Abstract
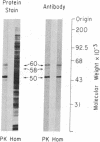
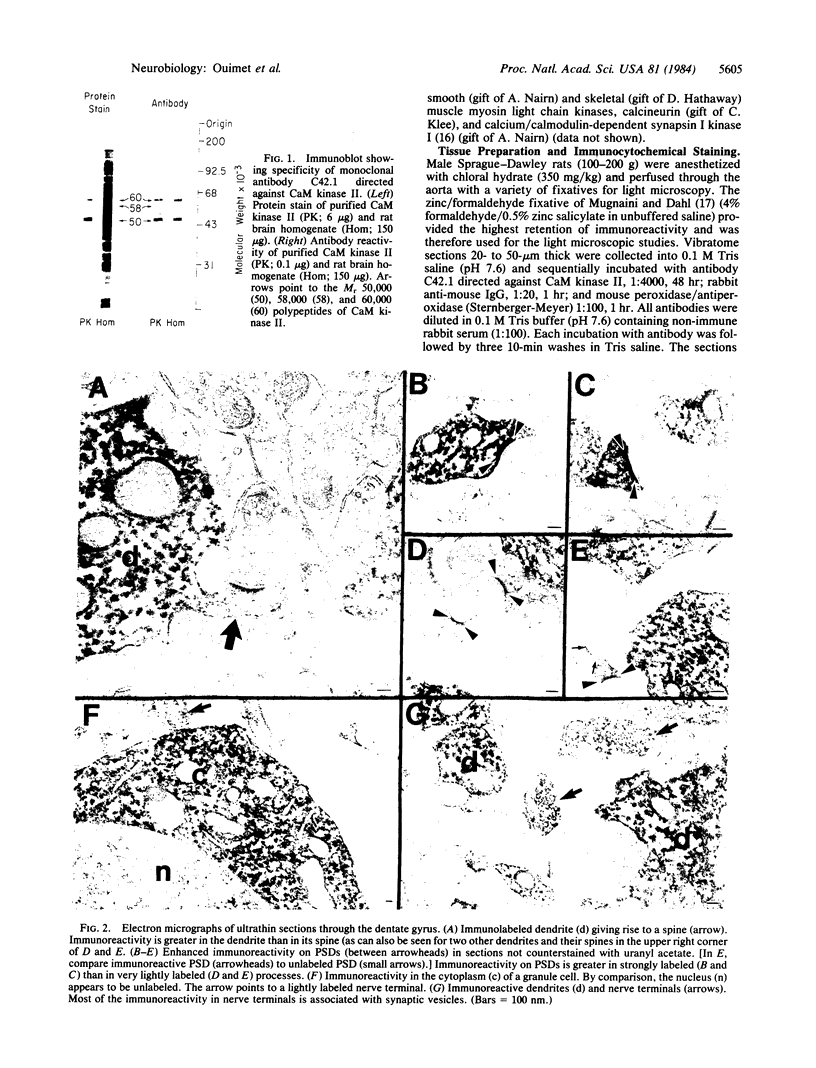
Calcium/calmodulin-dependent protein kinase II (CaM kinase II) is a prominent enzyme in mammalian brain capable of phosphorylating a variety of substrate proteins. In the present investigation, the subcellular and regional distribution of CaM kinase II has been studied by light and electron microscopic immunocytochemistry using an antibody that recognizes the Mr 50,000 and 60,000/58,000 subunits of the enzyme. Light microscopy demonstrates strong immunoreactivity in neuronal somata and dendrites and weak immunoreactivity in axons. Electron microscopy, in addition to confirming light microscopic observations, reveals moderate immunoreactivity in spines and weak immunoreactivity in nerve terminals. An accumulation of immunoreaction product is also present on postsynaptic densities. The presence of CaM kinase II in diverse structures throughout the neuron supports the view that this enzyme may be involved in mediating a variety of calcium-dependent physiological processes. CaM kinase II immunoreactivity is present in neurons throughout the brain, but a marked regional variation in the strength of the immunoreactivity exists. Overall, there is a gradient of staining intensity with the strongest immunoreactivity in the telencephalon and the weakest in the myelencephalon. The most heavily labeled regions of the telencephalon are the hippocampal formation, lateral septum, cortical regions, neostriatum, and amygdaloid complex.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bennett M. K., Erondu N. E., Kennedy M. B. Purification and characterization of a calmodulin-dependent protein kinase that is highly concentrated in brain. J Biol Chem. 1983 Oct 25;258(20):12735–12744. [PubMed] [Google Scholar]
- De Camilli P., Ueda T., Bloom F. E., Battenberg E., Greengard P. Widespread distribution of protein I in the central and peripheral nervous systems. Proc Natl Acad Sci U S A. 1979 Nov;76(11):5977–5981. doi: 10.1073/pnas.76.11.5977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldenring J. R., McGuire J. S., Jr, DeLorenzo R. J. Identification of the major postsynaptic density protein as homologous with the major calmodulin-binding subunit of a calmodulin-dependent protein kinase. J Neurochem. 1984 Apr;42(4):1077–1084. doi: 10.1111/j.1471-4159.1984.tb12713.x. [DOI] [PubMed] [Google Scholar]
- Kelly P. T., McGuinness T. L., Greengard P. Evidence that the major postsynaptic density protein is a component of a Ca2+/calmodulin-dependent protein kinase. Proc Natl Acad Sci U S A. 1984 Feb;81(3):945–949. doi: 10.1073/pnas.81.3.945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy M. B., Bennett M. K., Erondu N. E. Biochemical and immunochemical evidence that the "major postsynaptic density protein" is a subunit of a calmodulin-dependent protein kinase. Proc Natl Acad Sci U S A. 1983 Dec;80(23):7357–7361. doi: 10.1073/pnas.80.23.7357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy M. B., McGuinness T., Greengard P. A calcium/calmodulin-dependent protein kinase from mammalian brain that phosphorylates Synapsin I: partial purification and characterization. J Neurosci. 1983 Apr;3(4):818–831. doi: 10.1523/JNEUROSCI.03-04-00818.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Köhler G., Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975 Aug 7;256(5517):495–497. doi: 10.1038/256495a0. [DOI] [PubMed] [Google Scholar]
- McGuinness T. L., Lai Y., Greengard P., Woodgett J. R., Cohen P. A multifunctional calmodulin-dependent protein kinase. Similarities between skeletal muscle glycogen synthase kinase and a brain synapsin I kinase. FEBS Lett. 1983 Nov 14;163(2):329–334. doi: 10.1016/0014-5793(83)80846-1. [DOI] [PubMed] [Google Scholar]
- Miller P., Walter U., Theurkauf W. E., Vallee R. B., De Camilli P. Frozen tissue sections as an experimental system to reveal specific binding sites for the regulatory subunit of type II cAMP-dependent protein kinase in neurons. Proc Natl Acad Sci U S A. 1982 Sep;79(18):5562–5566. doi: 10.1073/pnas.79.18.5562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mugnaini E., Dahl A. L. Zinc-aldehyde fixation for light-microscopic immunocytochemistry of nervous tissues. J Histochem Cytochem. 1983 Dec;31(12):1435–1438. doi: 10.1177/31.12.6355290. [DOI] [PubMed] [Google Scholar]
- Pintar J. E., Levitt P., Salach J. I., Weyler W., Rosenberg M. B., Breakefield X. O. Specificity of antisera prepared against pure bovine MAO-B. Brain Res. 1983 Oct 3;276(1):127–139. doi: 10.1016/0006-8993(83)90554-1. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walaas S. I., Nairn A. C., Greengard P. Regional distribution of calcium- and cyclic adenosine 3':5'-monophosphate-regulated protein phosphorylation systems in mammalian brain. I. Particulate systems. J Neurosci. 1983 Feb;3(2):291–301. doi: 10.1523/JNEUROSCI.03-02-00291.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walaas S. I., Nairn A. C., Greengard P. Regional distribution of calcium- and cyclic adenosine 3':5'-monophosphate-regulated protein phosphorylation systems in mammalian brain. II. Soluble systems. J Neurosci. 1983 Feb;3(2):302–311. doi: 10.1523/JNEUROSCI.03-02-00302.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]