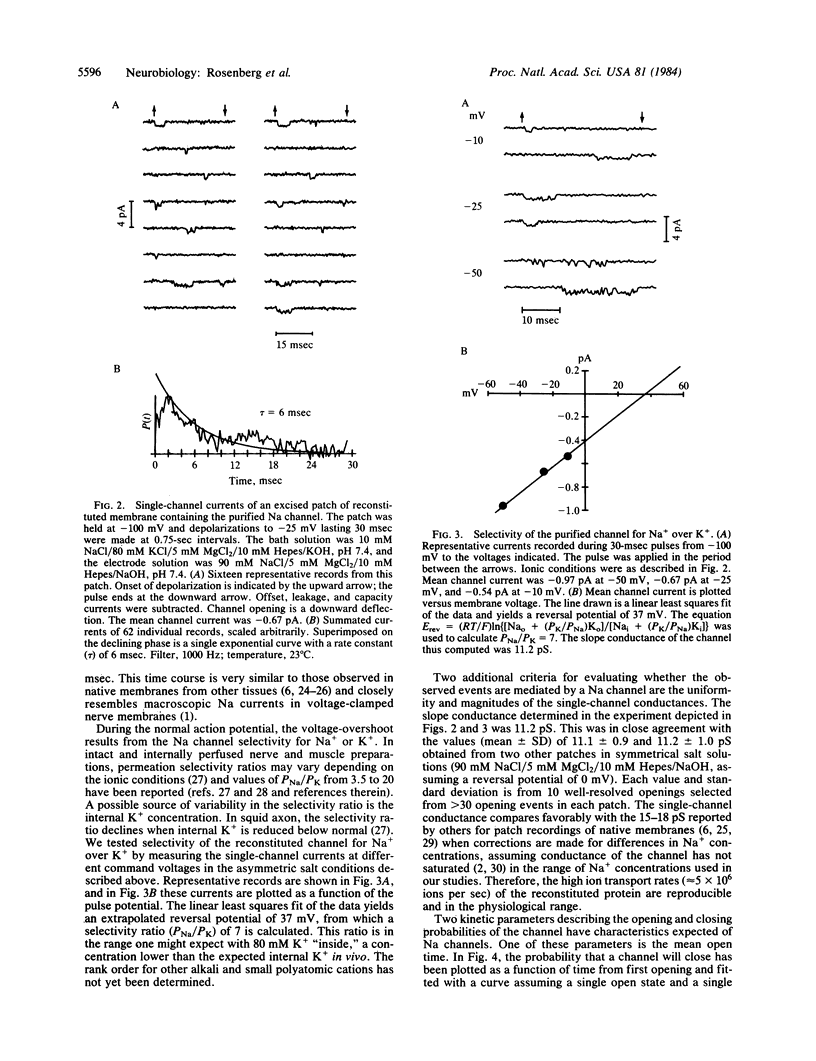
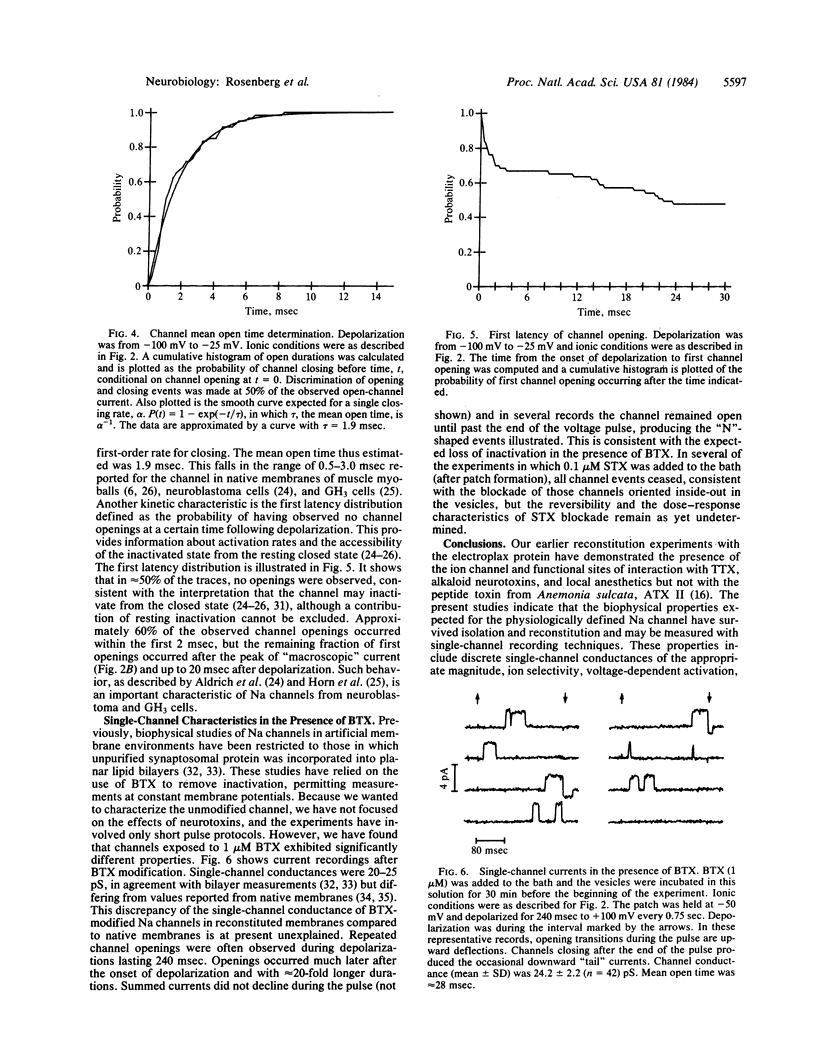
Abstract
The tetrodotoxin-binding protein purified from electroplax of Electrophorus electricus has been reincorporated into multilamellar vesicles that were used for patch recording. When excised patches of these reconstituted membranes were voltage clamped in the absence of neurotoxins, voltage-dependent single-channel currents were recorded. These displayed properties qualitatively and quantitatively similar to those reported for Na channels from nerve and muscle cells, including uniform single-channel conductances of the appropriate magnitude (approximately equal to 11 pS in 95 mM Na+), mean open times of approximately equal to 1.9 msec, and 7-fold selectively for Na+ over K+. Currents averaged from many depolarizations showed initial voltage-dependent activation and subsequent inactivation. In the presence of batrachotoxin, channels were observed with markedly different properties, including conductances of 20-25 pS (95 mM Na+), mean open times of approximately equal to 28 msec, and no indication of inactivation. Collectively, these findings indicate that the tetrodotoxin-binding protein of electroplax is a voltage-regulated sodium channel.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Agnew W. S., Levinson S. R., Brabson J. S., Raftery M. A. Purification of the tetrodotoxin-binding component associated with the voltage-sensitive sodium channel from Electrophorus electricus electroplax membranes. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2606–2610. doi: 10.1073/pnas.75.6.2606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agnew W. S. Voltage-regulated sodium channel molecules. Annu Rev Physiol. 1984;46:517–530. doi: 10.1146/annurev.ph.46.030184.002505. [DOI] [PubMed] [Google Scholar]
- Aldrich R. W., Corey D. P., Stevens C. F. A reinterpretation of mammalian sodium channel gating based on single channel recording. Nature. 1983 Dec 1;306(5942):436–441. doi: 10.1038/306436a0. [DOI] [PubMed] [Google Scholar]
- Armstrong C. M. Sodium channels and gating currents. Physiol Rev. 1981 Jul;61(3):644–683. doi: 10.1152/physrev.1981.61.3.644. [DOI] [PubMed] [Google Scholar]
- Barchi R. L., Cohen S. A., Murphy L. E. Purification from rat sarcolemma of the saxitoxin-binding component of the excitable membrane sodium channel. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1306–1310. doi: 10.1073/pnas.77.3.1306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barchi R. L. Protein components of the purified sodium channel from rat skeletal muscle sarcolemma. J Neurochem. 1983 May;40(5):1377–1385. doi: 10.1111/j.1471-4159.1983.tb13580.x. [DOI] [PubMed] [Google Scholar]
- Cahalan M., Begenisich T. Sodium channel selectivity. Dependence on internal permeant ion concentration. J Gen Physiol. 1976 Aug;68(2):111–125. doi: 10.1085/jgp.68.2.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catterall W. A. Activation of the action potential Na+ ionophore by neurotoxins. An allosteric model. J Biol Chem. 1977 Dec 10;252(23):8669–8676. [PubMed] [Google Scholar]
- Catterall W. A. The molecular basis of neuronal excitability. Science. 1984 Feb 17;223(4637):653–661. doi: 10.1126/science.6320365. [DOI] [PubMed] [Google Scholar]
- French R. J., Worley J. F., 3rd, Krueger B. K. Voltage-dependent block by saxitoxin of sodium channels incorporated into planar lipid bilayers. Biophys J. 1984 Jan;45(1):301–310. doi: 10.1016/S0006-3495(84)84156-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. Currents carried by sodium and potassium ions through the membrane of the giant axon of Loligo. J Physiol. 1952 Apr;116(4):449–472. doi: 10.1113/jphysiol.1952.sp004717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamill O. P., Marty A., Neher E., Sakmann B., Sigworth F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981 Aug;391(2):85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Hartshorne R. P., Catterall W. A. Purification of the saxitoxin receptor of the sodium channel from rat brain. Proc Natl Acad Sci U S A. 1981 Jul;78(7):4620–4624. doi: 10.1073/pnas.78.7.4620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartshorne R. P., Messner D. J., Coppersmith J. C., Catterall W. A. The saxitoxin receptor of the sodium channel from rat brain. Evidence for two nonidentical beta subunits. J Biol Chem. 1982 Dec 10;257(23):13888–13891. [PubMed] [Google Scholar]
- Hille B. The permeability of the sodium channel to metal cations in myelinated nerve. J Gen Physiol. 1972 Jun;59(6):637–658. doi: 10.1085/jgp.59.6.637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holloway P. W. A simple procedure for removal of Triton X-100 from protein samples. Anal Biochem. 1973 May;53(1):304–308. doi: 10.1016/0003-2697(73)90436-3. [DOI] [PubMed] [Google Scholar]
- Horn R., Patlak J., Stevens C. F. Sodium channels need not open before they inactivate. Nature. 1981 Jun 4;291(5814):426–427. doi: 10.1038/291426a0. [DOI] [PubMed] [Google Scholar]
- Horn R., Patlak J., Stevens C. F. The effect of tetramethylammonium on single sodium channel currents. Biophys J. 1981 Nov;36(2):321–327. doi: 10.1016/S0006-3495(81)84734-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horn R., Vandenberg C. A., Lange K. Statistical analysis of single sodium channels. Effects of N-bromoacetamide. Biophys J. 1984 Jan;45(1):323–335. doi: 10.1016/S0006-3495(84)84158-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang L. Y., Moran N., Ehrenstein G. Gating kinetics of batrachotoxin-modified sodium channels in neuroblastoma cells determined from single-channel measurements. Biophys J. 1984 Jan;45(1):313–322. doi: 10.1016/S0006-3495(84)84157-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krueger B. K., Worley J. F., 3rd, French R. J. Single sodium channels from rat brain incorporated into planar lipid bilayer membranes. Nature. 1983 May 12;303(5913):172–175. doi: 10.1038/303172a0. [DOI] [PubMed] [Google Scholar]
- Latorre R., Miller C. Conduction and selectivity in potassium channels. J Membr Biol. 1983;71(1-2):11–30. doi: 10.1007/BF01870671. [DOI] [PubMed] [Google Scholar]
- Miller J. A., Agnew W. S., Levinson S. R. Principal glycopeptide of the tetrodotoxin/saxitoxin binding protein from Electrophorus electricus: isolation and partial chemical and physical characterization. Biochemistry. 1983 Jan 18;22(2):462–470. doi: 10.1021/bi00271a032. [DOI] [PubMed] [Google Scholar]
- Quandt F. N., Narahashi T. Modification of single Na+ channels by batrachotoxin. Proc Natl Acad Sci U S A. 1982 Nov;79(21):6732–6736. doi: 10.1073/pnas.79.21.6732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg R. L., Tomiko S. A., Agnew W. S. Reconstitution of neurotoxin-modulated ion transport by the voltage-regulated sodium channel isolated from the electroplax of Electrophorus electricus. Proc Natl Acad Sci U S A. 1984 Feb;81(4):1239–1243. doi: 10.1073/pnas.81.4.1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigworth F. J., Neher E. Single Na+ channel currents observed in cultured rat muscle cells. Nature. 1980 Oct 2;287(5781):447–449. doi: 10.1038/287447a0. [DOI] [PubMed] [Google Scholar]
- Talvenheimo J. A., Tamkun M. M., Catterall W. A. Reconstitution of neurotoxin-stimulated sodium transport by the voltage-sensitive sodium channel purified from rat brain. J Biol Chem. 1982 Oct 25;257(20):11868–11871. [PubMed] [Google Scholar]
- Tamkun M. M., Catterall W. A. Ion flux studies of voltage-sensitive sodium channels in synaptic nerve-ending particles. Mol Pharmacol. 1981 Jan;19(1):78–86. [PubMed] [Google Scholar]
- Tamkun M. M., Talvenheimo J. A., Catterall W. A. The sodium channel from rat brain. Reconstitution of neurotoxin-activated ion flux and scorpion toxin binding from purified components. J Biol Chem. 1984 Feb 10;259(3):1676–1688. [PubMed] [Google Scholar]
- Tanaka J. C., Eccleston J. F., Barchi R. L. Cation selectivity characteristics of the reconstituted voltage-dependent sodium channel purified from rat skeletal muscle sarcolemma. J Biol Chem. 1983 Jun 25;258(12):7519–7526. [PubMed] [Google Scholar]
- Tank D. W., Miller C., Webb W. W. Isolated-patch recording from liposomes containing functionally reconstituted chloride channels from Torpedo electroplax. Proc Natl Acad Sci U S A. 1982 Dec;79(24):7749–7753. doi: 10.1073/pnas.79.24.7749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsien R. W. Calcium channels in excitable cell membranes. Annu Rev Physiol. 1983;45:341–358. doi: 10.1146/annurev.ph.45.030183.002013. [DOI] [PubMed] [Google Scholar]
- Udenfriend S., Stein S., Böhlen P., Dairman W., Leimgruber W., Weigele M. Fluorescamine: a reagent for assay of amino acids, peptides, proteins, and primary amines in the picomole range. Science. 1972 Nov 24;178(4063):871–872. doi: 10.1126/science.178.4063.871. [DOI] [PubMed] [Google Scholar]
- Weigele J. B., Barchi R. L. Functional reconstitution of the purified sodium channel protein from rat sarcolemma. Proc Natl Acad Sci U S A. 1982 Jun;79(11):3651–3655. doi: 10.1073/pnas.79.11.3651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto D., Yeh J. Z., Narahashi T. Voltage-dependent calcium block of normal and tetramethrin-modified single sodium channels. Biophys J. 1984 Jan;45(1):337–344. doi: 10.1016/S0006-3495(84)84159-4. [DOI] [PMC free article] [PubMed] [Google Scholar]



