EVI1 is a transcriptional regulator with an important function in haematopoiesis and self-renewal.1 Aberrant overexpression of EVI1 has been firmly established as one of the most adverse prognostic markers in acute myeloid leukaemia (AML),2 implying that EVI1 is one of the most aggressive oncogenes in AML. Importantly, a recent report in Leukemia from Konantz et al.3 suggests that EVI1 might also have a role in paediatric acute lymphoblastic leukaemia (ALL), where high expression confers apoptosis resistance, and possibly also an adverse prognosis. Rearrangements of the 3q26 region, which encompasses the MECOM (MDS–EVI1 complex) gene that encodes EVI1 transcripts and that are commonly associated with EVI1 overexpression in adult AML, rarely occur in childhood ALL or AML.2, 4 However, when EVI1 is expressed in childhood AML it seems to be predominate in MLL-rearranged cases,4, 5 which may then confer an adverse prognosis, as illustrated by the correlation with the t(6;11) subtype,4 and with complex karyotype cases. However, in general high EVI1 expression is not seen in AML with good prognosis cytogenetics such as core-binding factor-rearranged AML with t(8;21) or inv(16)4, 5 (Figure 1a). In addition, in chronic myeloid leukaemia, the BCR–ABL fusion tyrosine kinase sustains EVI1 expression.6
Figure 1.
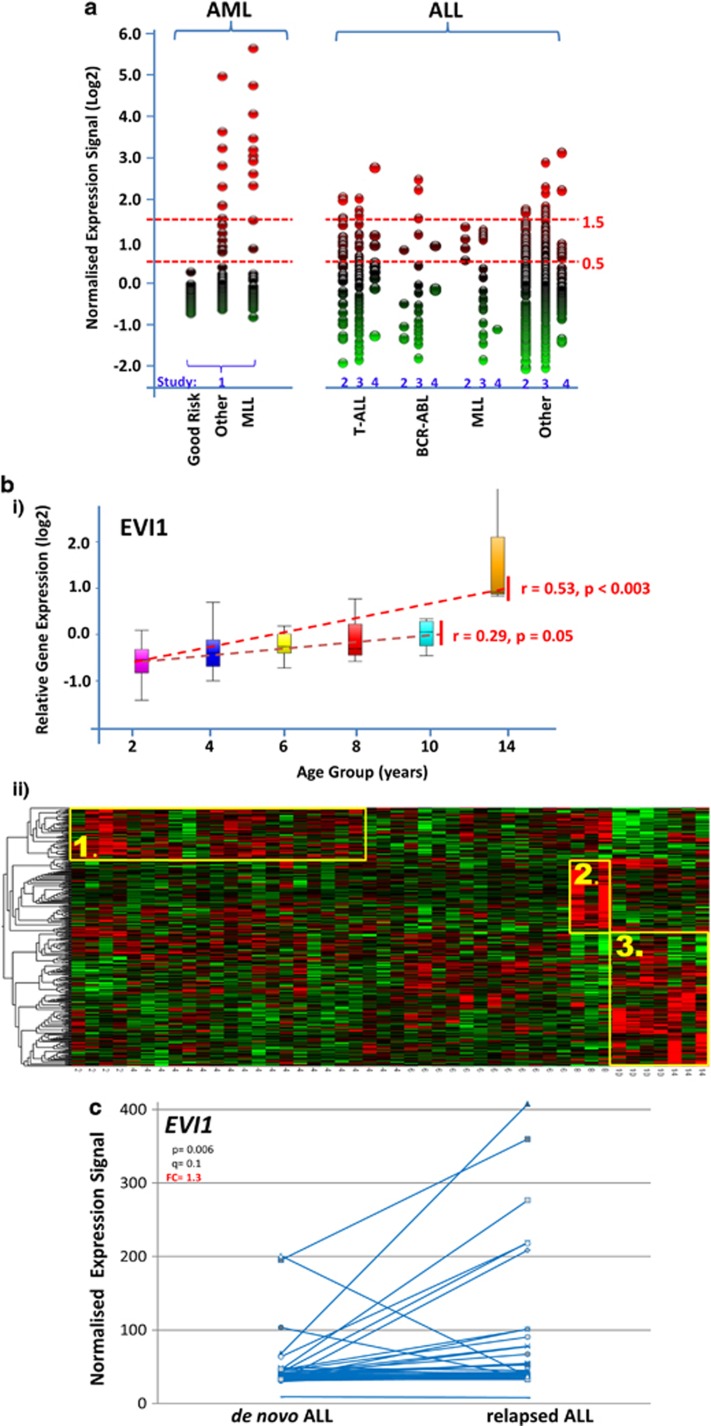
(a) Variability of EVI1 expression in paediatric leukaemia. Comparison of EVI1 expression of using the 221884_at Affymetrix gene probe. In an AML data set (1 GSE17855, n=237), Good Risk=good risk cytogenetics inv(16), t(15;17) and t(8;21) (n=74), Other=remaining cytogenetics (n=116) and MLL=MLL gene rearrangement-positive (n=47). The comparison shows three data sets of ALL (two GSE 13425 (n=190), three GSE28497 (n=255) and four Manchester (n=62)); BCR–ABL=BCR–ABL fusion (Philadelphia)-positive (n=5, n=16, n=3); T-ALL=T-cell ALL (n=36, n=45, n=9); MLL=MLL gene rearrangement-positive (n=4, n=18, n=1), Other=other ALL (n=145, n=176, n=49). (b) Age-associated gene expression in ALL panel (I). Age-related expression of EVI1 as measured by 221884_at Affymetrix gene expression probe set in 46 individuals with ALL (4.1, 1.1–13.0 years of ages (median, range); 23 female individuals); T-ALL, BCR–ABL and MLL-rearranged groups were removed. Box plot by age group (upper limit of bin shown), dotted line=median, whiskers show top and bottom quartiles. Rank regression by age, P-value and r-value shown. Panel (II) Heat map of age-associated changes in gene expression from cells obtained from diagnostic bone marrow aspirates of 46 individuals with ALL. Gene probe sets associated with age by multigroup analysis of variance with gender as covariate, P<0.01, identifying 415 probe sets as age related corresponding to 341 unique genes. Unsupervised hierarchical clustering using Euclidean metric with each variable normalised to mean 0 and variance 1. The horizontal axis ranked by age group (in years), age-related clusters derived from the dendrogram (vertical axis) highlighted in yellow; cluster 1=infancy/early childhood, cluster 2=late childhood, cluster 3=late childhood/puberty. Age specificity of clusters was confirmed using random substitution of probe sets (Qlucore Omics Explorer 2.3). (c) EVI1 expression in paired samples of de novo and relapse of ALL. Comparison of gene expression at diagnosis and at relapse of ALL from GSE28460 (Hogan et al.11). Expression of EVI1 was measured using the 221884_at Affymetrix gene probe set. Affymetrix gene expression microarray data normalisation was confirmed using multidimensional scaling in Qlucore Omics Explorer 2.3 and analysis of gene expression change was undertaken using a paired t-test with gender as an eliminated factor; q=false discovery rate-modified P-value, FC=fold change.
To complement the work reported by Konantz et al.,3 we analysed gene expression data generated from nucleated cells obtained from diagnostic bone marrow aspirates of 70 de novo ALL (31 female subjects, median age at diagnosis: 4.4 years, range: 1.1–14.6 years), using the Affymetrix U133+2 platform from our previously published and verified data set.7, 8 We analysed nine T-cell ALL, three BCR–ABL-rearranged Philadelphia-positive, one MLL-rearranged and 57 ALL with other cytogenetics, of which most were hyperdiploid. We focussed on EVI1 transcripts targeted by probes 226420_at and 221884_at, which detect exonic sequences of EVI1 transcripts.5 We also included two large and sufficiently annotated gene expression data sets in our analysis, together comprising 455 de novo ALL samples (GEO Data sets GES284979 and GES1342510). To determine whether high EVI1 expression in ALL is also associated with specific chromosomal changes involving BCR/ABL or MLL rearrangements, or T-cell phenotype, we divided de novo childhood ALL into four subgroups. We analysed T-cell ALL separately from BCR–ABL fusion (Philadelphia)-positive, MLL gene rearrangement-positive and ALL with other cytogenetic abnormalities (the largest group). In addition, and separately, we also analysed paired data sets of de novo and subsequently relapsed childhood ALL (GSE28460).11
EVI1 expression is variable in de novo paediatric ALL. However, the range of EVI1 expression levels is much smaller as compared with childhood AML (Figure 1a). When applying the criteria used for paediatric AML with samples considered EVI1-positive (+) when EVI1 expression is higher than log0.5 normalised fold change, the proportion of EVI1+ ALLs is larger than AML and includes cases of T-cell ALL. There was no clear association of higher EVI1 expression levels with BCR–ABL or MLL rearrangements in the analysis of our own samples and results of published data sets (Figure 1a). The variability of EVI1 expression in the MLL-rearranged ALL group might be partly dependent on specific MLL gene rearrangements, as AMLs with MLL/AF6 and MLL/AF9 fusions are associated in particular with high EVI1 expression in adults and children.5, 12 We found no obvious possible cause for variability of EVI1 expression in ALL of other cytogenetic subgroups. Given the rarity of 3q rearrangements in ALL, high EVI1 expression is likely to be a secondary event. Cytogenetic features of high EVI1-expressing ALL, which include hyperdiploid and normal karyotype disease, are listed in Supplementary Table 1.
Konatz et al.3 observed that EVI1 modulated the expression of apoptosis-related genes in paediatric ALL. In accordance with their data,3 we identified BCLX (fold change −1.8, P=0.007) and PUMA (FC=−1.9, P=0.005) in T-cell ALL when determining EVI1-co-regulated genes by selecting for significantly changed (P<0.01, analysis of variance) low and high expressed genes in paediatric T- and B-cell ALL with EVI1 expression >log1.5. To determine the overlap in global expression patterns associated with high EVI1 transcripts, we chose a >log1.5 cutoff to analyse a meaningful sample size for comparison. In general, the overlap was greater between T-cell ALL and B-cell ALL than either of these groups with AML (Supplementary Information, Figure 1). Only 12 genes were significantly co-regulated in all the subgroups of childhood leukaemia (P<0.01, analysis of variance) (Supplementary Information, Figure 1), with seven regulated in different directions between the subgroups. This included SMARCA5, of which the encoded protein recently has been shown to directly interact with the EVI1 protein in SKOV ovarian cancer cells and K562 leukaemia cells.13
Importantly, we noted that several of the high EVI1-expressing ALLs were from patients in late childhood or adolescence. To further explore a possible association of EVI1 expression with age in de novo childhood ALL, we carried out rank regression analysis by age on our B-cell ALL cases, which is the largest group of our age- and sex-annotated data set (n=51), excluding those with MLL or BCR–ABL rearrangements. We found a highly significant increase in EVI1 expression with age at diagnosis of ALL (Figure 1bi, upper panel) during childhood years (age 1–10, r=0.29, P=0.05) but in particular obvious with the onset of adolescence (age 1–14, r=0.53, P<0.003). To further explore the relationship of patient age on gene expression patterns in childhood ALL we applied this approach to the entire data set. We identified 415 probe sets corresponding to 341 genes with significant age-associated changes in expression levels (P=0.01) (Figure 1bii, lower panel). Of these, 130 genes have the highest expression in adolescence (cluster 3). When we associated the upstream regulators of these 130 genes (Ingenuity pathway analysis), we found that 21 are regulated directly by transforming growth factor-β (P=1.8 × 10−3). This resembles our findings with respect to the impact of age on gene expression patterns in general14 and implies that transforming growth factor-β might in particular contribute to age-dependent changes in ALL gene expression observed in the adolescent age group. As EVI1 also has been shown to have an impact on transforming growth factor-β signalling,15 we have started to investigate this in more detail. When we analysed the age association of gene expression patterns in our AML data set (also excluding MLL-rearranged cases), we detected some striking differences to ALL. Although age has an impact on gene expression patterns also distinctly in AML, we see a negative correlation of EVI1 with increasing age (Supplementary Figure 2).
The data set by Hogan et al.11 shows that in de novo ALL that subsequently relapses EVI1 expression has a significantly wider range compared with the de novo ALL cases that are not selected for subsequent relapse in the other studies (P<0.01). Importantly, paired analysis suggests that at ALL relapse, expression of EVI1 is on average higher than in the corresponding de novo sample (1.3-fold increased, P<0.006, Figure 1c).
In summary, our analysis confirms high EVI1 expression in a group of paediatric ALL, which does not appear to be confined to distinct cytogenetic subtypes. The limited overlap of gene expression profiles associated with high EVI1 expression in different forms of childhood leukaemia and the different levels of expression of some co-regulated genes implies the possibility of tissue specificity, and the impact of cell of origin for EVI1-mediated transcriptional regulation, which has also been suggested for MLL-rearranged AML subtypes.16 High EVI1 expression itself is likely to be a secondary event in paediatric lymphoblastic and myeloid leukaemia. The higher expression in older children might be linked to the worse prognosis of ALL in this age group. Whereas higher EVI1 expression in relapsed disease might be a function of increasing age, it also implies potentially a more general role in ALL stem cell survival. It will be important to prospectively investigate EVI1 in paediatric ALL for prognosis on current treatment regimens, and potential therapeutic benefit. In addition, our analysis strongly suggests an impact of age at diagnosis on gene expression patterns in childhood leukaemia. As we are uncertain to what extent this reflects the chronological age of the patient, or that of the cell of origin in childhood leukaemia, we have started to investigate this more comprehensively.
Acknowledgments
SM is supported by Leukaemia Lymphoma Research, UK, and the Kay Kendall Leukaemia Fund. CDL is supported by ESPE Research Fellowship, sponsored by Novo Nordisk A/S.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on Blood Cancer Journal website (http://www.nature.com/bcj)
Supplementary Material
References
- Goyama S, Yamamoto G, Shimabe M, Sato T, Ichikawa M, Ogawa S, et al. Evi-1 is a critical regulator for hematopoietic stem cells and transformed leukemic cells. Cell Stem Cell. 2008;3:207–220. doi: 10.1016/j.stem.2008.06.002. [DOI] [PubMed] [Google Scholar]
- Lugthart S, van Drunen E, van Norden Y, van Hoven A, Erpelinck CA, Valk PJ, et al. High EVI1 levels predict adverse outcome in acute myeloid leukemia: prevalence of EVI1 overexpression and chromosome 3q26 abnormalities underestimated. Blood. 2008;111:4329–4337. doi: 10.1182/blood-2007-10-119230. [DOI] [PubMed] [Google Scholar]
- Konantz M, Andre MC, Ebinger M, Grauer M, Wang H, Grzywna S, et al. EVI-1 modulates leukemogenic potential and apoptosis sensitivity in human acute lymphoblastic leukemia. Leukemia. 2013;27:56–65. doi: 10.1038/leu.2012.211. [DOI] [PubMed] [Google Scholar]
- Ho PA, Alonzo TA, Gerbing RB, Pollard JA, Hirsch B, Raimondi SC, et al. High EVI1 expression is associated with MLL rearrangements and predicts decreased survival in paediatric acute myeloid leukaemia: a report from the children's oncology group. Br J Haematol. 2013;162:670–677. doi: 10.1111/bjh.12444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balgobind BV, Lugthart S, Hollink IH, Arentsen-Peters ST, van Wering ER, de Graaf SS, et al. EVI1 overexpression in distinct subtypes of pediatric acute myeloid leukemia. Leukemia. 2010;24:942–949. doi: 10.1038/leu.2010.47. [DOI] [PubMed] [Google Scholar]
- Roy S, Jorgensen HG, Roy P, Abed El Baky M, Melo JV, Strathdee G, et al. BCR-ABL1 tyrosine kinase sustained MECOM expression in chronic myeloid leukaemia. Br J Haematol. 2012;157:446–456. doi: 10.1111/j.1365-2141.2012.09078.x. [DOI] [PubMed] [Google Scholar]
- Strefford JC, van Delft FW, Robinson HM, Worley H, Yiannikouris O, Selzer R, et al. Complex genomic alterations and gene expression in acute lymphoblastic leukaemia with intrachromosomal amplification of chromosome 21. Proc Natl Acad Sci USA. 2006;103:8167–8172. doi: 10.1073/pnas.0602360103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel N, Krishnan S, Offman MN, Krol M, Moss CX, Leighton C, et al. A dyad of lymphoblastic lysosomal cysteine proteases degrades the antileukemic drug L-asparaginase. J Clin Invest. 2009;119:1964–1973. doi: 10.1172/JCI37977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coustan-Smith E, Song G, Clark C, Key L, Liu P, Mehrpooya M, et al. New markers for minimal residual disease detection in acute lymphoblastic leukemia. Blood. 2011;117:6267–6276. doi: 10.1182/blood-2010-12-324004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Den Boer ML, van Slegtenhorst M, De Menezes RX, Cheok MH, Buijs-Gladdines JG, Peters ST, et al. A subtype of childhood acute lymphoblastic leukaemia with poor treatment outcome: a genome-wide classification study. Lancet Oncol. 2009;10:125–134. doi: 10.1016/S1470-2045(08)70339-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hogan LE, Meyer JA, Yang J, Wang J, Wong N, Yang W, et al. Integrated genomic analysis of relapsed childhood acute lymphoblastic leukemia reveals therapeutic strategies. Blood. 2011;118:5218–5226. doi: 10.1182/blood-2011-04-345595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bindels EM, Havermans M, Lugthart S, Erpelinck C, Wocjtowicz E, Krivtsov AV, et al. EVI1 is critical for the pathogenesis of a subset of MLL-AF9-rearranged AMLs. Blood. 2012;119:5838–5849. doi: 10.1182/blood-2011-11-393827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bard-Chapeau EA, Gunaratne J, Kumar P, Chua BQ, Muller J, Bard FA, et al. EVI1 oncoprotein interacts with a large and complex network of proteins and integrates signals through protein phosphorylation. Proc Natl Acad Sci USA. 2013;110:15. doi: 10.1073/pnas.1309310110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens A, Hanson D, Whatmore A, Destenaves B, Chatelain P, Clayton P. Human growth is associated with distinct patterns of gene expression in evolutionarily conserved networks. BMC Genomics. 2013;14:547. doi: 10.1186/1471-2164-14-547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Chen L, Ko TC, Fields AP, Thompson EA. Evi1 is a survival factor which conveys resistance to both TGFbeta- and taxol-mediated cell death via PI3K/AKT. Oncogene. 2006;25:3565–3575. doi: 10.1038/sj.onc.1209403. [DOI] [PubMed] [Google Scholar]
- Krivtsov AV, Figueroa ME, Sinha AU, Stubbs MC, Feng Z, Valk PJ, et al. Cell of origin determines clinically relevant subtypes of MLL-rearranged AML. Leukemia. 2013;27:852–860. doi: 10.1038/leu.2012.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.