Abstract
The usefulness of magnifying endoscopy with narrow-band imaging (ME-NBI) for the diagnosis of early gastric cancer is well known, however, there are no evaluation criteria. The aim of this study was to devise and evaluate a novel diagnostic algorithm for ME-NBI in depressed early gastric cancer. Between August, 2007 and May, 2011, 90 patients with a total of 110 depressed gastric lesions were enrolled in the study. A diagnostic algorithm was devised based on ME-NBI microvascular findings: microvascular irregularity and abnormal microvascular patterns (fine network, corkscrew and unclassified patterns). The diagnostic efficiency of the algorithm for gastric cancer and histological grade was assessed by measuring its mean sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy. Furthermore, inter- and intra-observer variation were measured. In the differential diagnosis of gastric cancer from non-cancerous lesions, the mean sensitivity, specificity, PPV, NPV, and accuracy of the diagnostic algorithm were 86.7, 48.0, 94.4, 26.7, and 83.2%, respectively. Furthermore, in the differential diagnosis of undifferentiated adenocarcinoma from differentiated adenocarcinoma, the mean sensitivity, specificity, PPV, NPV, and accuracy of the diagnostic algorithm were 61.6, 86.3, 69.0, 84.8, and 79.1%, respectively. For the ME-NBI final diagnosis using this algorithm, the mean κ values for inter- and intra-observer agreement were 0.50 and 0.77, respectively. In conclusion, the diagnostic algorithm based on ME-NBI microvascular findings was convenient and had high diagnostic accuracy, reliability and reproducibility in the differential diagnosis of depressed gastric lesions.
Keywords: magnifying endoscopy, narrow-band imaging, early gastric cancer, microvascular architecture, diagnostic algorithm
Introduction
Gastric cancer is one of the most commonly occurring malignancies worldwide (1,2). The detection of early-stage gastric cancer is associated with improved survival and the potential for curative resection (3,4). Conventional endoscopy with white-light imaging is an important method for the screening and detection of early-stage gastric cancer. Furthermore, recent advanced endoscopic technologies, including chromoendoscopy, high-resolution endoscopy and magnification endoscopy (ME), have greatly improved endoscopic image quality. However, formal histological diagnosis is difficult, even with these endoscopic techniques and a biopsy of the targeted lesion is necessary (5).
The narrow band imaging (NBI) system is a recently developed advanced endoscopic imaging technique based on the light of two specific wavelengths (blue: 390–445 nm and green: 530–550 nm). Magnifying endoscopy combined with narrow band imaging (ME-NBI) enables a clear visualization of the microvascular architecture and the microsurface structure of the superficial section of the mucosa. Certain studies have demonstrated that ME-NBI findings are closely associated with the histological type of gastric mucosal lesion (2,6–9). In particular, irregularities of microstructures have been revealed to be crucial for the endoscopic diagnosis of early gastric cancer (10–14). However, the evaluation of combined irregular microvascular and microstructural patterns is complicated and may lead to a large variation in diagnostic results among observers. Therefore, it is necessary to construct simple evaluation criteria for ME-NBI in the diagnosis of depressed early gastric cancer. Yao et al reported that ME findings based on microvascular architecture showed high diagnostic accuracy for flat early gastric cancer (15). However, no report evaluating the diagnostic accuracy of ME-NBI findings based on microvascular architecture for early gastric cancer is available.
The aim of this study was to devise a novel diagnostic algorithm based on ME-NBI findings relating to microvascular architecture and to evaluate the potential usefulness of this algorithm in the differential diagnosis of depressed gastric lesions.
Patients and methods
Patients
This retrospective study was performed at the Kurume University School of Medicine. Between August 2007 and May 2011, 110 lesions from 90 patients [62 males and 28 females with a median age of 70 years (range, 28–89)] were detected using conventional white light endoscopy as depressed gastric lesions and were included in the study. Of the 110 lesions, 68 were diagnosed as differentiated adenocarcinoma (well- and/or moderately differentiated adenocarcinoma), 32 as undifferentiated adenocarcinoma (poorly differentiated adenocarcinoma and/or signet ring cell carcinoma), and 10 as non-cancerous by the histopathological evaluation of the biopsy specimens. The histological findings were classified according to the Japanese classification (16). The invasion depth of the 100 cancerous lesions was examined by conventional white light endoscopy, endoscopic ultrasonography, and upper gastrointestinal X-ray. Following the evaluation of invasion depth, the lesions diagnosed as early gastric cancer were endoscopically resected with the endoscopic submucosal dissection (ESD) technique according to the Japanese gastric cancer treatment guidelines (17,18), and pathologic evaluation of the resected specimens confirmed complete resection and curative treatment. The study protocol was approved by the ethics committee of Kurume University (Kurume, Japan).
Endoscopy procedures
The patients submitted their written informed consent prior to examination and treatment. A high resolution magnifying upper gastrointestinal endoscope (GIF-Q240Z, GIF-H260Z; Olympus, Tokyo, Japan) and an electronic endoscopy system (EVIS Lucera Spectrum; Olympus) were used in the screening endoscopic examinations. Screening endoscopy procedures were as follows: i) examination of a target lesion with conventional white light endoscopy, ii) examination with chromoendoscopy with an indigo carmine dye solution, and iii) examination with ME-NBI. The endoscopic images of the entire procedure were recorded and stored in a digital filing system for subsequent evaluation.
Assessment of ME-NBI
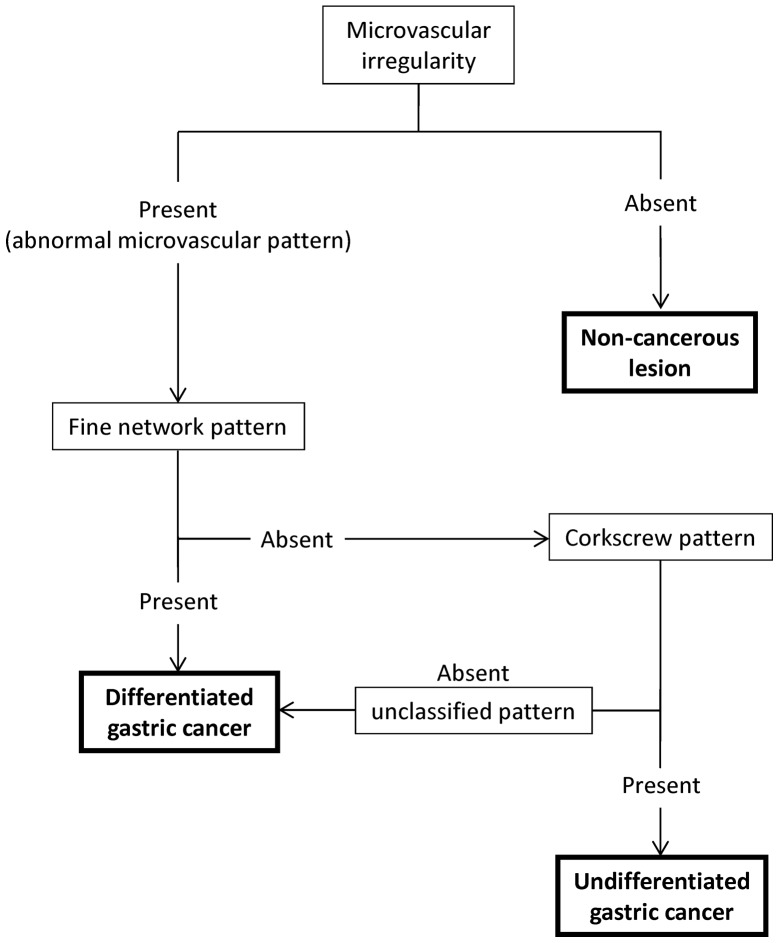
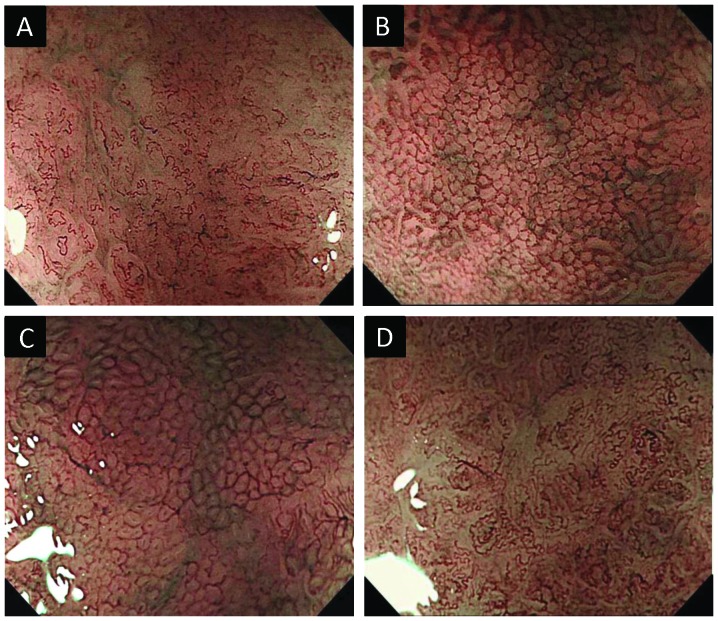
The recorded ME-NBI images were evaluated independently by five experienced endoscopists (H.S., Y.W., K.M., M.M and T.S.) blinded to patient information and histological features. In order to evaluate the depressed gastric lesions included in this study, a diagnostic algorithm based on the microvascular findings of ME-NBI was devised (Fig. 1). The diagnostic algorithm was composed of ME-NBI microvascular findings of the superficial mucosa as follows: i) microvascular irregularity (Fig. 2A and B) and ii) abnormal microvascular patterns (10) [fine network pattern (Fig. 2C), corkscrew pattern (Fig. 2D), or unclassified pattern)]. The definitions of micro-vascular irregularity were denoted in two previous reports [Yao et al (11) and Kaise et al (12)]. A lesion was considered to have microvascular irregularity if the ME-NBI findings were as follows: ‘heterogeneity of shape’ and ‘asymmetries and irregularities of arrangement and distribution’ (11) as well as ‘dilation’, ‘tortuousness’ and ‘abrupt caliber alteration’ as the abnormalities of individual microvessels (12). If the lesion had no microvascular irregularity, it was considered a non-cancerous lesion. If the lesion had microvascular irregularity, we assessed its abnormal microvascular pattern of ME-NBI to predict the histological grade of gastric cancer. If it had a fine network pattern or was regarded as an unclassified pattern, we identified it as a differentiated adenocarcinoma. If it had a corkscrew pattern, we identified it as an undifferentiated adenocarcinoma.
Figure 1.
Diagnostic algorithm based on magnifying endoscopy with narrow-band imaging microvascular findings in the differential diagnosis of depressed-type gastric lesion.
Figure 2.
Typical examples of magnifying endoscopy with narrow-band imaging (ME-NBI) microvascular findings evaluated in diagnostic algorithm. (A) A lesion with microvascular irregularity. (B) A lesion without microvascular irregularity. (C) A lesion with fine network pattern. (D) A lesion with corkscrew pattern.
Statistical analysis
Continuous variables were expressed as the median (range). Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and accuracy were calculated in order to evaluate the diagnostic efficiency of the algorithm for gastric cancer and histological grade based on the microvascular findings of ME-NBI. Cohen’s κ statistics were calculated for intra- and inter-observer agreement. Inter-observer variation was calculated from the results of the first reading, with 10 pairs in total. Intra-observer variation was determined by comparing the first and the second assessments for each endoscopist, with 5 pairs in all. A κ value of ≤0.20 indicated poor agreement; 0.21–0.40, fair agreement; 0.41–0.60, moderate agreement; 0.61–0.80, good agreement; and 0.81–1.00, excellent agreement. Statistical analysis was performed with SPSS software version 20 (SPSS Inc., Chicago, IL, USA).
Results
The patients analyzed included 62 males (69%) and 28 females (31%), with a median age of 70 years (range, 28–89). The median lesion size was 14 mm (range, 3–30). The location was the gastric lower third (L) for 60 lesions (55%), gastric middle third (M) for 33 lesions (30%), and gastric upper third (U) for 17 lesions (15%) (16).
In the predictions of gastric cancer by the five endoscopists, the ME-NBI diagnostic algorithm had a mean sensitivity, specificity, PPV, NPV, and accuracy of 86.7, 48.0, 94.4, 26.7 and 83.2%, respectively (Table I). Furthermore, in the prediction of the histological grade of gastric cancer by the five endoscopists, the ME-NBI diagnostic algorithm had a mean sensitivity, specificity, PPV, NPV, and accuracy of 61.6, 86.3, 69.0, 84.8 and 79.1%, respectively (Table II).
Table I.
Performance factors of the ME-NBI diagnostic algorithm in predicting gastric cancer.
| Factors | Observer A (%) | Observer B (%) | Observer C (%) | Observer D (%) | Observer E (%) | Mean (%) |
|---|---|---|---|---|---|---|
| Sensitivity | 88.0 | 88.5 | 85.0 | 81.0 | 91.0 | 86.7 |
| Specificity | 70.0 | 25.0 | 50.0 | 55.0 | 40.0 | 48.0 |
| PPV | 96.7 | 92.2 | 94.5 | 94.8 | 93.9 | 94.4 |
| NPV | 37.0 | 17.7 | 25.3 | 22.7 | 31.0 | 26.7 |
| Accuracy | 86.4 | 82.7 | 81.8 | 78.7 | 86.4 | 83.2 |
ME-NBI, magnifying endoscopy with narrow-band imaging; PPV, positive predictive value; NPV, negative predictive value. The values of sensitivity, specificity, PPV, NPV, and accuracy, i.e., the first and second reading/2.
Table II.
Performance factors of the ME-NBI diagnostic algorithm in predicting histological grade of gastric cancer.
| Factors | Observer A (%) | Observer B (%) | Observer C (%) | Observer D (%) | Observer E (%) | Mean (%) |
|---|---|---|---|---|---|---|
| Sensitivity | 57.8 | 73.5 | 64.1 | 65.6 | 46.9 | 61.6 |
| Specificity | 91.1 | 68.6 | 87.8 | 87.8 | 96.2 | 86.3 |
| PPV | 74.9 | 49.0 | 68.6 | 69.0 | 83.6 | 69.0 |
| NPV | 84.2 | 86.3 | 85.6 | 86.2 | 81.5 | 84.8 |
| Accuracy | 81.4 | 70.0 | 80.9 | 81.4 | 81.8 | 79.1 |
ME-NBI, magnifying endoscopy with narrow-band imaging; PPV, positive predictive value; NPV, negative predictive value. The values of sensitivity, specificity, PPV, NPV, and accuracy, i.e, first and second reading/2.
For evaluations of the ME-NBI features and the final diagnosis, the mean κ values for inter-observer agreement were as follows: microvascular irregularity, 0.49 (range, 0.32–0.74); fine network pattern, 0.55 (range, 0.38–0.65); corkscrew pattern, 0.60 (range, 0.35–0.83); and the ME-NBI final diagnosis, 0.50 (range, 0.32–0.67). The mean κ values for intra-observer agreement were as follows: microvascular irregularity, 0.61 (range, 0.39–0.84); fine network pattern, 0.75 (range, 0.64–0.84); corkscrew pattern, 0.78 (range, 0.64–0.86); and ME-NBI final diagnosis, 0.77 (range, 0.66–0.84) (Table III).
Table III.
Inter- and intra-observer agreements for evaluations of ME-NBI features and ME-NBI final diagnosis.
| Variables | Inter-observer κ value mean (range) | Intra-observer κ value mean (range) |
|---|---|---|
| ME-NBI features | ||
| Microvascular irregularity | 0.49 (0.32–0.74) | 0.61 (0.39–0.84) |
| Fine network pattern | 0.55 (0.38–0.65) | 0.75 (0.64–0.84) |
| Corkscrew pattern | 0.60 (0.35–0.83) | 0.78 (0.64–0.86) |
| ME-NBI final diagnosis | 0.50 (0.32–0.67) | 0.77 (0.66–0.84) |
ME-NBI, magnifying endoscopy with narrow-band imaging; κ, Cohen’s κ coefficient.
Discussion
Early gastric cancers can be classified macroscopically into elevated and depressed types. It is reported that the frequency of the depressed type (∼60–70%) was higher than that of the elevated type in early gastric cancer (19–21). Therefore, it is important to enhance the diagnostic accuracy of early depressed gastric cancers. ME-NBI yields clear visualization of the microvasculature and microsurface structure of the gastrointestinal mucosa. In a previous study it was demonstrated that the diagnostic accuracy was significantly higher with ME-NBI than with conventional endoscopy with white light imaging for the differential diagnosis of depressed gastric lesions (13). Thus, we devised a diagnostic algorithm based on ME-NBI findings to evaluate depressed gastric lesions. ME-NBI findings, such as irregular microvascular and microstructural patterns, are closely associated with gastric cancer. As the microsurface structure of depressed gastric lesions is frequently unclear, objective evaluation is difficult by ME-NBI. Kaise et al reported that disappearance of the microsurface structure was observed frequently in depressed gastric cancers (12). As a consequence, this algorithm was constructed based on ME-NBI findings relating to microvasculature alone.
In this study, ME-NBI diagnosis with microvascular irregularities demonstrated a high sensitivity (86.7%) and accuracy (83.2%) for predicting gastric cancer. Previous studies revealed that ME-NBI exhibited high sensitivity (69–93%), specificity (85–95%) and accuracy (79–90%) in the differential diagnosis of depressed gastric cancers from non-cancerous lesions (12–14). Those studies evaluated the ME-NBI features of combined irregular microvascular and microstructural patterns. By contrast, we evaluated only ME-NBI microvascular findings and obtained good sensitivity and accuracy equivalent to previous studies. However, the specificity of ME-NBI diagnosis based on microvascular irregularity was unsatisfactory. This result suggested that our definition of microvascular irregularity included the non-specific findings, thus, the highly specific findings for predicting gastric cancer should be investigated. The ME-NBI diagnosis based on abnormal microvascular patterns (fine network pattern, corkscrew pattern, or unclassified pattern) showed high specificity (86.3%) and accuracy (79.1%) in distinguishing undifferentiated from differentiated adenocarcinoma. Nakayoshi et al reported that differentiated adenocarcinoma frequently showed a fine network pattern, and differentiated adenocarcinoma frequently showed a corkscrew pattern on ME-NBI (10). In depressed early gastric cancers, this study showed that 105 of 109 differentiated lesions (96.3%) had a fine network pattern or unclassified pattern, and 48 of 56 undifferentiated lesions (85.7%) had a corkscrew pattern. Therefore, the ME-NBI diagnosis based on abnormal microvascular patterns was useful for predicting the histological grade of depressed gastric cancers.
We also investigated inter- and intra-observer agreements in the ME-NBI diagnosis with microvascular findings. To the best of our knowledge, no complete study on the reliability and the reproducibility of an ME-NBI diagnosis with micro-vascular findings for depressed gastric lesions is currently available. In the evaluation of depressed gastric lesions by the diagnostic algorithm, inter-observer reproducibility of the ME-NBI microvascular findings (κ value 0.49–0.60) and the ME-NBI final diagnosis (κ value 0.50) showed fair agreement. Furthermore, the intra-observer reproducibility of the ME-NBI microvascular findings (κ value 0.61–0.78) and the ME-NBI final diagnosis (κ value 0.77) exhibited a moderate agreement. Kaise et al reported that the κ value of inter-observer concordance for an ME-NBI diagnosis was 0.47 in the evaluation of depressed gastric lesions based on the ME-NBI features of combined irregular microvascular and microstructural patterns (12). Consequently, our results are satisfactory and representative of the observer agreement to be expected in clinical practice when ME-NBI findings are assessed with this diagnostic algorithm.
In conclusion, the diagnostic algorithm utilized in this study, based on ME-NBI microvascular findings was convenient and had high diagnostic accuracy, reliability and reproducibility in the differential diagnosis of depressed gastric lesions. Based on these results, a prospective study is essential to evaluate the benefit of this diagnostic algorithm.
References
- 1.Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108. doi: 10.3322/canjclin.55.2.74. [DOI] [PubMed] [Google Scholar]
- 2.Catalano V, Labianca R, Beretta GD, Gatta G, de Braud F, Van Cutsem E. Gastric cancer. Crit Rev Oncol Hematol. 2009;71:127–164. doi: 10.1016/j.critrevonc.2009.01.004. [DOI] [PubMed] [Google Scholar]
- 3.Oliveira FJ, Ferrao H, Furtado E, Batista H, Conceicao L. Early gastric cancer: report of 58 cases. Gastric Cancer. 1998;1:51–56. doi: 10.1007/s101200050054. [DOI] [PubMed] [Google Scholar]
- 4.Isomoto H, Shikuwa S, Yamaguchi N, et al. Endoscopic submucosal dissection for early gastric cancer: a large-scale feasibility study. Gut. 2009;58:331–336. doi: 10.1136/gut.2008.165381. [DOI] [PubMed] [Google Scholar]
- 5.Tajiri H, Ohtsu A, Boku N, et al. Routine endoscopy using electronic endoscopes for gastric cancer diagnosis: retrospective study of inconsistencies between endoscopic and biopsy diagnoses. Cancer Detect Prev. 2001;25:166–173. [PubMed] [Google Scholar]
- 6.Capelle LG, Haringsma J, de Vries AC, et al. Narrow band imaging for the detection of gastric intestinal metaplasia and dysplasia during surveillance endoscopy. Dig Dis Sci. 2010;55:3442–3448. doi: 10.1007/s10620-010-1189-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tahara T, Shibata T, Nakamura M, et al. Gastric mucosal pattern by using magnifying narrow-band imaging endoscopy clearly distinguishes histological and serological severity of chronic gastritis. Gastrointest Endosc. 2009;70:246–253. doi: 10.1016/j.gie.2008.11.046. [DOI] [PubMed] [Google Scholar]
- 8.Tsuji Y, Ohata K, Sekiguchi M, et al. Magnifying endoscopy with narrow-band imaging helps determine the management of gastric adenomas. Gastric Cancer. 2012;15:414–418. doi: 10.1007/s10120-011-0133-2. [DOI] [PubMed] [Google Scholar]
- 9.Hirata I, Nakagawa Y, Ohkubo M, Yahagi N, Yao K. Usefulness of magnifying narrow-band imaging endoscopy for the diagnosis of gastric and colorectal lesions. Digestion. 2012;85:74–79. doi: 10.1159/000334642. [DOI] [PubMed] [Google Scholar]
- 10.Nakayoshi T, Tajiri H, Matsuda K, Kaise M, Ikegami M, Sasaki H. Magnifying endoscopy combined with narrow band imaging system for early gastric cancer: correlation of vascular pattern with histopathology (including video) Endoscopy. 2004;36:1080–1084. doi: 10.1055/s-2004-825961. [DOI] [PubMed] [Google Scholar]
- 11.Yao K, Anagnostopoulos GK, Ragunath K. Magnifying endoscopy for diagnosing and delineating early gastric cancer. Endoscopy. 2009;41:462–467. doi: 10.1055/s-0029-1214594. [DOI] [PubMed] [Google Scholar]
- 12.Kaise M, Kato M, Urashima M, et al. Magnifying endoscopy combined with narrow-band imaging for differential diagnosis of superficial depressed gastric lesions. Endoscopy. 2009;41:310–315. doi: 10.1055/s-0028-1119639. [DOI] [PubMed] [Google Scholar]
- 13.Ezoe Y, Muto M, Horimatsu T, et al. Magnifying narrow-band imaging versus magnifying white-light imaging for the differential diagnosis of gastric small depressive lesions: a prospective study. Gastrointest Endosc. 2010;71:477–484. doi: 10.1016/j.gie.2009.10.036. [DOI] [PubMed] [Google Scholar]
- 14.Kato M, Kaise M, Yonezawa J, et al. Magnifying endoscopy with narrow-band imaging achieves superior accuracy in the differential diagnosis of superficial gastric lesions identified with white-light endoscopy: a prospective study. Gastrointest Endosc. 2010;72:523–529. doi: 10.1016/j.gie.2010.04.041. [DOI] [PubMed] [Google Scholar]
- 15.Yao K, Iwashita A, Tanabe H, et al. Novel zoom endoscopy technique for diagnosis of small flat gastric cancer: a prospective, blind study. Clin Gastroenterol Hepatol. 2007;5:869–878. doi: 10.1016/j.cgh.2007.02.034. [DOI] [PubMed] [Google Scholar]
- 16.Japanese Gastric Cancer Association Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101–112. doi: 10.1007/s10120-011-0041-5. [DOI] [PubMed] [Google Scholar]
- 17.Ono H, Kondo H, Gotoda T, et al. Endoscopic mucosal resection for treatment of early gastric cancer. Gut. 2001;48:225–229. doi: 10.1136/gut.48.2.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Japanese Gastric Cancer Association Japanese gastric cancer treatment guidelines 2010 (ver. 3) Gastric Cancer. 2011;14:113–123. doi: 10.1007/s10120-011-0042-4. [DOI] [PubMed] [Google Scholar]
- 19.Xuan ZX, Ueyama T, Yao T, Tsuneyoshi M. Time trends of early gastric carcinoma. A clinicopathologic analysis of 2846 cases. Cancer. 1993;72:2889–2894. doi: 10.1002/1097-0142(19931115)72:10<2889::aid-cncr2820721006>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 20.Hirota T, Itabashi M, Daibo M, et al. Chronological changes in the morphological features of early gastric cancer, especially recent changes in macroscopic findings. Jpn J Clin Oncol. 1984;14:181–199. [PubMed] [Google Scholar]
- 21.Hyung WJ, Cheong JH, Kim J, Chen J, Choi SH, Noh SH. Application of minimally invasive treatment for early gastric cancer. J Surg Oncol. 2004;85:181–185. doi: 10.1002/jso.20018. [DOI] [PubMed] [Google Scholar]