Summary
Background and objectives
Urine neutrophil gelatinase-associated lipocalin (uNGAL) has been shown to accurately predict and allow early detection of AKI, as assessed by an increase in serum creatinine in children and adults. The present study explores the accuracy of uNGAL for the prediction of severe AKI-related outcomes in neonates and infants undergoing cardiac surgery: dialysis requirement and/or death within 30 days.
Design, setting, participants, & measurements
Prospective, observational cohort study conducted in a tertiary referral pediatric cardiac intensive care unit, including 75 neonates and 125 infants undergoing surgery with cardiopulmonary bypass between August 1, 2010, and May 31, 2011. Urine samples were collected before surgery and at median of five time points within 48 hours of bypass. Urine NGAL was quantified as absolute concentration, creatinine-normalized concentration, and absolute excretion rate, and a clusterization algorithm was applied to the individual uNGAL kinetics. The accuracy for the prediction of the outcome was assessed using receiver-operating characteristic areas, likelihood ratios, diagnostic odds ratios, net reclassification index, integrated reclassification improvement, and number needed to screen.
Results
A total of 1176 urine samples were collected. Of all patients, 8% required dialysis and 4% died. Three clusters of uNGAL kinetics were identified, including patients with significantly different outcomes. The uNGAL level peaked between 1 and 3 hours of bypass and remained high in half of all patients who required dialysis or died. The uNGAL levels measured within 24 hours of bypass accurately predicted the outcome and performed best after normalization to creatinine, with varying cutoffs according to the time elapsed since bypass. The number needed to screen to correctly identify the risk of dialysis or death in one patient varied between 1.5 and 2.6 within 12 hours of bypass.
Conclusions
uNGAL is a valuable predictive tool of dialysis requirement and death in neonates and infants with AKI after cardiac surgery.
Introduction
AKI requiring renal replacement therapy (RRT) represents the strongest independent risk factor for death in adults undergoing cardiac surgery (1). In children undergoing cardiac surgery, severe AKI is an independent predictor of postoperative death, as strong as the functional single ventricle and the requirement for postoperative circulatory support (2). The currently available data suggest a trend toward reduced mortality and better renal recovery with earlier initiation of RRT (3,4). The primary challenge in improving AKI outcomes over the past decade has been the late detection of AKI by the variation in serum creatinine (sCr), and substantial research has been focused on the discovery and validation of biomarkers to detect AKI before the rise in sCr (5). The most popular to date is neutrophil gelatinase-associated lipocalin (NGAL) (5).
The pathogenesis of AKI following cardiac surgery with cardiopulmonary bypass (CPB) is complex, and it is largely assumed that the pathologic lesion is acute tubular necrosis (6). Oxidative stress due to the generation of free hemoglobin and iron through hemolysis (7), ischemia, and inflammation are thought to be the most prominent mechanisms contributing to the tubular injury during CPB. NGAL is a protective siderophore expressed in the proximal tubule and secreted in the urine in the presence of epithelial injury and inflammation (8). Several studies in children undergoing surgery with CPB have reported good discrimination between patients with and those without AKI by the concentration of urine NGAL (uNGAL), with receiver-operating characteristic (ROC) areas ranging between 0.75 and 0.99 (8,9–13). Because of the shortcomings of sCr as an assessor of AKI, it has been suggested assess AKI severity by hard outcomes, such as dialysis requirement and/or death (14). However, most studies of NGAL in children had small sample sizes; focused on the surrogate endpoint of a ≥50% increase in sCr for the definition of AKI; and didn’t allow assessment of associations with hard outcomes of AKI, such as the requirement for RRT and the occurrence of death. Because of the particularly challenging definition of AKI early after birth, most studies excluded neonates. Besides, most analyses have been based on a single specimen, yet the timing of specimen collection with respect to the kidney insult may significantly affect the predictive values of a biomarker (12). A majority did not provide clinically meaningful cutoff values that can be used at the bedside. Finally, the expression of the biomarker has not been standardized, and the expression best suitable for prediction has not been established.
This study aimed to (1) describe the temporal elevation of uNGAL in patients younger than 1 year of age undergoing surgery with CPB in order to establish the expression level of the biomarker that provides the best predictive performance and (2) explore the accuracy for the prediction of two hard clinical outcomes of AKI: the requirement for RRT and the occurrence of death.
Materials and Methods
The study was performed at the Necker-Enfants Malades University Hospital, Paris, France, after approval by the regional ethics committee (2009–524 CPP02 Ile de France) and registration under the Clinical Trial Registry (NCT01219998). Although de-identification of patient information and samples (urine collected through a urinary catheter that would have otherwise been discarded) enabled waiver of written consent, we sought verbal consent from all the parents. The study design was prospective, and the measurement of the biomarker was blinded to the postoperative outcome.
Patient care and monitoring were performed as part of the hospital’s standard protocol, and no specific intervention, other than the collection of the urine samples, was required for this research. Surgery was conducted with normothermic nonpulsatile CPB unless reconstruction of the aortic arch required hypothermic circulatory arrest. Surgical complexity was accounted for by the Aristotle score (15). Oliguria and AKI stage were defined according to the Acute Kidney Injury Network (AKIN) criteria (16). The only RRT used in the study population was peritoneal dialysis. Dialysis was initiated when patients met the commonly accepted criteria (17,18): evidence of fluid overload and positive fluid balance, inadequate urine output (<1 ml/kg per hour) or anuria unresponsive to fluid challenge and intravenous furosemide for at least 4 hours, concomitant low-cardiac-output syndrome, and acid-base or electrolyte disturbances (pH<7.30, serum K+>5.5 mmol/L).
Urine Sampling and uNGAL Measurement
According to the hospital’s standard protocol for surgery with CPB, every patient had a Foley catheter set after induction of anesthesia. Urine samples were collected preoperatively (when the catheter was set) and intraoperatively (at weaning from CPB [urine excreted during CPB] or within 1 hour of weaning from CPB). After surgery, urine excretion was monitored hourly using the urometer of the Foley system, and one hourly excretion was collected by the attending nurse on admission to the intensive care unit (ICU), at 6 pm, midnight, 6 am, and noon, concomitant with the hospital’s standard blood sampling protocol. A further sample was collected within 48 hours of surgery in all neonates and in patients with postoperative complications.
Fresh urine was stored at 4°C for <48 hours, than centrifuged at 5000×g for 4 minutes to remove cellular debris. The urine supernatant was then aliquoted into bar-coded cryovials and the samples stored at −80°C with no additional thaw cycle until biomarker measurement. No additives or protease inhibitors were added. We measured uNGAL using the ARCHITECT i2000 platform (Abbott Diagnostics, Abbott Park, IL) (19) and quantified as absolute concentration, urine creatinine–normalized concentration, and absolute excretion rate. The normalized uNGAL concentration was derived by dividing the absolute concentration by the urine creatinine concentration (millimole per liter), measured by the enzymatic method on the ARCHITECT C16000 platform. The absolute excretion rate was derived by multiplying the absolute concentration by the sample volume.
All baseline, intraoperative, and postoperative variables were collected prospectively. Serum creatinine was measured before surgery, on admission, and daily during the ICU stay. With the exception of uNGAL and urine creatinine, no additional measurement was conducted for the purpose of this research.
Statistical Analyses
Samples were grouped within time intervals according to the time elapsed since weaning from bypass. First, an exploratory analysis was conducted to assess overall uNGAL kinetics and associations with baseline, intraoperative and outcome characteristics. It used a clusterization algorithm for longitudinal data (20) that was applied to the normalized uNGAL concentration, shown to be the best predictor of postoperative events in adults with cardiac surgery. Baseline, intraoperative, and postoperative variables were compared between clusters; the cluster with the best outcome was the reference. Continuous variables were compared using the Mann-Whitney test, and categorical variables were compared using the chi-square or Fisher test. According to the Bonferroni correction, the statistical significance was set to 0.025.
Next, a composite outcome variable was defined as the requirement for dialysis and/or the occurrence of in-hospital death. For each time interval, the probability of correctly discriminating between patients with and without the outcome was quantified by the area under the ROC curve, and the optimal threshold was identified using the Youden index (21). The validity of uNGAL as a diagnostic test was assessed using sensitivity, specificity, and likelihood ratios (22) and the diagnostic odds ratios (23). All these metrics are unaffected by the prevalence of the event and allow extrapolation to other populations and comparison between studies. To judge the performance of uNGAL in the setting of this study, the number needed to screen was also calculated: It represents the number of patients required to yield one correct identification (case or noncase) beyond those misidentified (24). All 95% confidence intervals were estimated by bootstrapping with 1000 resamples. According to the latest recommendations (25), the net reclassification improvement (NRI) and the integrated discrimination improvement (IDI) (26) were calculated after adding the biomarker as a predictor in a model that included the duration of CPB, a choice guided by previous literature (12). Only the continuous NRI, unaffected by event incidence, was used here (27). To establish the best biomarker quantification method, the procedure was applied to the normalized and absolute concentrations and to the excretion rate.
The full statistical methods are presented as Supplemental Material. All analyses used R 2.10.1 software. Clusterization used the kml package, ROC curves and biomarker thresholds were estimated using the pROC package, and IDI and continuous NRI were calculated using the Design package in R.
Results
The study included 75 neonates (age, within 28 days of birth) and 125 infants undergoing surgery with CPB between August 1, 2010, and May 31, 2011. The main procedures they had undergone along with the surgical complexity are listed in Table 1. A total of 1176 samples were collected within 10 time intervals. The median number of samples per patient was 6 (interquartile range, 5–6). No patient had complete anuria after surgery, but 8 patients had anuria within 3 hours of CPB, and 2 patients had anuria between 3 and 6 hours of CPB. Eleven patients had oliguria, 16 patients required dialysis, and 8 died.
Table 1.
Surgical procedures performed in the study population
| Surgical Procedure | Patients (n) | Aristotle Score |
|---|---|---|
| Ventricular septal defect closure | 37 | 6 |
| Arterial switch operation | 34 | 10 |
| Tetralogy of Fallot repair | 27 | 8 |
| Arterial switch operation and ventricular septal defect closure | 20 | 11 |
| Pulmonary atresia, palliation | 14 | 7.5 |
| Complete atrioventricular canal repair | 11 | 9 |
| Total anomalous pulmonary vein connection repair | 8 | 9 |
| Bidirectional Glenn anastomosis | 7 | 7.5 |
| Interrupted aortic arch repair | 6 | 10.8 |
| Truncus arteriosus repair | 4 | 11 |
| Other | 32 |
The complexity of each cardiac condition is graded according to the Aristotle surgical complexity score (18), ranging between 1.5 and 15.
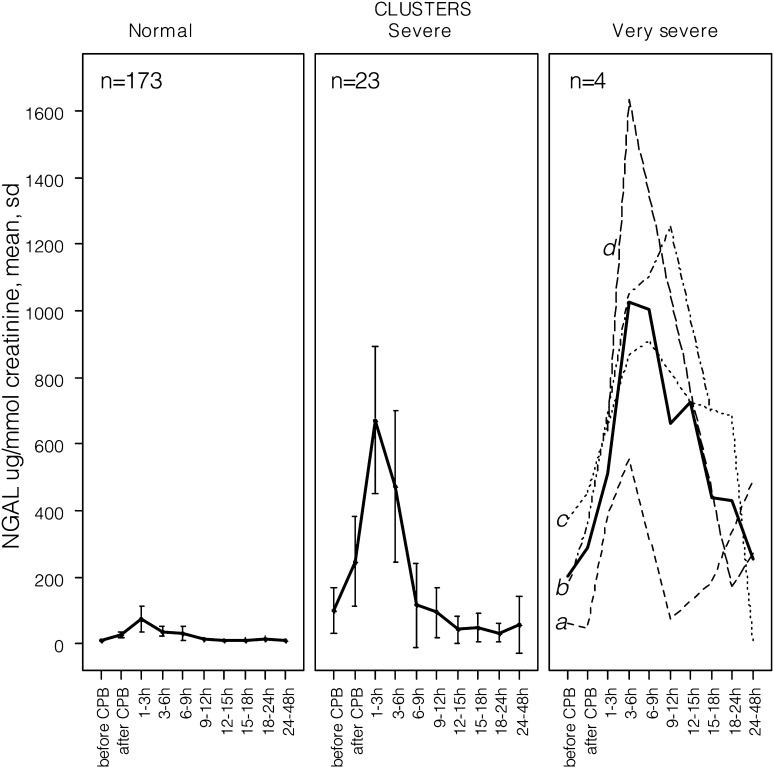
Clusterization resulted in three clusters of creatinine normalized uNGAL trajectories: normal (n=173), severe (n=23), and very severe (n=4), shown in Figure 1. As shown in Table 2, the normal cluster, taken as the reference, included patients who were less likely to undergo preoperative respiratory or hemodynamic support, underwent the less complex procedures, and had a majority of short and uncomplicated recoveries. Their uNGAL concentrations peaked at <100 μg/mmol creatinine within 3 hours of bypass, than decreased rapidly. The severe cluster included 23 patients with complex surgeries, with a 6-fold higher dialysis rate, a 4-days-longer ICU stay, and an almost 4-fold higher mortality rate. Their uNGAL peaked at about 700 μg/mmol within 3 hours of bypass, then decreased but remained above 100 μg/mmol creatinine for several hours. Interestingly, their preoperative uNGAL concentration was higher, and two patients required inotropic support before surgery. The very severe cluster included 4 patients with complex procedures and severe postoperative complications. One patient (denoted “a” in Figure 1) required reoperation for bleeding, then developed myocardial infarction, underwent angioplasty, required extracorporeal membrane oxygenation (ECMO), dialysis, and survived; uNGAL peaked at 3 hours, decreased, and rose again, concomitant with the occurrence of myocardial infarction. Another patient (“b” in Figure 1), with preoperative sepsis and multiorgan failure, developed nonoliguric AKI after surgery and was discharged from the ICU with persistent AKIN stage 2 AKI. The third patient (“c” in Figure 1) had sepsis and multiorgan failure (251 μmol/L sCr) before surgery, required ECMO within 2 hours of CPB, underwent dialysis, and died 13 days later. The last patient (“d” in Figure 1), developed myocardial infarction, required ECMO at the end of surgery, and died 34 days later. In these patients uNGAL concentrations rose abruptly within 3 hours of CPB and remained high during the postoperative events.
Figure 1.
Variation of the mean urine creatinine–normalized neutrophil gelatinase-associated lipocalin (NGAL) concentration within 48 hours of surgery. All urine creatinine–normalized uNGAL concentrations were included in individual trajectories of variation. Running the algorithm of clusterization on the individual trajectories resulted in three clusters of variation: normal (n=173), severe (n=23), and very severe (n=4) clusters. Mean concentrations are shown using a thick line, and 95% confidence intervals are shown using error bars. Individual trajectories in the very severe cluster are shown using dotted lines and are identified by letters (see Results). Letters in the right panel denote the individual uNGAL trajectories of the patients in the very severe cluster. CBP, cardiopulmonary bypass.
Table 2.
Patient baseline, intraoperative, and postoperative characteristics within the clusters
| Variable | Normal Cluster (n=173) | Severe Cluster (n=23) | P Value | Very Severe Cluster (n=4) | P Value |
|---|---|---|---|---|---|
| Demographic characteristics | |||||
| Age (d) | 84 (9–163) | 74 (7.5–119) | 0.33 | 30 (11.7–72.7) | 0.61 |
| <28 d | 65 (37.6) | 8 (34.8) | 0.79 | 2 (50) | 0.63 |
| <48 h | 3 (1.7) | 2 (8.7) | 0.10 | 0 | 0.99 |
| Weight (kg) | 4.2 (3.2–10.3) | 3.4 (3.2–4.6) | 0.18 | 3.4 (3.3–3.9) | 0.58 |
| Medical history before surgery | |||||
| Genetic syndrome | 8 (4.6) | 0 | 0.59 | 0 | 0.99 |
| Resternotomy | 15 (8.7) | 3 (13) | 0.45 | 0 | 0.99 |
| Cardiopulmonary resuscitation | 0 | 1 (4.3) | 0.12 | 1 (25) | 0.02 |
| Mechanical ventilation | 7 (4) | 2 (8.7) | 0.28 | 2 (50) | 0.01 |
| Inotropic support | 0 | 2 (8.7) | 0.01 | 1 (25) | 0.02 |
| Infection | 15 (8.7) | 3 (13) | 0.45 | 1 (25) | 0.32 |
| Enteropathy | 3 (1.7) | 1 (4.3) | 0.39 | 0 | 0.99 |
| Medication before surgery | |||||
| Furosemide | 36 (20.8) | 6 (26.1) | 0.33 | 0 | 0.58 |
| Spironolactone | 9 (5.2) | 3 (13) | 0.15 | 0 | 0.99 |
| Angiotensin-converting enzyme inhibitors | 26 (15) | 2 (8.7) | 0.54 | 0 | 0.99 |
| β-Blockers | 10 (5.8) | 2 (8.7) | 0.64 | 0 | 0.99 |
| Prostaglandin E2 | 37 (21.4) | 5 (21.7) | 0.99 | 2 (50) | 0.21 |
| Aminoglycoside antibiotics | 24 (13.9) | 6 (26.1) | 0.13 | 1 (25) | 0.46 |
| Vancomycin | 13 (7.5) | 4 (17.4) | 0.12 | 1 (25) | 0.28 |
| Angiography within 1 wk before surgery | 19 (11) | 2 (8.7) | 0.99 | 1 (25) | 0.37 |
| Intraoperative variables | |||||
| Aristotle complexity score | 8 (6–14.5) | 10 (6.25–11) | 0.08 | 11 (10.7–11) | 0.007 |
| Duration of cardiopulmonary bypass (min) | 107 (77–133) | 122 (105–180) | 0.008 | 259 (192–338) | 0.007 |
| Surgery requiring aortic crossclamping | 163 (94.2) | 23 (100) | 0.61 | 4 (100) | 0.99 |
| Duration of aortic crossclamping (min) | 61 (37–82) | 68 (56–78) | 0.24 | 128 (106–134) | 0.02 |
| Ultrafiltration rate (ml/kg per min of bypass) | 1.3 (0.9–1.8) | 1.1 (0.9–1.5) | 0.26 | 1.0 (0.8–1.2) | 0.20 |
| Surgery requiring deep hypothermic circulatory arrest | 10 (5.8) | 7 (30.4) | <0.001 | 1 (25) | 0.23 |
| Blood product transfusions on day 0 and day 1 | |||||
| Packed red blood cells (ml) | 511 (314–566) | 542 (505–567) | 0.05 | 573 (522–760) | 0.11 |
| Fresh frozen plasma (ml) | 219 (208–246) | 219 (212–347) | 0.23 | 425 (371–502) | 0.03 |
| Platelets (ml) | 45 (0–59) | 44 (25–88) | 0.21 | 143 (83–202) | 0.02 |
| Postoperative variables | |||||
| Requiring delayed sternal closure | 27 (15.6) | 8 (34.8) | 0.02 | 3 (75) | 0.02 |
| Requiring extracorporeal membrane oxygenation | 0 | 0 | 1 | 3 (75) | <0.001 |
| Left ventricular ejection fraction within 2 h of bypass (%) | 60 (60–65) | 60 (45–65) | 0.47 | 37.5 (30–45) | <0.001 |
| Peak lactatemia within 6 h of surgery (mmol/L) | 2.9 (2.1–4) | 3.0 (2.5–5.5) | 0.21 | 5.2 (3.8–7.5) | 0.02 |
| Peak lactatemia within 48 h of surgery (mmol/L) | 3.3 (2.3–4.4) | 3.6 (2.6–5.8) | 0.17 | 5.2 (3.8–7.5) | 0.04 |
| Inotropic support within 48 h of surgery | |||||
| Epinephrine (μg/kg per min) | 0.10 (0.08–0.14) | 0.14 (0.07–0.20) | 0.23 | 0.15 (0.08–0.21) | 0.45 |
| Milrinone (μg/kg per min) | 0.48 (0.35–0.57) | 0.44 (0.26–0.54) | 0.10 | 0.46 (0.43–0.51) | 0.95 |
| Hydrocortisone for low-cardiac–output syndrome | 30 (17.3) | 7 (30.4) | 0.13 | 2 (50) | 0.15 |
| Delay to sternal closure (d) | 2 (1.5–4) | 5 (2.7–7.2) | 0.05 | 13 (7.5–14) | 0.08 |
| Postoperative renal variables | |||||
| AKI according to the serum creatinine criteriaa | 23 (13.3) | 8 (34.8) | 0.008 | 3 (75.0) | 0.01 |
| AKI according to the urine output criteriaa | 6 (3.4) | 3 (13) | 0.07 | 2 (50) | 0.01 |
| Negative fluid balance (ml/kg per 24 h)b | 33.7 (18.9–50.1) | 14.4 (8.2–24.8) | 0.01 | 9.3 (3.6–21.9) | 0.09 |
| Peritoneal dialysis | 8 (4.6) | 6 (27.2) | <0.001 | 2 (50) | 0.02 |
| Length of mechanical ventilation (d) | 1 (0–4) | 5 (0.7–9.7) | 0.006 | 13.5 (10.2–19) | 0.009 |
| Length of intensive care unit stay (d) | 4 (2–6) | 8 (4–14.5) | 0.002 | 16 (11–22.7) | 0.01 |
| In-hospital death | 4 (2.3) | 2 (8.7) | 0.15 | 2 (50) | 0.006 |
Data are shown as medians (interquartile ranges) or as numbers and percentages. The normal cluster was taken as reference for the calculation of all P values. According to the Bonferroni correction, the significance level was set to 0.025.
According to the Acute Kidney Injury Network classification, with preoperative serum creatinine accounting for baseline.
Within 24 hours of surgery.
Estimated areas under the ROC curves for the three quantification methods are shown in Table 3. The estimated cutoffs, together with sensitivity, specificity, likelihood ratios, diagnostic odds ratios, numbers needed to screen, NRI, and IDI are shown in Table 4 for the creatinine-normalized uNGAL concentrations, and as additional material for the other quantification methods. The uNGAL concentration was predictive of the outcome before surgery and within 24 hours of surgery, and the threshold varied with the time elapsed since separation from bypass. Because of the small number of events, all estimated 95% confidence intervals were large and overlapping and did not allow demonstration of the most suitable quantization method for the prediction of the outcome. However, the slightly higher ROC areas and positive likelihood ratios, the slightly lower negative likelihood ratios and numbers needed to screen, and a better improvement in reclassification suggest that the normalized concentration is the most accurate for the prediction of dialysis and death early after bypass. The number needed to screen in order to correctly identify the risk of dialysis or death in one patient varied between 1.5 and 2.6 within 12 hours of surgery. Beyond 12 hours of surgery, according to the different quantization methods, the number needed to screen increased and became negative late after CPB, suggesting a misleading effect.
Table 3.
Relationship between timing of urine neutrophil gelatinase-associated lipocalin measurements and prediction accuracy
| Variable | Before CPB | Within 1 h of CPB | Within 1–3 h of CPB | Within 3–6 h of CPB | Within 6–12 h of CPB | Within 12–18 h of CPB | Within 18–24 h of CPB | Within 24–48 h of CPB |
|---|---|---|---|---|---|---|---|---|
| Measurements (n) | 155 | 166 | 180 | 115 | 199 | 176 | 80 | 105 |
| Outcome events (n) | 14 | 11 | 9 | 13 | 16 | 16 | 12 | 14 |
| Urine creatinine–normalized NGAL concentration (μg/mmol creatinine) | ||||||||
| ROC area (95% CI)a | 0.689 (0.552 to 0.886) | 0.736 (0.581 to 0.881) | 0.719 (0.541 to 0.897) | 0.755 (0.553 to 0.958) | 0.762 (0.622 to 0.901) | 0.775 (0.662 to 0.888) | 0.747 (0.625 to 0.869) | 0.441 (0.268 to 0.614) |
| Absolute urine NGAL concentration (ng/ml) | ||||||||
| ROC area (95% CI)a | 0.679 (0.512 to 0.847) | 0.656 (0.458 to 0.853) | 0.675 (0.510 to 0.841) | 0.796 (0.632 to 0.960) | 0.818 (0.708 to 0.927) | 0.783 (0.675 to 0.892) | 0.729 (0.572 to 0.887) | 0.466 (0.288 to 0.645) |
| Urine NGAL excretion rate (ng/hr) | ||||||||
| ROC area (95% CI)a | 0.583 (0.418 to 0.749) | 0.725 (0.572 to 0.878) | 0.668 (0.454 to 0.882) | 0.700 (0.525 to 0.876) | 0.672 (0.531 to 0.812) | 0.594 (0.445 to 0.743) | 0.638 (0.473 to 0.803) | 0.548 (0.349 to 0.746) |
The outcome events were dialysis requirement and/or in-hospital death. CPB, cardiopulmonary bypass; NGAL, neutrophil gelatinase-associated lipocalin; ROC, receiver-operating characteristic; 95% CI, 95% confidence interval.
95% CIs were estimated by boot-strapping with 1000 resamples.
Table 4.
Accuracy of urine neutrophil gelatinase-associated lipocalin normalized concentration for the prediction of dialysis or in-hospital death
| Variable | Before CPB | Before CPB | Within 1 h of CPB | Within 1–3 h of CPB | Within 3–6 h of CPB | Within 6–12 h of CPB | Within 12–18 h of CPB | Within 18–24 h of CPB |
|---|---|---|---|---|---|---|---|---|
| Urine creatinine–normalized NGAL concentration cutoff (μg/mmol creatinine) | 12.5 | 21.2 | 186.5 | 126.0 | 9.5 | 7.6 | 7.1 | 7.2 |
| Sensitivity (95% CI)a | 0.64 (0.53 to 1.00) | 0.81 (0.58 to 1.00) | 0.55 (0.47 to 1.00) | 0.61 (0.43 to 0.93) | 0.81 (0.56 to 1.00) | 0.87 (0.62 to 1.00) | 0.92 (0.83 to 1.00) | 0.57 (0.32 to 1.00) |
| Specificity (95% CI)a | 0.76 (0.32 to 0.84) | 0.68 (0.40 to 0.94) | 0.85 (0.31 to 0.96) | 0.85 (0.56 to 0.99) | 0.68 (0.53 to 0.92) | 0.61 (0.47 to 0.94) | 0.58 (0.38 to 0.82) | 0.51 (0.21 to 0.99) |
| Positive likelihood ratio (95% CI)a | 2.73 (1.33 to 4.52) | 2.56 (1.45 to 8.52) | 3.61 (1.39 to 14.17) | 4.26 (1.72 to 44.62) | 2.54 (1.90 to 8.24) | 2.25 (1.76 to 11.59) | 2.18 (1.63 to 5.28) | 1.16 (1.17 to 23.67) |
| Negative likelihood ratio (95% CI)a | 0.47 (0.00 to 0.68) | 0.27 (0.00 to 0.59) | 0.52 (0.00 to 0.66) | 0.45 (0.00 to 0.68) | 0.27 (0.00 to 0.55) | 0.20 (0.00 to 0.53) | 0.14 (0.00 to 0.33) | 0.84 (0.00 to 0.85) |
| Diagnostic odds ratio (95% CI)a | 5.85 (2.17 to ∞) | 9.57 (3.32 - ∞) | 6.87 (2.04 - ∞) | 9.48 (4.01 - ∞) | 9.24 (4.19 to ∞) | 11.03 (4.93 to ∞) | 15.17 (7.94 to ∞) | 1.37 (0.47 to ∞) |
| Net reclassification index (95% CI) | 0.81 (0.26 to 1.36) | 0.99 (0.38 to 1.61) | 0.80 (0.13 to 1.47) | 0.94 (0.36 to 1.52) | 0.97 (0.27 to 1.29) | 0.97 (0.46 to 1.49) | 0.99 (0.38 to 1.61) | 0.53 (-0.05 to 1.10) |
| P value | 0.004 | 0.001 | 0.02 | 0.001 | <0.001 | 0.003 | 0.001 | 0.07 |
| Integrated discrimination improvement (95% CI) | 0.07 (0.02 to 0.13) | 0.07 (0.03 to 0.10) | 0.03 (-0.006 to 0.07) | 0.08 (0.02 to 1.51) | 0.06 (0.03 to 0.08) | 0.06 (0.02 to 0.09) | 0.08 (0.03 to 0.12) | 0.04 (-0.001 to 0.08) |
| P value | 0.01 | <0.001 | 0.10 | 0.006 | <0.001 | <0.001 | <0.001 | 0.05 |
| Number needed to screen (95% CI)a | 1.97 (1.55 to 2.75) | 2.63 (1.95 to 4.13) | 1.51 (1.30 to 1.83) | 1.53 (1.28 to 1.96) | 2.60 (1.95 to 4.02) | 3.65 (2.41 to 8.14) | 3.86 (2.11 to 20.00) | −27.00 (−77 to ∞) |
NGAL, neutrophil gelatinase-associated lipocalin.
95% CIs were estimated by boot-strapping with 1000 resamples.
Discussion
The present study included the largest population of patients younger than 1 year of age undergoing cardiac surgery in whom uNGAL has been studied to date. It demonstrates the accuracy of the biomarker measured within 24 hours of bypass for the prediction of two severe postoperative AKI-related outcomes: dialysis requirement and death. It establishes a temporal pattern of uNGAL elevation after CPB and provides clinically meaningful cutoff values that can be used at the bedside. Finally, it suggests that the urine creatinine–normalized NGAL concentration is best suited for predicting severe AKI in neonates and infants.
It is admitted that critically ill patients die rather “of” than “with” AKI (28), and there is evidence that severe AKI is an independent predictor of postoperative death in children with cardiac surgery (2). The use of hard outcomes, such as dialysis requirement and death, in the present study was guided by a recent recommendation (14) because serum creatinine is acknowledged as an inadequate gold standard for the diagnosis of AKI. Even though the causal relationship among surgery with CPB, AKI, and the increase in uNGAL cannot be issued on the basis of the present results, there is an obvious temporal relationship between the surgery and/or the postoperative complications, the increase in uNGAL, and the occurrence of the outcome events, suggesting that uNGAL is a marker of severe AKI and a potential predictor of AKI-related outcomes.
Predictive Performance of uNGAL for Dialysis Requirement and Death
The meta-analysis by Haase et al. (29) pooled together more than 2500 cases across several settings and reported uNGAL diagnostic odds ratios of 12.9 and 8.8 for RRT and in-hospital mortality, and ROC areas of 0.782 and 0.706, respectively. The uNGAL cutoff concentration of 278 ng/ml identified AKI necessitating RRT with 76% sensitivity and 80% specificity. However, the meta-analysis included only four children with RRT following cardiac surgery (1), three patients who died (1), and no neonates. This is probably the result of the challenging definition of neonatal AKI due to unreliable sCr concentrations and improving filtration rate during the first week of life (30).
Few studies have reported the accuracy of NGAL for the prediction of hard AKI-related outcomes (Table 5). A pilot study of uNGAL in patients with hemolytic-uremic syndrome (31) found that a cutoff of 200 ng/ml measured on the day of diagnosis provides a relative risk of 10.6 for RRT requirement. Bennett et al. (9) found uNGAL>150 ng/ml to be predictive of the need for RRT and postoperative death, with ROC areas of 0.86 and 0.91. Parikh et al. (13) found that uNGAL concentrations>72 ng/ml within 6 hours after commencement of CPB in children were significantly associated with at least a 4-fold increased odds for severe AKI, as defined by receipt of dialysis or a doubling in sCr from baseline and identified severe AKI with 42% sensitivity and 85% specificity. Together, data in the literature imply that uNGAL may have an important interaction with conventional criteria to aid in the clinical decision to initiate RRT. In this study, the positive likelihood ratios observed across the study period were <10, suggesting that uNGAL alone cannot rule in the diagnosis of severe AKI that necessitates dialysis or predict AKI-related death. The results in Table 2 show that uNGAL rose abruptly and remained high in half of all patients who required dialysis or died. Nevertheless, uNGAL measured within 24 hours of surgery added discrimination and predictive ability to the model including CPB duration, suggesting uNGAL is a potentially useful tool for decision-making when considered together with the clinical criteria. On the other hand, the low negative likelihood ratio observed after 12 hours of bypass, close to 0.1, suggests that low uNGAL concentrations at approximately 12–24 hours after CPB render the diagnosis of severe AKI unlikely, a finding that has not been reported with other biomarkers to date. The number of patients needed to screen varied between 1.5 and 2.6 when uNGAL was measured within 12 hours of surgery. Because the prevalence of the composite event here is consistent with previous literature (32), it is possible to extrapolate these results to other populations. Additionally, preoperative uNGAL concentrations were predictive of the occurrence of the composite endpoint, consistent with a previous report (12) and suggesting that some of the patients had subclinical preoperative AKI, which may have predisposed them to severe AKI after surgery.
Table 5.
Predictive ability of urine neutrophil gelatinase-associated lipocalin NGAL for dialysis requirement and/or death in pediatric studies
| Study, Year | Setting | Endpoint Event | Timing of Urine Collection | ROC Area | Cutoff (ng/ml) | Sensitivity | Specificity | Positive LR | Negative LR | Diagnostic OR |
|---|---|---|---|---|---|---|---|---|---|---|
| Trachtman et al., 2006 | 34 children with diarrhea-associated hemolytic-uremic syndrome | RRT (n=10) | Day of diagnosis | 200 | 0.90 | 0.54 | 1.96 | 0.18 | 10.89 | |
| Bennett et al., 2007 | 194 children with cardiac surgery | RRT (n=4) | Within 2 h of bypass | 0.86 | 150 | |||||
| Within 4 h of bypass | 0.80 | |||||||||
| Within 6 h of bypass | 0.90 | |||||||||
| In-hospital death (n=3) | Within 2 h of bypass | 0.91 | 150 | |||||||
| Within 4 h of bypass | 0.81 | |||||||||
| Within 6 h of bypass | 0.67 | |||||||||
| Parikh et al., 2011 | 311 children with cardiac surgery | Doubling of sCr (n=53) or RRT (n=5) | Within 6 h of initiation of bypass (median, 30 min of ICU admission) | 0.71 | 72 | 0.42 | 0.85 | 2.8 | 0.68 | 4.12 |
LR, likelihood ratio; OR, odds ratio, RRT, renal replacement therapy; sCr, serum creatinine; ICU, intensive care unit.
The study of uNGAL that included the largest neonatal subgroup to date (12,33) showed similar kinetics but larger excretion compared with older infants. However, by using different AKI definitions in neonates and older infants, the study could have captured patients with different AKI severity. Here, age did not differ between clusters (Table 2), suggesting that NGAL excretion was similar in neonates and in older infants in response to similar injury.
Normalization to urine creatinine is standard practice in chronic conditions but may result in overestimation of AKI by tubular biomarkers because of the decreased GFR (34). To date, there is no consensus about how results should be reported. Our results are consistent with the report by Ralib et al. (35), showing the normalized rate to perform best for the prediction of death, dialysis, and subsequent AKI in adults.
Temporal Patterns of uNGAL Concentration
The timing of specimen collection with respect to the relevant insult may significantly affect the predictive value of a biomarker (12,36). In this study we show that uNGAL concentrations at various time intervals within 24 hours of surgery can identify severe AKI but that the threshold for accurate prediction differs according to the time elapsed since weaning from CPB. Our findings are consistent with previous reports, showing a peak of uNGAL at 2 hours after CPB in children (1,8,10,12,13,33). The peak concentration was more than 6-fold higher in the severe cluster, where 27% of patients required dialysis, and remained high afterward in half of them, consistent with previous literature (12,37). Importantly, clusterization identified a group of four patients with an abrupt and persistent increase, of whom three were in a life-threatening situation and required ECMO. Tubular ischemia was probably ongoing and worsening in these patients after weaning from bypass because of low cardiac output. Together, these data suggest that both the concentration and the kinetics of uNGAL are predictive of the severity of postoperative AKI. The accuracy of the biomarker for identification of patients with severe AKI dropped with the time elapsed since the occurrence of the kidney insult.
Study Strengths
The study enrolled a homogeneous cohort of patients in whom the most proximate cause of AKI was cardiac surgery and bypass. The study design allowed for a precise determination of the temporal rise in uNGAL following bypass. By having the timing of the sample collection coincide with routine blood sampling, and by involving nurses who were not dedicated to the study protocol, the study design is representative of “real-life” conditions and therefore strengthens the impact of the results.
Study Limitations
The results of this single-center study need to be validated at the multicenter level. Because the number of patients who required dialysis or died was small, the results are reported with large confidence intervals, and multivariable analysis of the risk was not performed. Urine collection using the Foley system could have resulted in inaccurate measurement on the urometer and in potentially significant contamination of the hourly collected sample with previously excreted urine, due to a dead-space effect. Thus, the accuracy of both urine flow rates and of uNGAL and creatinine concentrations can be questioned in patients with reduced urine output.
NGAL excretion after cardiac surgery in patients younger than 1 year of age is predictive of two severe AKI-related outcomes: dialysis requirement and death. The predictive ability and the threshold of uNGAL concentration vary with the time elapsed since bypass, and the best performance is attained early after surgery. The present data suggest that the urine creatinine–normalized NGAL concentration is most suitable for predicting dialysis requirement or death in neonates and infants with AKI.
Disclosures
None.
Supplementary Material
Acknowledgments
We are indebted to our nurses for their assistance and to our patients and their families for their participation. We acknowledge the Clinical Research Unit-Paris Centre Cochin Necker, Assistance Publique–Hopitaux de Paris, France, for methodologic support.
This work was performed with the financial support of the Direction de la Recherche Clinique, Assistance Publique–Hôpitaux de Paris, Paris, France.
Study collaborators: Michel Reviron, MD (Department of Hematology and Blood Center, Necker-Enfants Malades Hospital, Assistance Publique–Hôpitaux de Paris, Paris, France); Maria Constanza Basto Duarte, MD, Claire Martinon-Siringo, MD, Marie-Ange Silicani, MD, and Leticia Vaccaroni, MD (Department of Anesthesiolgy and Critical Care, Necker-Enfants Malades Hospital, Assistance Publique–Hôpitaux de Paris, Paris, France); and Natalia Ermak, MD, and Bernard Lacour, MD, PhD (Department of Biochemistry, Necker-Enfants Malades Hospital, Assistance Publique–Hôpitaux de Paris, Paris, France).
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.04730513/-/DCSupplemental.
References
- 1.Chertow GM, Levy EM, Hammermeister KE, Grover F, Daley J: Independent association between acute renal failure and mortality following cardiac surgery. Am J Med 104: 343–348, 1998 [DOI] [PubMed] [Google Scholar]
- 2.Blinder JJ, Goldstein SL, Lee VV, Baycroft A, Fraser CD, Nelson D, Jefferies JL: Congenital heart surgery in infants: Effects of acute kidney injury on outcomes. J Thorac Cardiovasc Surg 143: 368–374, 2012 [DOI] [PubMed] [Google Scholar]
- 3.Bojan M, Gioanni S, Vouhé PR, Journois D, Pouard P: Early initiation of peritoneal dialysis in neonates and infants with acute kidney injury following cardiac surgery is associated with a significant decrease in mortality. Kidney Int 82: 474–481, 2012 [DOI] [PubMed] [Google Scholar]
- 4.Seabra VF, Balk EM, Liangos O, Sosa MA, Cendoroglo M, Jaber BL: Timing of renal replacement therapy initiation in acute renal failure: A meta-analysis. Am J Kidney Dis 52: 272–284, 2008 [DOI] [PubMed] [Google Scholar]
- 5.Waikar SS, Bonventre JV: Biomarkers for the diagnosis of acute kidney injury. Nephron Clin Pract 109: c192–c197, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rosner MH, Okusa MD: Acute kidney injury associated with cardiac surgery. Clin J Am Soc Nephrol 1: 19–32, 2006 [DOI] [PubMed] [Google Scholar]
- 7.Haase M, Haase-Fielitz A, Bagshaw SM, Ronco C, Bellomo R: Cardiopulmonary bypass-associated acute kidney injury: A pigment nephropathy? Contrib Nephrol 156: 340–353, 2007 [DOI] [PubMed] [Google Scholar]
- 8.Mishra J, Dent C, Tarabishi R, Mitsnefes MM, Ma Q, Kelly C, Ruff SM, Zahedi K, Shao M, Bean J, Mori K, Barasch J, Devarajan P: Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365: 1231–1238, 2005 [DOI] [PubMed] [Google Scholar]
- 9.Bennett M, Dent CL, Ma Q, Dastrala S, Grenier F, Workman R, Syed H, Ali S, Barasch J, Devarajan P: Urine NGAL predicts severity of acute kidney injury after cardiac surgery: A prospective study. Clin J Am Soc Nephrol 3: 665–673, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dent CL, Ma Q, Dastrala S, Bennett M, Mitsnefes MM, Barasch J, Devarajan P: Plasma neutrophil gelatinase-associated lipocalin predicts acute kidney injury, morbidity and mortality after pediatric cardiac surgery: A prospective uncontrolled cohort study. Crit Care 11: R127, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Han WK, Waikar SS, Johnson A, Betensky RA, Dent CL, Devarajan P, Bonventre JV: Urinary biomarkers in the early diagnosis of acute kidney injury. Kidney Int 73: 863–869, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Krawczeski CD, Goldstein SL, Woo JG, Wang Y, Piyaphanee N, Ma Q, Bennett M, Devarajan P: Temporal relationship and predictive value of urinary acute kidney injury biomarkers after pediatric cardiopulmonary bypass. J Am Coll Cardiol 58: 2301–2309, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Parikh CR, Devarajan P, Zappitelli M, Sint K, Thiessen-Philbrook H, Li S, Kim RW, Koyner JL, Coca SG, Edelstein CL, Shlipak MG, Garg AX, Krawczeski CD, TRIBE-AKI Consortium : Postoperative biomarkers predict acute kidney injury and poor outcomes after pediatric cardiac surgery. J Am Soc Nephrol 22: 1737–1747, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Waikar SS, Betensky RA, Emerson SC, Bonventre JV: Imperfect gold standards for kidney injury biomarker evaluation. J Am Soc Nephrol 23: 13–21, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lacour-Gayet F, Clarke D, Jacobs J, Comas J, Daebritz S, Daenen W, Gaynor W, Hamilton L, Jacobs M, Maruszsewski B, Pozzi M, Spray T, Stellin G, Tchervenkov C, Mavroudis And C, Aristotle Committee : The Aristotle score: A complexity-adjusted method to evaluate surgical results. Eur J Cardiothorac Surg 25: 911–924, 2004 [DOI] [PubMed] [Google Scholar]
- 16.Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, Levin A, Acute Kidney Injury Network : Acute Kidney Injury Network: Report of an initiative to improve outcomes in acute kidney injury. Crit Care 11: R31, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chan KL, Ip P, Chiu CS, Cheung YF: Peritoneal dialysis after surgery for congenital heart disease in infants and young children. Ann Thorac Surg 76: 1443–1449, 2003 [DOI] [PubMed] [Google Scholar]
- 18.Pedersen KR, Hjortdal VE, Christensen S, Pedersen J, Hjortholm K, Larsen SH, Povlsen JV: Clinical outcome in children with acute renal failure treated with peritoneal dialysis after surgery for congenital heart disease. Kidney Int Suppl S81–S86, 2008 [DOI] [PubMed] [Google Scholar]
- 19.Grenier FC, Ali S, Syed H, Workman R, Martens F, Liao M, Wang Y, Wong PY: Evaluation of the ARCHITECT urine NGAL assay: Sssay performance, specimen handling requirements and biological variability. Clin Biochem 43: 615–620, 2010 [DOI] [PubMed] [Google Scholar]
- 20.Genolini C, Falissard B: KmL: A package to cluster longitudinal data. Comput Methods Programs Biomed 104: e112–e121, 2011 [DOI] [PubMed] [Google Scholar]
- 21.Youden WJ: Index for rating diagnostic tests. Cancer 3: 32–35, 1950 [DOI] [PubMed] [Google Scholar]
- 22.Deeks JJ, Altman DG: Diagnostic tests 4: Likelihood ratios. BMJ 329: 168–169, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Glas AS, Lijmer JG, Prins MH, Bonsel GJ, Bossuyt PM: The diagnostic odds ratio: A single indicator of test performance. J Clin Epidemiol 56: 1129–1135, 2003 [DOI] [PubMed] [Google Scholar]
- 24.Mitchell AJ: Implementing a screening programme for distress in cancer settings. Psycho-Oncology Information and Help. Available at: www.psycho-oncology.info Accessed ??
- 25.Siew ED, Ware LB, Ikizler TA: Biological markers of acute kidney injury. J Am Soc Nephrol 22: 810–820, 2011 [DOI] [PubMed] [Google Scholar]
- 26.Pencina MJ, D’Agostino RB, Sr, D’Agostino RB, Jr, Vasan RS: Evaluating the added predictive ability of a new marker: From area under the ROC curve to reclassification and beyond. Stat Med 27: 157–172, discussion 207–212, 2008 [DOI] [PubMed] [Google Scholar]
- 27.Pencina MJ, D’Agostino RB, Sr, Steyerberg EW: Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat Med 30: 11–21, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hoste EA, De Corte W: Clinical consequences of acute kidney injury. Contrib Nephrol 174: 56–64, 2011 [DOI] [PubMed] [Google Scholar]
- 29.Haase M, Bellomo R, Devarajan P, Schlattmann P, Haase-Fielitz A, NGAL Meta-analysis Investigator Group : Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: A systematic review and meta-analysis. Am J Kidney Dis 54: 1012–1024, 2009 [DOI] [PubMed] [Google Scholar]
- 30.Hogg RJ, Furth S, Lemley KV, Portman R, Schwartz GJ, Coresh J, Balk E, Lau J, Levin A, Kausz AT, Eknoyan G, Levey AS, National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative : National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative clinical practice guidelines for chronic kidney disease in children and adolescents: Evaluation, classification, and stratification. Pediatrics 111: 1416–1421, 2003 [DOI] [PubMed] [Google Scholar]
- 31.Trachtman H, Christen E, Cnaan A, Patrick J, Mai V, Mishra J, Jain A, Bullington N, Devarajan P, Investigators of the HUS-SYNSORB Pk Multicenter Clinical Trial : Urinary neutrophil gelatinase-associated lipocalcin in D+HUS: A novel marker of renal injury. Pediatr Nephrol 21: 989–994, 2006 [DOI] [PubMed] [Google Scholar]
- 32.Pedersen KR, Povlsen JV, Christensen S, Pedersen J, Hjortholm K, Larsen SH, Hjortdal VE: Risk factors for acute renal failure requiring dialysis after surgery for congenital heart disease in children. Acta Anaesthesiol Scand 51: 1344–1349, 2007 [DOI] [PubMed] [Google Scholar]
- 33.Krawczeski, CD, Woo, JG, Wang, Y, Bennett, MR, Ma, Q & Devarajan, P: Neutrophil gelatinase-associated lipocalin concentrations predict development of acute kidney injury in neonates and children after cardiopulmonary bypass. J Pediatr, 158: 1009-1015 e1, 2011. [DOI] [PubMed]
- 34.Waikar SS, Sabbisetti VS, Bonventre JV: Normalization of urinary biomarkers to creatinine during changes in glomerular filtration rate. Kidney Int 78: 486–494, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ralib AM, Pickering JW, Shaw GM, Devarajan P, Edelstein CL, Bonventre JV, Endre ZH: Test characteristics of urinary biomarkers depend on quantitation method in acute kidney injury. J Am Soc Nephrol 23: 322–333, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Thurman JM, Parikh CR: Peeking into the black box: New biomarkers for acute kidney injury. Kidney Int 73: 379–381, 2008 [DOI] [PubMed] [Google Scholar]
- 37.Wagener G, Jan M, Kim M, Mori K, Barasch JM, Sladen RN, Lee HT: Association between increases in urinary neutrophil gelatinase-associated lipocalin and acute renal dysfunction after adult cardiac surgery. Anesthesiology 105: 485–491, 2006 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.