Abstract
Thiopeptides, or thiazolyl peptides, are a relatively new family of antibiotics that already counts with more than one hundred different entities. Although they are mainly isolated from soil bacteria, during the last decade, new members have been isolated from marine samples. Far from being limited to their innate antibacterial activity, thiopeptides have been found to possess a wide range of biological properties, including anticancer, antiplasmodial, immunosuppressive, etc. In spite of their ribosomal origin, these highly posttranslationally processed peptides have posed a fascinating synthetic challenge, prompting the development of various methodologies and strategies. Regardless of their limited solubility, intensive investigations are bringing thiopeptide derivatives closer to the clinic, where they are likely to show their veritable therapeutic potential.
Keywords: thiopeptides, antibiotics, natural products, peptides, heterocycles, thiazole, oxazole, dehydroamino acids, pyridine
1. Introduction
Since the discovery of the first antibiotics and their golden age in mid 20th century, there has been a dramatic change in the way we face the development of new antimicrobials [1,2]. At first, it seemed that the many classes of naturally occurring antibiotics could be sufficient to fight against bacterial infections, whereas, for the last few decades it was thought that semi-synthetic modifications of those natural products would be enough to overcome pathogen resistance. However, we are now facing a new age, where the discovery of novel scaffolds and new modes of action is required to fight against the emergence of resistances and cross-resistances that make previously treatable infections a new threat.
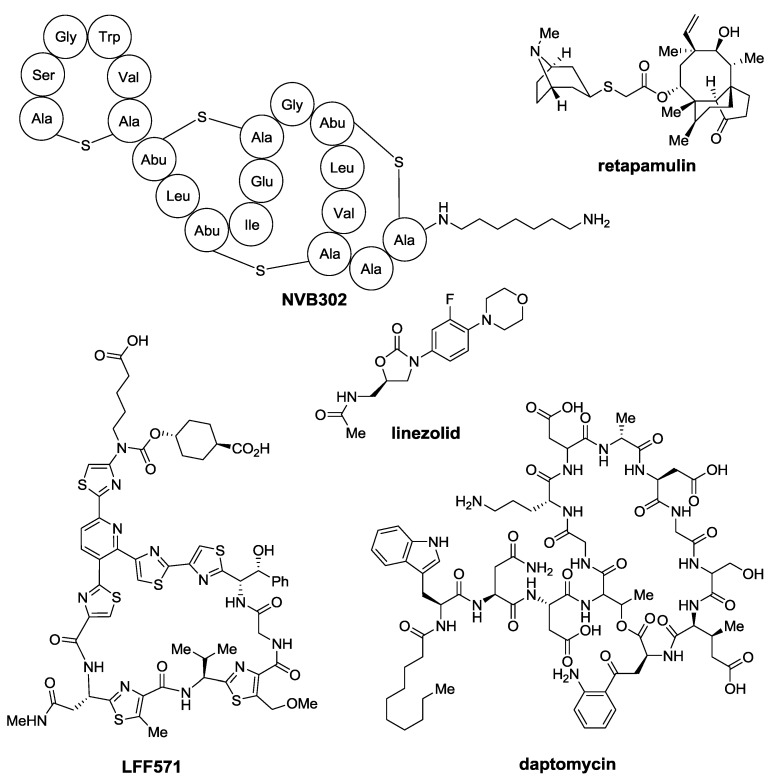
Most of the antibacterial scaffolds known to date were discovered from late 1930s to early 1960s. After that period, almost forty years followed without new bactericide architectures appearing in the market. During those years, semi-synthetic modifications of the already known compounds were used to fight antibacterial resistance. However, with the new century, a batch of new antibiotic scaffolds got closer to the clinic (Figure 1). These include oxazolidinones (linezolid, 2000), lipopeptides (daptomycin, 2003) and mutilins (retapamulin, 2007). In parallel, other types of antibiotics, such as lantibiotics [3] (NVB302) and thiopeptides [4] (LFF571) [5], are under study and some members of these groups are already in clinical trials for the treatment of human infections.
Figure 1.
Members of new classes of antibiotics. Abu = aminobutyric acid.
Among these new families of antibiotics, thiopeptides have gathered much attention due to their potent in vitro activity against Gram-positive bacteria and their intriguing structures. During the last two decades, intensive investigations on known thiopeptides, their semisynthetic modification and the discovery of new members of this class of antibiotics have been the focus of the efforts of many research groups.
2. Thiopeptides
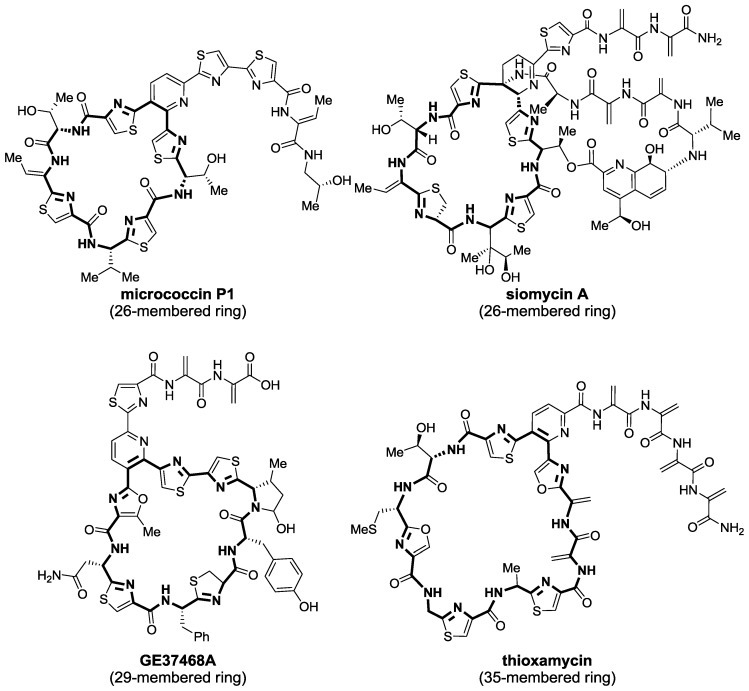
Thiopeptides, or thiazolyl peptides [4], are highly modified sulfur-rich peptides of ribosomal origin. They all share a series of common motifs that differentiate them from other peptide-derived and/or azole-containing natural products. Their most characteristic feature is the central nitrogen-containing six-membered ring, which can be found in many different oxidation states. This central ring serves as scaffold to at least one macrocycle and a tail, and both can be decorated with various dehydroamino acids and azoles, such as thiazoles, oxazoles, and thiazolines. All these moieties are formed though dehydration/dehydrosulfanylation of Ser, Thr, and Cys residues. Their impressive in vitro profile against Gram-positive bacteria, and their new mechanisms of action, have gathered the attention of many groups, both in academia and industry, as they pose an alternative to other antibiotics presently facing resistance by old pathogens. To date, more than one hundred members of this family of natural products have been identified; however, their very large molecular size and their poor aqueous solubility have been a major drawback to introduce them into the clinic. This has become their major limitation and has restricted their use to topic treatments, and so far only for pet skin infections (thiostrepton, Panolog).
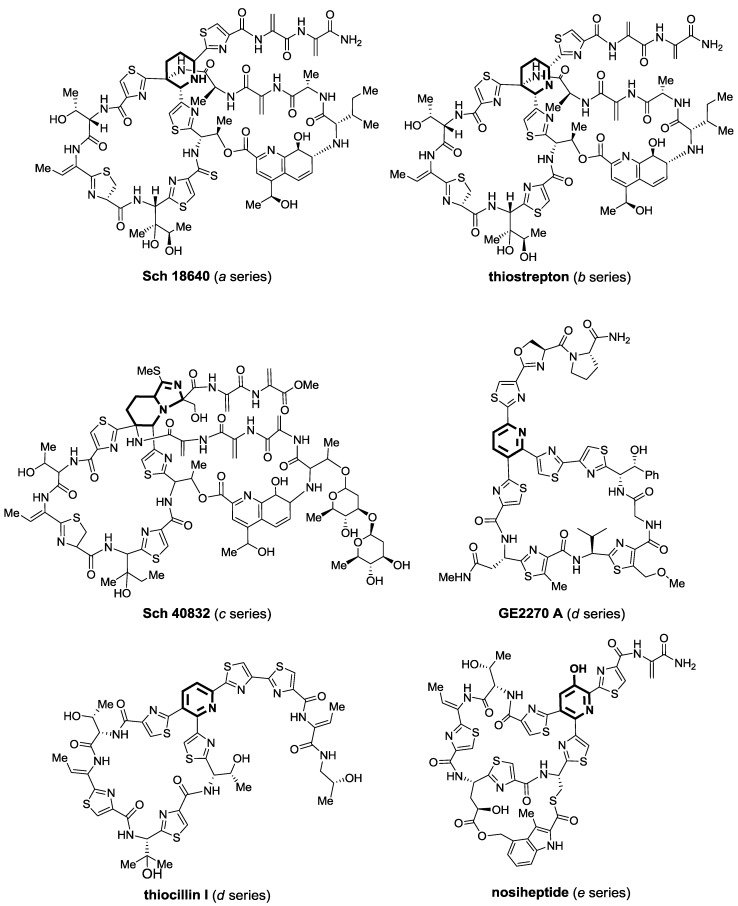
Given the different oxidation state the central ring of thiopeptides can be found in, they have been classified into different series (Figure 2) [4]. Thus, the a series presents a totally reduced central piperidine, whereas the b series is oxidized further and contains a 1,2-dehydropiperidine ring. Only one thiopeptide of the c series has been isolated to date and its core moiety is somewhat unexpected, as it displays a piperidine ring fused with imidazoline. All members of series a, b, and c have a second macrocycle, which contains a quinaldic acid moiety. The d series goes further on the oxidation state rank and shows a trisubstituted pyridine ring, which is the landmark of this subgroup, the most numerous among thiopeptides. In a sense, the e series is even more oxidized and is easily differentiated for the hydroxyl group in the central pyridine, which is now tetrasubstituted. The e series also presents a very characteristic second macrocycle appending from the main one and formed by a modified 3,4-dimethylindolic acid moiety.
Figure 2.
Classification of thiopeptide antibiotics into different series. Their characteristic central six-member ring is highlighted in bold.
2.1. Isolation and Structure Elucidation
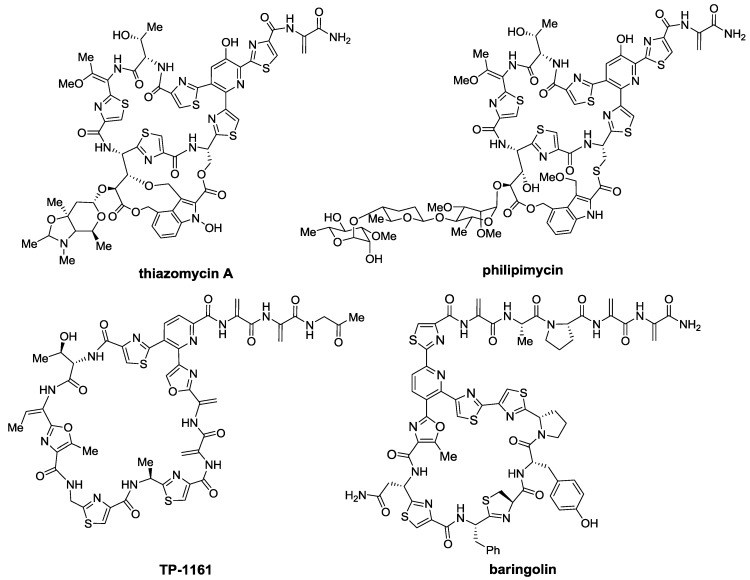
Thiopeptides have been isolated from diverse sources. In 1948, the first known member of the family, micrococcin [6], was isolated from a sample of Oxford’s sewage waters. Accounting for the highly diverse origin of thiopeptides, micrococcin P1 was more recently isolated from a completely different source, a French cheese [7]. However, more conventional samples, such as from soil are the main source of most thiopeptides. In fact, thiostrepton, the most famous member of the family, has been isolated from different soil samples [8,9,10], including one from Hawaii in 1955 [11], shortly after it was first discovered in 1954 [8,9,10]. Although a few more thiopeptides were isolated during the following years, it was from the 1980s, especially during the 1990s, that most of the known members were discovered. Nonetheless, many novel entities have also been described during the last decade. Remarkably, the first thiopeptide antibiotics isolated from a marine source were YM-266183 and YM-266184, discovered as late as 2003, in Japan [12]. During the last few years some more thiopeptides have been isolated and characterized; these include the thiazomycins (2007) [13,14,15,16], philipimycin (2008) [17], thiomuracins (2009) [18], TP-1161 (2010) [19,20], baringolin (2012) [21], and kocurin (2013) [22] (Figure 3).
Figure 3.
Some of the most recently described thiopeptides.
The assignment of thiopeptide structures can be a very complex task, as exemplified by thiostrepton, of which structure elucidation was originally addressed by degradation studies and structure determination of fragments [23]. However, the later use of X-ray diffraction was essential to elucidate both connectivity and stereochemistry [24,25]. Although the development of NMR spectroscopy techniques has permitted the elucidation of many structures of thiopeptides, a high degree of uncertainty remains until further evidence is provided. This was clearly the case of micrococcin P1 [26]. Early studies on its constitution by hydrolysis [27,28,29] of the natural extract permitted the identification of most moieties present in micrococcin; however, there was no clear evidence of its connectivity. Later on, NMR studies [30,31,32] and synthesis of proposed structures [33,34,35,36] of the natural compound resulted in better hypotheses for its constitution and stereochemistry, although none of the synthesized products was identical to the natural one. It was not until its total synthesis was achieved by Ciufolini in 2009, 51 years after its discovery, that micrococcin P1 structure and stereochemistry were finally confirmed [37].
The structure of most thiopeptides has been investigated by a combination of degradation, mass spectrometry and NMR studies. The impossibility to obtain crystals for the vast majority of them prompted the assignment of their structure without a clear evidence of their stereochemistry. In spite of this limitation, in many cases their configuration has been proposed by analogy with similar isolates [38,39], via amino acid analysis [40,41,42] or via isotopic labeling through feeding with labeled amino acids [43,44]. In some cases, less conventional techniques have been chosen. Such are the cases of promoinducin and thiotipin, where chiral TLC was used to determine the configuration of l-Thr from an acidic hydrolysate [45,46]. Absolute configurations have also been reported after NMR spectroscopy studies and chiral capillary electrophoresis [47].
As exemplified by micrococcin P1, a synthetic approach to the problem can serve as the ultimate confirmation for both connectivity and stereochemistry. This strategy also includes the comparison of fragments with their synthetic counterparts; such was the case of GE2270A [48]. Synthesis of thiopeptides polyheterocyclic cores has been used to confirm the structure of the corresponding degradation products, while, at the same time, it has also permitted the development of the necessary synthetic methodology [49].
2.2. Biosynthesis
The biosynthetic pathway of thiopeptides has been very elusive for a long time; however, recent discoveries have put light on the synthesis of these highly modified peptides. Peptide-based natural products can have two distinct origins depending on how their amino acids are condensed together to form the parent peptides. These can be either synthesized on the ribosome as product of mRNA translation or can be assembled by nonribosomal peptide synthases (NRPSs). Though most highly modified peptide-derived natural products are synthesized by NRPSs, there was no evidence of such origin for thiopeptides. Surprisingly, very recent discoveries by four different groups have demonstrated that parent pre-peptide of thiopeptide is ribosomally synthesized and, thus, is genetically encoded [19,50,51,52].
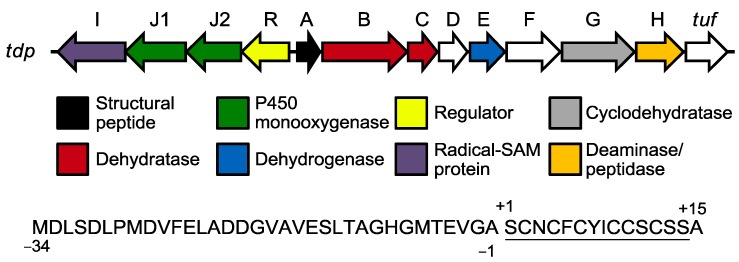
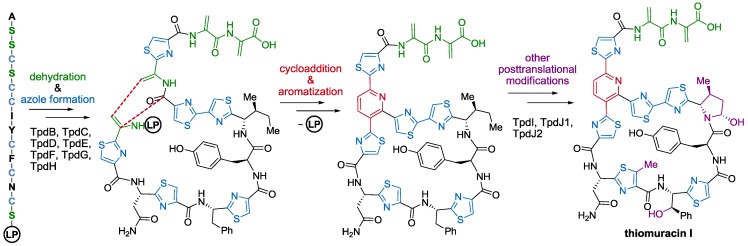
Joint efforts of bio-informatics and genome mining have been essential for the identification of genes that encode the precursor peptide and the enzymatic machinery necessary for its subsequent tailoring [18,20,50,51,52,53,54,55,56,57,58,59]. The gene encoding the precursor peptide has been identified for many thiopeptides and, in all cases, there is a perfect agreement with the expected amino acid sequence. This precursor peptide is divided in two different regions, a structural peptide of 12 to 17 residues at the C-terminus, which contains the amino acids that will constitute the thiopeptide itself, and a leading peptide of 34 to 55 residues at the N-terminus, which is cleaved during the bio-synthetic process. In some cases, the C-terminal structural peptide contains one or two extra residues that are cleaved during the tailoring to confer each thiopeptide its characteristic C-terminus [60,61]. All necessary enzymes for pre-peptide tailoring are encoded in genes surrounding that of the precursor peptide, forming a gene cluster (see Figure 4 for an example on thiomuracins’ gene cluster (tdp) and precursor peptide [18]; in the gene cluster, genes appear as arrows and are named with letters. Each gene (tdpX) codes a gene product, a protein/enzyme (TdpX)).
Figure 4.
The biosynthetic gene cluster of tiomuracins and their precursor peptide sequence, which is coded in the structural gene. In the precursor peptide sequence, the structural peptide is numbered with positive figures and the leading peptide with negative ones. Residues that appear in the mature thiopeptide are underlined.
The role of most enzymes present in some thiopeptides gene clusters has been already discovered. Similarity with known enzymes of the same function, gene deletions, and characterization of products that result from transformations with isolated enzymes, have permitted to establish which transformations and in which order they take place [51,58,59,62,63,64,65,66,67,68]. Apparently, oxazole, thiazole, and thiazoline rings are formed first through cyclization, dehydration, and, if required, oxidation of Ser, Thr, and Cys residues. In a second step, Ser and Thr phosphorylation and elimination yields the corresponding dehydroalanine (Dha) and dehydrobutyrine (Dhb) residues, respectively. Finally, intramolecular aza-Diels-Alder-like cycloaddition between distant Dha residues occurs, followed by dehydration, and, when required, elimination to constitute the central six-membered ring. Further side-chain modifications, such as oxidations, cyclizations, methylations, and incorporation of indolic or quinaldic acid moieties seem to occur in later stages of the bio-synthetic pathway (see Scheme 1 for an example on thiomuracin I biosynthesis [18]).
Scheme 1.
Biosynthetic pathway of thiomuracin I. LP = leading peptide. Enzymes involved in the biosynthetic pathway (TdpX) are named according to their corresponding gene (tdpX) in thiomuracins’ gene cluster (tdp).
Both quinaldic and indolic acid moieties found in a–c and e series thiopeptides are synthesized from l-Trp and are part of the second macrocycle found in these compounds. This was first demonstrated by labeling [69,70,71,72] and enzyme function [73,74] experiments and, more recently, also using genetic engineering methods [63,65,66,67].
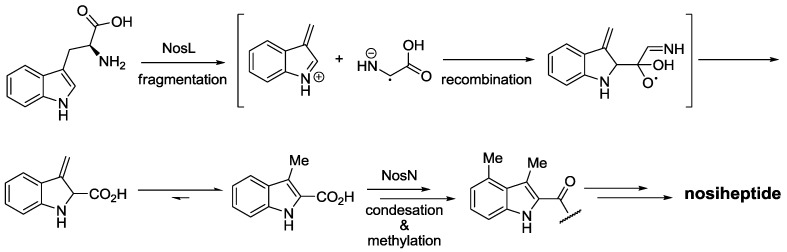
In the case of indolic acid formation, Trp undergoes a radical-mediated rearrangement and Cα migrates to position 2 of indole (Scheme 2). Subsequently, S-adenosylmethionine-dependent 4-methylation of the aromatic scaffold after condensation with the structural peptide yields an advanced intermediate of the mature thiopeptide [65,75].
Scheme 2.
Biosynthesis of indolic acid moietiy from Trp and incorporation into nosiheptide. Enzymes involved in the biosynthetic pathway (NosX) are named according to their corresponding gene (nosX) in gene cluster of nosiheptide (nos).
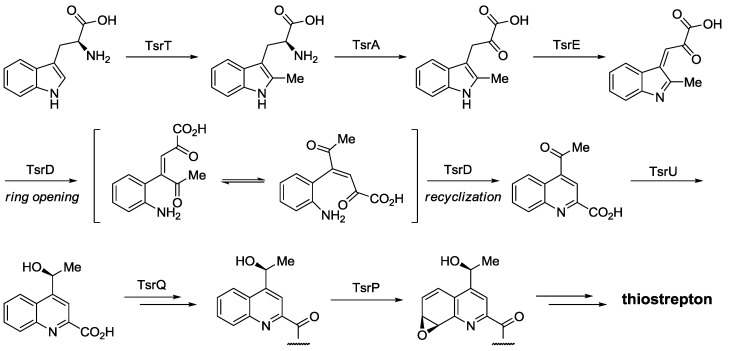
Alternatively, quinaldic acid synthesis starts with S-adenosylmethionine-mediated methylation of Trp (Scheme 3) [52,66]. Deamination/oxidation steps follow and after ring opening, recyclization yields the quinaldic acid moiety. This is then reduced, attached to the structural peptide, and epoxidized. Upon epoxide opening, the second macrocycle of thiostrepton is formed.
Scheme 3.
Biosynthesis of quinaldic acid moietiy from Trp and incorporation into thiostrepton.
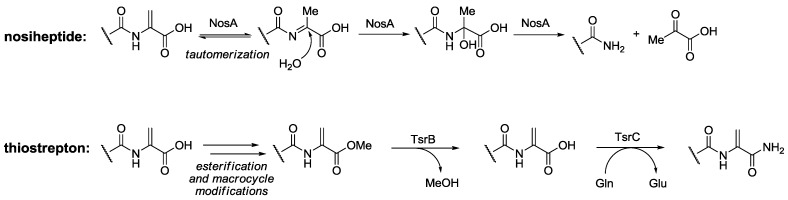
C-terminal tailoring is one of the last steps in thiopeptides maturation. In those cases where a C-terminal amide is present, two distinct mechanisms have been described for its formation (Scheme 4). Nosiheptide structural peptide contains an extra C-terminal Ser residue, which is lost during tail maturation, giving rise to a C-terminal amide [60]. By contrast, structural peptide of thiostrepton does not contain any extra amino acids and its C-terminal Ser residue can be methylated to form the corresponding ester (Scheme 4). The C-terminal amide is formed by deesterification and subsequent amidation using Gln as nitrogen donor [61].
Scheme 4.
Proposed mechanisms for C-terminal amide formation during nosiheptide and thiostrepton maturation.
2.3. Biological Activity
Thiopeptides are best regarded as antibacterial agents, however, their therapeutic potential is surprisingly broad and have been found to posses anticancer [76,77,78,79,80,81,82], antiplasmodial [83,84,85,86,87,88], immunosuppressive [89], renin inhibitory [90], RNA polymerase inhibitory [91], and antifungal [92] activities. This wide variety of biological functions has resulted in a very prolific literature outcome, positioning the macrocyclic scaffold of thiopeptides as a veritable privileged structure.
2.3.1. Antibacterial Activity
It is already well established that thiopeptides exert their antibacterial function via the inhibition of ribosomal protein synthesis. However, this is the result of different mechanisms of action that depend on macrocycle size. Thiopeptides exhibit macrocycles of three different sizes, 26-, 29-, and 35-membered rings, depending on the number of residues present. On one hand, thiopeptides of 26-member macrocycles, such as that of micrococcin P1 and the siomycins (Figure 5), are known to bind the GTPase-associated region of the ribosome/L11 protein complex. By doing so, the thiopeptide blocks the binding region of elongation factor G (EF-G) and does not allow translocation of the growing-peptide/tRNA complex in the ribosome to occur [93,94,95]. On the other hand, those thiopeptides with a 29-membered ring, in the fashion of GE37468A, bind to elongation factor Tu (EF-Tu), blocking its tRNA/amino acyl complex binding site [96,97,98]. As a consequence, the complex cannot be delivered into the ribosome and peptide elongation does not take place. Compounds with the largest macrocycles, those with 35-membered rings, maintain potent antibacterial activity; however, their molecular target still remains unknown.
Figure 5.
Thiopeptides have macrocycles of different sizes that determine their mode of action.
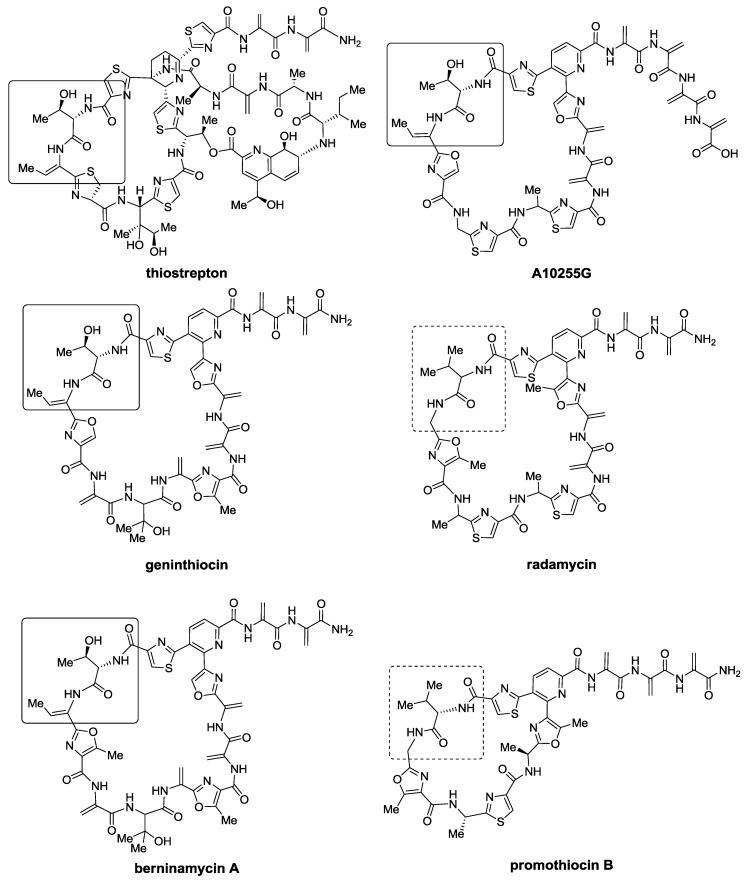
Somewhat related to antibacterial activity is tipA gene promotion, which encodes two thiostrepton-induced proteins (Tip), TipAL and TipAS [99]. The latter, TipAS, serves as a mechanism of defense for bacteria, as it sequesters and covalently binds a thiopeptide molecule, which can no longer inhibit ribosomal protein synthesis. TipA promotion has been used to identify thiopeptides in a high-throughput screening program, which detected transcription of the promoter of tipA (ptipA) and led to the discovery of geninthiocin [100] (Figure 6). Other thiopeptides, such as thiotipin [46] and thioxamycin [42], and promothiocins [101] were also discovered thanks to their tipA promoting activity. Interestingly, the 35-membered thiopeptide radamycin is completely devoid of antibacterial activity, but is a very strong inducer of tipA gene expression (Figure 6). Various tipA promoting thiopeptides are depicted in Figure 6, where very preserved regions, associated with key interactions for binding with ribosome/L11 complex [102], are highlighted. Although those residues are different in radamycin, promothiocin B displays those same, not-preserved residues in a smaller 26-membered macrocycle and retains potent antibacterial activity. Apparently, tipA promotion activity is more dependent on the presence of a dehydroalanine-containing tail close to the six-membered central scaffold [103].
Figure 6.
Thiopeptides with tipA promoting activity. A very preserved region, which has been shown to interact with the ribosome/L11 complex, is highlighted (solid squares). Radamycin, devoid of antibacterial activity, has a mutated sequence in the previously mentioned region (hashed squares). Promothiocin B possesses the same mutated residues, but maintains antibacterial activity, though in a 26-membered macrocycle.
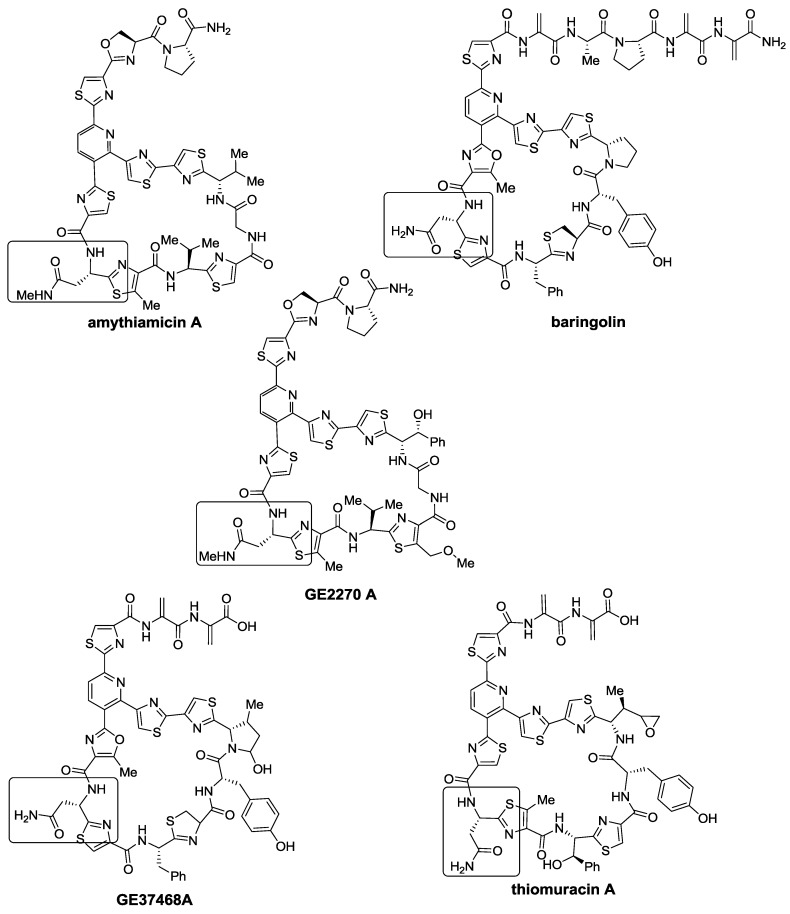
Thiopeptides known or presumed to bind to EF-Tu, those with 29-membered macrocycles, also possess a very preserved residue in their macrocycle, pointing to a key interaction required to exert their biological activity. The amythiamicins, baringolin, the GE2270 series, GE37468 A, and the thiomuracins, all belonging to the d series, preserve an Asn residue in the same position that is either methylated or not (Figure 7). Codon randomization experiments by Walsh et al. [104] produced various GE37468 A analogs that substituted Asn with Ala, Cys, His, and Ser; however, none of these analogs retained antibacterial activity, supporting the key role of this residue. Apart from providing a key contact with EF-Tu, the Asn residue participates in the stabilization of the bioactive conformation of the macrocycle, providing a transannular H-bond [105].
Figure 7.
29-membered thiopeptides with a very preserved Asn residue highlighted with a square.
2.3.2. Other Activities
One of the biological properties of thiopeptides of major interest, apart from the antibacterial one, is anticancer activity. In this regard, thiostrepton was found to selectively kill cancer cells without showing any cytotoxicity against healthy tissues [106]. Such a promising effect has been demonstrated to arise from selective inhibition of transcription factor forkhead box M1 (FOXM1) [78,81,82]. FOXM1 overexpression is associated with the development and progression of cancer and its selective targeting is a very large achievement, as transcription factors have been considered undruggable for a long time [107,108].
During synthetic efforts of Nicolaou’s group, it was discovered that the central core of thiostrepton (1) retained some antibacterial, but an increased potency against all cancer cell lines tested (Table 1) [76].
Table 1.
Thiostrepton fragment 1 outperforms its parent compound against various cancer cell lines.
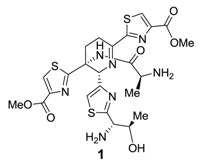
| Compound | LC50 (μM) | IC50 (μM) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| NCI-H460 | HCT-116 | SK-OV-3 | MCF-7 | K-562 | 1A9 | PTX10 | A8 | AD10 | |
| thiostrepton | 1.5 | 1.6 | 2.8 | 3.8 | 1.7 | 0.96 | 1.1 | 0.9 | 91.0 |
| 1 | 0.9 | 0.6 | 1.2 | 0.9 | 0.8 | 0.07 | 0.1 | 0.2 | 0.4 |
Many thiopeptides have been found to possess anti-malaria activity [83,84,85,86,87]. Although Plasmodium falciparum parasite cell is eukaryote, thiopeptides target apicoplast protein synthesis [109], which resembles that of prokaryotic organisms. Although it has been demonstrated that thiostrepton binds to the apicoplast 23S rRNA [83], thiopeptides of the d series, such as micrococcin P1 and amythiamicin A, are much more potent inhibitors of P. falciparum growth [84].
Very recently, the use of thiostrepton semi-synthetic analogs has demonstrated that it targets both the apicoplast ribosomes and the proteasome of P. falciparum [88]. This dual mode of action could make thiostrepton and similar thiopeptides less prone to resistance development than single-target drugs.
A screening program in search of immune-suppressants identified siomycin as inhibitor of antibody production by murine B-cells [89]. Comparison with thiostrepton showed the superior behavior of the structurally similar siomycin. Both thiopeptides are thought to possess a different mechanism of action than that of FK506, a common immunosuppressant drug, and would act directly on B-cells.
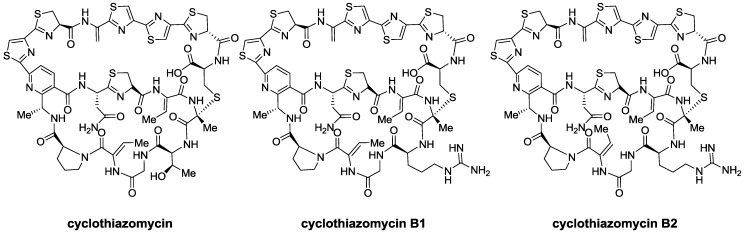
Clyclothiazomycin is a very unique thiopeptide that does not possess a tail (Figure 8). In fact, the azole-containing branch that would serve as tail is linked to the macrocycle, forming a second ring, different from those found in thiopeptides of series a–c and e. However, due to its central tri-substituted pyridine ring, it is still considered a member of the d series. Perhaps due to its peculiar structure, different activities have been found for it. The first one to be described was human plasma renin inhibitory activity [90]. Renin is an enzyme associated with hypertension [110], diabetes [111], and Alzheimer’s disease [112] and is a rate-limiting enzyme in a cascade that starts with the cleavage of angiotensiogen and ends with the formation of angiotensin II. Due to this, renin is regarded as one of the most effective targets to treat hypertension.
Figure 8.
Cyclothiazomycin and its more recently isolated analogs B1 and B2.
Thiopeptides with a very similar structure to cyclothioazomycin were recently isolated and characterized and were named after the parent compound (Figure 8) [91]. In particular, cyclothiazomycin B1 was found to inhibit transcription by bacteriophage RNA polymerase. Such result might serve to further understand transcription at the molecular level.
Cyclothiazomycin B1 (Figure 8) also exhibits antifungal activity and inhibits the growth of various filamentous fungi. It presumably does so by binding to chitin, causing cell wall fragility [92].
Saramycetin has also been described as an antifungal thiopeptide [113]; however, it was not fully characterized and might not fulfill the structural requirements to fall into this family of natural products.
2.4. Conformation and Binding
The structure of thiopeptides has been studied using different techniques, which have also been used to elucidate the active conformation and the key contacts required to exert their biological activity. Though thiopeptides are relatively large molecules with big macrocycles, they possess many azoles, dehydroamino acids and amide bonds that confer the required rigidity for efficient binding [114]. First conformational studies were performed by NMR and provided solution structures of the promothiocins [115], nocathiacin I [47] and amythiamicin D [105]. For amythiamicin D, an intramolecular H-bond was detected [105], which seems to favor the bioactive conformation. The same H-bond interaction can be observed between the equivalent positions of active GE2270A and thiomuracin A analogs [116,117]. NMR studies of the thiostrepton/L11/rRNA complex [102] identified key contacts in the 26-membered ring of thiostrepton that led to the design of small analogs that maintained their binding capability [118].
X-ray analysis of the L11/rRNA complex and superimposition with optimized thiopeptide structures have determined that compounds targeting this complex bind to a cleft between the ribosome and L11 protein [94,95,119]. This region, the so-called GTP associated center (GAC), is also the binding site of elongation factor G (EF-G), which is responsible for translocation during ribosomal protein synthesis.
Alternatively, mutation studies have been carried out to study the thiostrepton/L11/rRNA ternary complex. Site-directed mutation of L11 to introduce a Cys residue in a suitable surface position, permitted proximity induced covalent capture (PICC) experiments to be carried out (Figure 9) [93]. In a PICC experiment, the newly introduced Cys should perform a 1,4-conjugated addition to a Dha residue of the thiopeptide tail. These experiments suggested a slightly different binding mode for thiostrepton, which would not sit right inside the cleft between L11 and the 23S rRNA, but closer to the ribosome surface. Further mutation studies of either L11 or 23S rRNA were in agreement with this alternative binding mode, demonstrating that mutations on L11 did not avoid thiostrepton binding, whereas mutations on the ribosome diminished affinity between the complex and the thiopeptide [120].
Figure 9.
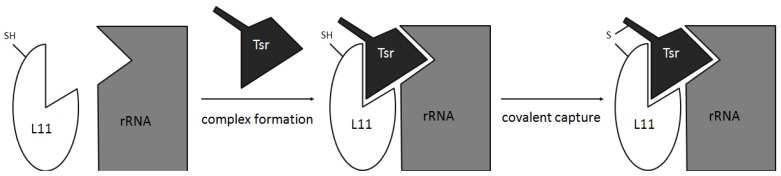
Proximity induced covalent capture. Mutated L11 protein has an external Cys residue in the area where interaction with the tail of thiostrepton is expected. Tsr = thiostrepton.
2.5. Chemical Synthesis
The complex architectures and challenging structures of thiopeptides have gathered the attention of many groups that have envisioned different strategies to accomplish their syntheses [49]. Pioneering work by the groups of Kelly [121,122,123,124], Shin [35], Moody [125,126], Nicolaou [127,128], Ciufolini [36,37], and Bach [127,129,130] developed different methodologies that led to the total synthesis of various thiopeptides. However, most efforts have been devoted to the construction of the central polyheterocyclic core [121,122,123,124,131,132,133,134,135,136,137,138], which have been synthesized by two well-distinguished main strategies: modification of an existing pyridine and construction of the central ring.
2.5.1. Modification of Pyridine
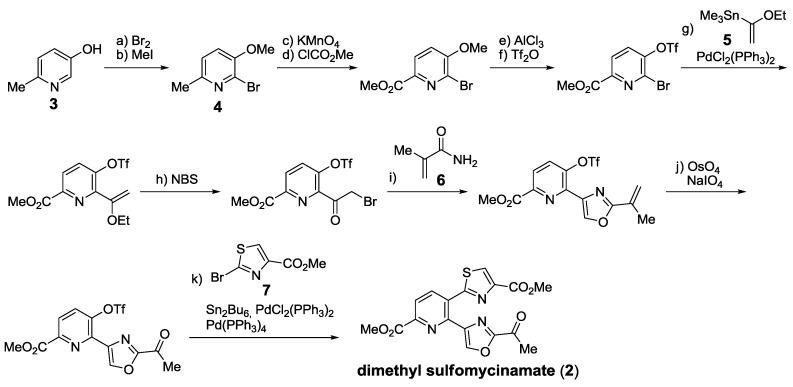
Early efforts by Kelly’s group focused on the synthesis of different thiopeptide cores [121,122,123,124] (Scheme 5). One of his landmark achievements was the synthesis of dimethyl sulfomycinamate (2), a product of acidic methanolysis of sulfomycin [139]. Selective bromination of 3 provided pyridine 4, which contained three well-differentiated positions. Stille cross-coupling with 5, oxazole formation using acrylamide 6 and an in situ stannation/cross-coupling protocol with bromothiazole 7 permitted the installment of the two azole rings present in 2.
Scheme 5.
Kelly’s synthesis of dimethyl sulfomycinamate. Reagents and conditions: (a) Br2, pyridine, rt, 77%; (b) MeI, K2CO3, acetone, reflux, overnight 88%; (c) KMnO4, 90 °C, 3 h; (d) ClCO2Me, Et3N, DMAP, CH2Cl2, 0 °C to rt, 2 h, 65%; (e) AlCl3, CH2Cl2, reflux, 2 days, 93%; (f) Tf2O, 2,6-lutidine, CH2Cl2, 0 °C, 5 min, 95%; (g) 5, PdCl2(PPh3)2, 1,4-dioxane, 100 °C, overnight 97%; (h) NBS, THF, H2O, rt, 10 min, 95%; (i) 6, THF sealed tube, 100 °C, 3 days, 65%; (j) OsO4, NaIO4, 1,4-dioxane, H2O, rt 3 h, 85%; (k) 7, Sn2Bu6, PdCl2(PPh3)2, Pd(PPh3)4, LiCl, 1,4-dioxane, 100 °C, overnight, 35%.
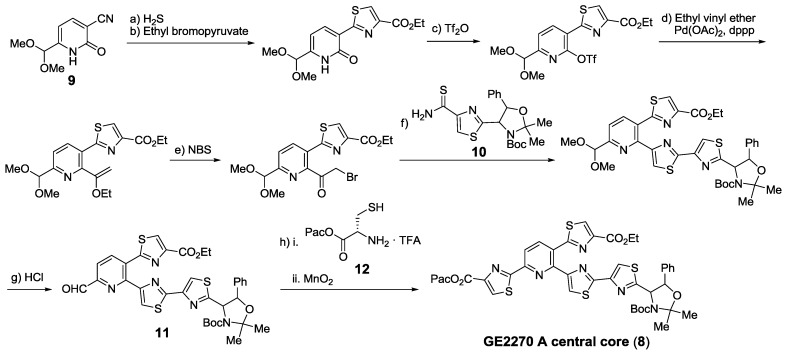
Shin and co-workers developed their own strategies and synthesized many polyheterocyclic cores and fragments, such as GE2270 A central core (8) [140,141,142,143,144,145,146,147]. Most of their syntheses start from a pre-functionalized pyridine or pyridone of the like of 9 (Scheme 6). For the synthesis of 8, thiazoles were constructed using different methods, including Hantzsch syntheses with ethyl bromopyruvate and thioamide 10, followed by condensation of aldehyde 11 with Cys 12 and subsequent oxidation with activated manganese (II) oxide.
Scheme 6.
Shin’s synthesis of GE2270A central core. Reagents and conditions: (a) H2S, DMAP, Et3N, pyridine, rt, 3 days, 90%; (b) i. KHCO3, BrCH2COCO2Et, THF, 0 °C, then rt, overnight; ii. TFAA, pyridine, THF, 0 °C, 1 h, then rt, overnight, 53%; (c) Tf2O, DMAP, pyridine, 0 °C, 1 h, then rt, overnight, 93%; (d) ethyl vinyl ether, Et3N, dppp, Pd(OAc)2, toluene, reflux, overnight, 73%; (e) NBS, THF, H2O, rt, 5 min; (f) i. 10, KHCO3, DME, 0 °C, 1 h, then rt, overnight; ii. TFAA, pyridine, 0 °C, 2 h, 63% (2 steps); (g) 2 M HCl in THF, rt, 24 h; (h) i. 12, Et3N, toluene, rt, 15 min; ii. MnO2, toluene, rt, 12 h, 41% (2 steps). dppp = 1,3-bis(diphenylphosphino)propane; Pac = Phenacyl.
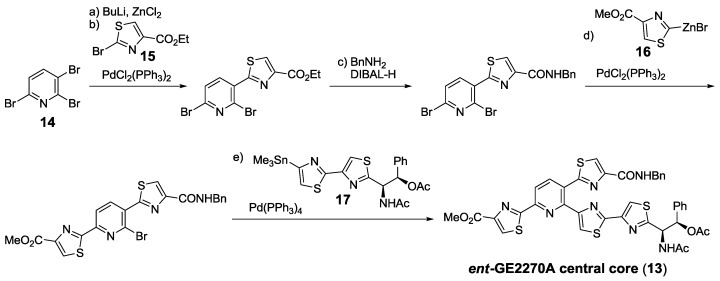
Bach’s group described a very convergent approach to the enantiomer of GE2270 A core (13), which started from 2,3,6-tribromopyridine (14), and was fully based on sequential cross-coupling reactions with thiazoles 15−17 [135] (Scheme 7). This work served for confirmation of the polyheterocyclic core stereochemistry and also set the methodology for further syntheses of GE2270A [129] and amythiamicins A and D [148].
Scheme 7.
The synthesis of ent-GE2270A central core by Bach. Reagents and conditions: (a) BuLi, ZnCl2, THF; (b) 15, PdCl2(PPh3)2, THF, 81% (2 steps); (c) BnNH2, DiBAL-H, THF, CH2Cl2, 86%; (d) 16, PdCl2(PPh3)2, THF, DMA, 78%; (e) 17, Pd(PPh3)4, 1,4-dioxane, 80 °C, 61%.
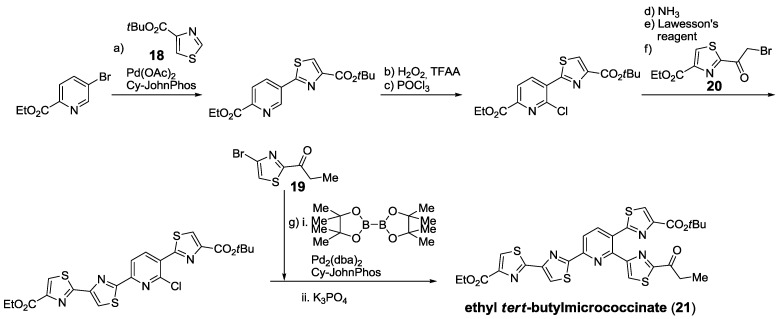
More recently, the group of Hoarau optimized conditions for the cross-coupling of thiazole-4-carboxylates (18) with pyridines using direct C-H activation on thiazole position 2 [137] and also formation of pinacolboronic acid and in situ cross-coupling on thiazole position 4, which is limited to 2-acylthiazoles, such as 19 [138]. Using, both, cross-coupling methodologies and Hantzsch thiazole synthesis with bromoketone 20, the synthesis of ethyl tert-butylmicrococcinate (21) was achieved [138] (Scheme 8).
Scheme 8.
Synthesis of ethyl tert-butylmicrococcinate using direct C-H activation and one-pot borylation/cross-coupling of thiazoles. Reagents and conditions: (a) 18, Pd(OAc)2, Cy-JohnPhos, Cs2CO3, DMF, 110 °C, 18 h, 74%; (b) CO(NH2)2, H2O2, TFAA, MeCN, 0 °C, 30 min; (c) POCl3, toluene, DMF, rt, 6 h, 78% (2 steps); (d) NH4OH, THF, rt, 82%; (e) Lawesson’s reagent, toluene, 2 h, 76%; (f) 20, EtOH, THF (1:1), 65%; (g) i. 19, bispinacolatodiboron, Pd2(dba)2, Cy-JohnPhos, KOAc, dioxane, 110 °C, 30 min; ii. K3PO4, dioxane, H2O, 110 °C, 12 h, 87%.
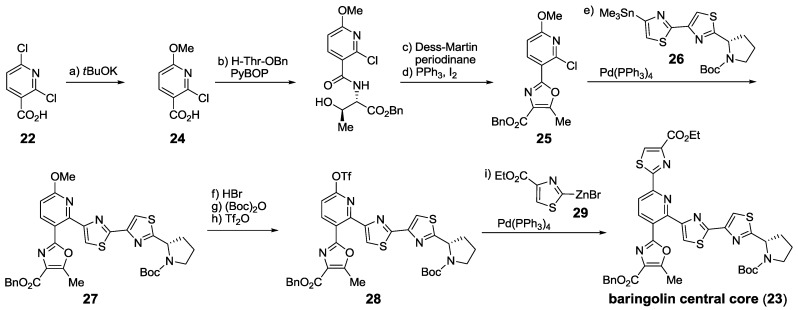
The last example of cross-coupling-based construction of thiopeptide polyheterocyclic cores was reported by Álvarez’s group [149] and used the regioselective differentiation of the two α positions in 2,6-dichloronicotinic acid (22) [150]. This strategy gave access to baringolin’s central core (23), facilitating its total synthesis [151] (Scheme 9). The carboxylic acid present in 24 was used to construct oxazole 25 and subsequent Stille cross-coupling with 26 [152] provided methoxypyridine 27. After conversion of 27 to the corresponding triflate 28, Negishi cross-coupling with 29 yielded 23.
Scheme 9.
Synthesis of baringolin’s central polyheterocyclic core. Reagents and conditions: (a) tBuOK, MeOH, 65 °C, 4 days, 85%; (b) H-Thr-OBn, pyBOP, DIPEA, THF, 0 °C, 3 h, 89%; (c) Dess-Martin periodinane, CH2Cl2, rt, 6 h, 95%; (d) PPh3, I2, NEt3, CH2Cl2, 0 °C to rt, 15 h, 78%; (e) 26, Pd(PPh3)4, 1,4-dioxane, 80 °C, 48 h, 88%; (f) HBr, AcOH, rt, 28 h, 73%; (g) (Boc)2O, NEt3, CH2Cl2, 0 °C, 4 h, 94%; (h) Tf2O, 2,6-lutidine, DMAP, CH2Cl2, 0 °C to rt, 3 h, 88%; (i) 29, Pd(PPh3)4, DMA, 45 °C, 1 h, quant.
2.5.2. Construction of the Central Ring
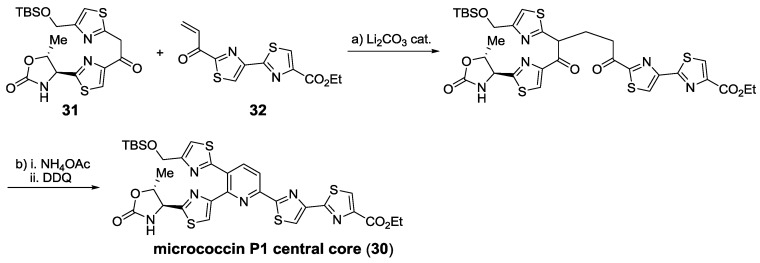
One of the most appealing contributions and the first one to rely on construction of the pyridine ring was Ciufolini’s synthesis of micrococcin’s central polyheterocylic core (30) [131] (Scheme 10). With the required thiazole building blocks, 31 and 32, in hand, the pyridine ring is formed in two steps in almost quantitative yield. This led to the synthesis of the Bycroft-Gowland structure of micrococcin P1, which had been miss-assigned [36]. It took ten more years to finally synthesize the true structure of micrococcin P1 and confirm its stereochemistry [37]. Using the same strategy, thiocillin I has been recently synthesized [153].
Scheme 10.
The synthesis of the polyheterocyclic core of micrococcin P1 by Ciufolini. Reagents and conditions: (a) cat. Li2CO3, EtOAc, 92%; (b) NH4OAc, EtOH then DDQ, toluene, 97%.
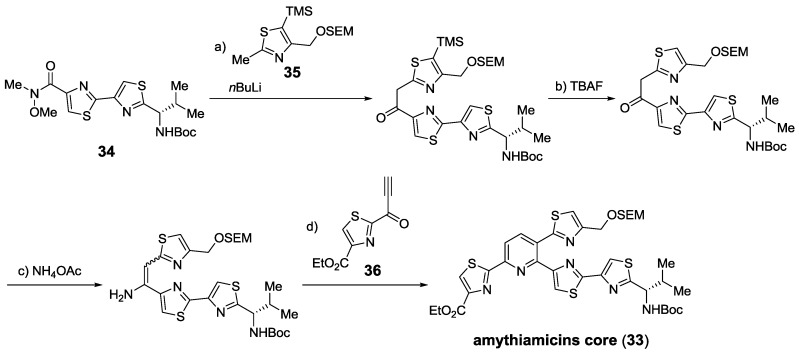
Bagley, first in Moody’s group, and co-workers also described their own syntheses of the thiopeptide cores, such as the amythiamicins core (33), constructing the pyridine ring with advanced thiazole-containing building blocks 34−36. Using Bohlmann-Rahtz pyridine synthesis, various thiopeptide cores were obtained [134,154,155,156,157,158,159] (Scheme 11). In addition, a very early total synthesis of promothiocin A was achieved [125,160,161].
Scheme 11.
Synthesis of amythiamicins pyridine cluster using Bohlmann-Rahtz pyridine formation. Reagents and conditions: (a) 35, nBuLi; H2O, 91%; (b) TBAF, THF, rt, 1 h, 93%; (c) NH4OAc, microwave, 120 °C (100 W), toluene, 30 min, 76%; (d) 36, EtOH, 60 °C; toluene, AcOH, 70 °C, 85% (93% ee). SEM = 2-(trimethylsilyl)ethoxymethyl.
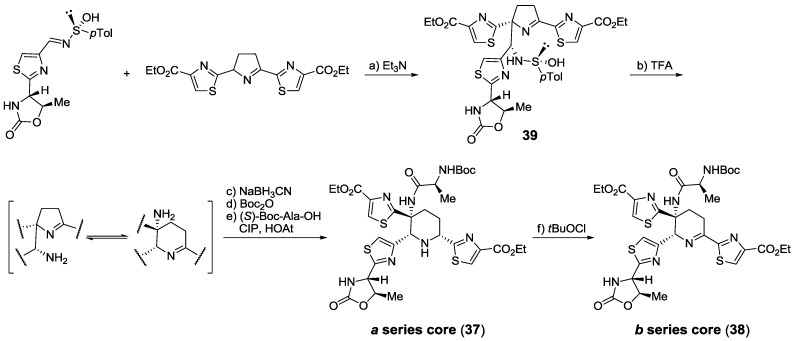
The first synthesis of a polyheterocyclic core from the a (37) and b (38) series was reported by the group of Hashimoto. Their approach was based on formation of a substituted central 1-pyrroline 39 that underwent ring-expansion to form a piperidine ring 37 (a series) that could be selectively oxidized for form the corresponding 2,3,4,5-tetrahydropyridine 38 (b series) [162] (Scheme 12). In further reports they described the synthesis of other fragments [163,164] of b series thiopeptides and finally achieved the total synthesis of siomycin A [165,166].
Scheme 12.
Synthesis of a and b series central core by Hashimoto and co-workers. Reagents and conditions: (a) Et3N, THF, −25 °C, 71%; (b) TFA, EtOH; (c) NaBH3CN, AcOH, EtOH, 52% (2 steps); (d) Boc2O, DMAP, Et3N, THF, 0 °C, 84%; (e) (S)-Boc-Ala-OH, CIP, HOAt, DIPEA, CH2Cl2, 93%; (f) tBuOCl, THF, −78 °C, then cat. DMAP, Et3N, 95%. CIP = 2-chloro-1,3-dimethylimidazolidium hexafluorophosphates.
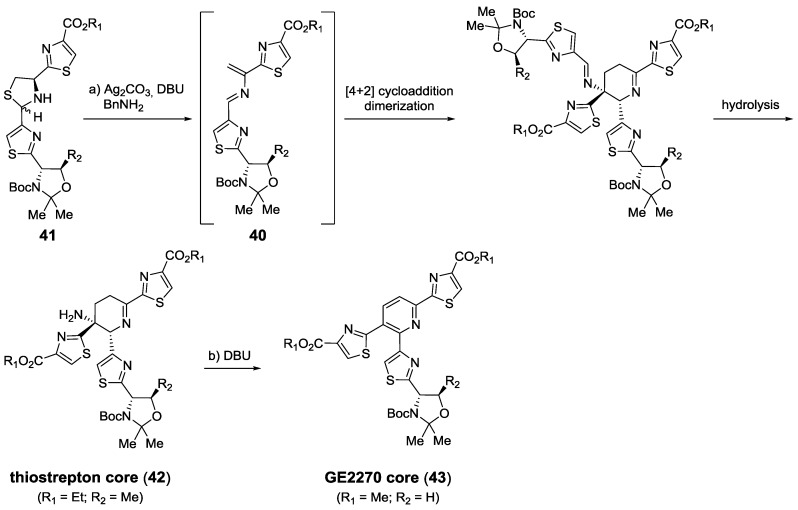
Cycloadditions have also been used for the synthesis of various central fragments. Almost simultaneously, the groups of Nicolaou [167] and Moody [132] developed two different bio-inspired strategies to obtain their target cores via an aza-Diels-Alder reaction. Nicolaou’s strategy was based on the silver-promoted formation of an aza-diene 40 from a precursor thiazolidine 41, which would dimerize in a [4 + 2] cycloaddition (Scheme 13). The 2,3,4,5-tetrahydropyridine 42 (b series) thus obtained after hydrolysis of the cycloaddition adduct could be oxidized to form the corresponding fully unsaturated d series core (43).
Scheme 13.
Bio-inspired synthesis of b and d series polyheterocyclic cores. Reagents and conditions: (a) Ag2CO3, BnNH2, DBU, pyridine, −15 °C, 1 h; then H2O/EtOAc (1:1), 1 h, 60%; (b) DBU, EtOAc, reflux, 5 h, 50%.
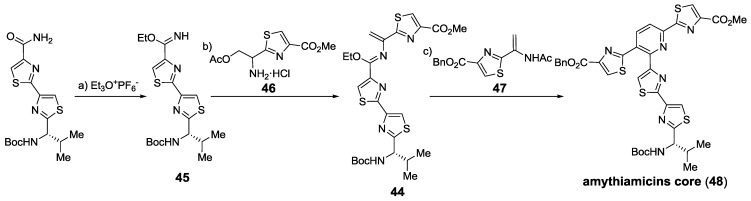
Using Nicolaou’s methodology various total syntheses were achieved, including the challenging thiostrepton [127,128,168], but also GE2270A and GE2270T [169], and various amythiamicins [170]. On the other hand, the strategy by Moody relied on a previously formed aza-diene 44, formed from the condensation of thiazoles 45 and 46 that could react with a different dienophile (47), yielding the aromatized cycloaddition product 48 [126,171] (Scheme 14).
Scheme 14.
Bio-mimetic aza-Diels-Alder synthesis of 2,3,6-trisubstituted pyridine cores. Reagents and conditions: (a) Et3O+PF6−, CH2Cl2, 100%; (b) 46, CH2Cl2, then DBU, CHCl3, 63%; (c) 47, toluene, microwave, 120 °C, 33%.
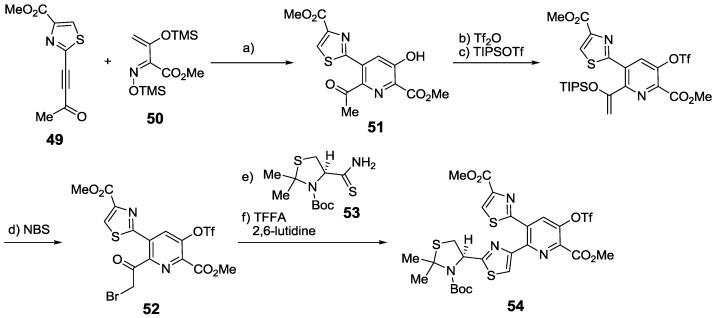
Alternatively, Arndt designed another aza-Diels-Alder strategy that was not based on the biosynthetic pathway, but used an alkyne 49 and a protected α,β-unsaturated oxime 50 as starting materials, giving access to 3-hydroxypyridines, such as 51. After a modified Hantzsch thiazole synthesis between 52 and 53, a precursor of the central core of nosiheptide (54) could be accessed [136] (Scheme 15). Using this strategy, the main macrocycle of nosiheptide could be synthesized [172].
Scheme 15.
Synthesis of nosiheptide core via an aza-Diels-Alder cycloaddition. Reagents and conditions: (a) toluene, 180 °C, 55%; (b) Tf2O, Et3N, CH2Cl2, 0 °C; (c) TIPSOTf, Et3N, CH2Cl2,0 °C, 79% (2 steps); (d) NBS, THF/ H2O, 97%; (e) 53, KHCO3, THF, −40 °C; (f) TFAA, 2,6-lutidine, −20 °C, 60% (2 steps).
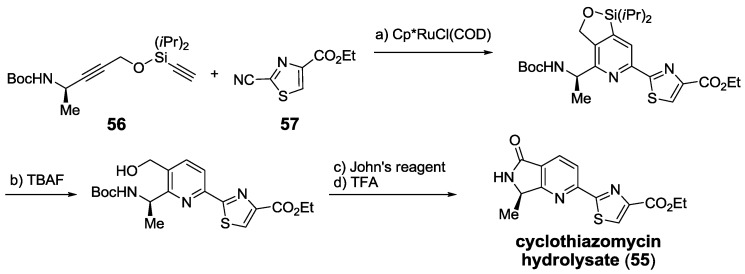
Finally, one of the newest and most innovative contributions to the field is the synthesis of the central core of cyclothiazomycin hydrolysate 55 through a [2 + 2 + 2] ruthenium-catalyzed cyclotrimerization reaction between 56 and 57 [173] (Scheme 16). This yielded a product identical to that previously described by Bagley, thus confirming its identity [158].
Scheme 16.
Syntehsis of cyclothiazomycin central core hydrolysate via a [2 + 2 + 2] ruthenium-catalyzed cycloaddition. Reagents and conditions: (a) Cp*RuCl(COD), 1,2-dichloroethane, 60 °C, 82%; (b) TBAF, THF, rt, 97%; (c) John’s reagent, acetone, 0 °C to rt; (d) TFA, CH2Cl2, rt, 80% (2 steps). Cp* = pentamethylcyclopentadienyl. TBAF = tetrabutylammonium fluoride.
Future advances in the preparation of substituted pyridines will surely facilitate the total synthesis of thiopeptides that still pose a challenge, but most importantly, will give easy access to fragments and analogs for the development of thiopeptide-based drugs with an improved pharmacokinetic profile.
2.6. In the Market
To date, only two thiopeptides have hit the market, thiostrepton and nosiheptide. Although both are exclusively devoted to veterinary use, their applications are very different. The lack of thiopeptide formulations for human use stems from their low aqueous solubility, a limitation that also restricts their use in animals.
Thiostrepton is used as one of the ingredients of an ointment for the treatment of cats and dogs skin infections. This ointment always has the same formulation (nystatin, 100,000 units; neomycin sulfate, 2.5 mg; thiostrepton, 2500 units; triamcinolone acetonide, 1 mg) and is sold by different companies with a variety of brand names: Animax (Dechra), Resortin (Hannah), Panolog (Novartis), Dermalone (Vedco), etc.
Alternatively, nosiheptide, is used as an animal growth promoter. This application was first described shortly after this thiopeptide was first isolated [174]. Nowadays, tons of pre-mixed animal food with the antibiotic are produced and commercialized.
Given the bad pharmacokinetic profile of thiopeptides, various analogs have been produced, mainly to improve their aqueous solubility. LFF571 (Figure 1), developed by Novartis, is a semi-synthetic analog of GE2270A currently under clinical trials for the treatment of Clostridium difficile intestinal infections in humans [9,175,176].
When considering the great therapeutic potential of thiopeptides and the efforts carried out by many research groups, it can be expected that their discoveries will lead to the development of analogs suitable for the treatment of more threatening systemic infections. The discovery of new thiopeptides might also hold the key to the future development of these natural products into suitable drugs. Moreover, the still vastly unexplored oceans are likely to become a fruitful source of the new thiopeptides to come.
3. Summary
A promising new family of antibacterial compounds, possessing other interesting bio-activities has been revised. Pioneering works from their isolation until the arrival to the marked though its biosynthesis, bio-activity and synthesis show that the thiopeptide are a wide range of investigation for scientists from different areas.
Acknowledgments
We gratefully acknowledge support from the Spanish Science and Innovation Ministry, CICYT (CTQ2012-30930) and the Generalitat de Catalunya (2009SGR 1024). XJ thanks ISCIII for a PFIS grant.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Fischbach M.A., Walsh C.T. Antibiotics for emerging pathogens. Science. 2009;325:1089–1093. doi: 10.1126/science.1176667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Demain A.L., Sanchez S. Microbial drug discovery: 80 years of progress. J. Antibiot. 2009;62:5–16. doi: 10.1038/ja.2008.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kumar N. Lantibiotics a novel antimicrobial agents: A review. Pharma Sci. Monit. 2012;3:2990–3009. [Google Scholar]
- 4.Bagley M.C., Dale J.W., Merritt E.A., Xiong X. Thiopeptide antibiotics. Chem. Rev. 2005;105:685–714. doi: 10.1021/cr0300441. [DOI] [PubMed] [Google Scholar]
- 5.LaMarche M.J., Leeds J.A., Amaral A., Brewer J.T., Bushell S.M., Deng G., Dewhurst J.M., Ding J., Dzink-fox J., Gamber G., et al. Discovery of LFF571: An investigational agent for Clostridium difficile infection. J. Med. Chem. 2012;6:2376–2387. doi: 10.1021/jm201685h. [DOI] [PubMed] [Google Scholar]
- 6.Su T.L. Micrococcin, an antibacterial substance formed by a strain of Micrococcus. Br. J. Exp. Pathol. 1948;29:473–481. [PMC free article] [PubMed] [Google Scholar]
- 7.Carnio M.C., Stachelhaus T., Francis K.P., Scherer S. Pyridinyl polythiazole class peptide antibiotic micrococcin P1, secreted by foodborne Staphylococcus equorum WS2733, is biosynthesized nonribosomally. Eur. J. Biochem. 2001;268:6390–6340. doi: 10.1046/j.0014-2956.2001.02591.x. [DOI] [PubMed] [Google Scholar]
- 8.Pagano J.F., Weinstein M.J., Stout H.A., Donovick R. Thiostrepton, a new antibiotic. I. In vitro studies. Antibiot. Ann. 1955–1956;3:554–559. [PubMed] [Google Scholar]
- 9.Vandeputte J., Dutcher J.D. Thiostrepton, a new antibiotic. II. Isolation and chemical characterization. Antibiot. Ann. 1955–1956;3:560–561. [PubMed] [Google Scholar]
- 10.Steinberg B.A., Jambor W.P., Suydam L.O., Soriano A. Thiostrepton, a new antibiotic. III. In vivo studies. Antibiot. Ann. 1955–1956;3:562–565. [PubMed] [Google Scholar]
- 11.Cron M.J., Whitehead D.F., Hooper I.R., Heinemann B., Lein J. Bryamycin, a new antibiotic. Antibiot. Chemother. 1956;6:63–67. [PubMed] [Google Scholar]
- 12.Nagai K., Kamigiri K., Arao N., Suzumura K.-I., Kawano Y., Yamaoka M., Zhang H., Watanabe M., Suzuki K. YM-266183 and YM-266184, novel thiopeptide antibiotics produced by Bacillus cereus isolated from a marine sponge. I. Taxonomy, fermentation, isolation, physico-chemical properties and biological properties. J. Antibiot. 2003;56:123–128. doi: 10.7164/antibiotics.56.123. [DOI] [PubMed] [Google Scholar]
- 13.Jayasuriya H., Herath K., Ondeyka J.G., Zhang C., Zink D.L., Brower M., Gailliot F.P., Greene J., Birdsall G., Venugopal J., et al. Isolation and structure elucidation of thiazomycin, a potent thiazolyl peptide antibiotic from Amycolatopsis fastidiosa. J. Antibiot. 2007;60:554–564. doi: 10.1038/ja.2007.70. [DOI] [PubMed] [Google Scholar]
- 14.Singh S.B., Occi J., Jayasuriya H., Herath K., Motyl M., Dorso K., Gill C., Hickey E., Overbye K.M., Barrett J.F. Antibacterial evaluations of thiazomycin—a potent thiazolyl peptide antibiotic from Amycolatopsis fastidiosa. J. Antibiot. 2007;60:565–571. doi: 10.1038/ja.2007.71. [DOI] [PubMed] [Google Scholar]
- 15.Zhang C., Zink D.L., Ushio M., Burgess B., Onishi R., Masurekar P., Barrett J.F., Singh S.B. Isolation, structure, and antibacterial activity of thiazomycin A, a potent thiazolyl peptide antibiotic from Amycolatopsis fastidiosa. Bioorg. Med. Chem. 2008;16:8818–8823. doi: 10.1016/j.bmc.2008.08.079. [DOI] [PubMed] [Google Scholar]
- 16.Zhang C., Herath K., Jayasuriya H., Ondeyka J.G., Zink D.L., Occi J., Birdsall G., Venugopal J., Ushio M., Burgess B., et al. Thiazomycins, thiazolyl peptide antibiotics from Amycolatopsis fastidiosa. J. Nat. Prod. 2009;72:841–847. doi: 10.1021/np800783b. [DOI] [PubMed] [Google Scholar]
- 17.Zhang C., Occi J., Masurekar P., Barrett J.F., Zink D.L., Smith S., Onishi R., Ha S., Salazar O., Genilloud O., et al. Isolation, structure, and antibacterial activity of philipimycin, a thiazolyl peptide discovered from Actinoplanes philippinensis MA7347. J. Am. Chem. Soc. 2008;130:12102–12110. doi: 10.1021/ja803183u. [DOI] [PubMed] [Google Scholar]
- 18.Morris R.P., Leeds J.A., Naegeli H.U., Oberer L., Memmert K., Weber E., LaMarche M.J., Parker C.N., Burrer N., Esterow S., et al. Ribosomally synthesized thiopeptide antibiotics targeting elongation factor Tu. J. Am. Chem. Soc. 2009;131:5946–5955. doi: 10.1021/ja900488a. [DOI] [PubMed] [Google Scholar]
- 19.Engelhardt K., Degnes K.F., Kemmler M., Bredholt H., Fjaervik E., Klinkenberg G., Sletta H., Ellingsen T.E., Zotchev S.B. Production of a new thiopeptide antibiotic, TP-1161, by a marine Nocardiopsis species. Appl. Environ. Microbiol. 2010;76:4969–4976. doi: 10.1128/AEM.00741-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Engelhardt K., Degnes K.F., Zotchev S.B. Isolation and characterization of the gene cluster for biosynthesis of the thiopeptide antibiotic TP-1161. Appl. Environ. Microbiol. 2010;76:7093–7101. doi: 10.1128/AEM.01442-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cañedo Hernández L.M., Romero Millan F., Fernández Medarde A., Fernández Chimeno R.I., Hidalgo Villar J.C. Peptides as bioactive compounds. WO 2012062906A1. Patent. 2012 May 18;
- 22.Palomo S., González I., de la Cruz M., Martín J., Tormo J.R., Anderson M., Hill R.T., Vicente F., Reyes F., Genilloud O. Sponge-derived Kocuria and Micrococcus spp. as sources of the new thiazolyl peptide antibiotic kocurin. Mar. Drugs. 2013;11:1071–7086. doi: 10.3390/md11041071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bodanzky M., Fried J., Sheehan J.T., Williams N.J., Alicino J., Cohen A.I., Keeler B.T., Birkhimer C.A. Thiostrepton. degradation products and structural features. J. Am. Chem. Soc. 1964;86:2478–2490. doi: 10.1021/ja01066a035. [DOI] [Google Scholar]
- 24.Anderson B., Hodgkin D.C., Viswamitra M.A. Structure of thiostrepton. Nature. 1970;225:133–235. doi: 10.1038/225233a0. [DOI] [PubMed] [Google Scholar]
- 25.Hall G.E., Sheppard N., Walker J. Chemistry of micrococcin P. X. Proton magnetic resonance spectrum of dimethyl micrococcinate, and the probable mode of biosynthesis of micrococcinic acid. J. Chem. Soc. C. 1966;16:1371–1373. doi: 10.1039/j39660001371. [DOI] [PubMed] [Google Scholar]
- 26.Ciufolini M.A., Lefranc D. Micrococcin P: Structure, biology and synthesis. Nat. Prod. Rep. 2010;27:330–342. doi: 10.1039/b919071f. [DOI] [PubMed] [Google Scholar]
- 27.Brookes P., Fuller A.T., Walker J. Chemistry of micrococcin P. Part I. J. Chem. Soc. 1957:689–699. doi: 10.1039/JR9570000689. [DOI] [Google Scholar]
- 28.Mijović M.P.V., Walker J. Chemistry of micrococcin P. Part II. J. Chem. Soc. 1960:909–916. doi: 10.1039/JR9600000909. [DOI] [Google Scholar]
- 29.Brookes P., Clark R.J., Fuller A.T., Mijović M.P.V., Walker J. Chemistry of micrococcin P. Part III. J. Chem. Soc. 1960:916–925. doi: 10.1039/JR9600000916. [DOI] [Google Scholar]
- 30.Walker J., Olesker A., Valente L., Rabanal R., Lukacs G. Total structure of the polythiazole-containing antibiotic micrococcin P. A carbon-13 nuclear magnetic resonance study. J. Chem. Soc. Chem. Commun. 1977:706–708. doi: 10.1039/C39770000706. [DOI] [Google Scholar]
- 31.Bycroft B.W., Gowland M.S. The structures of the highly modified peptide antibiotics micrococcin P1 and P2. J. Chem. Soc. Chem. Commun. 1978:256–258. doi: 10.1039/C39780000256. [DOI] [Google Scholar]
- 32.Fenet B., Pierre F., Cundliffe M., Ciufolini M.A. The constitution of micrococcin P1: The Bycroft—Gowland hypothesis confirmed. Tetrahedron Lett. 2002;43:2367–2370. doi: 10.1016/S0040-4039(02)00266-6. [DOI] [Google Scholar]
- 33.Dean B.M., Mijović M.P.V., Walker J. Chemistry of micrococcin P. Part VI. Racemisation of 2-(l-amino-2-methylpropyl)thiazole-4-carboxylic acid, and related studies. J. Chem. Soc. 1961:3394–3400. doi: 10.1039/JR9610003394. [DOI] [Google Scholar]
- 34.Shin C., Okumura K., Shigekuni M., Nakamura Y. Total synthesis of antibiotic, micrococcin P, from 2,3,6-polyazolesubstituted pyridine skeleton [fragmentA–C] Chem. Lett. 1998;27:139–140. [Google Scholar]
- 35.Okumura K., Ito A., Yoshioka D., Shin C. Total synthesis of macrocyclic antibiotic, Micrococcin P1. Heterocycles. 1998;48:1319–1324. doi: 10.3987/COM-98-8152. [DOI] [Google Scholar]
- 36.Ciufolini M.A., Shen Y.C. Synthesis of the Bycroft-Gowland structure of micrococcin P1. Org. Lett. 1999;1:1843–1846. doi: 10.1021/ol991115e. [DOI] [PubMed] [Google Scholar]
- 37.Lefranc D., Ciufolini M.A. Total synthesis and stereochemical assignment of micrococcin P1. Angew. Chem. Int. Ed. 2009;48:4198–4201. doi: 10.1002/anie.200900621. [DOI] [PubMed] [Google Scholar]
- 38.Tori K., Tokura K., Yoshimura Y., Terui Y., Okabe K., Otsuka H., Matsushita K., Inagaki F., Miyazama T. Structures of siomycin-B and -C and thiostrepton-B determined by NMR spectroscopy and carbon-13 signal assignments of siomycins, thiostreptons and thiopeptin-Ba. J. Antibiot. 1981;36:124–129. doi: 10.7164/antibiotics.34.124. [DOI] [PubMed] [Google Scholar]
- 39.Puar M.S., Ganguly A.K., Afonso A., Brambilla R., Mangiaracina P., Sarre O., MacFarlane R.D. Sch 18640. A new thiostrepton-type antibiotic. J. Am. Chem. Soc. 1981;36:5231–5233. [Google Scholar]
- 40.Muramatsu I., Motoki Y., Aoyama M. Amino acids and derivatives of thiazole-4-carboxylic acid as constituents of thiopeptin B. J. Antibiot. 1977;30:383–387. doi: 10.7164/antibiotics.30.383. [DOI] [PubMed] [Google Scholar]
- 41.Puar M.S., Chan T.M., Hedge V., Patel M., Bartner P., Ng K.J., Pramanik B.N. Sch 40832: A novel thiostrepton from Micromonospora carbonacea. J. Antibiot. 1998;51:221–224. doi: 10.7164/antibiotics.51.221. [DOI] [PubMed] [Google Scholar]
- 42.Yun B.-S., Hidaka T., Furihata K., Seto H. Microbial metabolites with tipA promoter inducing activity. III. thioxamycin and its novel derivative, thioactin, two thiopeptides produced by Streptomyces sp. DP94. J. Antibiot. 1994;47:1541–1545. doi: 10.7164/antibiotics.47.1541. [DOI] [PubMed] [Google Scholar]
- 43.Favret M.E., Paschal J.W., Elzey T.K., Boeck L.D. Biosynthesis of thiopeptide antibiotic A10255, incorporation of isotopically-labeled precursors. J. Antibiot. 1992;45:1499–1511. doi: 10.7164/antibiotics.45.1499. [DOI] [PubMed] [Google Scholar]
- 44.Fate G.D., Benner C.P., Grode S.H., Gilbertson T.J. The biosynthesis of sulfomycin elucidated by isotopic labeling studies. J. Am. Chem. Soc. 1996;118:11363–11368. doi: 10.1021/ja961864g. [DOI] [Google Scholar]
- 45.Yun B.-S., Seto H. Promoinducin, a novel thiopeptide produced by Streptomyces sp. SF2741. Biosci. Biotechol. Biochem. 1995;59:876–880. doi: 10.1271/bbb.59.876. [DOI] [PubMed] [Google Scholar]
- 46.Yun B.-S., Hidaka T., Furihata K., Seto H. Thiotipin, a novel thiopeptide with a tipA promoter inducing activity produced by Streptomyces sp. DT31. Tetrahedron. 1994;50:11659–11664. doi: 10.1016/S0040-4020(01)85660-X. [DOI] [Google Scholar]
- 47.Constantine K.L., Mueller L., Huang S., Abid S., Lam K.S., Li W., Leet J.E. Conformation and absolute configuration of nocathiacin I determined by NMR spectroscopy and chiral capillary electrophoresis. J. Am. Chem. Soc. 2002;124:7284–7285. doi: 10.1021/ja026249t. [DOI] [PubMed] [Google Scholar]
- 48.Tavecchia P., Gentili P., Kurz M., Sottani C., Bonfichi R., Selva E., Lociuro S., Restelli E., Ciabatti R. Degradation studies of antibiotic MDL 62,879 (GE2270A) and revision of the structure. Tetrahedron. 1995;51:4867–4890. doi: 10.1016/0040-4020(95)00171-4. [DOI] [Google Scholar]
- 49.Hughes R.A., Moody C.J. From amino acids to heteroaromatics—thiopeptide antibiotics, nature’s heterocyclic peptides. Angew. Chem. Int. Ed. 2007;46:7930–7954. doi: 10.1002/anie.200700728. [DOI] [PubMed] [Google Scholar]
- 50.Wieland Brown L.C., Acker M.G., Clardy J., Walsh C.T., Fischbach M.A. Thirteen posttranslational modifications convert a 14-residue peptide into the antibiotic thiocillin. Proc. Natl. Acad. Sci. USA. 2009;106:2549–2553. doi: 10.1073/pnas.0900008106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liao R., Duan L., Lei C., Pan H., Ding Y., Zhang Q., Chen D., Shen B., Yu Y., Liu W. Thiopeptide biosynthesis featuring ribosomally synthesized precursor peptides and conserved posttranslational modifications. Chem. Biol. 2009;16:141–147. doi: 10.1016/j.chembiol.2009.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kelly W.L., Pan L., Li C. Thiostrepton biosynthesis: Prototype for a new family of bacteriocins. J. Am. Chem. Soc. 2009;131:4327–4334. doi: 10.1021/ja807890a. [DOI] [PubMed] [Google Scholar]
- 53.Arndt H.-D., Schoof S., Lu J.-Y. Thiopeptide antibiotic biosynthesis. Angew. Chem. Int. Ed. 2009;48:6770–6773. doi: 10.1002/anie.200901808. [DOI] [PubMed] [Google Scholar]
- 54.Li C., Kelly W.L. Recent advances in thiopeptide antibiotic biosynthesis. Nat. Prod. Rep. 2010;27:153–164. doi: 10.1039/b922434c. [DOI] [PubMed] [Google Scholar]
- 55.Walsh C.T., Acker M.G., Bowers A.A. Thiazolyl peptide antibiotic biosynthesis: A cascade of post-translational modifications on ribosomal nascent proteins. J. Biol. Chem. 2010;285:27525–27531. doi: 10.1074/jbc.R110.135970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wang J., Yu Y., Tang K., Liu W., He X., Huang X., Deng Z. Identification and analysis of the biosynthetic gene cluster encoding the thiopeptide antibiotic cyclothiazomycin in Streptomyces hygroscopicus 10–22. Appl. Environ. Microbiol. 2010;76:2335–2344. doi: 10.1128/AEM.01790-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Young T.S., Walsh C.T. Identification of the thiazolyl peptide GE37468 gene cluster from Streptomyces ATCC 55365 and heterologous expression in Streptomyces lividans. Proc. Natl. Acad. Sci. USA. 2011;108:13053–13058. doi: 10.1073/pnas.1110435108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yu Y., Duan L., Zhang Q., Liao R., Ding Y., Pan H., Wendt- E., Tang G., Shen B., Liu W. Nosiheptide biosynthesis featuring a unique indole side ring formation on the characteristic thiopeptide framework. ACS Chem Biol. 2009;4:855–864. doi: 10.1021/cb900133x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ding Y., Yu Y., Pan H., Guo H., Li Y., Liu W. Moving posttranslational modifications forward to biosynthesize the glycosylated thiopeptide nocathiacin I in Nocardia sp. ATCC202099. Mol. BioSyst. 2010;6:1180–1185. doi: 10.1039/c005121g. [DOI] [PubMed] [Google Scholar]
- 60.Yu Y., Guo H., Zhang Q., Duan L., Ding Y., Liao R., Lei C., Shen B., Liu W. NosA catalyzing carboxyl-terminal amide formation in nosiheptide maturation via an enamine dealkylation on the serine-extended precursor. J. Am. Chem. Soc. 2010;132:16324–16326. doi: 10.1021/ja106571g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Liao R., Liu W. Thiostrepton maturation involving a deesterification-amidation way to process the C-terminally methylated peptide backbone. J. Am. Chem. Soc. 2011;133:2852–2855. doi: 10.1021/ja1111173. [DOI] [PubMed] [Google Scholar]
- 62.Bowers A.A., Walsh C.T., Acker M.G. Genetic interception and structural characterization of thiopeptide cyclization precursors from Bacillus cereus. J. Am. Chem. Soc. 2010;132:12182–12184. doi: 10.1021/ja104524q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhang Q., Li Y., Chen D., Yu Y., Duan L., Shen B., Liu W. Radical-mediated enzymatic carbon chain fragmentation-recombination. Nat. Chem. Biol. 2011;7:154–160. doi: 10.1038/nchembio.512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wei M., Deng J., Wang S., Liu N., Chen Y. A simple reverse genetics approach to elucidating the biosynthetic pathway of nocathiacin. Biotechnol. Lett. 2011;33:585–591. doi: 10.1007/s10529-010-0460-0. [DOI] [PubMed] [Google Scholar]
- 65.Zhang Q., Chen D., Lin J., Liao R., Tong W., Xu Z., Liu W. Characterization of NocL involved in thiopeptide nocathiacin I biosynthesis: a [4Fe-4S] cluster and the catalysis of a radical S-adenosylmethionine enzyme. J. Biol. Chem. 2011;286:21287–21294. doi: 10.1074/jbc.M111.224832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pierre S., Guillot A., Benjdia A., Sandström C., Langella P., Berteau O. Thiostrepton tryptophan methyltransferase expands the chemistry of radical SAM enzymes. Nat. Chem. Biol. 2012;8:957–959. doi: 10.1038/nchembio.1091. [DOI] [PubMed] [Google Scholar]
- 67.Duan L., Wang S., Liao R., Liu W. Insights into quinaldic acid moiety formation in thiostrepton biosynthesis facilitating fluorinated thiopeptide generation. Chem. Biol. 2012;19:443–448. doi: 10.1016/j.chembiol.2012.02.008. [DOI] [PubMed] [Google Scholar]
- 68.Malcolmson S.J., Young T.S., Ruby J.G., Skewes-Cox P., Walsh C.T. The posttranslational modification cascade to the thiopeptide berninamycin generates linear forms and altered macrocyclic scaffolds. Proc. Natl. Acad. Sci. USA. 2013;110:8483–8488. doi: 10.1073/pnas.1307111110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Houck D.R., Chen L., Keller P.J., Beale J.M., Floss H.G. Biosynthesis of the modified peptide antibiotic nosiheptide in Streptomyces actuosus. J. Am. Chem. Soc. 1988;110:5800–5806. doi: 10.1021/ja00225a035. [DOI] [Google Scholar]
- 70.Mocek U., Zeng Z., O’Hagan D., Zhou P., Fan L.D.G., Beale J.M., Floss H.G. Biosynthesis of the modified peptide antibiotic thiostrepton in Streptomyces azureus and Streptomyces laurentii. J. Am. Chem. Soc. 1993;115:7992–8001. [Google Scholar]
- 71.Mocek U., Knaggs A.R., Tsuchiya R., Nguyen T., Beale J.M., Floss H.G. Biosynthesis of the modified peptide antibiotic nosiheptide in Streptomyces actuosus. J. Am. Chem. Soc. 1993;115:7557–7558. doi: 10.1021/ja00070a001. [DOI] [Google Scholar]
- 72.Priestley N.D., Smith T.M., Shipley P.R., Floss H.G. Studies on the biosynthesis of thiostrepton: Intermediate on the pathway to the quinaldic acid moiety. Bioorg. Med. Chem. 1996;4:1135–1147. doi: 10.1016/0968-0896(96)00126-5. [DOI] [PubMed] [Google Scholar]
- 73.Frenzel T., Zhou P., Floss H.G. Formation of 2-methyltryptophan in the biosynthesis of thiostrepton: Isolation of S-adenosylmethionine: Tryptophan 2-methyltransferase. Arch. Biochem. Biophys. 1990;278:35–40. doi: 10.1016/0003-9861(90)90227-p. [DOI] [PubMed] [Google Scholar]
- 74.Smith T.M., Priestley N.D., Knaggs A.R., Nguyen T., Floss H.G. 3,4-Dimethylindole-2-carboxylate and 4-(1-hydroxyethyl)quinoline-2-carboxylate activating enzymes from the nosiheptide and thiostrepton producers, Streptomyces actuosus and Streptomyces laurentii. J. Chem. Soc. Chem. Commun. 1993:1612–1614. doi: 10.1039/C39930001612. [DOI] [Google Scholar]
- 75.Zhang Q.I., van der Donk W.A., Liu W. Radical-mediated enzymatic methylation: A tale of two SAMS. Acc. Chem. Res. 2012;45:555–564. doi: 10.1021/ar200202c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Nicolaou K.C., Zak M., Rahimipour S., Estrada A.A., Lee S.H., O’Brate A., Giannakakou P., Ghadiri M.R. Discovery of a biologically active thiostrepton fragment. J. Am. Chem. Soc. 2005;127:15042–15044. doi: 10.1021/ja0552803. [DOI] [PubMed] [Google Scholar]
- 77.Radhakrishnan S.K., Bhat U.G., Hughes D.E., Wang I.-C., Costa R.H., Gartel A.L. Identification of a chemical inhibitor of the oncogenic transcription factor forkhead box M1. Cancer Res. 2006;66:9731–9735. doi: 10.1158/0008-5472.CAN-06-1576. [DOI] [PubMed] [Google Scholar]
- 78.Kwok J.M.-M., Myatt S.S., Marson C.M., Coombes R.C., Constantinidou D., Lam E.W.-F. Thiostrepton selectively targets breast cancer cells through inhibition of forkhead box M1 expression. Mol. Cancer Ther. 2008;7:2022–2032. doi: 10.1158/1535-7163.MCT-08-0188. [DOI] [PubMed] [Google Scholar]
- 79.Bowling B.D., Doudican N., Manga P., Orlow S.J. Inhibition of mitochondrial protein translation sensitizes melanoma cells to arsenic trioxide cytotoxicity via a reactive oxygen species dependent mechanism. Cancer Chemother. Pharmacol. 2008;63:37–43. doi: 10.1007/s00280-008-0705-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Bhat U.G., Zipfel P.A., Tyler D.S., Gartel A.L. Novel anticancer compounds induce apoptosis in melanoma cells. Cell Cycle. 2008;7:1851–1855. doi: 10.4161/cc.7.12.6032. [DOI] [PubMed] [Google Scholar]
- 81.Bhat U.G., Halasi M., Gartel A.L. Thiazole antibiotics target FoxM1 and induce apoptosis in human cancer cells. PLoS One. 2009;4:e5592. doi: 10.1371/journal.pone.0005592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hegde N.S., Sanders D.A., Rodriguez R., Balasubramanian S. The transcription factor FOXM1 is a cellular target of the natural product thiostrepton. Nat. Chem. 2011;3:725–731. doi: 10.1038/nchem.1114. [DOI] [PubMed] [Google Scholar]
- 83.Clough B., Strath M., Preiser P., Denny P., Wilson I.R.J.M. Thiostrepton binds to malarial plastid rRNA. FEBS Lett. 1997;406:123–125. doi: 10.1016/S0014-5793(97)00241-X. [DOI] [PubMed] [Google Scholar]
- 84.Clough B., Rangachari K., Strath M., Preiser P.R., Iain Wilson R.J.M. Antibiotic inhibitors of organellar protein synthesis in Plasmodium falciparum. Protist. 1999;150:189–195. doi: 10.1016/S1434-4610(99)70021-0. [DOI] [PubMed] [Google Scholar]
- 85.Sullivan M., Li J., Kumar S., Rogers M.J., McCutchan T.F. Effects of interruption of apicoplast function on malaria infection, development, and transmission. Mol. Biochem. Parasitol. 2000;109:17–23. doi: 10.1016/S0166-6851(00)00226-7. [DOI] [PubMed] [Google Scholar]
- 86.Schlitzer M. Malaria chemotherapeutics part I: History of antimalarial drug development, currently used therapeutics, and drugs in clinical development. ChemMedChem. 2007;2:944–986. doi: 10.1002/cmdc.200600240. [DOI] [PubMed] [Google Scholar]
- 87.Goodman C.D., Su V., McFadden G.I. The effects of anti-bacterials on the malaria parasite Plasmodium falciparum. Mol. Biochem. Parasitol. 2007;152:181–191. doi: 10.1016/j.molbiopara.2007.01.005. [DOI] [PubMed] [Google Scholar]
- 88.Schoof S., Pradel G., Aminake M.N., Ellinger B., Baumann S., Potowski M., Najajreh Y., Kirschner M., Arndt H.-D. Antiplasmodial thiostrepton derivatives: Proteasome inhibitors with a dual mode of action. Angew. Chem. Int. Ed. 2010;49:3317–3321. doi: 10.1002/anie.200906988. [DOI] [PubMed] [Google Scholar]
- 89.Motoi U., Furukawa S., Abe F., Ushioda M., Fujine K., Johki S., Hatori H., Ueda J. Suppressive effect of antibiotic siomycin on antibody production. J. Antibiot. 2004;57:590–596. doi: 10.7164/antibiotics.57.590. [DOI] [PubMed] [Google Scholar]
- 90.Aoki M., Ohtsuka T., Yamada M., Ohba Y., Yoshizaki H., Yasuno H., Sano T., Watanabe J., Yokose K., Roche N. Cyclothiazomycin, a novel polythiazole-containing peptide with renin inhibitory activity: taxonomy, fermentation, isolation and physico-chemical characterization. J. Antibiot. 1991;44:582–588. doi: 10.7164/antibiotics.44.582. [DOI] [PubMed] [Google Scholar]
- 91.Hashimoto M., Murakami T., Funahashi K., Tokunaga T., Nihei K., Okuno T., Kimura T., Naoki H., Himeno H. An RNA polymerase inhibitor, cyclothiazomycin B1, and its isomer. Bioorg. Med. Chem. 2006;14:8259–8270. doi: 10.1016/j.bmc.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 92.Mizuhara N., Kuroda M., Ogita A., Tanaka T., Usuki Y., Fujita K.-I. Antifungal thiopeptide cyclothiazomycin B1 exhibits growth inhibition accompanying morphological changes via binding to fungal cell wall chitin. Bioorg. Med. Chem. 2011;19:5300–5310. doi: 10.1016/j.bmc.2011.08.010. [DOI] [PubMed] [Google Scholar]
- 93.Baumann S., Schoof S., Harkal S.D., Arndt H.-D. Mapping the binding site of thiopeptide antibiotics by proximity-induced covalent capture. J. Am. Chem. Soc. 2008;130:5664–5666. doi: 10.1021/ja710608w. [DOI] [PubMed] [Google Scholar]
- 94.Harms J.M., Wilson D.N., Schluenzen F., Connell S.R., Stachelhaus T., Zaborowska Z., Spahn C.M.T., Fucini P. Translational regulation via L11: Molecular switches on the ribosome turned on and off by thiostrepton and micrococcin. Mol. Cell. 2008;30:26–38. doi: 10.1016/j.molcel.2008.01.009. [DOI] [PubMed] [Google Scholar]
- 95.Walter J.D., Hunter M., Cobb M., Traeger G., Spiegel P.C. Thiostrepton inhibits stable 70S ribosome binding and ribosome-dependent GTPase activation of elongation factor G and elongation factor 4. Nucleic Acids Res. 2012;40:360–370. doi: 10.1093/nar/gkr623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Selva E., Montanini N., Stella S., Sofiientini A., Gastaldo L., Denaro M. Targeted screening of elonfation Factor Tu binding antibiotics. J. Antibiot. 1997;50:22–26. doi: 10.7164/antibiotics.50.22. [DOI] [PubMed] [Google Scholar]
- 97.Heffron S.E., Jurnak F. Structure of an EF-Tu complex with a thiazolyl peptide antibiotic determined at 2.35 A resolution: Atomic basis for GE2270A inhibition of EF-Tu. Biochemistry. 2000;39:37–45. doi: 10.1021/bi9913597. [DOI] [PubMed] [Google Scholar]
- 98.Parmeggiani A., Nissen P. Elongation factor Tu-targeted antibiotics: four different structures, two mechanisms of action. FEBS Lett. 2006;580:4576–4581. doi: 10.1016/j.febslet.2006.07.039. [DOI] [PubMed] [Google Scholar]
- 99.Murakami T., Holt T.G., Thompson C.J. Thiostrepton-induced gene expression in Streptomyces lividans. J. Bacteriol. 1989;171:1459–1466. doi: 10.1128/jb.171.3.1459-1466.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Yun B.-S., Hidaka T., Furihata K., Seto H. Microbial metabolites with tipA promoter inducing activity. II. Geninthiocin, a novel thiopeptide produced by Streptomyces sp. DD84. J. Antibiot. 1994;47:969–975. doi: 10.7164/antibiotics.47.969. [DOI] [PubMed] [Google Scholar]
- 101.Yun B.-S., Hidaka T., Furihata K., Seto H. Promothiocins A and B, novel thiopeptides with tipA promoter inducing activity produced by Streptomyces sp. SF2741. J. Antibiot. 1994;47:510–514. doi: 10.7164/antibiotics.47.510. [DOI] [PubMed] [Google Scholar]
- 102.Lentzen G., Klinck R., Matassova N., Aboul-ela F., Murchie A.I.H. Structural basis for contrasting activities of ribosome binding thiazole antibiotics. Chem. Biol. 2003;10:769–778. doi: 10.1016/S1074-5521(03)00173-X. [DOI] [PubMed] [Google Scholar]
- 103.Chiu M.L., Folcher M., Katoh T., Puglia A.M., Vohradsky J., Yun B.S., Seto H., Thompson C.J. Broad spectrum thiopeptide recognition specificity of the Streptomyces lividans TipAL protein and its role in regulating gene expression. J. Biol. Chem. 1999;274:20578–20586. doi: 10.1074/jbc.274.29.20578. [DOI] [PubMed] [Google Scholar]
- 104.Young T.S., Dorrestein P.C., Walsh C.T. Codon randomization for rapid exploration of chemical space in thiopeptide antibiotic variants. Chem. Biol. 2012;19:1600–1610. doi: 10.1016/j.chembiol.2012.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lewis R.J., Hughes R.A., Alcaraz L., Thompson S.P., Moody C.J. Solution structures of thiopeptide antibiotics. Chem. Commun. 2006:4215–4217. doi: 10.1039/b609282a. [DOI] [PubMed] [Google Scholar]
- 106.Kim J. Cancer therapeutic agent comprising thiopeptide with multiple thiazole rings. WO02066046. Patent. 2002 Aug 29;
- 107.Bauer R.A., Wurst J.M., Tan D.S. Expanding the range of “druggable” targets with natural product-based libraries: An academic perspective. Curr. Opin. Chem. Biol. 2010;14:308–314. doi: 10.1016/j.cbpa.2010.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Koehler A.N. A complex task? Direct modulation of transcription factors with small molecules. Curr. Opin. Chem. Biol. 2010;14:331–340. doi: 10.1016/j.cbpa.2010.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Chaubey S., Kumar A., Singh D., Habib S. The apicoplast of Plasmodium falciparum is translationally active. Mol. Microbiol. 2005;56:81–89. doi: 10.1111/j.1365-2958.2005.04538.x. [DOI] [PubMed] [Google Scholar]
- 110.Sleight P., Yusuf S. New evidence on the importance of the renin-angiotensin system in the treatment of higher-risk patients with hypertension. J. Hypertens. 2003;21:1599–1608. doi: 10.1097/00004872-200309000-00001. [DOI] [PubMed] [Google Scholar]
- 111.Prabhakar S.S. Inhibition of renin-angiotensin system: Implications for diabetes control and prevention. J. Invest. Med. 2013;61:551–557. doi: 10.2310/JIM.0b013e31828298ce. [DOI] [PubMed] [Google Scholar]
- 112.Yamazaki M., Katayama Y. Involvement of RAS in clinical pathophysiology. Alzheime’s disease. Angiotensin Res. 2013;10:116–120. [Google Scholar]
- 113.Cooper R., Truumees I., Barrett T., Patel M., Schwartz J., Puar M., Das P., Pramanik B. Saramycetin, a thiazolyl peptide from a Streptomyces sp., chemical characterization and molecular weight determination. J. Antibiot. 1990;43:897–900. doi: 10.7164/antibiotics.43.897. [DOI] [PubMed] [Google Scholar]
- 114.Bowers A.A., Acker M.G., Koglin A., Walsh C.T. Manipulation of thiocillin variants by prepeptide gene replacement: Structure, conformation, and activity of heterocycle substitution mutants. J. Am. Chem. Soc. 2010;132:7519–7527. doi: 10.1021/ja102339q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Yun B.-S., Fujita K.-I., Furihata K., Seto H. Absolute stereochemistry and solution conformation of promothiocins. Tetrahedron. 2001;57:9683–9687. [Google Scholar]
- 116.LaMarche M.J., Leeds J.A., Amaral K., Brewer J.T., Bushell S.M., Dewhurst J.M., Dzink-Fox J., Gangl E., Goldovitz J., Jain A., et al. Antibacterial optimization of 4-aminothiazolyl analogues of the natural product GE2270 A: Identification of the cycloalkylcarboxylic acids. J. Med. Chem. 2011;54:8099–8109. doi: 10.1021/jm200938f. [DOI] [PubMed] [Google Scholar]
- 117.LaMarche M.J., Leeds J.A., Dzink-Fox J., Gangl E., Krastel P., Neckermann G., Palestrant D., Patane M.A., Rann E.M., Tiamfook S., et al. Antibiotic optimization and chemical structure stabilization of thiomuracin A. J. Med. Chem. 2012;55:6934–6941. doi: 10.1021/jm300783c. [DOI] [PubMed] [Google Scholar]
- 118.Bower J., Drysdale M., Hebdon R., Jordan A., Lentzen G., Matassova N., Murchie A., Powles J., Roughley S. Structure-based design of agents targeting the bacterial ribosome. Bioorg. Med. Chem. Lett. 2003;13:2455–2458. doi: 10.1016/S0960-894X(03)00495-5. [DOI] [PubMed] [Google Scholar]
- 119.Jonker H.R.A., Baumann S., Wolf A., Schoof S., Hiller F., Schulte K.W., Kirschner K.N., Schwalbe H., Arndt H.-D. NMR structures of thiostrepton derivatives for characterization of the ribosomal binding site. Angew. Chem. Int. Ed. 2011;50:3308–3312. doi: 10.1002/anie.201003582. [DOI] [PubMed] [Google Scholar]
- 120.Baumann S., Schoof S., Bolten M., Haering C., Takagi M., Shin-ya K., Arndt H.-D. Molecular determinants of microbial resistance to thiopeptide antibiotics. J. Am. Chem. Soc. 2010;132:6973–6981. doi: 10.1021/ja909317n. [DOI] [PubMed] [Google Scholar]
- 121.Kelly T.R., Echevarren A., Chandrakumar N.S., Köksal Y. Synthesis of berninamycinic acid. Tetrahedron Lett. 1984;25:2127–2130. doi: 10.1016/S0040-4039(01)81179-5. [DOI] [Google Scholar]
- 122.Kelly T.R., Jagoe C.T., Gu Z. Synthesis of micrococcinic acid. Tetrahedron Lett. 1991;32:4263–4266. doi: 10.1016/S0040-4039(00)92144-0. [DOI] [Google Scholar]
- 123.Kelly T.R., Lang F. Total synthesis of dimethyl sulfomycinamate. Tetrahedron Lett. 1995;36:5319–5322. [Google Scholar]
- 124.Kelly T.R., Lang F. Total synthesis of dimethyl sulfomycinamate. J. Org. Chem. 1996;61:4623–4633. doi: 10.1021/jo960433d. [DOI] [PubMed] [Google Scholar]
- 125.Bagley M.C., Bashford K.E., Hesketh C.L., Moody C.J. Total synthesis of the thiopeptide promothiocin A. J. Am. Chem. Soc. 2000;122:3301–3313. doi: 10.1021/ja994247b. [DOI] [Google Scholar]
- 126.Hughes R.A., Thompson S.P., Alcaraz L., Moody C.J. Total synthesis of the thiopeptide antibiotic amythiamicin D. J. Am. Chem. Soc. 2005;127:15644–15651. doi: 10.1021/ja0547937. [DOI] [PubMed] [Google Scholar]
- 127.Nicolaou K.C., Safina B.S., Zak M., Estrada A.A., Lee S.H. Total synthesis of thiostrepton, part 1: Construction of the dehydropiperidine/thiazoline-containing macrocycle. Angew. Chem. Int. Ed. 2004;43:5087–5092. doi: 10.1002/anie.200461340. [DOI] [PubMed] [Google Scholar]
- 128.Nicolaou K.C., Zak M., Safina B.S., Lee S.H., Estrada A.A. Total synthesis of thiostrepton, part 2: Construction of the quinaldic acid macrocycle and final stages of the synthesis. Angew. Chem. Int. Ed. 2004;43:5092–5097. doi: 10.1002/anie.200461341. [DOI] [PubMed] [Google Scholar]
- 129.Müller H.M., Delgado O., Bach T. Total synthesis of the thiazolyl peptide GE2270 A. Angew. Chem. Int. Ed. 2007;46:4771–4774. doi: 10.1002/anie.200700684. [DOI] [PubMed] [Google Scholar]
- 130.Delgado O., Müller H.M., Bach T. Concise total synthesis of the thiazolyl peptide antibiotic GE2270 A. Chem. Eur. J. 2008;14:2322–2339. doi: 10.1002/chem.200701823. [DOI] [PubMed] [Google Scholar]
- 131.Ciufolini M.A., Shen Y.C. Studies toward thiostrepton antibiotics : assembly of the central pyridine-thiazole cluster of micrococcins. J. Org. Chem. 1997;3263:3804–3805. [Google Scholar]
- 132.Moody C.J., Hughes R.A., Thompson S.P., Alcaraz L. Biosynthesis inspired Diels-Alder route to pyridines: Synthesis of the 2,3-dithiazolylpyridine core of the thiopeptide antibiotics. Chem. Commun. 2002:1760–1761. doi: 10.1039/B204868J. [DOI] [PubMed] [Google Scholar]
- 133.Bagley M.C., Dale J.W., Xiong X., Bower J. Synthesis of dimethyl sulfomycinamate. Org. Lett. 2003;5:4421–4424. doi: 10.1021/ol0357144. [DOI] [PubMed] [Google Scholar]
- 134.Bagley M.C., Chapaneri K., Dale J.W., Xiong X., Bower J. One-pot multistep Bohlmann-Rahtz heteroannulation reactions: Synthesis of dimethyl sulfomycinamate. J. Org. Chem. 2005;70:1389–1399. doi: 10.1021/jo048106q. [DOI] [PubMed] [Google Scholar]
- 135.Heckmann G., Bach T. Synthesis of the heterocyclic core of the GE 2270 antibiotics and structure elucidation of a major degradation product. Angew. Chem. Int. Ed. 2005;44:1199–1201. doi: 10.1002/anie.200461715. [DOI] [PubMed] [Google Scholar]
- 136.Lu J.-Y., Arndt H.-D. Hetero Diels-Alder synthesis of 3-hydroxypyridines: Access to the nosiheptide core. J. Org. Chem. 2007;72:4205–4212. doi: 10.1021/jo0703505. [DOI] [PubMed] [Google Scholar]
- 137.Martin T., Verrier C., Hoarau C., Marsais F. Direct C-2 arylation of alkyl 4-thiazolecarboxylates: New insights in synthesis of heterocyclic core of thiopeptide antibiotics. Org. Lett. 2008;10:2909–2912. doi: 10.1021/ol801035c. [DOI] [PubMed] [Google Scholar]
- 138.Martin T., Laguerre C., Hoarau C., Marsais F. Highly efficient borylation Suzuki coupling process for 4-bromo-2-ketothiazoles: Straightforward access to micrococcinate and saramycetate esters. Org. Lett. 2009;11:3690–3693. doi: 10.1021/ol901525t. [DOI] [PubMed] [Google Scholar]
- 139.Abe H., Takaishi T., Okuda T., Aoe K., Date T. Methanolysis products of sulfomycin I. Tetrahedron Lett. 1978;19:2791–2794. doi: 10.1016/S0040-4039(01)94864-6. [DOI] [Google Scholar]
- 140.Okumura K., Shigekuni M., Nakamura Y., Shin C. Useful synthesis of 2,3,6-polythiazolesubstituted pyridine skeleton [Fragment A–C] of peptide antibiotic, micrococcin P. Chem. Lett. 1996;25:1025–1026. [Google Scholar]
- 141.Umemura K., Shin I., Yoshimura J., Okumura K., Saito H., Shin C. Synthesis of the central heterocyclic skeleton of an antibiotic, A10255. Chem. Lett. 1997;26:1203–1204. [Google Scholar]
- 142.Umemura K., Noda H., Yoshimura J., Konn A., Yonezawa Y., Shin C. The synthesis of fragment a of an antibiotic, nosiheptide. Tetrahedron Lett. 1997;38:3539–3542. doi: 10.1016/S0040-4039(97)00697-7. [DOI] [Google Scholar]
- 143.Okumura K., Saito H., Shin C., Umemura K., Yoshimura J. Convenient synthesis of the central 3,6-di(2-thiazolyl)-2-(4-thiazolyl)pyridine skeleton of a macrocyclic antibiotic, GE2270 A. Bull. Chem. Soc. Jpn. 1998;71:1863–1870. doi: 10.1246/bcsj.71.1863. [DOI] [Google Scholar]
- 144.Shin C., Okabe A., Ito A., Ito A., Yonezawa Y. Novel synthesis of the main central 2,3,6-trisubstituted pyridine skeleton [Fragment A–B–C] of a macrobicyclic antibiotic, cyclothiazomycin. Bull. Chem. Soc. Jpn. 2002;75:1583–1596. doi: 10.1246/bcsj.75.1583. [DOI] [Google Scholar]
- 145.Shin C., Saito H., Yonezawa Y. Useful synthesis of the main central 2,3,6-trisubstituted pyridine skeleton of various thiostrepton-type macrocyclic antibiotics. Heterocycles. 2003;61:45–50. doi: 10.3987/COM-03-S27. [DOI] [Google Scholar]
- 146.Endoh N., Yonezawa Y., Shin C. Asymmetric synthesis of the main pyridine skeleton for a macrobicyclic antibiotic, cyclothiazomycin. Bull. Chem. Soc. Jpn. 2003;76:643–644. doi: 10.1246/bcsj.76.643. [DOI] [Google Scholar]
- 147.Suzuki S., Yonezawa Y., Shin C. Useful synthesis of fragment A–C–D of a thiostrepton-type macrocyclic antibiotic, thiocilline I. Chem. Lett. 2004;33:814–815. doi: 10.1246/cl.2004.814. [DOI] [Google Scholar]
- 148.Ammer C., Bach T. Total syntheses of the thiopeptides amythiamicin C and D. Chem. Eur. J. 2010;16:14083–14093. doi: 10.1002/chem.201002144. [DOI] [PubMed] [Google Scholar]
- 149.Just-Baringo X., Albericio F., Álvarez M. From 2,6-dichloronicotinic acid to thiopeptide cores. Eur. J. Org. Chem. 2013:6404–6419. doi: 10.1002/ejoc.201300877. [DOI] [Google Scholar]
- 150.Hirokawa Y., Fujiwara I., Suzuki K., Harada H., Yoshikawa T. Synthesis and structure-affinity relationships of novel potent serotonin 5-HT 3 and dopamine D2 receptor antagonistic activity. J. Med. Chem. 2003;46:702–715. doi: 10.1021/jm020270n. [DOI] [PubMed] [Google Scholar]
- 151.Just-Baringo X., Bruno P., Ottesen L.K., Cañedo L.M., Albericio F., Álvarez M. Total synthesis and stereochemical assignment of baringolin. Angew. Chem. Int. Ed. 2013;52:7818–7821. doi: 10.1002/anie.201302372. [DOI] [PubMed] [Google Scholar]
- 152.Just-Baringo X., Bruno P., Albericio F., Álvarez M. Highly efficient, multigram and enantiopure synthesis of (S)-2-(2,4′-bithiazol-2-yl)pyrrolidine. Tetrahedron Lett. 2011;52:5435–5437. doi: 10.1016/j.tetlet.2011.07.128. [DOI] [Google Scholar]
- 153.Aulakh V.S., Ciufolini M.A. Total synthesis and complete structural assignment of thiocillin I. J. Am. Chem. Soc. 2011;133:5900–5904. doi: 10.1021/ja110166x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Moody C.J., Bagley M.C. Studies on thiopeptide antibiotics: synthesis of an oxazole-thiazole-pyridine fragment related to promothiocin A. Synlett. 1998;1998:361–362. doi: 10.1055/s-1998-1670. [DOI] [Google Scholar]
- 155.Bagley M.C., Dale J.W., Hughes D.D., Ohnesorge M., Phillips N.G., Bower J. Synthesis of pyridines and pyrido[2,3-d]pyrimidines by the Lewis acid catalyzed Bohlmann-Rahtz heteroannulation reaction. Synlett. 2001;2001:1523–1526. doi: 10.1055/s-2001-17447. [DOI] [Google Scholar]
- 156.Xiong X., Bagley M.C., Chapaneri K. A new mild method for the one-pot synthesis of pyridines. Tetrahedron Lett. 2004;45:6121–6124. doi: 10.1016/j.tetlet.2004.06.061. [DOI] [Google Scholar]
- 157.Bagley M.C., Dale J.W., Jenkins R.L., Bower J. First synthesis of an amythiamicin pyridine cluster. Chem. Commun. 2004:102–103. doi: 10.1039/B310944E. [DOI] [PubMed] [Google Scholar]
- 158.Bagley M.C., Xiong X. Stereoselective Synthesis of the β-lactam hydrolysate of the thiopeptide cyclothiazomycin. Org. Lett. 2004;6:3401–3404. doi: 10.1021/ol0485870. [DOI] [PubMed] [Google Scholar]
- 159.Merritt E., Bagley M. Convergent synthesis of the central heterocyclic domain of micrococcin P1. Synlett. 2007;2007:954–958. doi: 10.1055/s-2007-973870. [DOI] [Google Scholar]
- 160.Moody C.J., Bagley M.C. The first synthesis of promothiocin A. Chem. Commun. 1998;4:2049–2050. doi: 10.1039/a805762a. [DOI] [Google Scholar]
- 161.Bagley M.C., Moody C.J. Synthesis of bioactive cyclic peptides; Proceedings of 216th ACS National Meeting; Boston, MA, USA. 23–27 August 1998. [Google Scholar]
- 162.Higashibayashi S., Hashimoto K., Nakata M. Synthetic studies on the thiostrepton family of peptide antibiotics: synthesis of the tetrasubstituted dehydropiperidine and piperidine cores. Tetrahedron Lett. 2002;43:105–110. doi: 10.1016/S0040-4039(01)02090-1. [DOI] [Google Scholar]
- 163.Higashibayashi S., Kohno M., Goto T., Suzuki K., Mori T., Hashimoto K., Nakata M. Synthetic studies on thiostrepton family of peptide antibiotics: synthesis of the pentapeptide segment containing dihydroxyisoleucine, thiazoline and dehydroamino acid. Tetrahedron Lett. 2004;45:3707–3712. doi: 10.1016/j.tetlet.2004.03.099. [DOI] [Google Scholar]
- 164.Mori T., Tohmiya H., Satouchi Y., Higashibayashi S., Hashimoto K., Nakata M. Synthetic studies on thiostrepton family of peptide antibiotics: synthesis of the cyclic core portion containing the dehydropiperidine, dihydroquinoline, l-valine, and masked dehydroalanine segments. Tetrahedron Lett. 2005;46:6423–6427. doi: 10.1016/j.tetlet.2005.07.122. [DOI] [Google Scholar]
- 165.Mori T., Higashibayashi S., Goto T., Kohno M., Satouchi Y., Shinko K., Suzuki K., Suzuki S., Tohmiya H., Hashimoto K., et al. Total synthesis of siomycin A. Tetrahedron Lett. 2007;48:1331–1335. doi: 10.1016/j.tetlet.2006.12.121. [DOI] [Google Scholar]
- 166.Mori T., Higashibayashi S., Goto T., Kohno M., Satouchi Y., Shinko K., Suzuki K., Suzuki S., Tohmiya H., Hashimoto K., et al. Total synthesis of siomycin A: Construction of synthetic segments. Chem. Asian J. 2008;3:984–1012. doi: 10.1002/asia.200800032. [DOI] [PubMed] [Google Scholar]
- 167.Nicolaou K.C., Nevalainen M., Safina B.S., Zak M., Bulat S. A biomimetically inspired synthesis of the dehydropiperidine domain of thiostrepton. Angew. Chem. Int. Ed. 2002;41:1941–1945. doi: 10.1002/1521-3773(20020603)41:11<1941::AID-ANIE1941>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 168.Nicolaou K.C. How thiostrepton was made in the laboratory. Angew. Chem. Int. Ed. 2012;51:12414–12436. doi: 10.1002/anie.201205576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Nicolaou K.C., Zou B., Dethe D.H., Li D.B., Chen D.Y.-K. Total synthesis of antibiotics GE2270A and GE2270T. Angew. Chem. Int. Ed. 2006;45:7786–7792. doi: 10.1002/anie.200602798. [DOI] [PubMed] [Google Scholar]
- 170.Nicolaou K.C., Dethe D.H., Chen D.Y.-K. Total syntheses of amythiamicins A, B and C. Chem. Commun. 2008:2632–2634. doi: 10.1039/B805069B. [DOI] [PubMed] [Google Scholar]
- 171.Hughes R.A., Thompson S.P., Alcaraz L., Moody C.J. Total synthesis of the thiopeptide amythiamicin D. Chem. Commun. 2004:946–948. doi: 10.1039/B401580K. [DOI] [PubMed] [Google Scholar]
- 172.Lu J.-Y., Riedrich M., Mikyna M., Arndt H.-D. Aza-Wittig-supported synthesis of the A ring of nosiheptide. Angew. Chem. Int. Ed. 2009;48:8137–8140. doi: 10.1002/anie.200903477. [DOI] [PubMed] [Google Scholar]
- 173.Zou Y., Liu Q., Deiters A. Synthesis of the pyridine core of cyclothiazomycin. Org. Lett. 2011;13:4352–4355. doi: 10.1021/ol201682k. [DOI] [PubMed] [Google Scholar]
- 174.Keppens L., De Groote G. Effect of a new antibiotic “nosiheptide” in broiler feeds. Rev. l’Agric. 1979;32:159–177. [Google Scholar]
- 175.Trzasko A., Leeds J.A., Praestgaard J., LaMarche M.J., McKenney D. Efficacy of LFF571 in a hamster model of Clostridium difficile infection. Antimicrob. Agents Chemother. 2012;56:4459–4462. doi: 10.1128/AAC.06355-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Leeds J.A., Sachdeva M., Mullin S., Dzink-Fox J., LaMarche M.J. Mechanism of action of and mechanism of reduced susceptibility to the novel anti-Clostridium difficile compound LFF571. Antimicrob. Agents Chemother. 2012;56:4463–4465. doi: 10.1128/AAC.06354-11. [DOI] [PMC free article] [PubMed] [Google Scholar]