Abstract
The canonical WNT pathway regulates the stability of the proto-oncogene β-catenin and is aberrantly activated in many cancer types. Studies in a wide range of experimental models confirm that β-catenin activity is required for tumor initiation in cancers where this pathway is deregulated. However, to date this pathway has proven to be challenging to target therapeutically. Moreover, several lines of evidence suggest that other components and regulators of β-catenin exist. Here we will describe recent structural and functional studies describing genomic alterations and new regulators of β-catenin that lead to aberrant activation of the WNT/β-catenin pathway. These findings provide new insights into the biology of WNT/β-catenin signaling and suggest potential therapeutic opportunities.
Keywords: WNT, cancer, β-catenin, YAP1, synthetic lethality
The canonical WNT pathway
WNT signaling is involved in diverse processes including embryonic development, maintenance of tissue homeostasis and cancer pathogenesis. The Wingless (WNT) gene was first identified in a random mutagenesis screen in Drosophila melanogaster [1]. Mutations in WNT resulted in loss of wing development and defects in larval segmentation. Subsequent experiments demonstrated that mutations in WNT or ARM (the D. melanogaster β-catenin ortholog) result in a similar segmentation phenotype [1]. Genetic complementation screens and biochemical studies led to the finding that WNT signaling inactivates a cytosolic protein complex (the destruction complex) that regulates β-catenin stability [1].
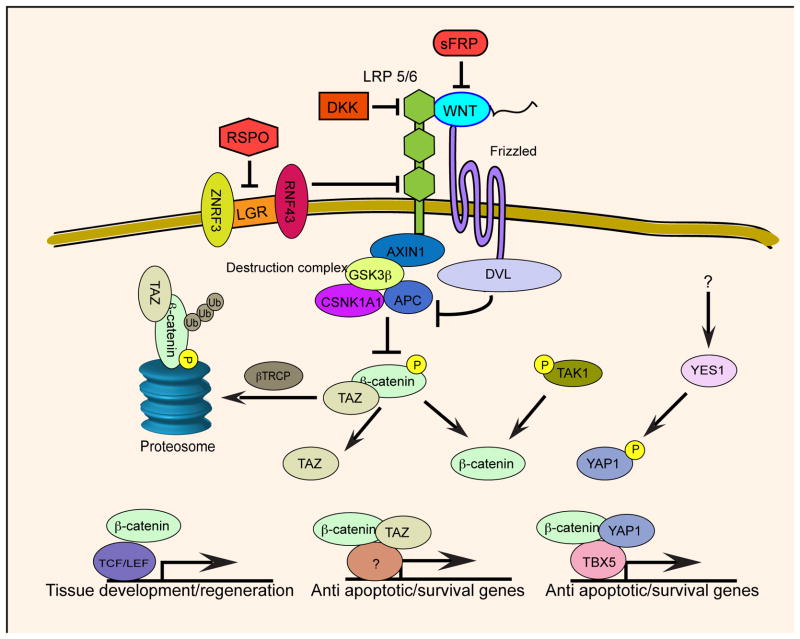
Binding of WNT ligands to the FZD/LPR6 receptor inactivates the destruction complex, which stabilizes β-catenin [2]. The destruction complex, composed of APC, AXIN1, GSK3β and CSNK1A1, phosphorylates serine residues in β-catenin leading to its ubiquitination by βTRCP and degradation by the proteasome. Activation of the WNT pathway was previously thought to result in disassembly of the destruction complex. However, recent work has shown that upon binding of WNT to the FZD/LPR6 receptor, the destruction complex remains intact and bound to AXIN1. In this state, newly synthesized β-catenin is not recognized by the destruction complex [3]. Stabilized β-catenin directly binds to nucleoproteins Nup62, Nup153 and RanBP2 facilitating its nuclear translocation [4]. In the nucleus, a β-catenin TCF/LEF complex regulates transcription of specific target genes [2] (Figure 1).
Figure 1.
Multiple pathways regulate β-catenin signaling. In normal homeostasis, the destruction complex regulates β-catenin stability (top). Mutations in components of the destruction complex lead to stabilization of β-catenin and activation of various context-dependent transcriptional complexes.
WNT in Development
Early studies implicated WNT signaling as a critical regulator of early vertebrate development [5]. Injection of Xenopus embryos with mRNA encoding positive regulators of the WNT pathway such as WNT1, β-catenin or LEF1 inhibited formation of the anterior posterior axis and resulted in body axis duplication [5]. In consonance with these observations, β-catenin deletion results in early lethality due to defects in the formation of the anterior posterior axis [5]. Studies in flies revealed that a gradient of WNT ligand found throughout the developing embryo determines the anterior-posterior axis by deferential activation of β-catenin [6]. The WNT pathway also regulates cell fate and is essential for differentiation of embryonic stem cells into the endoderm and mesoderm lineages [5]. In adult animals and humans, WNT signaling is also essential to maintain the stem cell compartment in self-renewing organs such as the intestine and hair follicle [5].
During development β-catenin regulates transcription by forming a complex with TCF7L2 (also known as TCF4) or LEF1 [2]. Indeed, TCF7L2 null mice fail to develop the small intestine and die within 24 hours of birth [7]. Moreover, both TCF7L2 and β-catenin have been shown to co-occupy many promoters [8].
WNT activity in cancer pathogenesis
WNT signaling plays an important role in the pathogenesis of several types of human cancers. In a seminal paper, Nusse and Varmus showed that integration of the mouse mammary tumor virus (MMTV) in the mammary epithelium induces mammary tumors by forcing the expression of the proto-oncogene Wnt1 [9]. Moreover, individuals carrying a germline APC mutation develop familial adenomatous polyposis (FAP). Patients affected by FAP develop hundreds of colonic polyps, which progress inevitably to malignant colon cancer [10]. Subsequent studies identified recurrent mutations in components of the WNT signaling pathway in sporadic colon cancers [11].
Recent large-scale sequencing efforts have identified several new recurrent mutations in components of the WNT signaling pathway [11]. Surprisingly, these efforts have identified co-occurrence of mutations in positive and negative regulators of the WNT pathway, suggesting that WNT signaling in cancer is more complex than was previously appreciated. In this review, we will describe emerging evidence suggesting that β-catenin is a modular transcription factor activating distinct context dependent transcriptional programs.
Genomic alterations in components of the WNT/β-catenin pathway
WNT ligands
WNTs are an evolutionary conserved family of secreted glycoproteins [12]. There are 19 distinct human WNTs that bind specific receptors and activate β-catenin-dependent and independent pathways [12], in part explaining the diverse pathways and biological processes regulated by WNT signaling [12]. Indeed, forced expression of 14 of the 19 WNT ligands in human cell lines stabilizes β-catenin [13].
Several WNTs have been reported to be involved in cancer initiation and progression through autocrine or paracrine mechanisms. For example, expression of Wnt1 in murine mammary tissue leads to development of mammary tumors [9]. WNT3A and WNT10B are overexpressed in triple negative breast cancer [14]. In multiple myeloma, secreted WNT3A has been reported to induce migration and invasion through activation of RhoA kinase and PKC [15].
In contrast to these WNTs, other WNT ligands do not appear to contribute to cancer initiation. A commonly used model to assess WNT transforming activity is the mouse mammary cell line C57MG. When transformed, C57MG cells undergo morphological changes and are able to form foci (clusters of cells) when seeded at low densities. A transformed phenotype was observed in C57MG cells following expression of Wnt1, 2, 3A and 5B but not Wnt6, 4 or 5A [16].
Why do some WNT ligands promote β-catenin stabilization yet not promote cancer? One possible explanation is that, in addition to control of β-catenin signaling, specific WNTs in particular contexts activate or inhibit pathways that contribute to or inhibit transformation. In support of this view, Green et al. reported that WNT3A promotes or inhibits tumor growth in a context dependent manner [17]. Specifically, co-injection of fibroblasts overexpressing WNT3A and patient-derived triple negative breast cancer into immunodeficient mice resulted in accelerated growth of some cancers, whereas others were inhibited. It is clear that further work will be necessary to identify the key factors that specify the response to WNT signaling.
R-Spondin (RSPO)
Using Xenopus-derived cDNA expression libraries, Kazanskaya et al. identified RSPO2 as a potent activator of WNT activity [18]. The human RSPO family consists of four members (RSPO1-4) that encode proteins with unique developmental functions [19]. Cells treated with both WNT and RSPO1-4 show a synergistic activation of WNT signaling [18] suggesting that all four RSPO family members potentiate WNT signaling. Furthermore, the expression pattern of RSPO closely resembles that of WNTs in mouse development [20], indicating a physiological role for RSPO in WNT signaling.
Two models have been proposed to explain the synergistic activity of WNTs and RSPO. The first model postulates that RSPO tethers the LGR4/5 and LPR6 receptors enabling more efficient activation of FZD receptors by WNTs [21]. The second model instead suggests that binding of RSPO to the LGR4/5 receptor stabilizes LPR6 by inhibiting the ubiquitin ligases RNF43 and ZNRF3 from targeting LPR6 to proteasomal degradation [22]. Treatment of cell lines with RSPO1 increased the interaction between ZNRF3 and LGR4 and suppressed degradation of the LPR6/FZD receptor [23]. Furthermore, colonic organoids derived from RNF43 and ZNRF3 compound knockout mice can be maintained in culture without addition of RSPO1, further supporting the second model [22].
Using RNA sequencing to analyze 70 tumor-normal primary colon cancers, Seshagiri et al. identified recurrent RSPO2 and RSPO3 fusions in 10% of colon cancer samples [24]. These fusions were mutually exclusive with APC mutations, suggesting that these two alterations activate the same pathway. Indeed, RSPO2 and RSPO3 fusions expressed in 293T cells were secreted into the culture media and synergized with WNT3A to stimulate β-catenin signaling [24].
β-catenin (CTNNB1)
The destruction complex phosphorylates serine and threonine residues on β-catenin leading to its degradation (Figure 1). Specifically, GSK3β phosphorylates Ser33 and Ser37 and CSNK1A1 phosphorylates Thr41 and Ser45 [25]. β-catenin mutations affecting these residues render such mutants refractory to regulation by the destruction complex and lead to constitutive β-catenin signaling. β-catenin mutations are found in various types of cancers, including 30% of hepatocellular and endometrial cancers [11, 26].
Experiments in a wide range of model systems have shown that both activating β-catenin mutations and inactivating APC mutations lead to increased β-catenin transcriptional activity (Figure 1). However, several lines of evidence suggest that APC and β-catenin have distinct functions in tumor development. Re-expression of WT APC in cell lines with inactivating APC mutations results in widespread apoptosis [27]. In contrast, suppression of β-catenin expression in the same cell lines results in a G1 cell cycle arrest and does not induce an apoptotic response [28]. Furthermore, although many tumor types harbor mutations in components of the WNT pathway, the prevalence of these mutations is organ specific [11]. For example, colon cancers harbor frequent APC but not β-catenin mutations, whereas in gastric and endometrial cancers, β-catenin mutations are more common, suggesting that these mutations have additional tissue specific functions.
APC
Germline APC mutations are found in individuals affected by FAP, and the majority of sporadic colon cancers harbor inactivating APC mutations [11]. Although less frequent, APC focal copy number loss is found in a subset of ovarian, lung, breast and colon cancers. APC homozygous deletions are rare (1–3% in colon cancer) suggesting a gain of function role for mutant APC. In support of this notion, Rai et al. reported that APC also regulates cell fate by inhibiting the activity of the DNA demethylase CtBP1 independent of its role in regulating β-catenin [29].
Massively parallel sequencing of 212 primary colorectal cancers identified reoccurring loss of function mutations in AMER1 (also known as FAM123B or WTX), a negative regulator of APC [11]. AMER1 mutations are found in 30% of Wilm’s tumors (a rare childhood kidney cancer) and have been shown to negatively regulate WNT signaling [30]. Interestingly, in colon cancer, APC and AMER1 mutations are not mutually exclusive, suggesting these proteins have distinct roles that are not related to WNT signaling Future studies are necessary determine the roles of AMER1 and APC in tumor progression.
AXIN1, CSNK1A1 and GSK3β
Deletion of APC or GSK3β in the murine intestine activates the WNT/β-catenin pathway, resulting in rapid development of adenomas and early lethality [31]. Similarly, intestine-specific deletion of Csk1a1 in mice leads to nuclear accumulation of β-catenin and increased transcription of WNT target genes [32]. However, in contrast to Apc null mice, these mice display normal gut homeostasis and do not exhibit an increased incidence of adenomas or carcinomas. Acidic β-gal staining, a commonly used marker of senescence [33], was observed in the intestine of Csk1a1 null mice, suggesting that activation of the WNT pathway by deletion of Csk1a1 resulted in oncogene-induced senescence. However, deletion of Csk1a1 failed to induce colon cancer unless accompanied by co-deletion of Trp53 [32], suggesting that inhibition of CSNK1A1 in tumors with wild-type p53 may induce senescence.
Unlike APC, recurrent AXIN1, CSNK1A1 and GSK3β mutations have not been identified in human cancers. AXIN1 deletions are found in a subset of cancers including colon, breast ovarian and melanoma [34].
TCF7L2 (TCF4)
Genomic characterization of human colorectal tumors identified recurrent inactivating mutations and focal copy number losses involving TCF7L2 that often co-occur with APC mutations [11]. Specifically, 26 of 212 colorectal cancers (12%) harbored TCF7L2 mutations. Surprisingly, of these 26 mutations, five samples had inactivating TCF7L2 mutations (frameshift or non-sense mutations) that result in truncation of TCF7L2 DNA binding domain and would be predicted to fail to activate β-catenin [35]. In addition, co-occurrence of TCF7L2 homozygous deletion and APC inactivating mutations was found in 4 tumors. These observations demonstrate that in a significant fraction of WNT deregulated colon cancers TCF7L2 is deleted or functionally inactivated.
Whole genome sequencing analysis on a subset of these cancers identified recurring translocations (3 cases) between TCF7L1 (also known as TCF3), a well-known β-catenin transcription regulator, and NAV2 [11]. These translocations are similar to a recurring translocation identified between TCF7L2 and VIIi [36]. Surprisingly, these translocations result in the expression of TCF7L1 or TCF7L2 that lack a β-catenin binding domain and are predicted to act as dominantly interfering alleles [35]. Indeed, suppressing the expression of the TCF7L2-VIIi fusion in colon cancer cell lines that harbor this translocation inhibited anchorage independent growth [36].
In consonance with these observations, colon specific, heterozygous deletion of TCF7L2 in Apc null mice [37] led to the unexpected finding that these mice developed aggressive colon cancers, compared to Apc null mice, which only developed colonic adenomas. Analysis of the aggressive tumors demonstrated loss of heterozygosity (LOH) of the remaining TCF7L2 allele. Together these observations suggest that although β-catenin and TCF family members are required for adenoma formation, loss of TCF family members is associated with tumor progression.
Functional genomics and WNT signaling
Functional studies in model organisms and cultured cells have been instrumental in deciphering the WNT/β-catenin pathway. The use of large scale loss of function approaches in cancer cell lines have enabled a detailed view of the components directly involved in regulation of the WNT/β-catenin pathway and the various pathways that interact with the WNT/β-catenin pathway in tumor development.
Cyclin-dependent kinase 8 (CDK8)
The serine-threonine kinase CDK8 was originally identified as a Cyclin C binding partner [38]. CDK8, together with Cyclin C, MED12, and MED13, form the kinase module of the Mediator complex, a large protein complex involved in transcription regulation [39]. Although CDK8 was initially described as a transcription repressor, further work showed that CDK8 may also activate transcription [40]. Furthermore, biochemical fractionation of CDK8 from cultured human cell lines identified a second CDK8 complex that is not associated to the mediator complex, suggesting that CDK8 may also have functions independent of its role as part of the mediator complex [41].
CDK8 was identified as a gene that regulates β-catenin driven reporter activity in a loss of function RNAi screen [42]. CDK8 is essential for both proliferation of β-catenin-dependent cancer cell lines and for β-catenin transcriptional activity. CDK8 is amplified and overexpressed in 60% of colorectal cancers. Furthermore, high levels of CDK8 are associated with nuclear β-catenin and colon cancer mortality [43, 44]. Together these observations suggest that CDK8 is an amplified oncogene. Recent studies have suggested that in addition to CDK8, co-amplification of CDX2 may also contribute to colon cancer progression [45].
Several roles have been suggested for CDK8 regulation of β-catenin activity. Studies in Drosophila flies suggest that MED12 and MED13 regulate β-catenin transcriptional activity through interactions with Pygopus (PYGO) a well-annotated β-catenin cofactor [46]. In addition, inhibition of WNT activity blocks CDK8-mediated cell transformation [42].
CDK8 has also been reported to attenuate E2F1 inhibition of β-catenin [47]. Inactivation of the RB pathway induces transcription of E2F1, a potent β-catenin inhibitor [47]. Studies in flies and human cultured cells demonstrated that CDK8 inactivates E2F1 by phosphorylation of E2F1 on serine 375 relieving the repression of E2F1 on β-catenin [47].
YAP1 and TAZ
Using genome scale loss-of-function screens, the transcriptional modulator YAP1 has been shown to bind β-catenin and modulate its function [48]. The YAP1-β-catenin complex also includes the SRC tyrosine kinase YES1 and the transcription regulator TBX5. This complex drives the expression of survival genes such as BCL2L1 and BIRC5 (Figure 1). Dasatinib, a small molecule inhibitor of YES1, was shown to inhibit the proliferation of β-catenin-dependent cell lines and tumors.
Over-expression of a mutant constitutively active form of YAP1 (YAP1 S127A) in the murine intestine leads to early morbidity due to loss of intestinal crypts [49], associated with inhibition of β-catenin signaling. Deletion of YAP1 in the murine intestine resulted in hyperproliferation of intestinal stem cells and increased WNT signaling following injury. Mechanistically, YAP1 was shown to inhibit WNT signaling by cytosolic retention of DVL2. These observations are consistent with the dual role β-catenin has in gut homeostasis and tumor progression. As discussed above, TCF7L2 is essential for the functions of β-catenin in gut development and interferes with β-catenin activity in tumorigenesis. Similarly, these observations suggest that YAP1 interferes with β-catenin functions in homeostasis and promotes β-catenin activity in tumor pathogenesis.
Similar to YAP1, a homolog TAZ, has been shown to be required in β-catenin driven cancers [50]. Phosphorylated β-catenin binds to TAZ, leading to proteasomal degradation of the two proteins (Figure 1). Mutations that inactivate the destruction complex stabilize TAZ, which subsequently activates transcriptional programs that lead to proliferation and survival of cancer cells.
Mutations in YAP1 or TAZ have not been identified in cancer. YAP1 amplifications are found in a subset of hepatocellular and ovarian cancers, and hepatocellular cancer cell lines that harbor YAP1 amplifications depend on YAP1 for survival [51].
MAPK pathway (KRAS and BRAF)
Activating KRAS and BRAF mutations are often found in cancers that harbor mutations that activate the WNT pathway [11]. Furthermore, functional studies suggest a synergistic interaction between the WNT and MAPK pathways.
Genetically engineered mice that harbor only loss of function APC mutations or gain of function KRAS mutations fail to develop malignant colon adenocarcinomas. Specifically, APC mutant mice develop adenomas that rarely progress to full adenocarcinoma, and expression of mutant KRAS (KRASV12G) in the murine intestine results in oncogene-induced senescence with no occurrence of cancer [52]. In contrast, compound genetically engineered mice harboring APC mutations and KRASV12G develop invasive colon adenocarcinoma accompanied by enhanced morbidity and mortality [53]. Tumors from these mice show increased nuclear β-catenin [52] and enhanced WNT reporter activity [53], suggesting an interaction between KRAS effector pathways and WNT signaling. Consistent with these observations, Jeong et al. [54] demonstrated that in murine and human colon cancers activation of the WNT pathway leads to activation of the MAPK pathway.
Further supporting the connection between WNT and MAPK signaling, Singh et al. [55] reported that MAP3K7 (TAK1) is upregulated and essential in WNT-deregulated colon cancer cell lines that are dependent on KRAS. Both KRAS and TAK1 regulate WNT signaling in KRAS-dependent colon cancer cell lines. In these cell lines, KRAS up-regulates the expression of BMP7 resulting in activation of the BMPR1A receptor. This feedback loop activates both TAK1 and β-catenin resulting in amplification of β-catenin activity (Figure 1). Furthermore, TAK1 upregulates NF-κB activity, resulting in additional proliferative and survival signals. In consonance with these findings, NF-κB signaling is increased in genetically engineered mice expressing a hyper-activated β-catenin allele, which promoted the proliferation of colonic stem cells [56] (Figure 1). Because TAK1 phosphorylates TBK1 [57], a serine-threonine kinase, that activates NF-κB and interferon signaling and is required for the survival of KRAS-driven cancers [58], these observations suggest that KRAS and WNT signaling cooperate at several levels to drive colon cancer progression.
Pharmacological targeting of the WNT pathway
The WNT/β-catenin pathway plays a key role in colon cancer pathogenesis, yet pharmacological targeting has proven to be challenging. Because β-catenin, the major effector of the WNT pathway, drives various context dependent transcriptional outputs that contribute to diverse phenotypes, effective targeting of β-catenin driven cancers will most likely require inhibition of multiple β-catenin-driven pathways.
Several new compounds have recently been described as inhibitors of different nodes of the β-catenin signaling pathway (Table 1). One class of β-catenin inhibitors restores the activity of the destruction complex (Figure 1). Using a WNT reporter screen Chen et al. identified IWR1-4 as a class of WNT inhibitors that enhance the activity of the destruction complex by stabilizing AXIN1 [59]. Using a similar approach Cong et al. reported that XAV939, a tankyrase 1/2 inhibitor, stabilized AXIN1, which led to β-catenin degradation [60]. Detailed genetic and biochemical studies demonstrated that tankyrase 1/2 stabilize AXIN1 through ADR ribosylation [61]. Recently, a more potent tankyrase inhibitor (G007-LK) has been reported to inhibit the growth of tumors in mice [62]. However, these inhibitors target β-catenin also in normal tissues such as the intestine and may lead to dose-limiting toxicity [62].
Table 1.
Genomic alterations in pathways that regulate β-catenin activity
| Pathway | Biological function | Components | Genomic alterations | Tissue | Compounds |
|---|---|---|---|---|---|
| Pathways that regulate β-catenin stability | |||||
| Destruction complex | Targets β-catenin for proteasomal degradation | APC | LOF Mutations and Deletions | Colorectal, Gastric, Liver, | |
| GSK3B | |||||
| AXIN1 | Deletions | Melanoma, Liver | XAV939, IWR, G007-LK | ||
| AXIN2 | LOF Mutations | ||||
| CSNK1A1 | |||||
| WNT ligand signaling | Inhibits the destruction complex and activates β-catenin | WNTs | Amplifications | Breast, Ovarian, Pancreas | |
| PORCN | IWP, LGK974 | ||||
| RNF43 | LOF mutations | Endometrial, Gastric,Colorectal, Pancreas | |||
| ZNRF3 | |||||
| RSPO | Translocations | ||||
| LRP5/6 | |||||
| FZD5/6 | Neutralizing antibody | ||||
| Pathways that interact with β-catenin signaling | |||||
| MAPK | Interacts with the WNT pathway and enhances the activity of WNT signaling | KRAS | GOF mutations and Amplifications | Colon, Pancreas, Gastric, Lung | |
| BRAF | GOF mutations | Melanoma, Colon | Vemurafenib | ||
| TAK1 | 5Z-7-oxozeaenol | ||||
| YAP1/TAZ | Forms a transcriptional complex with β-catenin | YES1/SRC | Dasatinib | ||
| YAP1 | Amplification | Liver, Medulloblastoma | |||
| TAZ | |||||
| TBX5 | |||||
| BCL2L1 | Navitoclax | ||||
| BIRC5 | YM-155 | ||||
| TCF/LEF | Development and adenoma formation | TCF7L2 | LOF mutations, Deletions, Translocation | Colorectal | |
| TCF7L1 | LOF Translocations | Colorectal | |||
| TCF7 | |||||
| LEF1 | |||||
A second class of WNT inhibitors target specific nodes within the WNT signaling pathway. Large scale profiling of small molecule inhibitors in a large panel of cancer cell lines identified Navitoclax, a BCL2L1 inhibitor, as having activity in β-catenin mutated cancers [63]. Other compounds such as dasatinib [48] or 5Z-7-oxozeaenol [55] inhibit other nodes of the WNT/β-catenin pathway (Table 1). Future studies using combination therapies that target multiple nudes of this pathway are likely to result in more potent and durable inhibition of WNT dependent cancers.
Concluding remarks
Although the WNT/β-catenin pathway has been extensively studied both in development and in cancer, recent genomic studies in colon cancer have identified new components and regulators of the canonical WNT/β-catenin pathway. These studies suggest that WNT/β-catenin signaling is more complex than anticipated and may be influenced by context. Further studies will be necessary to understand how WNT/β-catenin signaling contributes to both tumor initiation and progression.
Indeed, the identification of additional components and regulators of WNT/β-catenin signaling may also provide new approaches to therapeutically target WNT/β-catenin deregulated cancers. The identification of genetic alterations in both canonical and new components of WNT/β-catenin will inform studies to develop new modulators as well as identify cancers that are likely to respond to such manipulations.
Highlights.
Mutations in the WNT/β-catenin pathway occur in many types of human cancers
New components of the WNT/β-catenin pathway have been identified
B-catenin forms several transcriptional complexes
β-catenin and RAS signaling interact
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Klaus A, Birchmeier W. Wnt signalling and its impact on development and cancer. Nature Review Cancer. 2008;8:387–398. doi: 10.1038/nrc2389. [DOI] [PubMed] [Google Scholar]
- 2.Clevers H, Nusse R. Wnt/beta-catenin signaling and disease. Cell. 2012;149:1192–1205. doi: 10.1016/j.cell.2012.05.012. [DOI] [PubMed] [Google Scholar]
- 3.Li VS, et al. Wnt signaling through inhibition of beta-catenin degradation in an intact Axin1 complex. Cell. 2012;149:1245–1256. doi: 10.1016/j.cell.2012.05.002. [DOI] [PubMed] [Google Scholar]
- 4.Sharma M, et al. Specific armadillo repeat sequences facilitate beta-catenin nuclear transport in live cells via direct binding to nucleoporins Nup62, Nup153, and RanBP2/Nup358. J Biol Chem. 2012;287:819–831. doi: 10.1074/jbc.M111.299099. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 5.Grigoryan T, et al. Deciphering the function of canonical Wnt signals in development and disease: conditional loss- and gain-of-function mutations of beta-catenin in mice. Genes Dev. 2008;22:2308–2341. doi: 10.1101/gad.1686208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zecca M, et al. Direct and long-range action of a wingless morphogen gradient. Cell. 1996;87:833–844. doi: 10.1016/s0092-8674(00)81991-1. [DOI] [PubMed] [Google Scholar]
- 7.Korinek V, et al. Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat Genet. 1998;19:379–383. doi: 10.1038/1270. [DOI] [PubMed] [Google Scholar]
- 8.Yochum GS, et al. Serial analysis of chromatin occupancy identifies beta-catenin target genes in colorectal carcinoma cells. Proc Natl Acad Sci U S A. 2007;104:3324–3329. doi: 10.1073/pnas.0611576104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nusse R, Varmus HE. Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell. 1982;31:99–109. doi: 10.1016/0092-8674(82)90409-3. [DOI] [PubMed] [Google Scholar]
- 10.Kinzler KW, Vogelstein B. Lessons from hereditary colorectal cancer. Cell. 1996;87:159–170. doi: 10.1016/s0092-8674(00)81333-1. [DOI] [PubMed] [Google Scholar]
- 11.TCGA. Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487:330–337. doi: 10.1038/nature11252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Niehrs C. The complex world of WNT receptor signalling. Nat Rev Mol Cell Biol. 2012;13:767–779. doi: 10.1038/nrm3470. [DOI] [PubMed] [Google Scholar]
- 13.Najdi R, et al. A uniform human Wnt expression library reveals a shared secretory pathway and unique signaling activities. Differentiation. 2012;84:203–213. doi: 10.1016/j.diff.2012.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wend P, et al. WNT10B/beta-catenin signalling induces HMGA2 and proliferation in metastatic triple-negative breast cancer. EMBO Mol Med. 2013;5:264–279. doi: 10.1002/emmm.201201320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Qiang YW, et al. Wnts induce migration and invasion of myeloma plasma cells. Blood. 2005;106:1786–1793. doi: 10.1182/blood-2005-01-0049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wong GT, et al. Differential transformation of mammary epithelial cells by Wnt genes. Mol Cell Biol. 1994;14:6278–6286. doi: 10.1128/mcb.14.9.6278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Green JL, et al. Paracrine Wnt signaling both promotes and inhibits human breast tumor growth. Proc Natl Acad Sci U S A. 2013;110:6991–6996. doi: 10.1073/pnas.1303671110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kazanskaya O, et al. R-Spondin2 is a secreted activator of Wnt/beta-catenin signaling and is required for Xenopus myogenesis. Dev Cell. 2004;7:525–534. doi: 10.1016/j.devcel.2004.07.019. [DOI] [PubMed] [Google Scholar]
- 19.Jin YR, Yoon JK. The R-spondin family of proteins: emerging regulators of WNT signaling. Int J Biochem Cell Biol. 2012;44:2278–2287. doi: 10.1016/j.biocel.2012.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nam JS, et al. Dynamic expression of R-spondin family genes in mouse development. Gene Expr Patterns. 2007;7:306–312. doi: 10.1016/j.modgep.2006.08.006. [DOI] [PubMed] [Google Scholar]
- 21.Carmon KS, et al. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/beta-catenin signaling. Proc Natl Acad Sci U S A. 2011;108:11452–11457. doi: 10.1073/pnas.1106083108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Koo BK, et al. Tumour suppressor RNF43 is a stem-cell E3 ligase that induces endocytosis of Wnt receptors. Nature. 2012;488:665–669. doi: 10.1038/nature11308. [DOI] [PubMed] [Google Scholar]
- 23.Hao HX, et al. ZNRF3 promotes Wnt receptor turnover in an R-spondin-sensitive manner. Nature. 2012;485:195–200. doi: 10.1038/nature11019. [DOI] [PubMed] [Google Scholar]
- 24.Seshagiri S, et al. Recurrent R-spondin fusions in colon cancer. Nature. 2012;488:660–664. doi: 10.1038/nature11282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Valenta T, et al. The many faces and functions of beta-catenin. EMBO J. 2012;31:2714–2736. doi: 10.1038/emboj.2012.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tornesello ML, et al. Mutations in TP53, CTNNB1 and PIK3CA genes in hepatocellular carcinoma associated with hepatitis B and hepatitis C virus infections. Genomics. 2013;102:74–83. doi: 10.1016/j.ygeno.2013.04.001. [DOI] [PubMed] [Google Scholar]
- 27.Morin PJ, et al. Apoptosis and APC in colorectal tumorigenesis. Proc Natl Acad Sci U S A. 1996;93:7950–7954. doi: 10.1073/pnas.93.15.7950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huang WS, et al. ShRNA-mediated gene silencing of beta-catenin inhibits growth of human colon cancer cells. World J Gastroenterol. 2007;13:6581–6587. doi: 10.3748/wjg.v13.i48.6581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rai K, et al. DNA demethylase activity maintains intestinal cells in an undifferentiated state following loss of APC. Cell. 2010;142:930–942. doi: 10.1016/j.cell.2010.08.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Major MB, et al. Wilms tumor suppressor WTX negatively regulates WNT/beta-catenin signaling. Science. 2007;316:1043–1046. doi: 10.1126/science/1141515. [DOI] [PubMed] [Google Scholar]
- 31.van Amerongen R, Berns A. Knockout mouse models to study Wnt signal transduction. Trends Genet. 2006;22:678–689. doi: 10.1016/j.tig.2006.10.001. [DOI] [PubMed] [Google Scholar]
- 32.Elyada E, et al. CKIalpha ablation highlights a critical role for p53 in invasiveness control. Nature. 2011;470:409–413. doi: 10.1038/nature09673. [DOI] [PubMed] [Google Scholar]
- 33.Dimri GP, et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci U S A. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Beroukhim R, et al. The landscape of somatic copy-number alteration across human cancers. Nature. 2010;463:899–905. doi: 10.1038/nature08822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Roose J, Clevers H. TCF transcription factors: molecular switches in carcinogenesis. Biochim Biophys Acta. 1999;1424:M23–37. doi: 10.1016/s0304-419x(99)00026-8. [DOI] [PubMed] [Google Scholar]
- 36.Bass AJ, et al. Genomic sequencing of colorectal adenocarcinomas identifies a recurrent VTI1A-TCF7L2 fusion. Nature Genetics. 2011;43:964–968. doi: 10.1038/ng.936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Angus-Hill ML, et al. T-cell factor 4 functions as a tumor suppressor whose disruption modulates colon cell proliferation and tumorigenesis. Proc Natl Acad Sci U S A. 2011;108:4914–4919. doi: 10.1073/pnas.1102300108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tassan JP, et al. Identification of human cyclin-dependent kinase 8, a putative protein kinase partner for cyclin C. Proc Natl Acad Sci U S A. 1995;92:8871–8875. doi: 10.1073/pnas.92.19.8871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Taatjes DJ. The human Mediator complex: a versatile, genome-wide regulator of transcription. Trends Biochem Sci. 2010;35:315–322. doi: 10.1016/j.tibs.2010.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Galbraith MD, et al. CDK8: a positive regulator of transcription. Transcription. 2010;1:4–12. doi: 10.4161/trns.1.1.12373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Knuesel MT, et al. The human CDK8 subcomplex is a histone kinase that requires Med12 for activity and can function independently of mediator. Mol Cell Biol. 2009;29:650–661. doi: 10.1128/MCB.00993-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Firestein R, et al. CDK8 is a colorectal cancer oncogene that regulates beta-catenin activity. Nature. 2008;455:547–551. doi: 10.1038/nature07179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Firestein R, et al. CDK8 expression in 470 colorectal cancers in relation to beta-catenin activation, other molecular alterations and patient survival. Int J Cancer. 2010;126:2863–2873. doi: 10.1002/ijc.24908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kim MY, et al. Roles of cyclin-dependent kinase 8 and beta-catenin in the oncogenesis and progression of gastric adenocarcinoma. Int J Oncol. 2011;38:1375–1383. doi: 10.3892/ijo.2011.948. [DOI] [PubMed] [Google Scholar]
- 45.Salari K, et al. CDX2 is an amplified lineage-survival oncogene in colorectal cancer. Proc Natl Acad Sci U S A. 2012;109:E3196–3205. doi: 10.1073/pnas.1206004109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Carrera I, et al. Pygopus activates Wingless target gene transcription through the mediator complex subunits Med12 and Med13. Proc Natl Acad Sci U S A. 2008;105:6644–6649. doi: 10.1073/pnas.0709749105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Morris EJ, et al. E2F1 represses beta-catenin transcription and is antagonized by both pRB and CDK8. Nature. 2008;455:552–556. doi: 10.1038/nature07310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rosenbluh J, et al. beta-Catenin-driven cancers require a YAP1 transcriptional complex for survival and tumorigenesis. Cell. 2012;151:1457–1473. doi: 10.1016/j.cell.2012.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Barry ER, et al. Restriction of intestinal stem cell expansion and the regenerative response by YAP. Nature. 2013;493:106–110. doi: 10.1038/nature11693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Azzolin L, et al. Role of TAZ as mediator of Wnt signaling. Cell. 2012;151:1443–1456. doi: 10.1016/j.cell.2012.11.027. [DOI] [PubMed] [Google Scholar]
- 51.Zender L, et al. Identification and validation of oncogenes in liver cancer using an integrative oncogenomic approach. Cell. 2006;125:1253–1267. doi: 10.1016/j.cell.2006.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sansom OJ, et al. Loss of Apc allows phenotypic manifestation of the transforming properties of an endogenous K-ras oncogene in vivo. Proc Natl Acad Sci U S A. 2006;103:14122–14127. doi: 10.1073/pnas.0604130103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Janssen KP, et al. APC and oncogenic KRAS are synergistic in enhancing Wnt signaling in intestinal tumor formation and progression. Gastroenterology. 2006;131:1096–1109. doi: 10.1053/j.gastro.2006.08.011. [DOI] [PubMed] [Google Scholar]
- 54.Jeong WJ, et al. Ras stabilization through aberrant activation of Wnt/beta-catenin signaling promotes intestinal tumorigenesis. Sci Signal. 2012;5:ra30. doi: 10.1126/scisignal.2002242. [DOI] [PubMed] [Google Scholar]
- 55.Singh A, et al. TAK1 inhibition promotes apoptosis in KRAS-dependent colon cancers. Cell. 2012;148:639–650. doi: 10.1016/j.cell.2011.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schwitalla S, et al. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell. 2013;152:25–38. doi: 10.1016/j.cell.2012.12.012. [DOI] [PubMed] [Google Scholar]
- 57.Kawai T, Akira S. Innate immune recognition of viral infection. Nat Immunol. 2006;7:131–137. doi: 10.1038/ni1303. [DOI] [PubMed] [Google Scholar]
- 58.Barbie DA, et al. Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature. 2009;462:108–112. doi: 10.1038/nature08460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chen B, et al. Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer. Nat Chem Biol. 2009;5:100–107. doi: 10.1038/nchembio.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Huang SM, et al. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature. 2009;461:614–620. doi: 10.1038/nature08356. [DOI] [PubMed] [Google Scholar]
- 61.Zhang Y, et al. RNF146 is a poly(ADP-ribose)-directed E3 ligase that regulates axin degradation and Wnt signalling. Nat Cell Biol. 2011;13:623–629. doi: 10.1038/ncb2222. [DOI] [PubMed] [Google Scholar]
- 62.Lau T, et al. A novel tankyrase small-molecule inhibitor suppresses APC mutation-driven colorectal tumor growth. Cancer Res. 2013;73:3132–3144. doi: 10.1158/0008-5472.CAN-12-4562. [DOI] [PubMed] [Google Scholar]
- 63.Basu A, et al. An interactive resource to identify cancer genetic and lineage dependencies targeted by small molecules. Cell. 2013;154:1151–1161. doi: 10.1016/j.cell.2013.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]