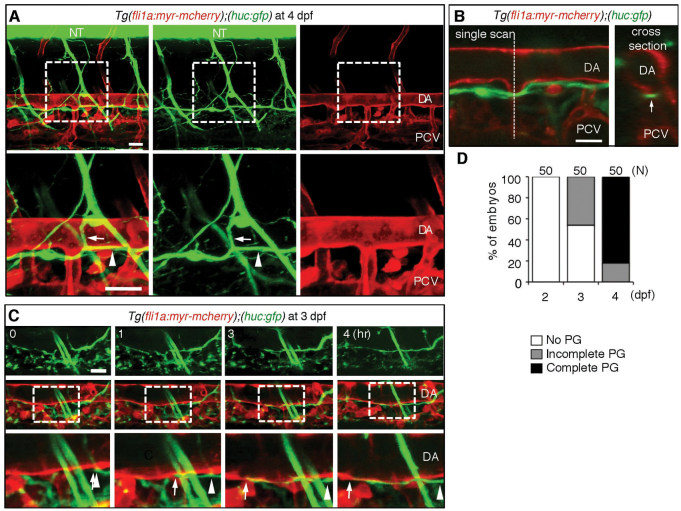
To examine the alignment of neurons and blood vessels during zebrafish embryogenesis, we monitored the development of neurons and blood vessels simultaneously using transgenic (Tg) fish expressing Myr-mCherry in blood vessels under the fli1a promoter and GFP in neurons under the huc promoter Tg(fli1a:myr-mcherry);(huc:gfp) (Park et al., 2000). We noticed GFP-positive axons just beneath the DA at 4 dpf (Fig. 1A). The axons beneath the DA seemed to be a single axon fascicle as confirmed by a single scan image of the lateral view and by a cross-section of confocal stack images of the lateral views (Fig. 1B). Therefore, to observe how and when the axon started to grow, we time-lapse imaged the extension of axons and found that the axons grew rostrally and caudally along the pre-existing DA from 3 dpf (Fig. 1C,D; supplementary material Movie 1).
Fig. 1.
Neuronal axons extend beneath the dorsal aorta in zebrafish. (A) 3D-rendered confocal stack fluorescence images (lateral view) of an Tg(fli1a:myr-mcherry);(huc:gfp) embryo at 4 dpf. Left column, merged images; center column, GFP images; right column, mCherry images. Anterior is to the left. Arrows and arrowheads indicate branches of motoneuron axons and those found beneath the DA. Lower panels show magnifications of the boxed areas in upper panels. (B) Left, a single scan confocal image from the stack shown in A. Right, a cross-section image of the stacked image shown in A at the level indicated by the dashed line in the left panel. Arrow denotes a single fascicle of axons. (C) 3D-rendered confocal time-sequential stack images of an Tg(fli1a:myr-mcherry);(huc:gfp) embryo. Elapsed time (hours) from the start point of time-lapse imaging (3 dpf) is indicated. Top panels, GFP images; middle panels, merged images of GFP and mCherry; bottom panels, enlarged images of boxed regions of middle panels. Arrows and arrowheads indicate the tip of the axon and the location of the tip when starting to extend, respectively. (D) Quantitative analyses of the parallel growth (PG) of axons with the DA at the time points indicated. ‘Complete’ indicates the complete continuity of the axon between the region above the rostral part of the yolk tube and that above the caudal part of the yolk tube. The number of embryos observed is indicated at the top. Scale bars: 25 μm. DA, dorsal aorta; NT, neural tube; PCV, posterior cardinal vein.
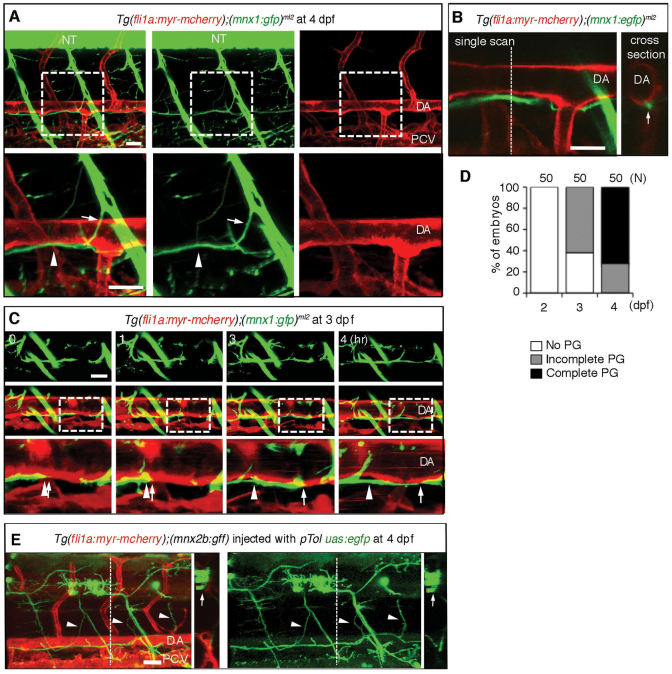
To ascertain whether axons beneath the DA developed from motoneurons or other neurons, we observed embryos expressing GFP under the mnx1 promoter, which is activated in motoneurons, crossed with fish expressing mCherry under the control of the fli1a promoter, Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 (Flanagan-Steet et al., 2005). We noticed that the axons derived from motoneurons marked by GFP located beneath the DA at 4 dpf (Fig. 2A). Therefore, we time-lapse imaged the embryos and found that the axons of motoneurons started developing caudally and rostrally as a single fascicle beneath the DA from 3 dpf (Fig. 2B-D; supplementary material Fig. S1A and Movie 2). To examine which motoneuron extends axons beneath the DA, we tried to label single cells by injecting pTol uas:egfp into Tg (fli1a:myr-mcherry);(mnx2b:gff) embryos. The motoneurons marked by EGFP in the neural tube extended axons towards the ventromedial area and beneath the dorsal aorta. In addition, when we performed the single-cell labeling experiment, those motoneurons seemed to exist in every somite and extend axons outside of the neural tube bilaterally (Fig. 2E; supplementary material Fig. S1B-D and Movie 3). Therefore, the subpopulation of the motoneurons we identified in the present study corresponds to the intermyotomal secondary motoneurons or the ventromedial secondary motoneurons, as previously reported (Asakawa et al., 2013; Menelaou and McLean, 2012).
Fig. 2.
Axons of motoneurons develop beneath the dorsal aorta. (A) 3D-rendered confocal stack of fluorescence images (lateral view) of an Tg(fli1a;myr-mcherry);(mnx1:gfp)ml2 embryo at 4 dpf. Left column, merged images; center column, GFP images; right column, mCherry images. Lower panels are enlarged images of the boxed regions in the upper panels. Anterior is to the left. Arrows indicate the branch of the descending axon of motoneurons. Arrowheads indicate the extension of axons beneath the DA. (B) Left, a single scan confocal image from the stack shown in A. Right, a cross-section image of the stacked image shown in A at the level of the dashed line in the left panel. Arrow denotes the single fascicle of axons. (C) 3D-rendered confocal time-sequential stack images of an Tg (fli1a:myr-mcherry);(mnx1:gfp)ml2embryo. Elapsed time (hours) from the start point of time-lapse imaging (3 dpf) is indicated. Top panels, GFP images; middle panels, merged images of GFP and mCherry; bottom panels, enlarged images of boxed regions of the middle panels. Arrows indicate the tip of a neuronal axon. Arrowheads denote the location of the tip when starting time-lapse imaging. (D) Parallel growth (PG) of motoneuron axons with the DA was quantitatively analyzed. ‘Complete’ indicates the complete continuity of the axon between the region above the rostral part of the yolk tube and that above the caudal part of the yolk tube. The number of embryos observed is indicated at the top. (E) Single-cell labeling experiment of an embryo of the indicated genotype. Left, 3D-rendered confocal merged image of mCherry and EGFP with a cross-section image at the level of the dashed line. Right, EGFP image with a cross-section image of the EGFP image at the level of the dashed line. Arrows and arrowheads denote the cell bodies of motoneurons in the neural tube and their axons extending beneath the DA, respectively. Scale bars: 25 μm. DA, dorsal aorta; NT, neural tube; PCV, posterior cardinal vein.
Motoneurons express Vegfr3
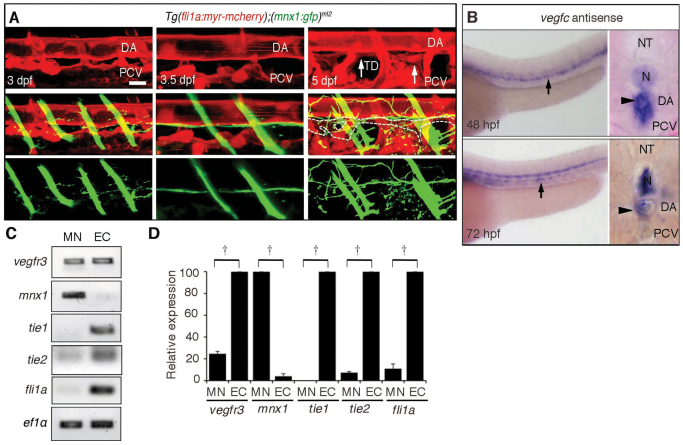
The thoracic duct (TD), a major lymphatic vessel, is known to develop between the DA and the PCV (Yaniv et al., 2006). Thus, we tried to examine the exact location of the TD and motoneuron axons beneath the DA. Motoneuron axons were located between the DA and TD. The TD formed at 5 dpf, whereas extension of motoneuron axons was completed by 3.5 dpf. Therefore, the extension of axons preceded the formation of the TD (Fig. 3A). In addition, when we looked closely at TD formation, it developed along the preformed axons of motoneurons in Tg(fli1a:egfp)y1 embryos transiently expressing mCherry by Tol2-mediated gene transfer using mnx2b promoter, which is activated in motoneurons (Asakawa et al., 2012; Kawakami et al., 2004) (supplementary material Fig. S2A). These results prompted us to assume that the alignment of the motoneuron axons and the DA might depend on the molecular mechanism by which the TD develops along the DA, which is probably dependent on Vegfc-mediated signaling, because lymphatic vessel development depends on Vegfc/Vegfr3 signaling in zebrafish (Küchler et al., 2006).
Fig. 3.
Motoneurons express Vegfr3. (A) 3D-rendered confocal stack of fluorescence images (lateral view) of a Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryo at the times indicated in the top panels. Top panels, blood vessels and lymphatic vessels marked by mCherry; middle panels, merged images of GFP and mCherry; bottom panels, motoneurons marked by GFP. Arrows indicate the TD. Dashed lines in the middle panel indicate the outline of the TD. Note that the axon extension preceded TD development and that the axons were formed between DA and TD. Scale bar: 25 μm. (B) In situ hybridization analyses of vegfc mRNA in embryos at the indicated stages. Arrows and arrowheads indicate vegfc mRNA detected in the DA in the lateral views (left) and cross-section views (right), respectively. (C) RT-PCR analyses using the primers indicated at the left and RNAs prepared from the Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos by FACS at 72 hpf. (D) Real-time quantitative RT-PCR analyses using RNAs prepared from embryos at 72 hpf by the primers as indicated at the bottom. †P<0.05. Error bars indicate s.d. DA, dorsal aorta; EC, endothelial cells; MN, motoneurons; N, notochord; NT, neural tube; PCV, posterior cardinal vein; TD, thoracic duct.
Vegfc is essential for development of the TD and is expressed in the DA (Covassin et al., 2006; Küchler et al., 2006; Villefranc et al., 2013). We confirmed the expression of vegfc mRNA in the DA of embryos at 24, 48 and 72 hpf by in situ hybridization (Fig. 3B; supplementary material Fig. S2B,C). Then, we hypothesized that motoneurons might express Vegfr3 similar to lymphatic endothelial cells and extend axons in response to Vegfc from the DA. To test this hypothesis, we examined the expression of vegfr3 mRNA by in situ hybridization. We could detect it in the PCV and in the intersomitic vessels (ISVs) but not in the neurons at 24 or 48 hpf (supplementary material Fig. S2D). At 72 hpf, even its expression in the PCV was not clear. vegfr3 mRNA expression was found in the neural tube when the reaction was extended (supplementary material Fig. S2E). Therefore, to analyze vegfr3 mRNA expression in embryos at 72 hpf, we performed RT-PCR. Motoneurons and vascular endothelial cells were collected by FACS from Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos. RNAs from the collected cells were first analyzed by RT-PCR. Motoneurons and endothelial cells expressed vegfr3 mRNA (Fig. 3C). The purity of RNA for RT-PCR was confirmed by the observation that mnx1 mRNA was detected exclusively in motoneurons and that tie1 mRNA, tie2 (Tek) mRNA and fli1a mRNA were detected only in endothelial cells (Fig. 3C). To examine quantitatively the expression of vegfr3 mRNA, we performed qRT-PCR and found that vegfr3 mRNA was detected in motoneurons although its expression was less than that in the endothelial cells (Fig. 3D).
Motoneuron expressing Vegfr3 responds to Vegfc
To test whether motoneurons respond to Vegfc, we examined neurite sprouts from motoneurons in an ex vivo model. The Tg(mnx1:gfp)ml2 embryos, from which heads and yolks were dissected from the trunks at 36 hpf, were incubated in matrigel containing recombinant human (h)VEGFC (Fig. 4A). We measured the number of sprouting neurites marked by GFP from embryos in five groups: those incubated without recombinant (h)VEGFC; those treated with control DMSO and incubated with (h)VEGFC; those treated with vegfr3 morpholino (MO) and incubated with (h)VEGFC; those treated with (h)VEGFC plus the VEGFR3 inhibitor maz51 (Ny et al., 2008); and those treated with (h)VEGFC plus the VEGFR2 inhibitor ki8751 (Kubo et al., 2005) (Fig. 4B,C; supplementary material Fig. S3A). (h)VEGFC-induced sprouting of neurites was inhibited by either knockdown or inhibition of Vegfr3 but not by inhibition of Vegfr2, suggesting that neurite outgrowth from motoneurons depends on Vegfc/Vegfr3 signaling. We then verified whether (z)Vegfr3 can respond to (h)VEGFC in cultured cells. Cells expressing (z)Vegfr3 exhibited phosphorylation of extracellular-regulated kinase (Erk) when stimulated with (h)VEGFC (Fig. 4D), confirming that motoneurons extend neurites in response to (h)VEGFC.
Fig. 4.
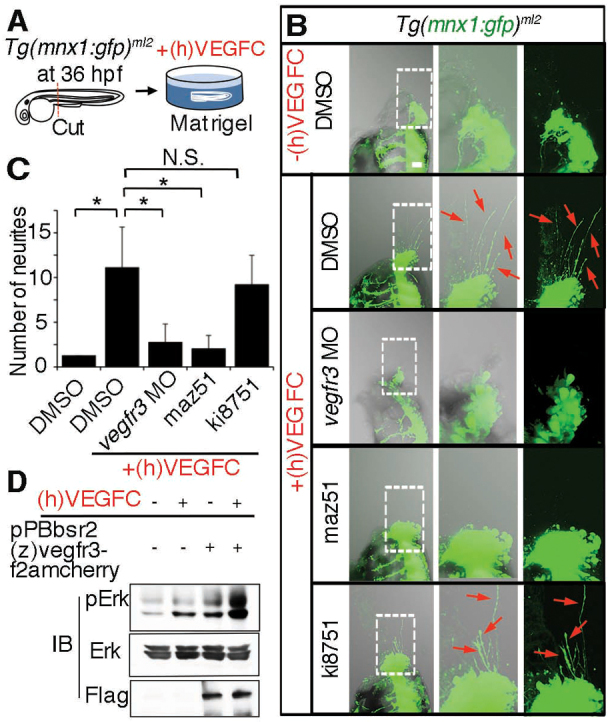
Motoneurons extend neurites in response to recombinant VEGFC. (A) Schematic of an ex vivo assay for examining neurite sprouting from motoneurons. Briefly, the Tg(mnx1:gfp)ml2 embryos were cut at the dorsoventral line between the main yolk and the rostrally extended yolk tube and was embedded in matrigel with or without recombinant human vascular endothelial growth factor-C, (h)VEGFC. (B) z-stack confocal images (left panel, merged image of transmitted light image and GFP image; center, enlarged image of the boxed region in the left panel; right, GFP image of the boxed region in the left panel) of Tg(mnx1:gfp)ml2 embryos incubated in matrigel. Upper panels, embryo in matrigel without (h)VEGFC; lower four panels, embryos in matrigel with (h)VEGFC and treated as indicated at the left. Red arrows indicate GFP-positive neurites from motoneurons. Anterior is at the top and dorsal to the right. (C) Quantitative analyses (ANOVA) of neurite number of the embryos grouped as in B. *P<0.05. Plotted is mean ± s.d. of more than ten embryos for each group. (D) Immunoblot analyses with the antibodies indicated at the left using lysates of parental HEK293 and those stably transfected with pPBbsr2(z)vegfr3-f2amcherry and treated with or without (h)VEGFC. A representative result of three independent experiments is shown.
Vegfc/Vegfr3 signaling augments axon growth of motoneurons
To test whether motoneurons respond to Vegfc in vivo, we examined the effects of overexpression of (z)Vegfc in arteries or (z)Vegfr3 in motoneurons on the axonal growth of motoneurons. We observed the growth of axons in Tg(mnx1:gfp)ml2 embryos injected with QDot 655 into the blood vessels at 4 dpf to visualize both motoneurons and blood vessels. We used the zebrafish flt1 promoter/enhancer and mnx2b promoter for transient overexpression of (z)Vegfc and (z)Vegfr3 in arteries and motoneurons, respectively (Asakawa et al., 2012; Bussmann et al., 2010; Ny et al., 2008). Artery-specific expression mediated by the flt1 promoter/enhancer and motoneuron-specific expression by mnx2b promoter was confirmed by transient expression of mCherry in Tg (fli1a:egfp)y1 and Tg (mnx1:gfp)ml2 embryos (supplementary material Fig. S4A,B). Forced expression of (z)Vegfc in arteries resulted in an increase in branching of motoneuron axons over the DA, whereas that of control (mCherry) did not (Fig. 5A,B,E). Similarly, overexpression of (z)Vegfr3 in motoneurons induced branching of motoneuron axons over the DA, whereas that of control (mCherry) did not alter any parallel growth of motoneuron axons with the DA (Fig. 5C-E). Overexpression of Flag-tagged (z)Vegfr3 was confirmed by expression of mCherry in the motoneurons in the neural tube (supplementary material Fig. S4C). These results indicate that gain of Vegfc/Vegfr3 signaling between the DA and motoneuron axons enhances their congruency.
Fig. 5.
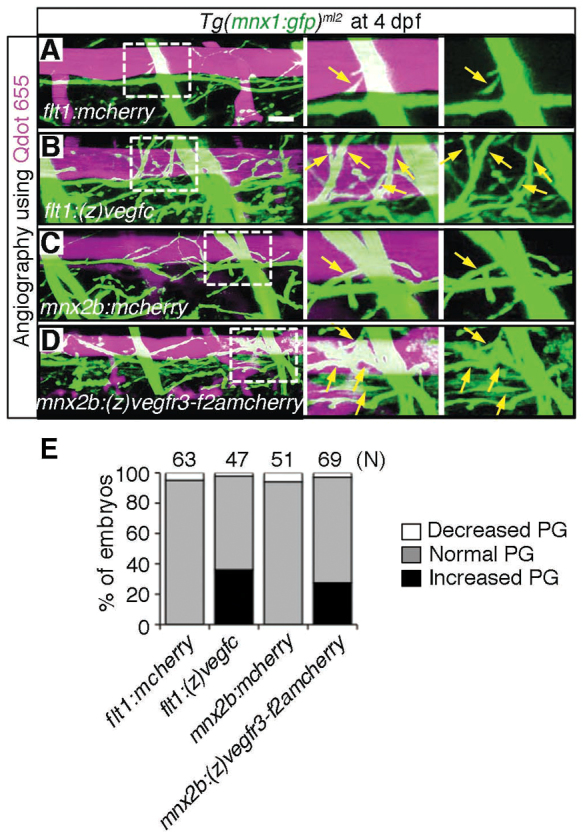
Overexpression of either Vegfc in the dorsal aorta or Vegfr3 in the motoneurons enhances axon branches of motoneurons over the dorsal aorta. (A-D) 3D-rendered confocal stack images (lateral view) of Tg(mnx1:gfp)ml2 embryos transiently expressing the molecules indicated at the bottom left of the left-hand panels. The embryos were intravascularly injected with Qdot 655. Left panels, merged images of GFP and Qdot 655 images; center panels, enlarged image of boxed region of the left panels; right panels, enlarged image of boxed region of the left panels showing GFP signal only. (A) Tg(mnx1:gfp)ml2 embryo transiently expressing mCherry in arteries under the control of the flt1 promoter by Tol2-mediated gene transfer. Scale bar: 25 μm. (B) Zebrafish expressing (z)Vegfc similar to A. (C) Embryo transiently expressing mCherry in motoneurons under the control of the mnx2b promoter by Tol2-mediated gene transfer. (D) Embryo transiently expressing zebrafish (z)Vegfr3 tagged with Flag (F) followed by 2A peptide and mCherry [(z)Vegfr3-F2AmCherry] similar to C. Arrows indicate branches of motoneurons. (E) Quantitative analyses of branching of axons over the DA. The number of the embryos showing the increased, normal or decreased parallel growth (PG) divided by the total number (n) of embryos counted (indicated at the top) is expressed as percentage of embryos.
Vegfc/Vegfr3 signaling is essential for axon growth of motoneurons beneath the dorsal aorta
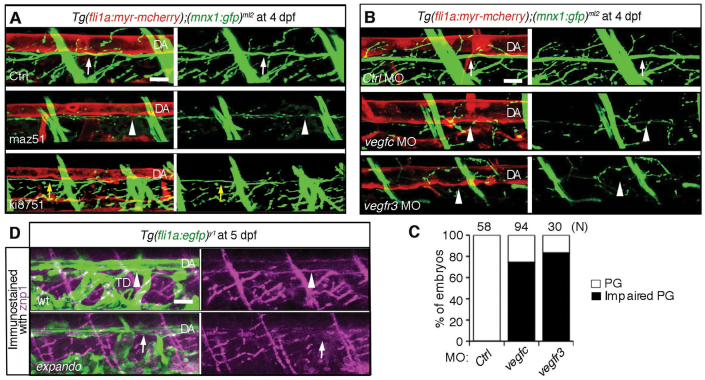
To examine whether Vegfc/Vegfr3 signaling between the DA and motoneurons is essential for the axon extension of motoneurons beneath the DA, we investigated the effect of inhibition of Vegfc/Vegfr3 signaling or depletion of Vegfc or Vegfr3 on the alignment of motoneuron axons and the DA. We observed the alignment of motoneurons beneath DA in Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos treated with maz51 and ki8751 to inhibit Vegfr3 and Vegfr2, respectively. Inhibition of Vegfr3 but not Vegfr2 clearly reduced the diameter of axons and the number of branches, indicating an important role for Vegfr3-mediated signaling in the growth of axons beneath the DA (Fig. 6A). We then injected either vegfc MO or vegfr3 MO into one-cell-stage Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos (Fig. 6B; supplementary material Fig. S3B,C). Depletion of either Vegfc or Vegfr3 resulted in less axon extension of motoneuron beneath the DA (Fig. 6B,C). Depletion of Vegfc did not affect the ventral axon growth of motoneurons (supplementary material Fig S3D). To examine the requirement of Vegfr3 for the growth of motoneuron axon beneath the DA, we examined the presence of motoneuron axons beneath the DA by immunohistochemistry of Tg(fli1a:egfp)y1 and Tg(fli1a:egfp)y1;flt4hu4602 (expando mutant;Vegfr3 I1034F) using znp1 antibody against synaptotagmin 2. Whereas motoneuron axons were detectable beneath the DA in the control embryos, those in expando mutants (Vegfr3 mutants) were hardly detectable (Fig. 6D). These data indicate the essential role for Vegfc/Vegfr3 signaling in the parallel growth of the motoneuron axons and DA.
Fig. 6.
Requirement of Vegfc and Vegfr3 for alignment of motoneuron axons and dorsal aorta. (A) 3D-rendered confocal stack images (lateral view) of Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos treated with control DMSO (Ctrl, upper panels), with maz51 (middle panels) or with ki8751 (lower panels) at 4 dpf. Left panels, merged images of mCherry and GFP; right panels, GFP images. White arrows indicate the motoneurons beneath the DA. Arrowheads indicate the reduced growth of motoneuron axons beneath the DA. Yellow arrows indicate the slight reduction of growth of motoneuron axon beneath the DA. (B) 3D-rendered confocal stack images (lateral view) of Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos treated with control MO (Ctrl, top panels), with vegfc MO (middle panels) or with vegfr3 MO (bottom panels). Left panels, merged images of mCherry and GFP; right panels, GFP images. Arrows indicate the motoneuron axons beneath the DA. Arrowheads indicate the reduced interaction of motoneurons and dorsal aorta. (C) Quantitative analyses of impairment of parallel growth (PG) of the embryos grouped as in B. The number (n) of the embryos examined for quantitative analyses is indicated at the top. (D) Immunohistochemical study of Tg(fli1a:egfp)y1 embryos (top) and vegfr3hu4602/wt(expando mutant);Tg(fli1a:egfp)y1 embryos using anti-znp1 antibody. Arrowheads and arrows indicate the presence or absence of axons beneath the dorsal aorta, respectively. Note that the TD is absent in the expando mutant. Scale bar: 25 μm. DA, dorsal aorta; TD, thoracic duct.
Motoneuron-specific inhibition of Vegfr3 reduces the parallel growth of motoneuron axons and dorsal aorta
Because the effect of MO is not restricted in either DA or motoneurons, we examined the effect of Vegfr3 inhibition specifically in motoneurons on axon extension beneath the DA. To inhibit Vegfr3, we overexpressed a mutant form of (h)VEGFR3 fused with human IgG Fc, (h)VEGFR3-Fc (Mäkinen et al., 2001; Ober et al., 2004). We first confirmed that this mutant could inhibit both (h)VEGFC- or (z)Vegfc-triggered signaling. HUVECs were stimulated with recombinant (h)VEGFC in the presence or absence of (h)VEGFR3-Fc prepared by the conditioned medium of cells transfected with the plasmid expressing (h)VEGFR3-Fc. (h)VEGFC-dependent phosphorylation of Erk was reduced in the cells treated with (h)VEGFR3-Fc compared with those without (h)VEGFR3-Fc (supplementary material Fig. S5A). We also tested whether this human VEGFR3 mutant could inhibit zebrafish Vegfc/Vegfr3 signaling by treating HEK293 cells stably expressing (z)Vegfr3 with (z)Vegfc in the presence or absence of (h)VEGFR3-Fc. (z)Vegfc-dependent phosphorylation of Erk via (z)Vegfr3 was inhibited by (h)VEGFR3-Fc (supplementary material Fig. S5B).
We, therefore, examined the effect of overexpression of (h)VEGFR3-Fc exclusively in motoneurons on the parallel growth of motoneurons along the DA. We transiently and specifically expressed (h)VEGFR3-Fc using the mnx2b promoter in the motoneurons of Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos. The embryos injected with the plasmid expressing (h)VEGFR3-Fc exhibited less axon extension of motoneurons than did the control embryos (Fig. 7A). The impairment of parallel growth of the motoneuron axons and DA was quantitatively analyzed (Fig. 7B). We further corroborated the essential role for Vegfr3 of motoneuron axons in the alignment with the DA by overexpressing another dominant-negative form of (z)Vegfr3. We tested the effect of overexpression of (z)Vegfr3 lacking the cytoplasmic kinase domain (z)Vegfr3deltaRTK, which is expressed on the cell surface by the transmembrane domain, on the alignment of the motoneuron axons and DA. Expression of (z)Vegfr3deltaRTK in the cells stably expressing (z)Vegfr3 led to a reduction of (z)Vegfc-induced phosphorylation of Erk (supplementary material Fig. S5C,D). Thus, we transiently expressed this (z)Vegfr3deltaRTK in motoneurons of Tg(mnx1:gfp)ml2 embryos and examined the alignment of motoneuron and DA by injecting Qdot 655 to visualize blood vessels. Overexpression of (z)Vegfr3deltaRTK resulted in reduced extension of motoneuron axons even though the (z)Vegfc was overexpressed in the arteries (supplementary material Fig. S5E). Collectively, these data indicate that Vegfr3 expression on motoneurons is essential for the extension of axons in response to Vegfc from the DA.
Fig. 7.
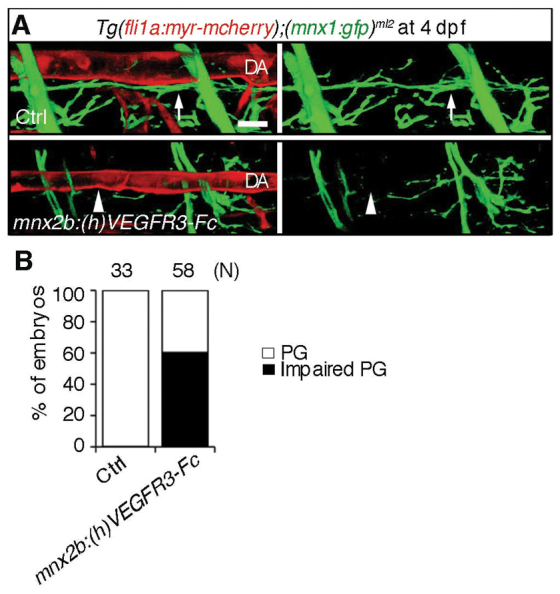
Inhibition of Vegfr3 in the motoneuron results in impairment of alignment of the motoneuron axons and dorsal aorta. (A) 3D-rendered confocal stack images (lateral view) of Tg(fli1a:myr-mcherry);(mnx1:gfp)ml2 embryos (upper panel) and those transiently expressing (h)VEGFR3-Fc in the motoneurons under the control of the mnx2b promoter by Tol2-mediated gene transfer (lower panels). Left panels, merged images of mCherry and GFP; right panels, GFP images. Arrows indicate the motoneuron axon beneath the dorsal aorta (DA). Arrowheads indicate the impairment of parallel growth of motoneuron axon with the DA. Scale bar: 25 μm. (B) Quantitative analyses of the impaired parallel growth (PG) of the embryos grouped as in A. The number (n) of the embryos observed indicated at the top.
DISCUSSION
In the present study, we provide evidence that Vegfc functions as a guidance molecule for the axon growth of motoneurons. We found that Vegfr3 was expressed in motoneurons and that forced expression of the Vegfc-trapping (h)VRGFR3 mutant resulted in the inhibition of parallel growth of motoneuron axons beneath the DA expressing Vegfc. The TD, a main lymphatic vessel formed between the DA and PCV, needs Vegfc to grow in zebrafish (Küchler et al., 2006; Villefranc et al., 2013; Yaniv et al., 2006). We found that the formation of motoneuron axons beneath the DA preceded that of the TD. These spatiotemporal data suggest that axon development employs the same mechanism underlying the development of the TD, because both lymphatic vessel and motoneurons express Vegfr3. We cannot completely exclude the involvement of Vegfr2 in motoneurons, because Vegfc activates the heterodimer complex of Vegfr2-Vegfr3 (Herbert and Stainier, 2011). We examined the effect of a Vegfr2 inhibitor (ki8751) on the parallel growth and noticed that the axon beneath the DA seemed to be slightly affected, although 0.5 μM ki8751 did not affect the secondary sprouting from the PCV that is promoted by Vegfc. Therefore, we can conclude at least that Vegfc/Vegfr3 signaling is required for the alignment of the axons and DA.
Why and how do the axons extend ventrally and grow beneath the DA? Although Vegfc is expressed in the DA at 48 hpf, it is expressed in the hypochord at an early stage (18-somite stage) (Covassin et al., 2006). We assumed that Vegfc from the hypochord might be essential for the ventral growth of axons and Vegfc from the DA for parallel growth of the axons beneath the DA. Knockdown of Vegfc did not affect the ventral growth but did affect the parallel growth with the DA, suggesting that the ventral axon growth depends on the other guidance cues. PlexinA3 expressed on motoneurons and sema3A2 (sema3ab) in somites, dcc on motoneurons and netrin in myoseptum have been reported to be involved in ventral growth of motoneuron axons from the cell body in the neural tube (Feldner et al., 2007; Lim et al., 2011). The axons of motoneurons grew more ventrally beyond the DA and grew beneath the DA. It is probable that some molecules from the PCV might attract the axons ventral to the DA. Whereas Cxcl12a is expressed in the PCV at 72 hpf (Cha et al., 2012), Cxcr4, a receptor for Cxcl12, is expressed on neuronal cells (Tiveron and Cremer, 2008). Cxcl12a from the PCV might attract the axons ventrally to the DA. In addition, some repulsive cues from the DA might prevent the axons from touching the DA. During the pathfinding of axon growth, neurons utilize Ephrin/Eph, Netrin/Unc5, Semaphorin/Plexin and NP signaling and the Slit/Robo system (Carmeliet and Tessier-Lavigne, 2005). Because the DA expresses ephrinB2, motoneuron axons might express EphB family receptors to prevent the budding of axon branches. These questions about the effect of Cxcl12 in the PCV on the axons and the expression of EphB family in the motoneuron axons need to be examined further.
VEGFA is a bipotential, angiogenic and neurotrophic molecule. VEGFA found as an angiogenic growth factor is capable of activating VEGFR2 expressed on neurons (Bearden and Segal, 2004; Jin et al., 2002). By contrast, VEGFA from neurons guides neovascularization in the skin, suggesting that a preformed network could give guidance cues to the other network (Cantarella et al., 2002; Mukouyama et al., 2002). Although these reports accelerate the research on VEGFA-mediated signaling in both blood vessels and neurons, involvement of other VEGFs, including VEGFC, in the congruence of blood vessels and neurons has been largely unknown. Therefore, this study provides the first evidence that Vegfc is involved in the pathfinding of axon growth in zebrafish.
Vegfc/Vegfr3 signaling might be important not only for pathfinding but also for the maintenance of motoneurons. When VEGFA is insufficient for activating VEGFR2-expressing motoneurons, they become degenerative, as proven in mice in which the Vegfa promoter is mutated (Oosthuyse et al., 2001). Consistently, mutations of the VEGFA promoter are reported in patients with amyotrophic lateral sclerosis associated with motoneuron degeneration (Lambrechts et al., 2003). Thus, if VEGFC can activate VEGFR3 expressed on motoneurons, VEGFC/VEGFR3 signaling might prevent motoneurons from regenerating in the patients with motoneuron diseases. VEGFR2 and its co-receptor NRP1 respond to VEGFA in vascular endothelial cells (Soker et al., 1998), whereas VEGFR3 and NRP2 are co-activated by VEGFC to induce sprouting of lymphatic vessels (Kärpänen et al., 2006). Nrp1a and Nrp1b are expressed in the DA and motoneurons, whereas Nrp2a and Nrp2b are expressed in the region surrounding the PCV in zebrafish (Martyn and Schulte-Merker, 2004). Although both are expressed in neural tissues, the expression of Np-2 in motoneurons has remained elusive. It will be interesting to explore whether VEGFR3 and NP-2 function not only for outgrowth of motoneuron axons but also for survival of motoneurons.
In summary, using transgenic zebrafish expressing GFP in motoneurons and mCherry in vascular endothelial cells, we revealed that a subpopulation of secondary motoneurons extended their axons along the preformed DA. This alignment is regulated by Vegfc/Vegfr3 signaling.
Acknowledgments
We thank Nathan Lawson (Universtiy of Massachusetts Medical School) for providing us with the fli1a promoter DNA and Tg(fli1a:egfp)y1 fish; Stefan Schulte-Merker (Hubrecht Institute-KNAW) for providing us with expando (Vegfr3) mutant fish; Allan Bradley (Wellcome Trust Sanger Institute) and Michiyuki Matsuda (Kyoto University) for providing us with piggyBac system and pPBbsr2, respectively; Didier Stainier (Max Planck Institute) for his comments and materials; James Pearson for critical reading; Manami Sone, Keiko Hiratomi, Wakana Koeda and Yuko Matsuura for excellent technical assistance; and Yukiko Shintani for fish care. We are grateful to the National BioResource Project from MEXT, Japan for several lines of fish.
Footnotes
Funding
This work was partly supported by Japan Society for the Promotion of Science KAKENHI grants [24370084 and 22122003 to N.M.; 22390040 and 22113009 to S.F.]; by Health Labor Science research grants [to N.M.]; by a grant from Takeda Science Foundation [to N.M. and S.F.]; by an AstraZeneca research grant [to N.M.]; and by funding from the Senshin Medical Research Foundation [to N.M.]. Deposited in PMC for immediate release.
Competing interests statement
The authors declare no competing financial interests.
Author contributions
H.-B.K., K. Asakawa, K. Ando and T.K. performed the experiments; S.F., K.K., M.H., Y.-G.K., K.-W.K. and N.M. designed the research; S.F. and K.K. analyzed the data; and K. Alitalo and N.M. wrote the paper.
Supplementary material
Supplementary material available online at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.091702/-/DC1
References
- Adams R. H., Eichmann A. (2010). Axon guidance molecules in vascular patterning. Cold Spring Harb. Perspect. Biol. 2, a001875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asakawa K., Suster M. L., Mizusawa K., Nagayoshi S., Kotani T., Urasaki A., Kishimoto Y., Hibi M., Kawakami K. (2008). Genetic dissection of neural circuits by Tol2 transposon-mediated Gal4 gene and enhancer trapping in zebrafish. Proc. Natl. Acad. Sci. USA 105, 1255–1260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asakawa K., Higashijima S., Kawakami K. (2012). An mnr2b/hlxb9lb enhancer trap line that labels spinal and abducens motor neurons in zebrafish. Dev. Dyn. 241, 327–332 [DOI] [PubMed] [Google Scholar]
- Asakawa K., Abe G., Kawakami K. (2013). Cellular dissection of the spinal cord motor column by BAC transgenesis and gene trapping in zebrafish. Front Neural Circuits. 7, 100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bearden S. E., Segal S. S. (2004). Microvessels promote motor nerve survival and regeneration through local VEGF release following ectopic reattachment. Microcirculation 11, 633–644 [DOI] [PubMed] [Google Scholar]
- Bearden S. E., Segal S. S. (2005). Neurovascular alignment in adult mouse skeletal muscles. Microcirculation 12, 161–167 [DOI] [PubMed] [Google Scholar]
- Bussmann J., Bos F. L., Urasaki A., Kawakami K., Duckers H. J., Schulte-Merker S. (2010). Arteries provide essential guidance cues for lymphatic endothelial cells in the zebrafish trunk. Development 137, 2653–2657 [DOI] [PubMed] [Google Scholar]
- Calvo C. F., Fontaine R. H., Soueid J., Tammela T., Makinen T., Alfaro-Cervello C., Bonnaud F., Miguez A., Benhaim L., Xu Y., et al. (2011). Vascular endothelial growth factor receptor 3 directly regulates murine neurogenesis. Genes Dev. 25, 831–844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantarella G., Lempereur L., Presta M., Ribatti D., Lombardo G., Lazarovici P., Zappalà G., Pafumi C., Bernardini R. (2002). Nervegrowth factor-endothelial cell interaction leads to angiogenesis in vitro and in vivo. FASEB J. 16, 1307–1309 [DOI] [PubMed] [Google Scholar]
- Carmeliet P., Tessier-Lavigne M. (2005). Common mechanisms of nerve and blood vessel wiring. Nature 436, 193–200 [DOI] [PubMed] [Google Scholar]
- Cha Y. R., Fujita M., Butler M., Isogai S., Kochhan E., Siekmann A. F., Weinstein B. M. (2012). Chemokine signaling directs trunk lymphatic network formation along the preexisting blood vasculature. Dev. Cell 22, 824–836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Covassin L. D., Villefranc J. A., Kacergis M. C., Weinstein B. M., Lawson N. D. (2006). Distinct genetic interactions between multiple Vegf receptors are required for development of different blood vessel types in zebrafish. Proc. Natl. Acad. Sci. USA 103, 6554–6559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichmann A., Makinen T., Alitalo K. (2005). Neural guidance molecules regulate vascular remodeling and vessel navigation. Genes Dev. 19, 1013–1021 [DOI] [PubMed] [Google Scholar]
- Feldner J., Reimer M. M., Schweitzer J., Wendik B., Meyer D., Becker T., Becker C. G. (2007). PlexinA3 restricts spinal exit points and branching of trunk motor nerves in embryonic zebrafish. J. Neurosci. 27, 4978–4983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flanagan-Steet H., Fox M. A., Meyer D., Sanes J. R. (2005). Neuromuscular synapses can form in vivo by incorporation of initially aneural postsynaptic specializations. Development 132, 4471–4481 [DOI] [PubMed] [Google Scholar]
- Guan K. L., Rao Y. (2003). Signalling mechanisms mediating neuronal responses to guidance cues. Nat. Rev. Neurosci. 4, 941–956 [DOI] [PubMed] [Google Scholar]
- Herbert S. P., Stainier D. Y. (2011). Molecular control of endothelial cell behaviour during blood vessel morphogenesis. Nat. Rev. Mol. Cell Biol. 12, 551–564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hogan B. M., Herpers R., Witte M., Heloterä H., Alitalo K., Duckers H. J., Schulte-Merker S. (2009). Vegfc/Flt4 signalling is suppressed by Dll4 in developing zebrafish intersegmental arteries. Development 136, 4001–4009 [DOI] [PubMed] [Google Scholar]
- Honma Y., Araki T., Gianino S., Bruce A., Heuckeroth R., Johnson E., Milbrandt J. (2002). Artemin is a vascular-derived neurotropic factor for developing sympathetic neurons. Neuron 35, 267–282 [DOI] [PubMed] [Google Scholar]
- Isogai S., Horiguchi M., Weinstein B. M. (2001). The vascular anatomy of the developing zebrafish: an atlas of embryonic and early larval development. Dev. Biol. 230, 278–301 [DOI] [PubMed] [Google Scholar]
- James J. M., Mukouyama Y. S. (2011). Neuronal action on the developing blood vessel pattern. Semin. Cell Dev. Biol. 22, 1019–1027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin K., Zhu Y., Sun Y., Mao X. O., Xie L., Greenberg D. A. (2002). Vascular endothelial growth factor (VEGF) stimulates neurogenesis in vitro and in vivo. Proc. Natl. Acad. Sci. USA 99, 11946–11950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karkkainen M. J., Haiko P., Sainio K., Partanen J., Taipale J., Petrova T. V., Jeltsch M., Jackson D. G., Talikka M., Rauvala H., et al. (2004). Vascular endothelial growth factor C is required for sprouting of the first lymphatic vessels from embryonic veins. Nat. Immunol. 5, 74–80 [DOI] [PubMed] [Google Scholar]
- Kärpänen T., Heckman C. A., Keskitalo S., Jeltsch M., Ollila H., Neufeld G., Tamagnone L., Alitalo K. (2006). Functional interaction of VEGF-C and VEGF-D with neuropilin receptors. FASEB J. 20, 1462–1472 [DOI] [PubMed] [Google Scholar]
- Kawakami K., Takeda H., Kawakami N., Kobayashi M., Matsuda N., Mishina M. (2004). A transposon-mediated gene trap approach identifies developmentally regulated genes in zebrafish. Dev. Cell 7, 133–144 [DOI] [PubMed] [Google Scholar]
- Komatsu N., Aoki K., Yamada M., Yukinaga H., Fujita Y., Kamioka Y., Matsuda M. (2011). Development of an optimized backbone of FRET biosensors for kinases and GTPases. Mol. Biol. Cell 22, 4647–4656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubo K., Shimizu T., Ohyama S., Murooka H., Iwai A., Nakamura K., Hasegawa K., Kobayashi Y., Takahashi N., Takahashi K., et al. (2005). Novel potent orally active selective VEGFR-2 tyrosine kinase inhibitors: synthesis, structure-activity relationships, and antitumor activities of N-phenyl-N′-4-(4-quinolyloxy)phenylureas. J. Med. Chem. 48, 1359–1366 [DOI] [PubMed] [Google Scholar]
- Küchler A. M., Gjini E., Peterson-Maduro J., Cancilla B., Wolburg H., Schulte-Merker S. (2006). Development of the zebrafish lymphatic system requires VEGFC signaling. Curr. Biol. 16, 1244–1248 [DOI] [PubMed] [Google Scholar]
- Lambrechts D., Storkebaum E., Morimoto M., Del-Favero J., Desmet F., Marklund S. L., Wyns S., Thijs V., Andersson J., van Marion I., et al. (2003). VEGF is a modifier of amyotrophic lateral sclerosis in mice and humans and protects motoneurons against ischemic death. Nat. Genet. 34, 383–394 [DOI] [PubMed] [Google Scholar]
- Larrivée B., Freitas C., Suchting S., Brunet I., Eichmann A. (2009). Guidance of vascular development: lessons from the nervous system. Circ. Res. 104, 428–441 [DOI] [PubMed] [Google Scholar]
- Lawson N. D., Weinstein B. M. (2002). In vivo imaging of embryonic vascular development using transgenic zebrafish. Dev. Biol. 248, 307–318 [DOI] [PubMed] [Google Scholar]
- Lawson N. D., Scheer N., Pham V. N., Kim C. H., Chitnis A. B., Campos-Ortega J. A., Weinstein B. M. (2001). Notch signaling is required for arterial-venous differentiation during embryonic vascular development. Development 128, 3675–3683 [DOI] [PubMed] [Google Scholar]
- Lawson N. D., Vogel A. M., Weinstein B. M. (2002). sonic hedgehog and vascular endothelial growth factor act upstream of the Notch pathway during arterial endothelial differentiation. Dev. Cell 3, 127–136 [DOI] [PubMed] [Google Scholar]
- Le Bras B., Barallobre M. J., Homman-Ludiye J., Ny A., Wyns S., Tammela T., Haiko P., Karkkainen M. J., Yuan L., Muriel M. P., et al. (2006). VEGF-C is a trophic factor for neural progenitors in the vertebrate embryonic brain. Nat. Neurosci. 9, 340–348 [DOI] [PubMed] [Google Scholar]
- Lewis K. E., Eisen J. S. (2003). From cells to circuits: development of the zebrafish spinal cord. Prog. Neurobiol. 69, 419–449 [DOI] [PubMed] [Google Scholar]
- Li W., Kohara H., Uchida Y., James J. M., Soneji K., Cronshaw D. G., Zou Y. R., Nagasawa T., Mukouyama Y. S. (2013). Peripheral nerve-derived CXCL12 and VEGF-A regulate the patterning of arterial vessel branching in developing limb skin. Dev. Cell 24, 359–371 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim A. H., Suli A., Yaniv K., Weinstein B., Li D. Y., Chien C. B. (2011). Motoneurons are essential for vascular pathfinding. Development 138, 3847–3857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mackenzie F., Ruhrberg C. (2012). Diverse roles for VEGF-A in the nervous system. Development 139, 1371–1380 [DOI] [PubMed] [Google Scholar]
- Mäkinen T., Jussila L., Veikkola T., Karpanen T., Kettunen M. I., Pulkkanen K. J., Kauppinen R., Jackson D. G., Kubo H., Nishikawa S., et al. (2001). Inhibition of lymphangiogenesis with resulting lymphedema in transgenic mice expressing soluble VEGF receptor-3. Nat. Med. 7, 199–205 [DOI] [PubMed] [Google Scholar]
- Makita T., Sucov H. M., Gariepy C. E., Yanagisawa M., Ginty D. D. (2008). Endothelins are vascular-derived axonal guidance cues for developing sympathetic neurons. Nature 452, 759–763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin P., Lewis J. (1989). Origins of the neurovascular bundle: interactions between developing nerves and blood vessels in embryonic chick skin. Int. J. Dev. Biol. 33, 379–387 [PubMed] [Google Scholar]
- Martyn U., Schulte-Merker S. (2004). Zebrafish neuropilins are differentially expressed and interact with vascular endothelial growth factor during embryonic vascular development. Dev. Dyn. 231, 33–42 [DOI] [PubMed] [Google Scholar]
- Menelaou E., McLean D. L. (2012). A gradient in endogenous rhythmicity and oscillatory drive matches recruitment order in an axial motor pool. J. Neurosci. 32, 10925–10939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukouyama Y. S., Shin D., Britsch S., Taniguchi M., Anderson D. J. (2002). Sensory nerves determine the pattern of arterial differentiation and blood vessel branching in the skin. Cell 109, 693–705 [DOI] [PubMed] [Google Scholar]
- Ny A., Koch M., Vandevelde W., Schneider M., Fischer C., Diez-Juan A., Neven E., Geudens I., Maity S., Moons L., et al. (2008). Role of VEGF-D and VEGFR-3 in developmental lymphangiogenesis, a chemicogenetic study in Xenopus tadpoles. Blood 112, 1740–1749 [DOI] [PubMed] [Google Scholar]
- Ober E. A., Olofsson B., Mäkinen T., Jin S. W., Shoji W., Koh G. Y., Alitalo K., Stainier D. Y. (2004). Vegfc is required for vascular development and endoderm morphogenesis in zebrafish. EMBO Rep. 5, 78–84 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsson A. K., Dimberg A., Kreuger J., Claesson-Welsh L. (2006). VEGF receptor signalling - in control of vascular function. Nat. Rev. Mol. Cell Biol. 7, 359–371 [DOI] [PubMed] [Google Scholar]
- Oosthuyse B., Moons L., Storkebaum E., Beck H., Nuyens D., Brusselmans K., Van Dorpe J., Hellings P., Gorselink M., Heymans S., et al. (2001). Deletion of the hypoxia-response element in the vascular endothelial growth factor promoter causes motor neuron degeneration. Nat. Genet. 28, 131–138 [DOI] [PubMed] [Google Scholar]
- Park H. C., Kim C. H., Bae Y. K., Yeo S. Y., Kim S. H., Hong S. K., Shin J., Yoo K. W., Hibi M., Hirano T., et al. (2000). Analysis of upstream elements in the HuC promoter leads to the establishment of transgenic zebrafish with fluorescent neurons. Dev. Biol. 227, 279–293 [DOI] [PubMed] [Google Scholar]
- Piltonen M., Planken A., Leskelä O., Myöhänen T. T., Hänninen A. L., Auvinen P., Alitalo K., Andressoo J. O., Saarma M., Männistö P. T. (2011). Vascular endothelial growth factor C acts as a neurotrophic factor for dopamine neurons in vitro and in vivo. Neuroscience 192, 550–563 [DOI] [PubMed] [Google Scholar]
- Soker S., Takashima S., Miao H. Q., Neufeld G., Klagsbrun M. (1998). Neuropilin-1 is expressed by endothelial and tumor cells as an isoform-specific receptor for vascular endothelial growth factor. Cell 92, 735–745 [DOI] [PubMed] [Google Scholar]
- Tiveron M. C., Cremer H. (2008). CXCL12/CXCR4 signalling in neuronal cell migration. Curr. Opin. Neurobiol. 18, 237–244 [DOI] [PubMed] [Google Scholar]
- Villefranc J. A., Nicoli S., Bentley K., Jeltsch M., Zarkada G., Moore J. C., Gerhardt H., Alitalo K., Lawson N. D. (2013). A truncation allele in vascular endothelial growth factor c reveals distinct modes of signaling during lymphatic and vascular development. Development 140, 1497–1506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westerfield M. (2000). The Zebrafish Book: A Guide for the Laboratory Use of Zebrafish (Danio rerio). Eugene, OR: University of Oregon Press; [Google Scholar]
- Yaniv K., Isogai S., Castranova D., Dye L., Hitomi J., Weinstein B. M. (2006). Live imaging of lymphatic development in the zebrafish. Nat. Med. 12, 711–716 [DOI] [PubMed] [Google Scholar]
- Yusa K., Rad R., Takeda J., Bradley A. (2009). Generation of transgene-free induced pluripotent mouse stem cells by the piggyBac transposon. Nat. Methods 6, 363–369 [DOI] [PMC free article] [PubMed] [Google Scholar]