Abstract
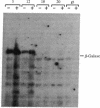
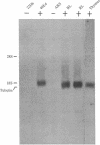
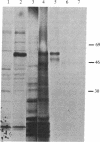
A cDNA library was prepared from a terminal deoxynucleotidyltransferase-containing thymoma in the lambda phage vector lambda gt11. By screening plaques with anti-terminal transferase antibody, positive clones were identified of which some had beta-galactosidase-cDNA fusion proteins identifiable after electrophoretic fractionation by immunoblotting with anti-terminal transferase antibody. The predominant class of cross-hybridizing clones was determined to represent cDNA for terminal transferase by showing that one representative clone hybridized to a 2200-nucleotide mRNA in close-matched enzyme-positive but not to enzyme-negative cells and that the cDNA selected a mRNA that translated to give a protein of the size and antigenic characteristics of terminal transferase. Only a small amount of genomic DNA hybridized to the longest available clone, indicating that the sequence is virtually unique in the mouse genome.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alt F. W., Baltimore D. Joining of immunoglobulin heavy chain gene segments: implications from a chromosome with evidence of three D-JH fusions. Proc Natl Acad Sci U S A. 1982 Jul;79(13):4118–4122. doi: 10.1073/pnas.79.13.4118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alt F. W., Enea V., Bothwell A. L., Baltimore D. Activity of multiple light chain genes in murine myeloma cells producing a single, functional light chain. Cell. 1980 Aug;21(1):1–12. doi: 10.1016/0092-8674(80)90109-9. [DOI] [PubMed] [Google Scholar]
- Alt F., Rosenberg N., Lewis S., Thomas E., Baltimore D. Organization and reorganization of immunoglobulin genes in A-MULV-transformed cells: rearrangement of heavy but not light chain genes. Cell. 1981 Dec;27(2 Pt 1):381–390. doi: 10.1016/0092-8674(81)90421-9. [DOI] [PubMed] [Google Scholar]
- Baltimore D. Is terminal deoxynucleotidyl transferase a somatic mutagen in lymphocytes? Nature. 1974 Mar 29;248(447):409–411. doi: 10.1038/248409a0. [DOI] [PubMed] [Google Scholar]
- Bittner M., Kupferer P., Morris C. F. Electrophoretic transfer of proteins and nucleic acids from slab gels to diazobenzyloxymethyl cellulose or nitrocellulose sheets. Anal Biochem. 1980 Mar 1;102(2):459–471. doi: 10.1016/0003-2697(80)90182-7. [DOI] [PubMed] [Google Scholar]
- Bollum F. J., Brown M. A high molecular weight form of terminal deoxynucleotidyl transferase. Nature. 1979 Mar 8;278(5700):191–192. doi: 10.1038/278191a0. [DOI] [PubMed] [Google Scholar]
- Boss M., Greaves M., Teich N. Abelson virus-transformed haematopoietic cell lines with pre-B-cell characteristics. Nature. 1979 Apr 5;278(5704):551–553. doi: 10.1038/278551a0. [DOI] [PubMed] [Google Scholar]
- Bothwell A. L., Paskind M., Reth M., Imanishi-Kari T., Rajewsky K., Baltimore D. Heavy chain variable region contribution to the NPb family of antibodies: somatic mutation evident in a gamma 2a variable region. Cell. 1981 Jun;24(3):625–637. doi: 10.1016/0092-8674(81)90089-1. [DOI] [PubMed] [Google Scholar]
- Chang L. M., Bollum F. J. Deoxynucleotide-polymerizing enzymes of calf thymus gland. V. Homogeneous terminal deoxynucleotidyl transferase. J Biol Chem. 1971 Feb 25;246(4):909–916. [PubMed] [Google Scholar]
- Chang L. M. Development of terminal deoxynucleotidyl transferase activity in embryonic calf thymus gland. Biochem Biophys Res Commun. 1971 Jul 2;44(1):124–131. doi: 10.1016/s0006-291x(71)80167-5. [DOI] [PubMed] [Google Scholar]
- Chang L. M., Plevani P., Bollum F. J. Proteolytic degradation of calf thymus terminal deoxynucleotidyl transferase. J Biol Chem. 1982 May 25;257(10):5700–5706. [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Coleman M. S., Hutton J. J., De Simone P., Bollum F. J. Terminal deoxyribonucleotidyl transferase in human leukemia. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4404–4408. doi: 10.1073/pnas.71.11.4404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis R. W., Hopkins N., Fleissner E. Biochemical analysis of murine leukemia viruses isolated from radiation-induced leukemias of strain BALB/c. J Virol. 1980 Feb;33(2):661–670. doi: 10.1128/jvi.33.2.661-670.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gearhart P. J., Johnson N. D., Douglas R., Hood L. IgG antibodies to phosphorylcholine exhibit more diversity than their IgM counterparts. Nature. 1981 May 7;291(5810):29–34. doi: 10.1038/291029a0. [DOI] [PubMed] [Google Scholar]
- Hedrick S. M., Nielsen E. A., Kavaler J., Cohen D. I., Davis M. M. Sequence relationships between putative T-cell receptor polypeptides and immunoglobulins. Nature. 1984 Mar 8;308(5955):153–158. doi: 10.1038/308153a0. [DOI] [PubMed] [Google Scholar]
- KRAKOW J. S., COUTSOGEORGOPOULOS C., CANELLAKIS E. S. Studies on the incorporation of deoxyribonucleic acid. Biochim Biophys Acta. 1962 May 14;55:639–650. doi: 10.1016/0006-3002(62)90842-9. [DOI] [PubMed] [Google Scholar]
- Kato K. I., Gonçalves J. M., Houts G. E., Bollum F. J. Deoxynucleotide-polymerizing enzymes of calf thymus gland. II. Properties of the terminal deoxynucleotidyltransferase. J Biol Chem. 1967 Jun 10;242(11):2780–2789. [PubMed] [Google Scholar]
- Kung P. C., Siverstone A. E., McCaffrey R. P., Baltimore D. Murine terminal deoxynucleotidyl transferase: cellular distribution and response to cortisone. J Exp Med. 1975 Apr 1;141(4):855–865. [PMC free article] [PubMed] [Google Scholar]
- McCaffrey R., Harrison T. A., Parkman R., Baltimore D. Terminal deoxynucleotidyl transferase activity in human leukemic cells and in normal human thymocytes. N Engl J Med. 1975 Apr 10;292(15):775–780. doi: 10.1056/NEJM197504102921504. [DOI] [PubMed] [Google Scholar]
- Nakamura H., Tanabe K., Yoshida S., Morita T. Terminal deoxynucleotidyltransferase of 60,000 daltons from mouse, rat, and calf thymus. Purification by immunoadsorbent chromatography and comparison of peptide structures. J Biol Chem. 1981 Aug 25;256(16):8745–8751. [PubMed] [Google Scholar]
- Okayama H., Berg P. High-efficiency cloning of full-length cDNA. Mol Cell Biol. 1982 Feb;2(2):161–170. doi: 10.1128/mcb.2.2.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce J. H., Aaronson S. A. BALB- and Harvey-murine sarcoma virus transformation of a novel lymphoid progenitor cell. J Exp Med. 1982 Sep 1;156(3):873–887. doi: 10.1084/jem.156.3.873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothenberg E., Triglia D. Clonal proliferation unlinked to terminal deoxynucleotidyl transferase synthesis in thymocytes of young mice. J Immunol. 1983 Apr;130(4):1627–1633. [PubMed] [Google Scholar]
- Silverstone A. E., Cantor H., Goldstein G., Baltimore D. Terminal deoxynucleotidyl transferase is found in prothymocytes. J Exp Med. 1976 Aug 1;144(2):543–548. doi: 10.1084/jem.144.2.543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverstone A., Sun L., Witte O. N., Baltimore D. Biosynthesis of murine terminal deoxynucleotidyltransferase. J Biol Chem. 1980 Jan 25;255(2):791–796. [PubMed] [Google Scholar]
- Wolf S. C., Kourides I. A., Good R. A., Silverstone A. E. Cell-free translation of the messenger RNA for calf terminal deoxynucleotidyltransferase. J Biol Chem. 1982 Apr 25;257(8):4013–4015. [PubMed] [Google Scholar]
- Young R. A., Davis R. W. Efficient isolation of genes by using antibody probes. Proc Natl Acad Sci U S A. 1983 Mar;80(5):1194–1198. doi: 10.1073/pnas.80.5.1194. [DOI] [PMC free article] [PubMed] [Google Scholar]