Abstract
Purpose
The role of metastasectomy has been debatable and unclear in the treatment for patients with metastatic gastric cancer. Therefore, this study was designed to evaluate the role of metastasectomy on the overall survival of these patients.
Materials and Methods
In 2,406 patients who underwent gastrectomy for gastric cancer between 1998 and 2010, 188 (7.8%) patients had their first surgery for metastatic gastric cancer. To minimize the bias of systemic chemotherapy, 99 patients who received postoperative chemotherapy (fewer than 2 cycles) were excluded. The primary gastrectomy or metastasectomy had not been enforced in the following cases. Patients with far advanced peritoneal dissemination, multiple liver and lung metastasis (more than 2), and a poor general condition (Eastern Cooperative Oncology Group>2) were excluded. Based on the metastasectomy, the patients were classified into two groups, gastrectomy with metastasectomy and gastrectomy only group.
Results
There was no significant difference between both groups in clinicopathological characteristics except for the mean age (P=0.047). The univariate analysis for overall survival show statistical significances in metastasectomy (P=0.026), distal gastrectomy (P=0.047), and combined resection of another organ (P=0.047) group. With a multivariate analysis, metastasectomy was a significant factor in patient survival after surgery (odds ratio 1.679; P=0.034).
Conclusions
Based on our results, we assume that a detailed strategy for surgery is needed to improve the overall survival of patients with metastatic gastric cancer. Therefore, we suggest that a metastasectomy can help prolong overall survival in some patients with metastatic gastric cancer.
Keywords: Gastric cancer, Distant metastasis, Metastasectomy, Overall survival
Introduction
The overall prognosis of gastric cancer patients with distant metastasis remains very poor despite the decline in incidence and mortality rate of gastric cancer in most parts of the world.1-4 Despite many investigators reporting that their surgical methods improve the overall survival of patients with gastric cancer and distant metastasis, the proper surgical management remains controversial.
Recently, several investigators suggested that a primary debulking surgery or a primary gastrectomy with metastasectomy could help prolong the survival of patients with gastric cancer patients and distant metastasis.5-10 On the other hand, other studies suggested an unfavorable overall survival after resection of metastatic gastric cancer.11-15 However, most studies were performed without consideration of the impact of systemic chemotherapy and the severity of the disease at time of treatment on the survival benefit.
Therefore, our study was designed to evaluate the impact of metastasectomy on survival of patients with gastric cancer patients and distant metastasis.
Materials and Methods
1. Patients and methods
We reviewed the data on 188 patients with metastatic gastric cancer who underwent gastrectomy and who had postoperative systemic chemotherapy that was collected prospectively between 1992 and 2012 at Hanyang University Hospital. To minimize the bias of systemic chemotherapy, 99 patients who received postoperative systemic chemotherapy of less than two cycles were excluded from this study. Based on the metastasectomy, all patients were divided into two groups (gastrectomy with metastasectomy [GM] and gastrectomy only [GO]).
This study included 89 patients with an optimal Eastern Cooperative Oncology Group (ECOG) performance status (measured as an ECOG<2). The purpose of the metastasectomy was to remove all metastatic lesions. In our approach to the metastasectomy, we did not attempt aggressive surgical procedures such as peritonectomy and hemihepatectomy, as these can increase the risk of postoperative morbidity and mortality. Therefore, the intent was to remove all visible metastatic lesions during the operation. The inclusion criteria for the metastasectomy group were a single metastasis in the liver or lung, no advanced peritoneal dissemination (P1 or P2, as per the Japanese Research Society for the Study of Gastric Cancer criteria). The inclusion criteria for the gastrectomy only were the presence of multiple liver metastasis, multiple lung metastasis, or advanced peritoneal dissemination (P3 as per the Japanese Research Society for the Study of Gastric Cancer criteria).
The exclusion criteria for this study were the rigid fixation of the pancreas head due to the primary gastric cancer, a palliative gastrectomy for uncontrolled bleeding, perforation or obstruction, and poor ECOG performance status (ECOG >2).
2. Statistical analysis
The primary end-point for this study was survival; the overall survival rates were estimated by using the Kaplan-Meier method. Statistical analysis was performed by using PASW Statistics ver. 18.0 for Windows (IBM Co., Armonk, NY, USA). All values are expressed as mean with standard deviation. Categorical variables were analyzed by using the chi-square test and all continuous variables were analyzed by using Student's t-test, depending on the data. The long-rank test was used to compare significant differences between subgroups by using aunivariate analysis. Multivariate analysis was performed to identify prognostic factors associated with overall survival. The Cox proportional hazards model was employed for multivariate regression analysis. The survival curve was calculated by using the Kaplan-Meier method. Hazard ratios with 95% confidence intervals were estimated for each variable in the multivariate analysis. A P<0.05 was considered statistically significant.
Results
1. Patient characteristics
The clinicopathological characteristics of the 89 gastric cancer patients with distant metastasis are presented in Table 1. There was no significant difference between both groups in most variable factors except for mean age (GM vs. GO group, 58.8 vs. 53.1, P=0.047).
Table 1.
Clinicopathological characteristics of patients with metastatic gastric cancer
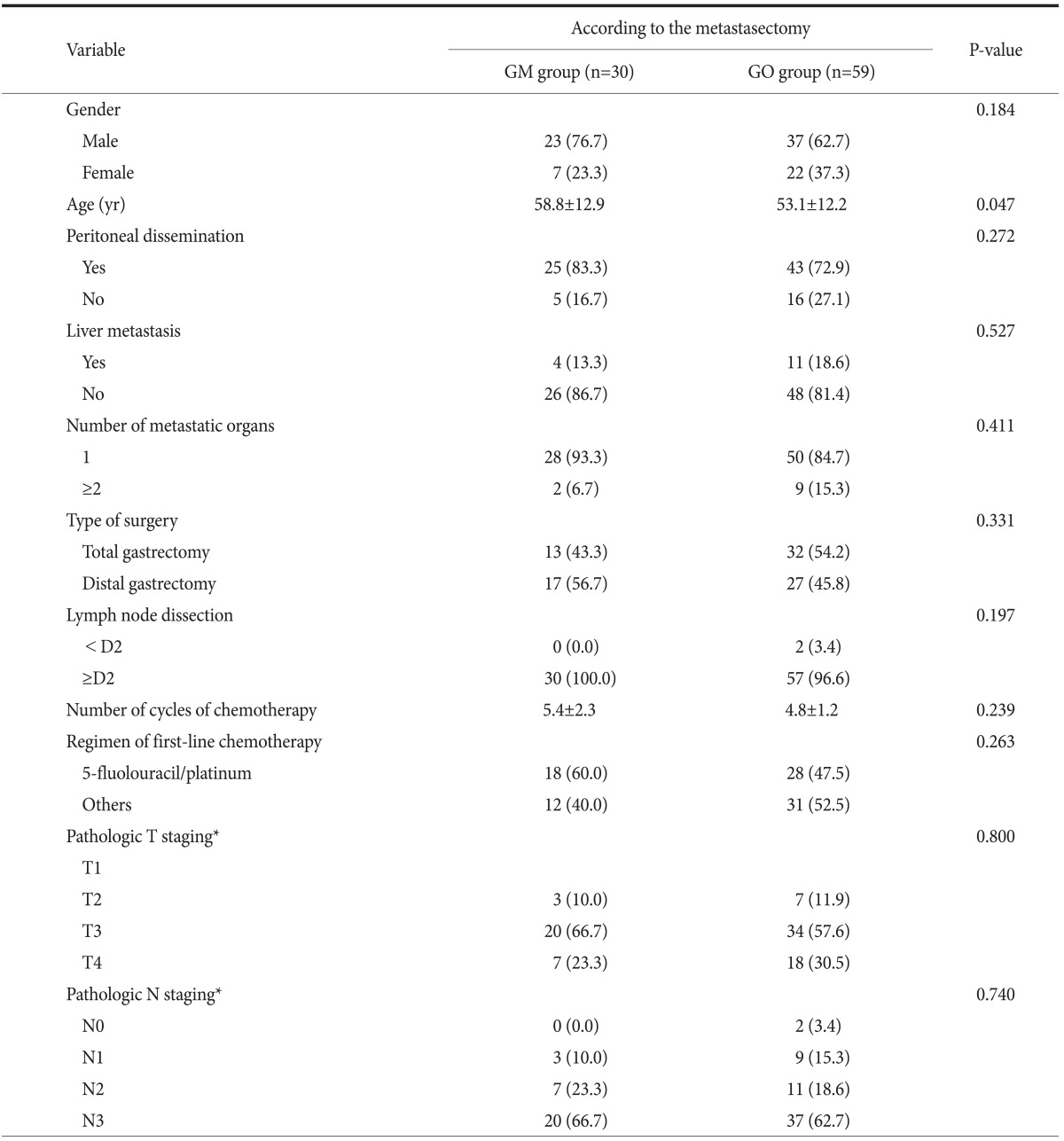
Values are presented as number (%) or mean±standard deviation. GM group = gastrectomy with metastasectomy group; GO group = gastrectomy only group. *American Joint Committee on Cancer (AJCC) 6th edition on gastric cancer staging system.
2. Surgical outcomes
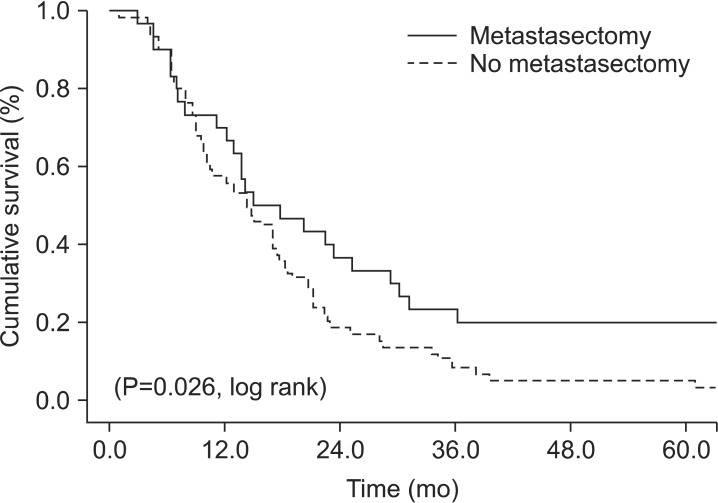
The median follow-up period was 14.9 months (range 1 to 157.6 months). The 3-year overall survival rates were 23.3 and 8.5% for the GM and GO groups, respectively (Fig. 1).
Fig. 1.
Survival of patients with metastatic gastric cancer who underwent gastrectomy with and without metastasectomy.
3. Analysis of prognostic factors for overall survival
In the univariate analysis for overall survival, there were significant differences in the 3-year survival rates for metastasectomy, distal gastrectomy, and combined resection of other organs was 23.3% vs. 8.5% (P=0.026), 17.8% vs. 9.1% (P=0.047) and 8.1% vs. 17.3% (P=0.047), respectively (Table 2). The multivariate analysis showed that a primary gastrectomy with metastasectomy (odds ratio=1.679, 95% confidence interval 1.040 to 2.711, P=0.034) was associated with survival (Table 3).
Table 2.
One- and three-year survival rates of patients with M1* gastric cancer after surgery
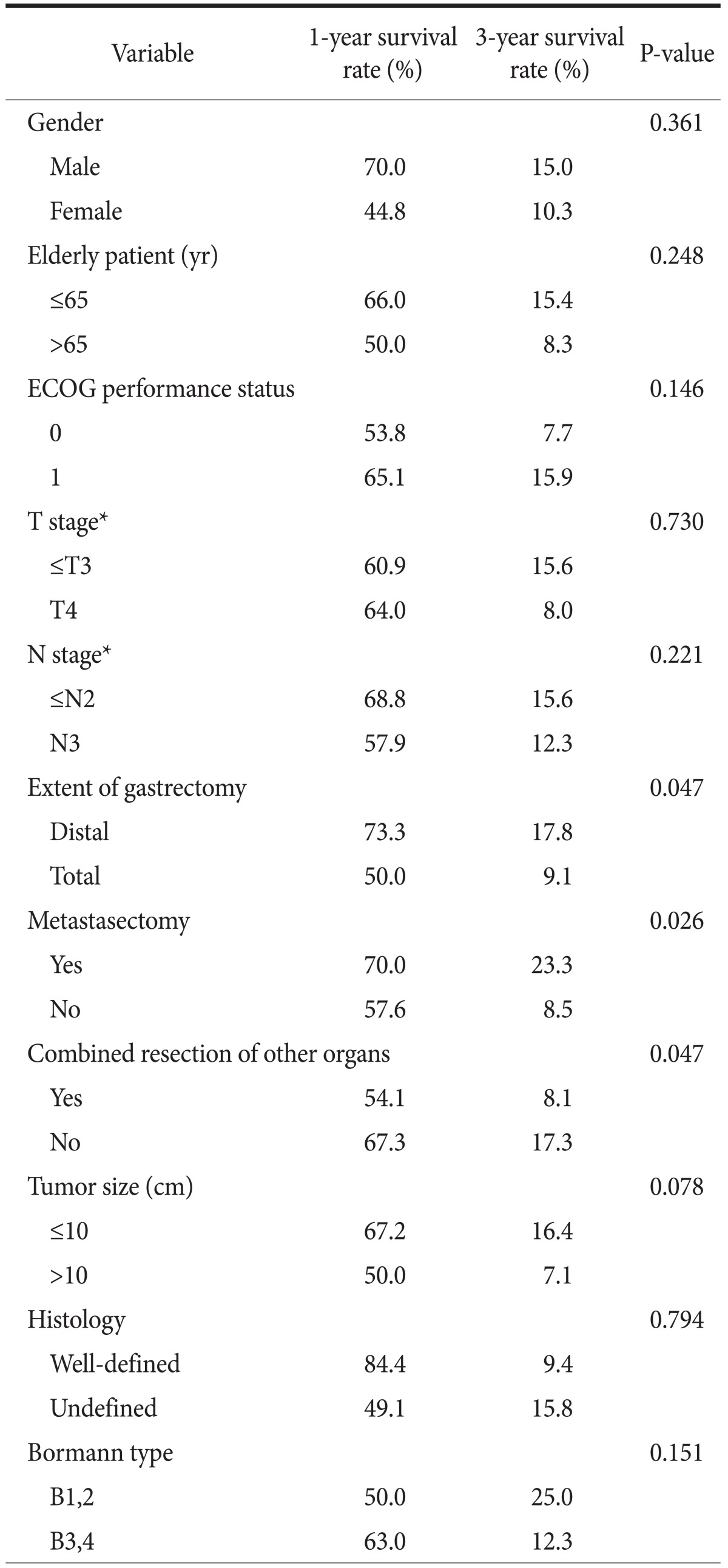
ECOG = Eastern Cooperative Oncology Group. *American Joint Committee on Cancer (AJCC) 6th edition on gastric cancer staging system.
Table 3.
Multivariate analysis of prognostic factors associated with overall survival
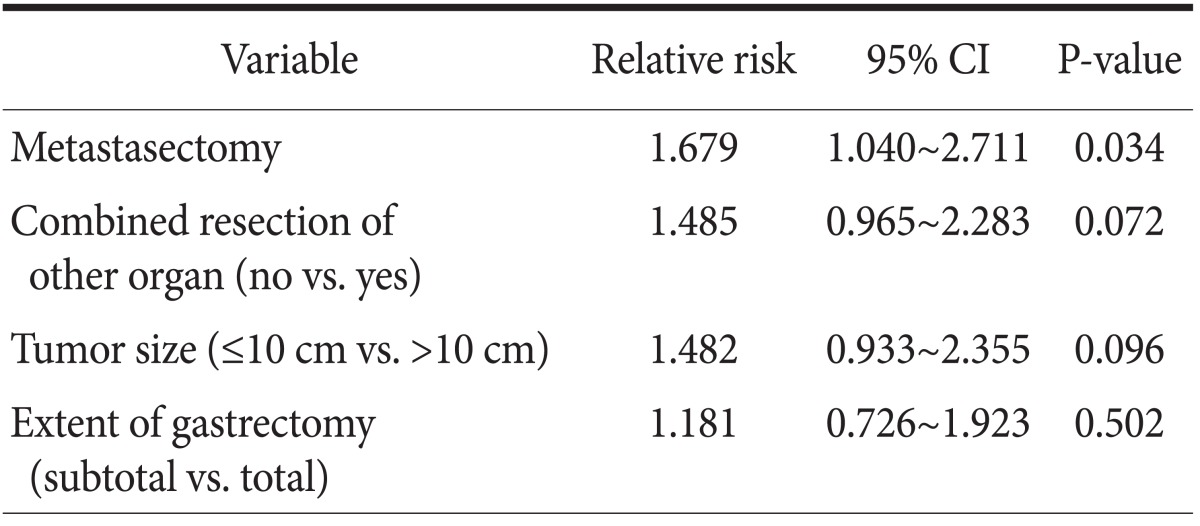
CI = confidence interval.
Discussion
Despite the extensive screening programs that have been operating under national health support for early detection of gastric cancer in Korea and Japan, many patients, at the time of diagnosis have very advanced gastric cancer with distant metastasis. A radical gastrectomy with a clear resection margin and D2 lymph node dissection has become a standard surgical method for advanced gastric cancer patients, but for patients with distant metastasis the role of surgery is still debatable.
Recently, the role of surgery in patients with metastatic gastric cancer is a matter of debate and controversy, as there is no current standard treatment for patients with metastatic gastric cancer. According to the National Comprehensive Cancer Network clinical practice guidelines, palliative systemic chemotherapy and best supportive care are recommended in patients with metastatic gastric cancer.16 Recently, however, several investigators have suggested that cytoreductive surgery or a primary gastrectomy with metastasectomy is associated with an increase in survival. The rationale for surgery is that the disease may respond to adjuvant treatment more effectively due to a reduction in tumor load. Theoretically, a reduction in the tumor burden can decrease the metabolic demands of the tumor. Also, because the tumor itself can produce an immunosuppressive cytokine, reducing the tumor burden may also have an immunologic benefit.3,13,17-19
In practice, however, the procedure is difficult to perform for many reasons Many experts on gastric cancer treatment suggested that patients with metastatic gastric cancer who had surgery did not experience an increase in overall survival. Also, many patients have a high associated mortality and morbidity. Based on these results, these experts suggested that the majority of patients with metastatic disease did not require surgery.
Based on our results of the overall survival of patients with gastric cancer, those patients with metastases show a poor overall 5-year survival rate (12.9%). We saw the need to reconsider the surgical strategy on the basis of the differences in the characteristics of these patients. Unlike most gastric cancer patients without metastasis, patients with metastases show a wide range of physiological presentations based on their specific medical condition and the stage of their disease. Therefore, an appropriate surgical strategy for improving the overall survival should be considered according to the specific condition of the individual patient.
Our institution, as previously mentioned, had implemented a strict method for patient selection for surgery in patients with metastatic gastric cancer. To the exclusion of palliative surgery for obstruction or bleeding, the primary gastrectomy with or without metastasectomy is rigorously enforced in patients <70 years of age, those with a good performance status (ECOG<2), patients with a resectable primary tumor (excluding fixed gastric cancers in the pancreas head), and in patients with a less aggressive cancer (<P2, solitary liver metastasis, etc.). Our most important policy in selecting patients for the procedure is the presence of the possibility for systemic chemotherapy after gastrectomy. The results of this study are to achieve better surgical results in improving the overall survival of patients with metastatic gastric cancer. In particular in this study we assumed that metastasectomy could be useful in improving the overall survival in a strictly selected patient group.
There are several limitations inherent in our retrospective study. Although the baseline characteristics of the patients between two groups were similar, there was a clear difference in the progression of disease (P1, P2 vs. P3, solitary liver metastasis vs. multiple liver metastasis). This difference may be a confounding factor in the evaluation of the impact of metastasectomy on overall survival. Unfortunately, we could not analyze the impact of surgery on overall survival according to the specific organ because of our low case number, therefore further studies are needed to evaluate the impact of surgery on overall survival according to the specific organ. Although our study is limited in its ability to evaluate surgical outcomes, our findings regarding the survival benefit of metastasectomy strongly support the hypothesis that metastasectomy can play a vital role in improving overall survival of patients with metastatic gastric cancer.
In our study we are not proposing that primary gastrectomy with metastasectomy is the gold standard in improving the overall survival for all patients with metastatic gastric cancer. Unlike those without metastasis, patients with metastases have a wide variation in their general health because of the range in disease progression. Therefore, we suggest that primary gastrectomy with metastasectomy can help improve overall survival in some patients with metastatic gastric cancer.
References
- 1.Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 2.Yang L, Parkin DM, Ferlay J, Li L, Chen Y. Estimates of cancer incidence in China for 2000 and projections for 2005. Cancer Epidemiol Biomarkers Prev. 2005;14:243–250. [PubMed] [Google Scholar]
- 3.Hartgrink HH, Putter H, Klein Kranenbarg E, Bonenkamp JJ, van de Velde CJ Dutch Gastric Cancer Group. Value of palliative resection in gastric cancer. Br J Surg. 2002;89:1438–1443. doi: 10.1046/j.1365-2168.2002.02220.x. [DOI] [PubMed] [Google Scholar]
- 4.Swan R, Miner TJ. Current role of surgical therapy in gastric cancer. World J Gastroenterol. 2006;12:372–379. doi: 10.3748/wjg.v12.i3.372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Maekawa S, Saku M, Maehara Y, Sadanaga N, Ikejiri K, Anai H, et al. Surgical treatment for advanced gastric cancer. Hepatogastroenterology. 1996;43:178–186. [PubMed] [Google Scholar]
- 6.Kikuchi S, Tsukamoto H, Mieno H, Sato K, Kobayashi N, Shimao H, et al. Results of resection of gastric cancer with distant metastases. Hepatogastroenterology. 1998;45:592–596. [PubMed] [Google Scholar]
- 7.Inada T, Ogata Y, Kubota T, Ishihara M, Tomikawa M, Ando J, et al. D2-lymphadenectomy improves the survival of patients with peritoneal cytology-positive gastric cancer. Anticancer Res. 2002;22:291–294. [PubMed] [Google Scholar]
- 8.Medina-Franco H, Contreras-Saldívar A, Ramos-De La Medina A, Palacios-Sanchez P, Cortés-González R, Ugarte JA. Surgery for stage IV gastric cancer. Am J Surg. 2004;187:543–546. doi: 10.1016/j.amjsurg.2003.12.045. [DOI] [PubMed] [Google Scholar]
- 9.Lin SZ, Tong HF, You T, Yu YJ, Wu WJ, Chen C, et al. Palliative gastrectomy and chemotherapy for stage IV gastric cancer. J Cancer Res Clin Oncol. 2008;134:187–192. doi: 10.1007/s00432-007-0268-z. [DOI] [PubMed] [Google Scholar]
- 10.Kobayashi Y, Fukui T, Ito S, Shitara K, Ito S, Hatooka S, et al. Pulmonary metastasectomy for gastric cancer: a 13-year single-institution experience. Surg Today. 2013;43:1382–1389. doi: 10.1007/s00595-012-0432-1. [DOI] [PubMed] [Google Scholar]
- 11.Doglietto GB, Pacelli F, Caprino P, Alfieri S, Carriero C, Malerba M, et al. Palliative surgery for far-advanced gastric cancer: a retrospective study on 305 consecutive patients. Am Surg. 1999;65:352–355. [PubMed] [Google Scholar]
- 12.Sarela AI, Yelluri S Leeds Upper Gastrointestinal Cancer Multidisciplinary Team. Gastric adenocarcinoma with distant metastasis: is gastrectomy necessary? Arch Surg. 2007;142:143–149. doi: 10.1001/archsurg.142.2.143. [DOI] [PubMed] [Google Scholar]
- 13.Chow LW, Lim BH, Leung SY, Branicki FJ, Gertsch P. Gastric carcinoma with synchronous liver metastases: palliative gastrectomy or not? Aust N Z J Surg. 1995;65:719–723. doi: 10.1111/j.1445-2197.1995.tb00544.x. [DOI] [PubMed] [Google Scholar]
- 14.Ouchi K, Sugawara T, Ono H, Fujiya T, Kamiyama Y, Kakugawa Y, et al. Therapeutic significance of palliative operations for gastric cancer for survival and quality of life. J Surg Oncol. 1998;69:41–44. doi: 10.1002/(sici)1096-9098(199809)69:1<41::aid-jso8>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 15.Kunisaki C, Shimada H, Akiyama H, Nomura M, Matsuda G, Ono H. Survival benefit of palliative gastrectomy in advanced incurable gastric cancer. Anticancer Res. 2003;23:1853–1858. [PubMed] [Google Scholar]
- 16.Ajani JA, Barthel JS, Bekaii-Saab T, Bentrem DJ, D'Amico TA, Das P, et al. NCCN Gastric Cancer Panel. Gastric cancer. J Natl Compr Canc Netw. 2010;8:378–409. doi: 10.6004/jnccn.2010.0030. [DOI] [PubMed] [Google Scholar]
- 17.McCarter MD, Fong Y. Role for surgical cytoreduction in multimodality treatments for cancer. Ann Surg Oncol. 2001;8:38–43. doi: 10.1007/s10434-001-0038-0. [DOI] [PubMed] [Google Scholar]
- 18.Pollock RE, Roth JA. Cancer-induced immunosuppression: implications for therapy? Semin Surg Oncol. 1989;5:414–419. doi: 10.1002/ssu.2980050607. [DOI] [PubMed] [Google Scholar]
- 19.Koga S, Kawaguchi H, Kishimoto H, Tanaka K, Miyano Y, Kimura O, et al. Therapeutic significance of noncurative gastrectomy for gastric cancer with liver metastasis. Am J Surg. 1980;140:356–359. doi: 10.1016/0002-9610(80)90167-1. [DOI] [PubMed] [Google Scholar]