Abstract
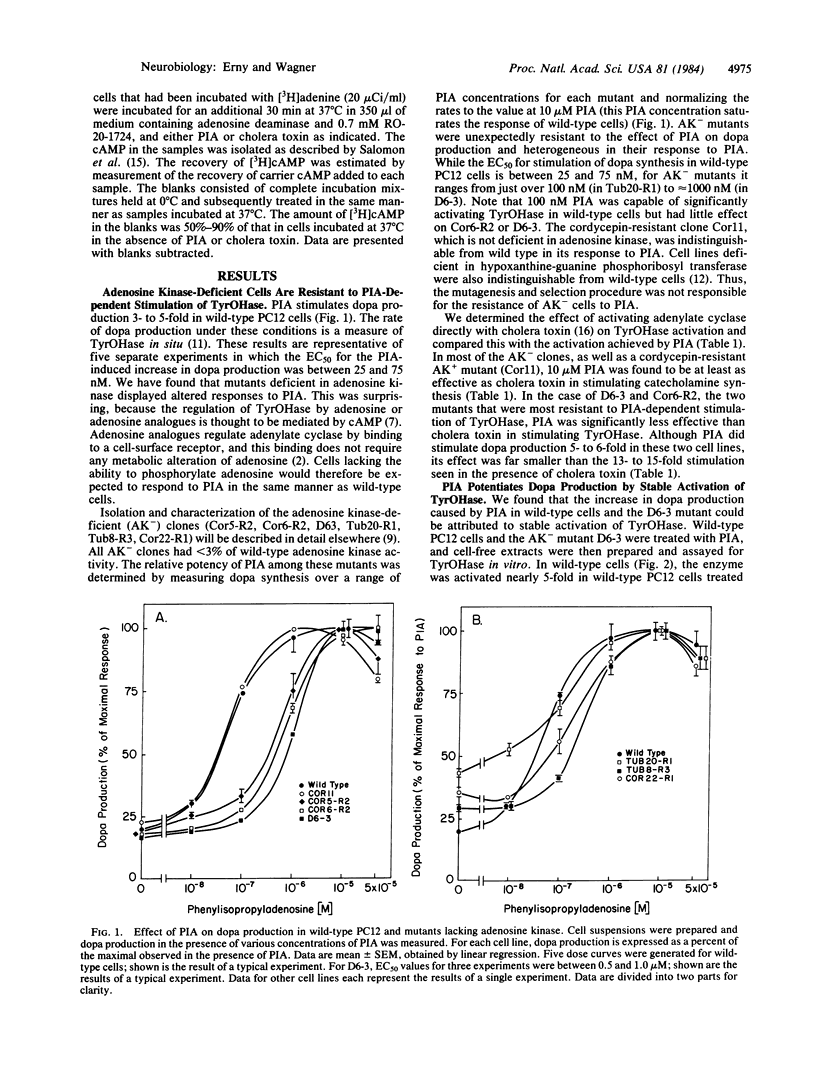
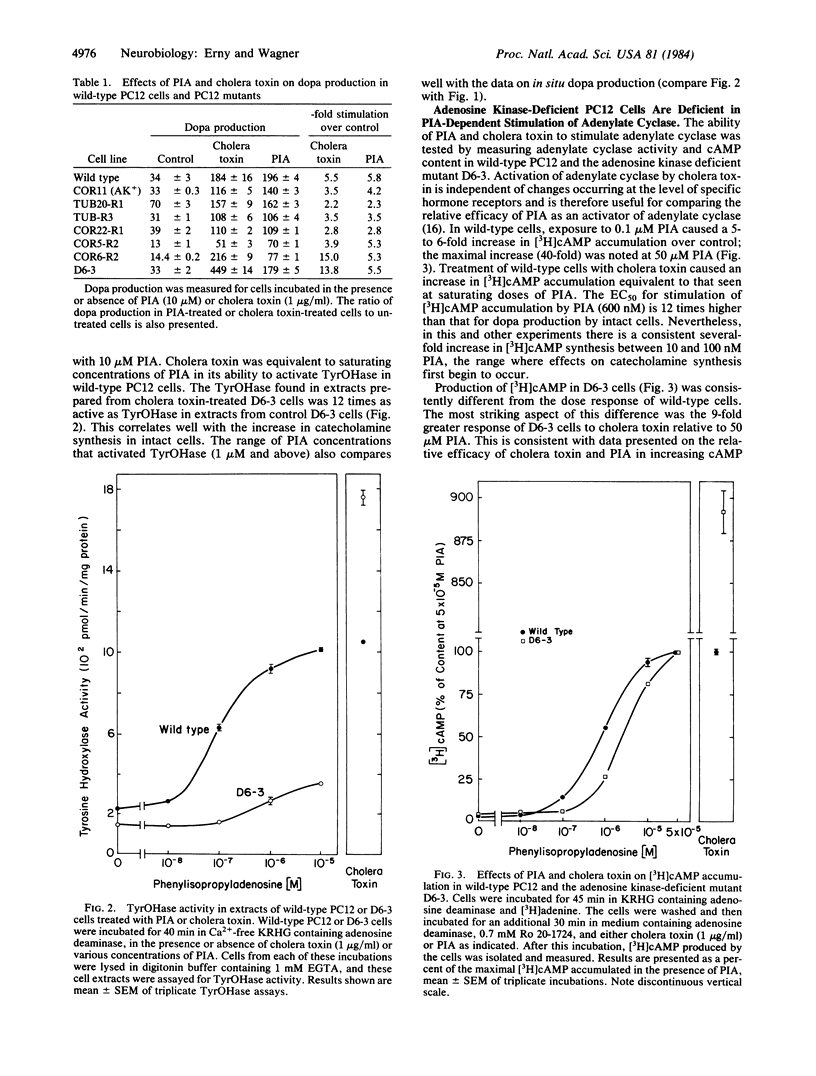
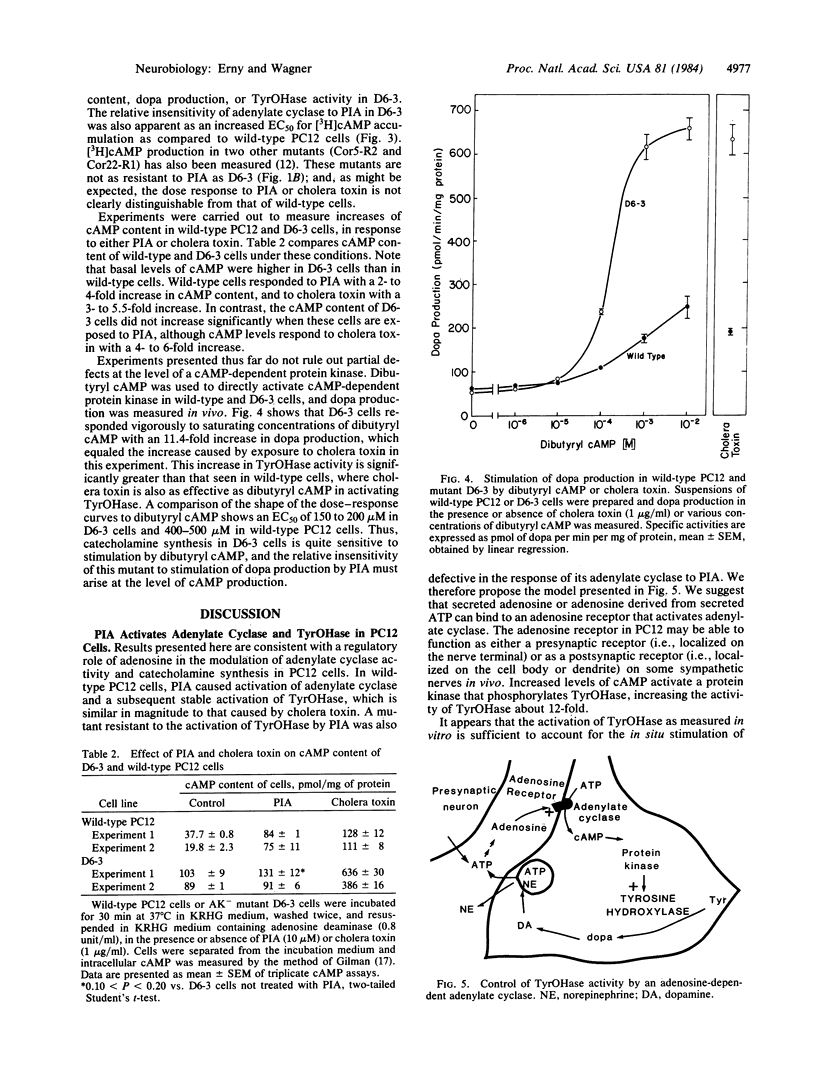
(R)-N6-Phenylisopropyladenosine (PIA) stimulates dopa production 3- to 5-fold in PC12 cells, with a half-maximal effective concentration (EC50) of 50 nM. This increase can be explained by a stable activation of tyrosine hydroxylase [TyrOHase; L-tyrosine, tetrahydropteridine:oxygen oxidoreductase (3-hydroxylating), EC 1.14.16.2] when it is phosphorylated by a cAMP-dependent protein kinase. The activation of TyrOHase is mediated by the adenosine-dependent activation of adenylate cyclase (EC50 = 600 nM). PIA (10 microM) is as effective as cholera toxin or dibutyryl cAMP in activating TyrOHase in wild-type cells. Adenosine kinase-deficient mutants of PC12 were found to be resistant to PIA-dependent activation of TyrOHase (EC50 = 100-1000 nM). This phenomenon was explored in detail in one adenosine kinase-deficient mutant and was shown to occur because the mutant was resistant to the adenosine-dependent activation of adenylate cyclase. In this mutant, TyrOHase was activated 14-fold by cholera toxin, suggesting that activated TyrOHase is about 14 times as active as unactivated TyrOHase. These studies with kinase-deficient PC12 cells provide genetic evidence that adenosine-dependent activation of TyrOHase is mediated by acute increases in cAMP. When the adenosine receptor found on PC12 cells is expressed in vivo, it might function as either a presynaptic (i.e., localized on the nerve terminal) or a postsynaptic (i.e., localized on the cell body or dendrite) receptor that regulates rates of transmitter synthesis in response to cell activity.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arch J. R., Newsholme E. A. The control of the metabolism and the hormonal role of adenosine. Essays Biochem. 1978;14:82–123. [PubMed] [Google Scholar]
- Blank C. L., Pike R. A novel, inexpensive, and sensitive method for analysis of tyrosine hydroxylase activity in tissue samples. Life Sci. 1976 Apr 15;18(8):859–865. doi: 10.1016/0024-3205(76)90013-8. [DOI] [PubMed] [Google Scholar]
- Burnstock G. Do some nerve cells release more than one transmitter? Neuroscience. 1976 Aug;1(4):239–248. doi: 10.1016/0306-4522(76)90054-3. [DOI] [PubMed] [Google Scholar]
- Erny R. E., Berezo M. W., Perlman R. L. Activation of tyrosine 3-monooxygenase in pheochromocytoma cells by adenosine. J Biol Chem. 1981 Feb 10;256(3):1335–1339. [PubMed] [Google Scholar]
- Exton J. H., Lewis S. B., Ho R. J., Robison G. A., Park C. R. The role of cyclic AMP in the interaction of glucagon and insulin in the control of liver metabolism. Ann N Y Acad Sci. 1971 Dec 30;185:85–100. doi: 10.1111/j.1749-6632.1971.tb45239.x. [DOI] [PubMed] [Google Scholar]
- Fox I. H., Kelley W. N. The role of adenosine and 2'-deoxyadenosine in mammalian cells. Annu Rev Biochem. 1978;47:655–686. doi: 10.1146/annurev.bi.47.070178.003255. [DOI] [PubMed] [Google Scholar]
- Gilman A. G. Protein binding assays for cyclic nucleotides. Adv Cyclic Nucleotide Res. 1972;2:9–24. [PubMed] [Google Scholar]
- Guroff G., Dickens G., End D., Londos C. The action of adenosine analogs on PC12 cells. J Neurochem. 1981 Dec;37(6):1431–1439. doi: 10.1111/j.1471-4159.1981.tb06312.x. [DOI] [PubMed] [Google Scholar]
- Harris J. E., Morgenroth V. H., 3rd, Roth R. H., Baldessarini R. J. Regulation of catecholamine synthesis in the rat brain in vitro by cyclic AMP. Nature. 1974 Nov 8;252(5479):156–158. doi: 10.1038/252156a0. [DOI] [PubMed] [Google Scholar]
- Joh T. H., Park D. H., Reis D. J. Direct phosphorylation of brain tyrosine hydroxylase by cyclic AMP-dependent protein kinase: mechanism of enzyme activation. Proc Natl Acad Sci U S A. 1978 Oct;75(10):4744–4748. doi: 10.1073/pnas.75.10.4744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kebabian J. W., Kuo J. F., Greengard P. Determination of relative levels of cyclic AMP in tissues or cells prelabeled with radioactive adenine. Adv Cyclic Nucleotide Res. 1972;2:131–137. [PubMed] [Google Scholar]
- Kenimer J. G., Nirenberg M. Desensitization of adenylate cyclase to prostaglandin E1 or 2-chloroadenosine. Mol Pharmacol. 1981 Nov;20(3):585–591. [PubMed] [Google Scholar]
- Rodbell M. The role of hormone receptors and GTP-regulatory proteins in membrane transduction. Nature. 1980 Mar 6;284(5751):17–22. doi: 10.1038/284017a0. [DOI] [PubMed] [Google Scholar]
- Saez J. M., Morera A. M., Dazord A. Mediators of the effects of ACTH on adrenal cells. Adv Cyclic Nucleotide Res. 1981;14:563–579. [PubMed] [Google Scholar]
- Salomon Y., Londos C., Rodbell M. A highly sensitive adenylate cyclase assay. Anal Biochem. 1974 Apr;58(2):541–548. doi: 10.1016/0003-2697(74)90222-x. [DOI] [PubMed] [Google Scholar]
- Sattin A., Rall T. W. The effect of adenosine and adenine nucleotides on the cyclic adenosine 3', 5'-phosphate content of guinea pig cerebral cortex slices. Mol Pharmacol. 1970 Jan;6(1):13–23. [PubMed] [Google Scholar]
- Soderling T. R., Corbin J. D., Park C. R. Regulation of adenosine 3',5'-monophosphate-dependent protein kinase. II. Hormonal regulation of the adipose tissue enzyme. J Biol Chem. 1973 Mar 10;248(5):1822–1829. [PubMed] [Google Scholar]
- Vulliet P. R., Langan T. A., Weiner N. Tyrosine hydroxylase: a substrate of cyclic AMP-dependent protein kinase. Proc Natl Acad Sci U S A. 1980 Jan;77(1):92–96. doi: 10.1073/pnas.77.1.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J. A. Cyclic AMP formation and thyroid secretion by incubated mouse thyroid lobes. Endocrinology. 1972 Dec;91(6):1411–1417. doi: 10.1210/endo-91-6-1411. [DOI] [PubMed] [Google Scholar]
- Wolff J., Londos C., Cooper D. M. Adenosine receptors and the regulation of adenylate cyclase. Adv Cyclic Nucleotide Res. 1981;14:199–214. [PubMed] [Google Scholar]


