Abstract
Objective:
To find out whether DTPA-DG complex can enhance clearance of intracellular free iron.
Materials and Methods:
Diethylenetriaminepentaacetic acid-D-deoxy-glucosamine (DTPA-DG) was synthesized and examined for its activity as a cell-permeable iron chelator in human hepatocellular carcinoma (HEPG2) cell line exposed to high concentration of iron sulfate and compared with deferoxamine (DFO), a prototype iron chelator. The effect of DTPA-DG on cell viability was monitored using the 3-(4,5-dimethythiazol-2-yl)-2,5-diphenyl tetrazolium bromide MTT assay as well.
Results:
There was a significant increase of iron level after iron overload induction in HEPG2 cell culture. DTPA-DG presented a remarkable capacity to iron burden reducing with estimated 50% inhibitory concentration value of 65.77 nM. In fact, glycosyl moiety was gained access of DTPA to intracellular iron deposits through glucose transporter systems.
Conclusion:
DTPA-DG, more potent than DFO to sequester deposits of free iron with no profound toxic effect. The results suggest the potential of DTPA-DG in chelating iron and permitting its excretion from primary organ storage.
Keywords: Cell-permeable, glycosyl moiety, human hepatocellular carcinoma, iron chelation, iron overload
INTRODUCTION
Although, iron is a vital element for the normal functioning of body organs; excessive amounts in the body are very toxic, especially for liver and heart. Iron-overload can result from the genetic disorder named hereditary hemochromatosis (HH) or secondary causes.[1]
HH is an autosomal recessive disorder that affects one in two hundred in Caucasian people and higher prevalence in Irish population.[2,3] High Fe gene mutation creates positive balance of iron. Secondary iron overload is an evitable response to repeated transfusions as in thalassemia and sickle cell anemia or overdose of iron intake.[4,5]
Iron chelators are mainstays of treatment for secondary hemochromatosis. Since 1970 until now,[6] the gold standard iron chelator is deferoxamine (DFO) that improve life expectancy of patients; it must be administered for long-term by parenteral infusion for 5-7 days/week. Regardless cumbersome administration, high cost and side-effects leading to poor patient compliance.[7,8,9] More recently literature has emerged to discover more safe, efficient and affordable alternative chelator to treat patients with iron overload.[10]
Diethylenetriaminepentaacetic acid (DTPA) is a member of a family of polyaminocarboxylic acid that is similar to ethylenediaminetetraacetic acid (EDTA). This molecule widely used as a chelating agent to treat internal contamination of multivalent cations like: plutonium and americium [Figure 1]. The penta-anion DTPA-5 is potentially an octadentate ligand, usually can form less than eight coordination bonds with divalent metal cations. Currently, DTPA has been approved by the Food and Drug Administration for chelating of transuranic elements including: Plutonium, americium and curium. Furthermore, DTPA can form complex with Gd3+, 111In and 99mTc in order to label chemical compounds to be used in nuclear medicine.[11,12,13]
Figure 1.
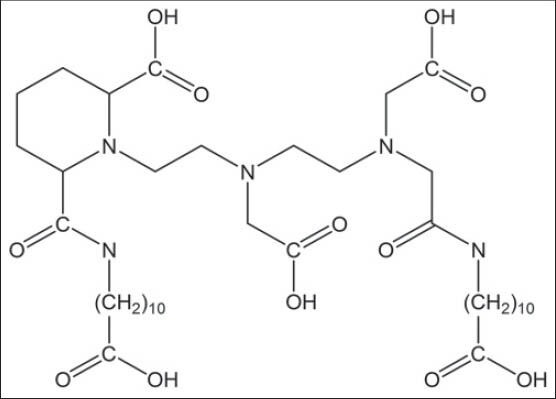
Structural formula of Puchel, lipophilic derivative of diethylenetriaminepentaacetic acid used for plutonium decorporation
The main drawback of this compound is impermeability to the cell membrane so that is classified as an extracellular fluid complexing agent.[14,15] To overcome this handicap, several structural derivatives have been synthesized and examined. For example, Puchel is a lipophilic derivative of DTPA is used in removing intracellular plutonium[16] or DTPA-conjugate compounds (DTPA-folate [Figure 2], DTPA-monoclonal antibody) that increase cellular uptake of chelator through receptor mediated endocytosis.[17,18]
Figure 2.
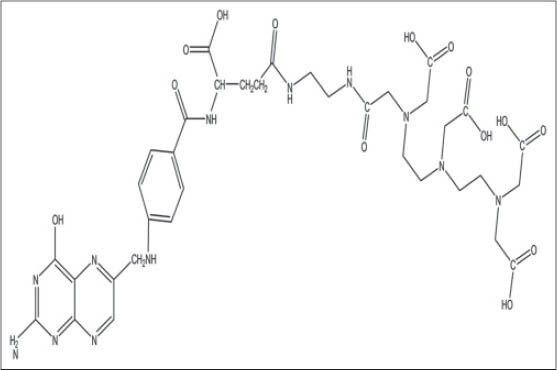
Diethylenetriaminepentaacetic acid-folate structure, conjugated form of DTPA for tumor targeting-delivery
Recently, we reported that glucosamine-DTPA conjugate diethylenetriaminepentaacetic acid D-deoxy-glucosamine (DTPA-DG) [Figure 3] could be penetrated to inside the cancer cells easily.[19] With this capability, cancerous cells are characterized by a high-contrast in magnetic resonance imaging technique.[19]
Figure 3.
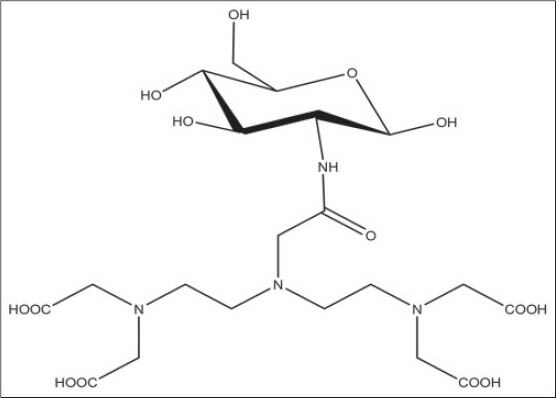
Chemical structure of diethylenetriaminepentaacetic acid-D-deoxy-glucosamine
The aim of this study is to report that DTPA-DG complex can enhance clearance of intracellular free iron.
MATERIALS AND METHODS
Iron chelators
The iron chelator DTPA-DG was synthesized in large scale using recently published method.[20] Various concentrations of DTPA-DG (20, 40, 60, 80,100 mM) were used in this investigation to evaluate the iron chelating capacity. DFO (DFO; Novartis-Pharma) at 10 and 20 μM concentration was used in parallel cultures for comparative purposes. All reagents in powder form were used without further purification.
Cell culture and induction of iron overload
The human hepatocellular carcinoma (HEPG2) obtained from American type culture collection (ATCC no: HB-8065). HEPG2 cells were grown in high glucose Dulbecco's modification of Eagle's medium; GIBCO® supplemented with 10% heat-inactivated fetal bovine serum, 1% glutamine, 10% penicillin/streptomycin and NaHCO3 (mg). The hepatocytes were cultured in 75 cm2 sterile polystyrene flasks (Seromed/Biochrom, Germany) in an incubator with saturating humidified atmosphere of 5% CO2 in the air at 37°C. For cell harvesting 0.05% trypsin solution, phosphate-buffered saline (PBS) is used. The viability and count of cells were determined by 0.4% trypan blue exclusion using hemocytometer. Iron overload was induced by adding 1 mM ferrous sulfate (FeSO4) solution to the media for 72 h. Cells were harvested and washed in PBS. To ensure removal of non-internalized iron that present in the media, cells were washed once in PBS, then in washing buffer including 500 μM DFO (DFO; Desferal; Novartis) and 1.5 mM EDTA in PBS and in PBS buffer again.[21]
Measurement of cell viability by MTT assay
This method is one of the more practical ways to determine cytotoxicity of test agent. The principle of MTT assay is that yellow tetrazolium salt converted to blue formazan crystals by mitochondrial dehydrogenases of living cells.[22] Cells (3 × 104) were placed in a 96-well plate (Seromed/Biochrom, Germany) and incubated for routinely 24 h at 37°C prior to use. After 24 h, they were treated with various concentrations of DTPA-DG for 24 h. Then remained media into wells were removed carefully and 100 μL of 0.5 mg/ml MTT reagent was added to each well. The plate covered with aluminum foil and incubated for 2 h until formazan crystal is formed. Finally, the formazan product in each well was dissolved in 100 μl of dimethyl sulfoxide completely.[22] The optical density of the 96-well plate was measured at 570 nm by microplate reader and the results were compared with untreated cell group.
Measurement of cellular iron content by atomic absorption spectrometry
The total amount of free iron in the presence or absence of chelator was determined by Perkin Elmer flame atomic absorption spectrophotometer (Varian Spectra AA-220Fs / Australia). In practice, iron loaded hepatocyte cultures incubated for 24-48 h in the presence of 20, 40, 60, 80, 100 nM DTPA-DG or 10, 20 μM DFO, then washed with isotonic buffer and subsequently lysed with glass beads, the lysate was centrifuged at 7000 rpm for 5 min. Aliquots were used for determination of free iron content.[13] The optical densities were recorded and data processed with Perkin atomic absorption spectrophotometer. Finally, iron chelating capacity of DTPA-DG was compared with the effect of DFO at several concentrations.
Statistical analysis
Data were expressed as mean ± standard deviation of three independent measurements (n = 3). Statistical analysis of in-vitro data was performed using the SPSS (version 19, SPSS Inc., Chicago, Illinois, USA) software package and Prism (version 5, GraphPad Software Inc., San Diego, California, USA) software for determination of 50% inhibitory concentration value (IC50). Measurement data were analyzed by Student's t-test and one-way ANOVA followed by Tukey's Post-hoc test. A value of P < 0.05 was considered as statistically significant.
RESULTS
Cell culture and induction of iron overload
The experimental model obtained by the addition of 1 mM FeSO4 to the culture media for 72 h. This procedure is in agreement with Perak's (2006) findings, which showed this iron amount can be tolerable for HEPG2 cells in short-term of cultivation.[21]
Following 72 h iron exposure duration, total intracellular iron was increased from 16 μg Fe/g of wet cell pellet to 98 μg Fe/g. In fact, there is increased 6-fold in iron level [Figure 4].
Figure 4.

Cellular iron level in control and iron-overload human hepatocellular carcinoma cell model as measurement by atomic absorption spectrometry. Measurements were performed with sample triplicates (n = 3)
Measurement of cell viability by MTT assay
MTT assay of cytotoxicity associated with various concentrations of DTPA-DG after 48 h demonstrated a negligible (P > 0.05) decrease of cell viability observed by MTT test [Figure 5].
Figure 5.
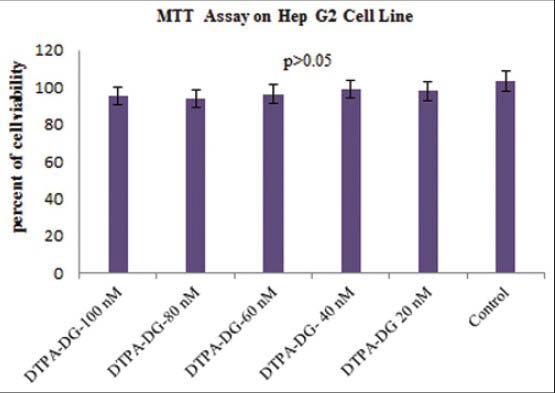
MTT assay of cytotoxicity associated with various concentrations of diethylenetriaminepentaacetic acid-D-deoxy-glucosamine after 48 h. Data are presented as the percentage of cell viability relative to the untreated normal human hepatocellular carcinoma (mean ± standard deviation, n = 3). As a result, a negligible (P > 0.05) decrease of cell viability observed by MTT test
Measurement of cellular iron content by atomic absorption spectrometry
In order to demonstrate the ability of chelator to decrease intracellular free iron content, the amount of free iron in the presence of several concentrations of DTPA-DG was quantified by using atomic absorption spectrophotometer. Exposure of iron overload HEPG2 cell model for about 24 h to different concentrations of DTPA-DG (20-100 nM) dramatically decreases free iron level [Figure 6]. The lowest cellular iron content was observed with the concentration 100 nM that approximately reached as much as control cell group (P < 0.05). For concentration higher than 40 nM DTPA-DG significant differences (P < 0.05, P = 0.022) versus untreated cell group were observed; can prove successful DTPA-DG's function as intracellular iron chelators in the low concentration range. The comparison of chelator efficacy with DFO exhibited that the highest rate of decrease in the iron content from HEPG2 cells occurred during 24 h treatment with DTPA-DG. The rate of decrease was significantly (P < 0.05, P = 0.011) higher in cell groups in which 60 nM DTPA-DG were injected than those of 20 μM DFO [Figure 7].
Figure 6.

A dose-dependent decrease of free iron content after 24 h incubation of human hepatocellular carcinoma cells at 37°C which treated with 20-100 nM of diethylenetriaminepentaacetic acid-D-deoxy-glucosamine. The decrease was significant (P < 0.05) at concentration equal to or higher than 40 nM of DTPA-DG in comparison with untreated cells (n = 3). Results are the mean ± standard deviation of three separate experiments
Figure 7.
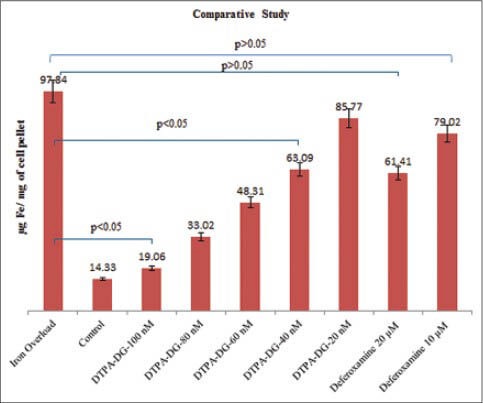
The comparison of two chelators diethylenetriaminepentaacetic acid-D-deoxy-glucosamine and deferoxamine efficacy in similar conditions after 24 h indicated statistically (P < 0.05) a higher reducing of cellular iron content with DTPA-DG than with DFO at much lower doses. Results are the mean ± standard deviation of three separate experiments (n = 3)
As a result of copper intracellular assay experiments, reducing in free iron level was significantly correlated (P < 0.05, P = 0.001, r2 = 0.994) with iron chelator concentration [Figure 8]. DTPA-DG was the most effective at decreasing free iron content in HEPG2 cell line with an IC50 value near 65.77 nM.
Figure 8.
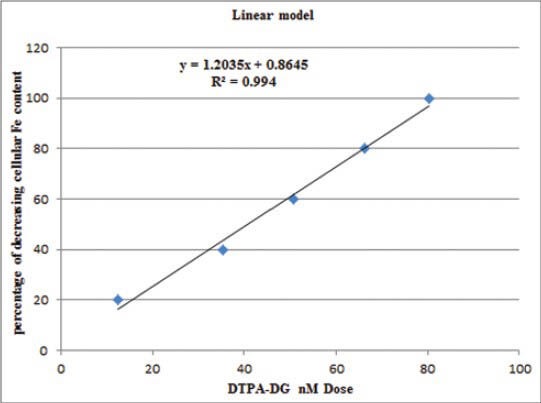
A significant (P < 0.05, r2 = 0.994) correlation with iron chelator dose and its success in reducing cellular iron content is shown in this figure. Diethylenetriaminepentaacetic acid D-deoxy-glucosamine at concentration of 65.77 nM has been able to reduce 50% of iron deposits
DISCUSSION
In this study, iron-clearing efficiency and safety profile of DTPA-DG was assessed using iron overload HEPG2 cell culture model. This model was chosen because the liver is primary iron storage site and HEPG2 are well-established in-vitro model for human hepatocytes.
Recently, we have reported that linking of glucosamine through its 2-amino group to DTPA increased remaining of DTPA-DG into cells, which is mechanistically when DTPA-DG enters the cell after 6-Phosphorilation by the hexokinase enzyme, is not metabolized further; then DTPA more efficiently reduces intracellular iron in the iron loaded HEPG2 cells.[19,23,24]
DTPA-DG was found to be much more efficient at iron decorporation than DFO in HEPG2 cell line; scilicet, the chelator has shown a significant effect at lower doses. Because glucose receptor mediated endocytosis pathway is led to high cell uptake of DTPA without cell destruction.[13]
Surveys such as studies conducted by Schibli et al. have shown that when glucose is linked to a molecule through C2 position, its affinity for cell surface receptor doesn’t alter essentially.[25] So, DTPA-DG can be transported through glucose channels with similar manner as unconjugated glucose.[25,26]
Another experiment was MTT colorimetric assay that is a suitable technique for determining cell viability after drug treatment. Our finding about HEPG2 cell cultures were subjected to the MTT assay after 48 h indicated no significant detrimental effect of DTPA-DG on cell viability at concentration 20-100 nM. In conclusion, DTPA-DG is a negligible toxicity profile.
Iron chelator activity was estimated by the IC50. Actually, the concentration of compound that is effective to reduce up to 50% of iron deposits was calculated by Prism software.
In 1963 published a paper by Walsh et al., in which they claimed that long-term use of DTPA could be a possible method for eliminating excessive iron in the urine.[27] The current study defines that glucose-targeted delivery of DTPA will be more successful in minimizing accumulation of iron than DTPA by itself. While future study with more focus on pharmacodynamics and pharmacokinetic of DTPA-DG will be required.
CONCLUSION
Based on a pressing need to design a new iron-chelating agent, this project was undertaken to evaluate the efficacy of a new DTPA-DG iron chelator in iron overload HEPG2 cell model. The stark decrease of iron deposits showed the high efficiency of DTPA-DG in iron decorporation from the main target of iron overload. We found that the glycosyl moiety facilitates entrance of DTPA into cells without any iron interaction capabilities. It was also shown DTPA-DG is a non-toxic compound that can be rescue cells from deleterious effects of free iron.
Finally, this new cell-permeable iron chelator can be decreased tissue iron deposits more effectively than the known chelator DFO. These promising results encourage us to continue more experiments on DTPA-DG complex for treatment of iron overload disease and animal experiments that will be published in near future.
ACKNOWLEDGMENT
This manuscript was extracted from a Pharm.D. Thesis of Ms. Mona Mosayebnia and was supported by Tehran University of Medical Sciences. Authors are thankful to Pasteur Institute of Iran for providing us cell culture facilities.
Footnotes
Source of Support: Tehran University of Medical Sciences
Conflict of Interest: None declared.
REFERENCES
- 1.Felitti VJ. Hemochromatosis: A common, rarely diagnosed disease. Perm J. 1999;3:10–22. [Google Scholar]
- 2.Merryweather-Clarke AT, Pointon JJ, Jouanolle AM, Rochette J, Robson KJ. Geography of HFE C282Y and H63D mutations. Genet Test. 2000;4:183–98. doi: 10.1089/10906570050114902. [DOI] [PubMed] [Google Scholar]
- 3.Ryan E, O’keane C, Crowe J. Hemochromatosis in Ireland and HFE. Blood Cells Mol Dis. 1998;24:428–32. doi: 10.1006/bcmd.1998.0211. [DOI] [PubMed] [Google Scholar]
- 4.Brissot P. Diagnosis and current treatments for primary iron overload. Am J Hematol. 2007;82:1140–1. doi: 10.1002/ajh.21071. [DOI] [PubMed] [Google Scholar]
- 5.Payne KA, Rofail D, Baladi JF, Viala M, Abetz L, Desrosiers MP, et al. Iron chelation therapy: Clinical effectiveness, economic burden and quality of life in patients with iron overload. Adv Ther. 2008;25:725–42. doi: 10.1007/s12325-008-0085-z. [DOI] [PubMed] [Google Scholar]
- 6.Hemochromatosis: Monitoring and treatment. National Center on Birth Defects and Developmental Disabilities (NCBDDD), 2007-11-01. [Retrieved on 2008 Mar 29]. Available from: http://www.cdc.gov/ncbddd/hemochromatosis/training/treatment/monitoring treatment.htm .
- 7.Olivieri NF, Brittenham GM. Iron-chelating therapy and the treatment of thalassemia. Blood. 1997;89:739–61. [PubMed] [Google Scholar]
- 8.Cao A, Galanello R, Rosatelli MC, Argiolu F, De Virgiliis S. Clinical experience of management of thalassemia: The Sardinian experience. Semin Hematol. 1996;33:66–75. [PubMed] [Google Scholar]
- 9.Olivieri NF, Buncic JR, Chew E, Gallant T, Harrison RV, Keenan N, et al. Visual and auditory neurotoxicity in patients receiving subcutaneous deferoxamine infusions. N Engl J Med. 1986;314:869–73. doi: 10.1056/NEJM198604033141402. [DOI] [PubMed] [Google Scholar]
- 10.Wong C, Richardson DR. Beta-thalassaemia: Emergence of new and improved iron chelators for treatment. Int J Biochem Cell Biol. 2003;35:1144–9. doi: 10.1016/s1357-2725(03)00046-3. [DOI] [PubMed] [Google Scholar]
- 11.Ou MH, Chen YM, Chang YH, Lu WK, Liu GC, Wang YM. Synthesis, complexation and water exchange properties of Gd (III)-TTDA-mono and bis (amide) derivatives and their binding affinity to human serum albumin. Dalton Trans. 2007;112:2749–59. doi: 10.1039/b703211k. [DOI] [PubMed] [Google Scholar]
- 12.Taylor DM, Hodgson SA, Stradling N. Treatment of human contamination with plutonium and americium: Would orally administered Ca- or Zn-DTPA be effective? Radiat Prot Dosimetry. 2007;127:469–71. doi: 10.1093/rpd/ncm299. [DOI] [PubMed] [Google Scholar]
- 13.Zakeri S, Afzal E, Yaghmaei P, Mirzaei M, Mehravi B, Aghasadeghi MR, et al. Nano-scaled diethylenetriamine pent acetic acid (N-DTPA): Novel anti-Wilson's disease cell model. Am J Biomed Sci. 2012;4:204–19. [Google Scholar]
- 14.Haririan I, Alavidjeh MS, Khorramizadeh MR, Ardestani MS, Ghane ZZ, Namazi H. Anionic linear-globular dendrimer-cis-platinum (II) conjugates promote cytotoxicity in vitro against different cancer cell lines. Int J Nanomedicine. 2010;5:63–75. doi: 10.2147/ijn.s8595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kainthan RK, Hester SR, Levin E, Devine DV, Brooks DE. In vitro biological evaluation of high molecular weight hyperbranched polyglycerols. Biomaterials. 2007;28:4581–90. doi: 10.1016/j.biomaterials.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 16.Volf V, Peter E. DTPA is superior to its lipophilic derivative Puchel in removing 234Th, 238,239Pu and 241Am from Chinese hamsters and rats. Health Phys. 1984;46:422–6. [PubMed] [Google Scholar]
- 17.Mathias CJ, Hubers D, Low PS, Green MA. Synthesis of (99m) Tc DTPA-folate and its evaluation as a folate-receptor-targeted radiopharmaceutical. Bioconjug Chem. 2000;11:253–7. doi: 10.1021/bc9901447. [DOI] [PubMed] [Google Scholar]
- 18.Brechbiel MW, Gansow OA, Atcher RW, Schlom J, Esteban J, Simpson D, et al. Synthesis of 1-(p-isothiocyanatobenzyl) derivatives of DTPA and EDTA. Antibody labeling and tumor-imaging studies. Inorg Chem. 1986;25:2772–81. [Google Scholar]
- 19.Amanlou M, Siadat SD, Ebrahimi SE, Alavi A, Aghasadeghi MR, Ardestani MS, et al. Gd (3+)-DTPA-DG: Novel nanosized dual anticancer and molecular imaging agent. Int J Nanomedicine. 2011;6:747–63. doi: 10.2147/IJN.S17648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shafiee-Ardestani M, Jabbari Arabzadeh A, Heidari Z, Hosseinzadeh A, Ebrahimi H, Hashemi E, et al. Novel and facile methods for the synthesis of DTPA-mono-amid: A new completely revised strategy in radiopharmaceutical chemistry. J Radioanal Nucl Chem. 2010;283:447–55. [Google Scholar]
- 21.Petrak J, Myslivcova D, Man P, Cmejla R, Cmejlova J, Vyoral D. Proteomic analysis of iron overload in human hepatoma cells. Am J Physiol Gastrointest Liver Physiol. 2006;290:G1059–66. doi: 10.1152/ajpgi.00469.2005. [DOI] [PubMed] [Google Scholar]
- 22.Cree LA, et al. 2nd ed. New York: Humana Press and Business Media; 2011. Cancer Cell Culture: Methods and Protocols; pp. 237–44. [Google Scholar]
- 23.Cheze-Le Rest C, Metges JP, Teyton P, Jestin-Le Tallec V, Lozac’h P, Volant A, et al. Prognostic value of initial fluorodeoxyglucose-PET in esophageal cancer: A prospective study. Nucl Med Commun. 2008;29:628–35. doi: 10.1097/MNM.0b013e3282f81423. [DOI] [PubMed] [Google Scholar]
- 24.Parente P, Coli A, Massi G, Mangoni A, Fabrizi MM, Bigotti G. Immunohistochemical expression of the glucose transporters Glut-1 and Glut-3 in human malignant melanomas and benign melanocytic lesions. J Exp Clin Cancer Res. 2008;27:34. doi: 10.1186/1756-9966-27-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schibli R, Dumas C, Petrig J, Spadola L, Scapozza L, Garcia-Garayoa E, et al. Synthesis and in vitro characterization of organometallic rhenium and technetium glucose complexes against Glut 1 and hexokinase. Bioconjug Chem. 2005;16:105–12. doi: 10.1021/bc049774l. [DOI] [PubMed] [Google Scholar]
- 26.Marshall S, Nadeau O, Yamasaki K. Glucosamine-induced activation of glycogen biosynthesis in isolated adipocytes. Evidence for a rapid allosteric control mechanism within the hexosamine biosynthesis pathway. J Biol Chem. 2005;280:11018–24. doi: 10.1074/jbc.M413499200. [DOI] [PubMed] [Google Scholar]
- 27.Walsh RJ, Perkins KW, Blackburn CR, Sanford R, Cantrill S. The use of DTPA in the diagnosis and management of idiopathic haemochromatosis. Australas Ann Med. 1963;12:192–6. doi: 10.1111/imj.1963.12.3.192. [DOI] [PubMed] [Google Scholar]