Abstract
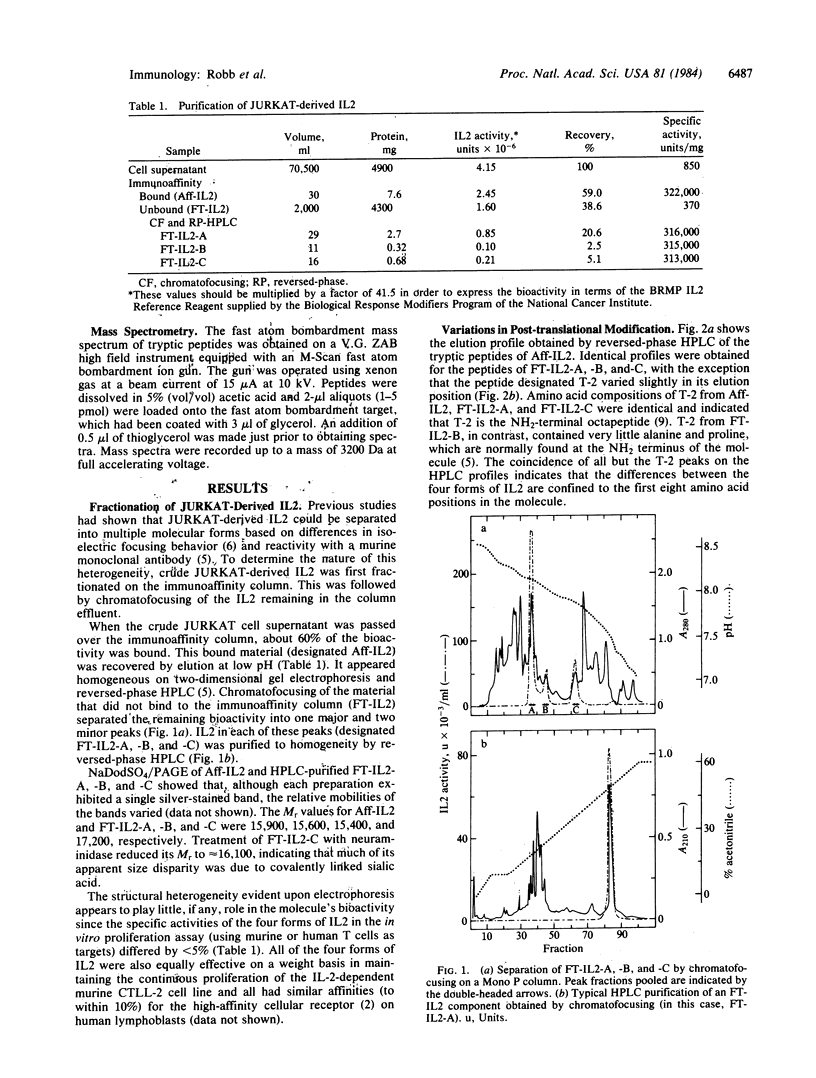
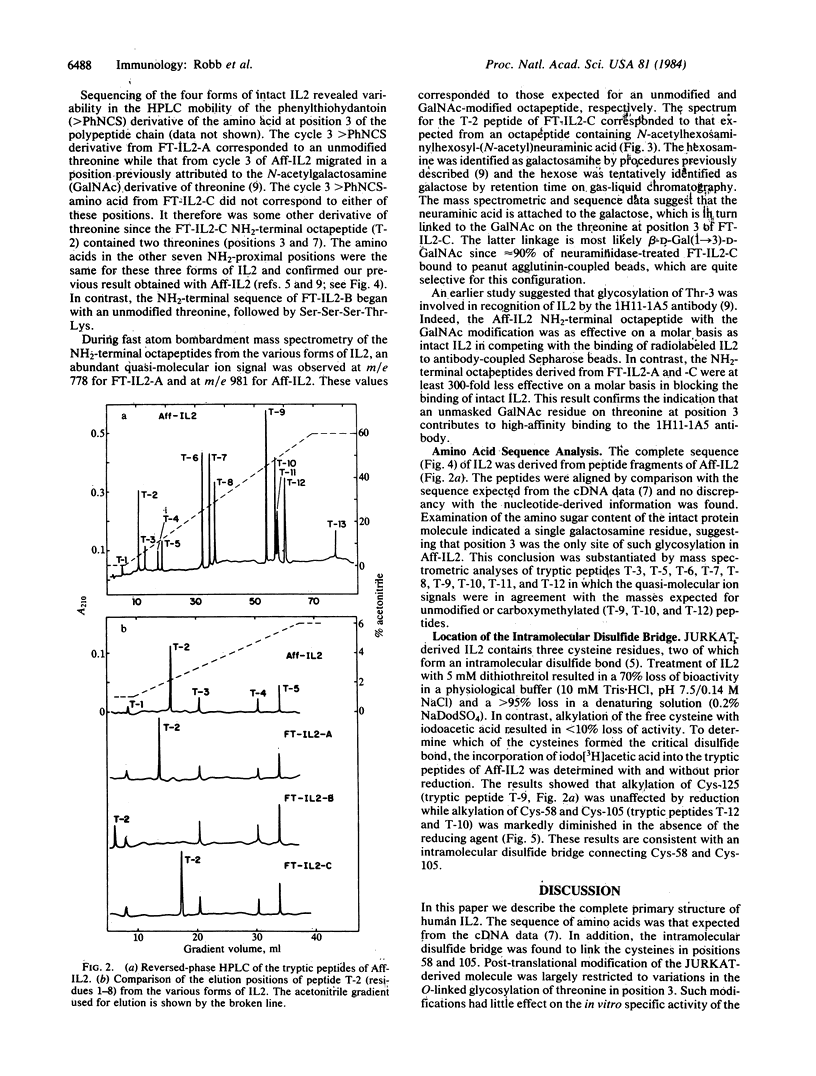
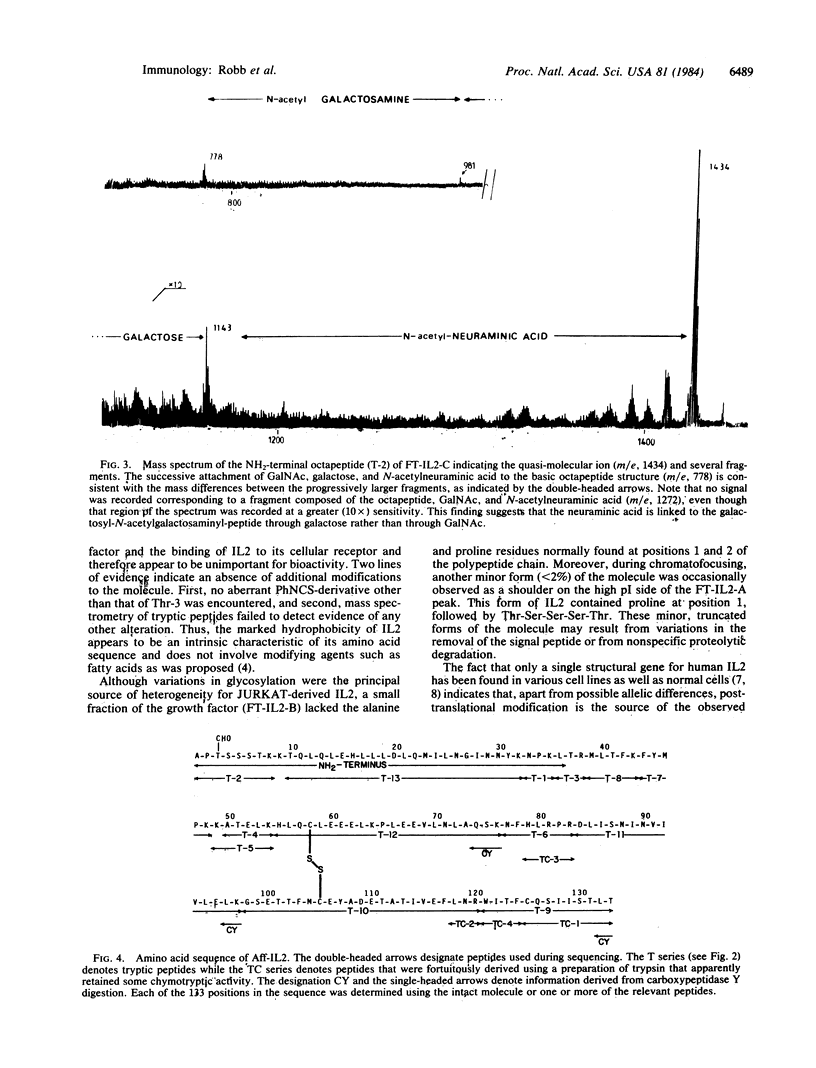
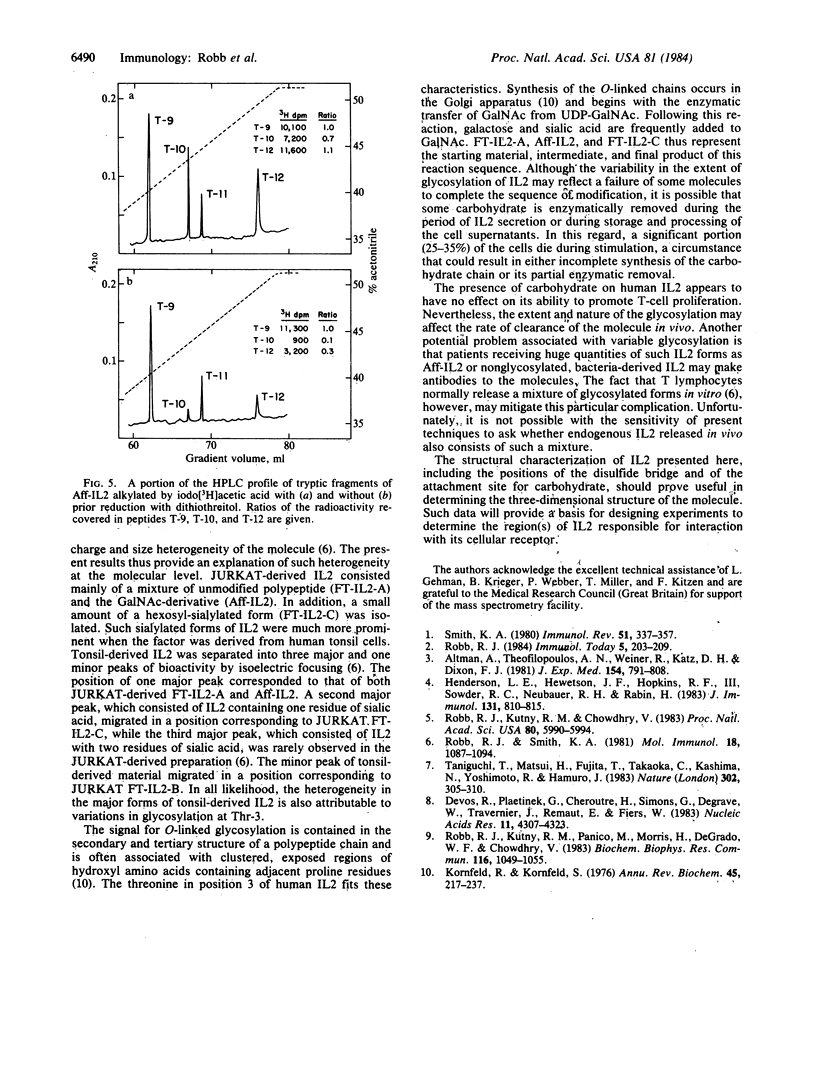
Human interleukin 2 was separated into multiple molecular forms by selective immunoaffinity chromatography and chromatofocusing. For the most part, this heterogeneity was attributed to variations in glycosylation of the threonine residue in position 3 of the polypeptide chain. The various molecular forms of interleukin 2 had nearly identical specific activities in the in vitro proliferation assay, indicating that the glycosylation had no significant effect on this response. The entire primary sequence of interleukin 2, including the location of the intramolecular disulfide bridge, was determined by a combination of peptide mapping and protein sequencing. This information should aid in the determination of the active site(s) of the molecule.
Full text
PDF
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Altman A., Theofilopoulos A. N., Weiner R., Katz D. H., Dixon F. J. Analysis of T cell function in autoimmune murine strains. Defects in production and responsiveness to interleukin 2. J Exp Med. 1981 Sep 1;154(3):791–808. doi: 10.1084/jem.154.3.791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devos R., Plaetinck G., Cheroutre H., Simons G., Degrave W., Tavernier J., Remaut E., Fiers W. Molecular cloning of human interleukin 2 cDNA and its expression in E. coli. Nucleic Acids Res. 1983 Jul 11;11(13):4307–4323. doi: 10.1093/nar/11.13.4307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson L. E., Hewetson J. F., Hopkins R. F., 3rd, Sowder R. C., Neubauer R. H., Rabin H. A rapid, large scale purification procedure for gibbon interleukin 2. J Immunol. 1983 Aug;131(2):810–815. [PubMed] [Google Scholar]
- Kornfeld R., Kornfeld S. Comparative aspects of glycoprotein structure. Annu Rev Biochem. 1976;45:217–237. doi: 10.1146/annurev.bi.45.070176.001245. [DOI] [PubMed] [Google Scholar]
- Robb R. J., Kutny R. M., Chowdhry V. Purification and partial sequence analysis of human T-cell growth factor. Proc Natl Acad Sci U S A. 1983 Oct;80(19):5990–5994. doi: 10.1073/pnas.80.19.5990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robb R. J., Kutny R. M., Panico M., Morris H., DeGrado W. F., Chowdhry V. Posttranslational modification of human T-cell growth factor. Biochem Biophys Res Commun. 1983 Nov 15;116(3):1049–1055. doi: 10.1016/s0006-291x(83)80248-4. [DOI] [PubMed] [Google Scholar]
- Robb R. J., Smith K. A. Heterogeneity of human T-cell growth factor(s) due to variable glycosylation. Mol Immunol. 1981 Dec;18(12):1087–1094. doi: 10.1016/0161-5890(81)90024-9. [DOI] [PubMed] [Google Scholar]
- Smith K. A. T-cell growth factor. Immunol Rev. 1980;51:337–357. doi: 10.1111/j.1600-065x.1980.tb00327.x. [DOI] [PubMed] [Google Scholar]
- Taniguchi T., Matsui H., Fujita T., Takaoka C., Kashima N., Yoshimoto R., Hamuro J. Structure and expression of a cloned cDNA for human interleukin-2. Nature. 1983 Mar 24;302(5906):305–310. doi: 10.1038/302305a0. [DOI] [PubMed] [Google Scholar]


