Abstract
Purpose
Echinochrome is a pigment present in the shells and spines of sea urchins. It has been reported to have several biologic protective effects, including in experimental models of myocardial ischemia/reperfusion injury, for which the proposed mechanisms are scavenging reactive oxygen species (ROS) and chelating iron. Endotoxin-induced uveitis (EIU) is an animal model of acute anterior segment intraocular inflammation that is induced by the injection of lipopolysaccharide (LPS). In this study, the therapeutic effect of echinochrome was examined in uveitis using the EIU model.
Methods
EIU was induced in Lewis rats via 200 μg subcutaneous injections of LPS from Escherichia coli. Echinochrome was administered intravenously in 10, 1, or 0.1 mg/kg doses suspended in PBS (controls were injected with PBS only). Twenty-four hours after LPS injection, the number of infiltrating cells and the protein concentration in aqueous humor were determined. Aqueous tumor necrosis factor α (TNF-α) concentration was quantified with enzyme-linked immunosorbent assay, eyes were stained with nuclear factor (NF) κB antibodies, and ROS production was determined by dihydroethidium staining in fresh frozen samples.
Results
The number of inflammatory aqueous cells and protein levels were lower in the groups treated with 10 and 1 mg/kg of echinochrome than in the untreated LPS group (p<0.01). Treatment with 10 and 1 mg/kg of echinochrome significantly reduced TNF-α concentrations in aqueous humor (p<0.01). The numbers of NFκB-positive cells and ROS signals were also reduced by echinochrome administration (p<0.05).
Conclusions
Echinochrome ameliorated intraocular inflammation caused by EIU by reducing ROS production, thereby also decreasing the expression of NFκB and TNF-α. As a natural pigment, echinochrome may therefore be a promising candidate for the safe treatment of intraocular inflammation. The use of sea urchin shells and spines in health foods and medical products is thus both economically and environmentally meaningful.
Introduction
Echinochrome, or 7(2)-ethyl-2,3,5,6,8-pentahydroxy-1,4-naphthoquinone, is one of several spinochromes that occur as pigments in the shells of sea urchins. Part of the chemical structure of echinochrome is appropriate for free-radical scavenging, as it includes 2,3,7-hydroxyl groups and contains a 1,4-naphthoquinone ring, similar to vitamin K. Unlike vitamin K, echinochrome is water soluble and does not affect blood coagulation [1,2]. Naturally derived echinochrome contains several isomers with different location of methyl group with varied proportions, with two most abundant are presented (Figure 1). However, it is considered that these isomers have a similar antioxidant effect, since this is expected to arise from their multiple hydroxyl groups. Echinochrome expression in larval and adult sea urchin tissues is related to seawater quality [3]: It increases when water quality decreases or contains harmful agents [4]. In adult sea urchins, spinochromes—including echinochrome—are mainly found in the shell and spines of the animal. Sea urchin spinochromes form part of the daily diet of sea otters [5], and lifelong oral consumption of these pigments leads to the pink or light-purple staining of the teeth and bones observed in sea otters [6].
Figure 1.
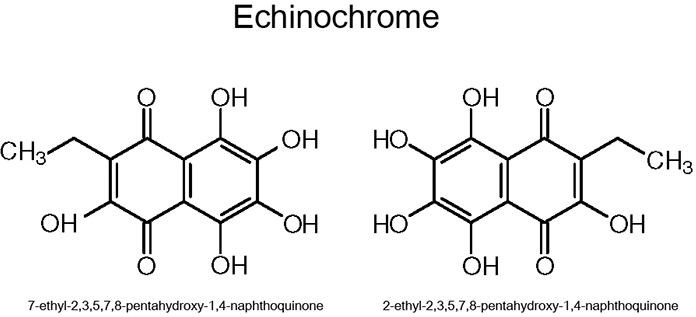
The chemical structure of echinochrome is shown. Echinochrome has two main isomers.
Echinochrome has been reported to have shas twoeveral biologic effects [7,8]. In animal experimental models of myocardial ischemia/reperfusion injury, echinochrome exhibits a considerable protective effect and reduces infarct size by up to 45% [9,10].
The proposed protective mechanisms of echinochrome are reactive oxygen species (ROS) scavenging and iron chelation [11]. In the Russian Federation, echinochrome is approved as an experimental treatment for acute infarction in humans as the commercially available drug Hystochrome® (Pacific Institute of Bioorganic Chemistry [PIBOC], Vladivostok, Russia) [12,13] and the dietary supplement Timarin® (PIBOC) [14].
Endotoxin-induced uveitis (EIU) is an animal model of acute anterior segment intraocular inflammation that is induced by the injection of an endotoxin, the lipopolysaccharide (LPS) component of the Gram-negative bacterial cell wall [15]. It has been reported that increased ROS production is one of the factors that triggers inflammation, leading to activation of nuclear factor (NF) κB; this is one of the most ubiquitous transcription factors, and it has been suggested that it has a key role in inflammatory reactions [16,17]. The inflammatory feedback loop progresses with cellular infiltration and protein leakage into the anterior chamber of the eye, which reaches a maximum 24 h after LPS injection [18]. Elevated expression of cytokines and chemokines has been observed to be concomitant with maximum EIU [18,19]. Therefore, reducing ROS expression in the eye may be beneficial in limiting inflammatory response. The purpose of this study was to examine the therapeutic effects of echinochrome in a rat EIU model.
Methods
Animals and reagents
Echinochrome was obtained from PIBOC. The substance was dissolved in PBS (NaCl 8 g, (Na2HPO4)12H2O 2.9 g, KCl 0.2 g, KH2PO4 0.2 g, H2O 1 l; pH 7.4) immediately before use.
Eight-week-old male Lewis rats (180–220 g) were used. All procedures involving animals were performed in accordance with the Association for Research in Vision and Ophthalmology resolution on the use of animals in research. EIU was induced by subcutaneous injection with 200 μg LPS from Escherichia coli (Sigma-Aldrich, St. Louis, MO) that had been diluted in 300 μl PBS. At the same time, the animals were injected intravenously with 10, 1, or 0.1 mg/kg of echinochrome diluted in 1.0 ml PBS. Control EIU rats were intravenously administered 1.0 ml PBS (no echinochrome). Euthanasia was performed by intravenous injection of 100 mg/kg of sodium pentobarbital (Sigma, Tokyo, Japan). All experiments were performed in triplicate, with four animals in each group.
Number of infiltrating cells, concentrations of protein, and tumor necrosis factor α in aqueous humor
Twenty-four hours after LPS injection, the rats were euthanatized, and the aqueous humor was collected immediately from both eyes via an anterior chamber puncture (25–50 μl/rat) using a 30-gauge needle (Terumo Corporation, Tokyo, Japan), under an Olympus SZ61 microscope (Olympus, Tokyo, Japan). To count the cells, the aqueous humor samples were suspended in an equal amount of Türk stain solution (Merck, Darmstadt, Germany). The cells were counted with a hemocytometer under an Olympus CKX31 microscope (Olympus), and the number of cells was obtained by averaging the results of four fields from each sample. The total protein concentration in the aqueous humor samples was measured with a bicinchoninic acid protein assay kit (Pierce, Rockford, IL). The aqueous humor samples were stored in ice water until testing, and cell counts and total protein concentrations were measured on the day of sample collection.
TNF-α levels in the aqueous humor were assessed with a commercially available rat TNF-α enzyme-linked immunosorbent assay kit according to the manufacturer’s instructions (Thermo Fisher Scientific, Waltham, MA). Each assay was performed in triplicate.
Immunohistochemical studies for nuclear factor κB
NFκB translocation precedes the development of systemic and ocular inflammation. Based on the preliminary results, the rats were deeply anesthetized by intravenous injection of sodium pentobarbital (50 mg/kg) 3 h after LPS injection, and the eyes were fixed by an intracardiac perfusion of Superfix 100 solution (Kurabo, Osaka, Japan). The eyes were then enucleated and immersed in the same fixative for 12 h, and then embedded in paraffin. Next, 5 μm sagittal sections were cut near the optic nerve head.
Sections were dewaxed with xylene and rehydrated with ethanol. Antigens were retrieved by heating sections in citrate buffer solution in a microwave oven. To evaluate the NFκB-positive cells in the iris/ciliary body (ICB), a 1:100 dilution of NFκB p65 antibody (Santa Cruz Biotechnology, Santa Cruz, CA) was applied to the sections for 12 h, after which they were washed with PBS. Following this, a 1:1,000 dilution of a secondary goat–anti-rabbit antibody dye conjugate (Invitrogen, Carlsbad, CA), which produces red fluorescence, was applied. Because NFκB is widely present in intercellular tissue, to detect its expression in cell nuclei, section slides were stained with a 1:1,000 dilution of YO-PRO®-1 (Invitrogen), which produces a green signal. YO-PRO-1 is a sensitive detector of membrane permeabilization, and a normally cell-impermeant, monomeric, cyanine dye with a strong binding affinity to nucleic acids. NFκB-positive nuclei (yellow) were quantified in merged images of the two ICB areas of the eyes, as we reported previously [20]. In brief, the results of two areas of each eye were averaged for each sample and in each group and then compared with the total number of stained nuclei in ICB areas to obtain percentage of NFκB colocalized cells. Unless stated otherwise, slide imaging and evaluations were performed under a BZ-9000 fluorescence microscope (Keyence, Osaka, Japan) using software bundled with the apparatus.
Detection of reactive oxygen species
The eyes were dissected from the rats 24 h after LPS injection and frozen fresh in FSC22 frozen section compound (Leica Microsystems, Wetzlar, Germany) with liquid nitrogen. Dihydroethidium (DHE, Sigma), an oxidative fluorescent dye, was used for immunohistochemical detection of cytosolic superoxide anion (O2−). ROS production was determined by conversion of DHE to ethidium bromide. Square areas of ICB microphotographs (1.44 megapixels in size) were selected, and mean gray values of the DHE signal were evaluated with ImageJ software, as we reported recently [21]. Two whole ICB areas were evaluated in each eye and then four eyes were examined in each group.
Statistical analysis
All values are expressed as the mean ± standard error of the mean (SEM) for the respective groups. Statistical significance was evaluated by an unpaired t test. P values less than 0.05 were considered statistically significant.
Results
Effect on cellular infiltration and protein concentration in aqueous humor
Severe uveitis was found in the anterior segments of the rats with EIU 24 h after LPS administration. In the untreated LPS group, the number of inflammatory cells was 288.0±25.0×104 cells/ml. The aqueous infiltrating cell numbers of the rats treated with 10, 1, and 0.1 mg/kg of echinochrome were 11.5±0.9×104, 19.8±2.2×104, and 308.0±16.0×104 cells/ml, respectively. The mean numbers of aqueous inflammatory cells were significantly reduced by treatment with 10 (p<0.01) and 1 (p<0.05) mg/kg of echinochrome (Figure 2A). No infiltrating cells were detected in the aqueous humor of the naïve rats.
Figure 2.
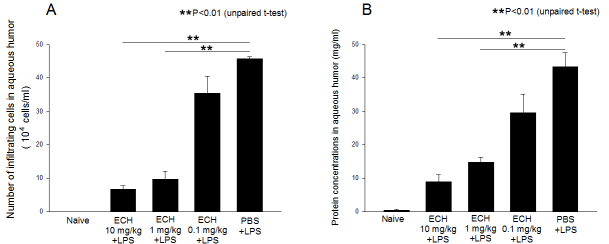
Effects of echinochrome (10, 1, and 0.1 mg/kg) on A: cellular infiltration and B: protein concentration in aqueous humor collected 24 h after lipopolysaccharide (LPS) treatment are shown. Data are presented as mean±standard error of the mean (SEM; n=4). **Groups treated with echinochrome 10 and 1 mg/kg have significantly less infiltrating cells and protein concentrations than untreated endotoxin-induced uveitis (EIU) controls (p<0.01).
A high protein level (43.3±4.2 mg/ml) was detected in the aqueous humor of the untreated EIU rats. The aqueous protein levels of the rats injected with 10, 1, and 0.1 mg/kg of echinochrome were 8.9±2.2, 14.8±1.5, and 29.5±5.5 mg/ml, respectively. Aqueous protein concentration was only 0.48±9.2 mg/ml in the naïve rats.
Protein concentrations in the rats treated with 10 mg/kg (p<0.01) and 1 mg/kg (p<0.01) of echinochrome were significantly lower than that of the untreated LPS group (Figure 2B). The decrease in aqueous cellular infiltration and protein levels indicates a dose-dependent response to echinochrome (Figure 2).
Effect of echinochrome on tumor necrosis factor α in aqueous humor
The aqueous humor of the untreated EIU rats showed considerable production of TNF-α, with a concentration of 457.0±29.0 pg/ml. The aqueous TNF-α levels of the rats treated with 10, 1, and 0.1 mg/kg of echinochrome were 225.7±9.4, 276.0±12.0, and 405.4±26.0 pg/ml, respectively. Treatment with 10 (p<0.05) and 1 mg/kg (p<0.01) of echinochrome significantly reduced the concentrations of TNF-α in the aqueous humor (Figure 3). No TNF-α was detected in the aqueous humor of the naïve controls (below the 15 pg/ml sensitivity threshold of the enzyme-linked immunosorbent assay kit).
Figure 3.
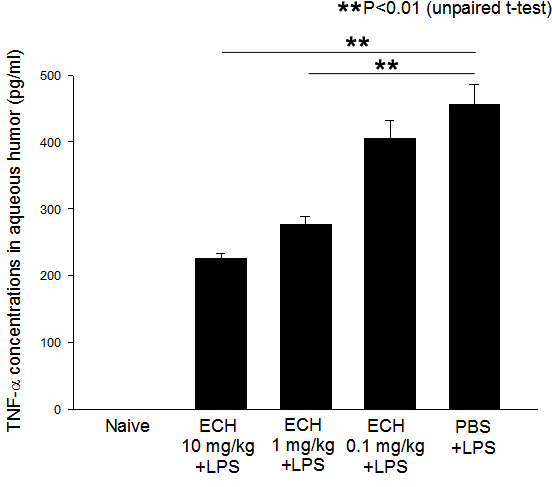
Effects of echinochrome (10, 1, and 0.1 mg/kg) on tumor necrosis factor-α (TNF-α) in aqueous humor collected 24 h after lipopolysaccharide treatment are shown. Data are presented as mean ± standard error of the mean (SEM; n=4). ** TNF-α concentration is significantly lower in echinochrome (10 and 1 mg/kg) treated groups than untreated endotoxin-induced uveitis (EIU) rats (p<0.01).
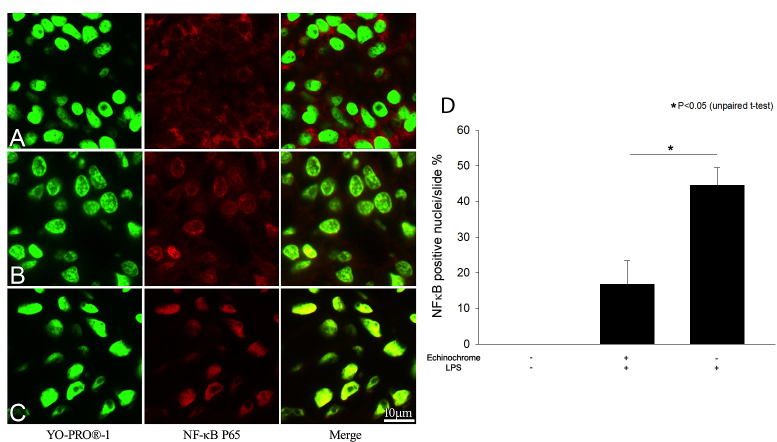
Immunohistochemistry of nuclear factor κB p65 translocation in the iris/ciliary body
Activated NFκB p65 immunoreactivity was examined 3 h after the LPS injections were administered. In the naïve controls, NFκB was stained only in the cytoplasm in its inactivated form, with no nuclear translocation detected (Figure 4A). A few activated NFκB-positive cells were observed in the ICB of the rats treated with echinochrome at 10 mg/kg (Figure 4B); in contrast, NFκB p65 was strongly expressed in the nuclei of the untreated EIU rats (Figure 4C). Active NFκB-positive nuclei were counted to obtain a quantitative measure of NFκB activity in the ICB (Figure 4D). Most of the stained cells were ICB cells. LPS injection resulted in a significant increase in the number of active NFκB-positive cells in the ICB after 3 h (44.5±5.0%). In the group treated with 10 mg/kg echinochrome, the number of active NFκB-positive cells decreased significantly (16.7±6.6; p<0.05). In the naïve control group, no active NFκB-translocated nuclei were detected in the ICB (Figure 4D).
Figure 4.
Effect of echinochrome on nuclear factor κB (NFκB) p65 (red) activation in the iris/ciliary body 3 h after lipopolysaccharide injection is shown. Dual-immunofluorescence labeling showed NFκB colocalization (yellow) in nuclei (green). A: Control naïve animals were not injected with lipopolysaccharide (LPS); an inactive NFκB signal was observed only in the cytoplasm area of cells and no nuclear colocalization of NFκB was detected. B: The group of endotoxin-induced uveitis (EIU) rats treated with echinochrome showed reduced NFκB colocalization compared to C: untreated EIU rats. D: Quantitative analysis of NFκB-positive cells in the iris/ciliary body (ICB). Data are shown as mean±standard error of mean (SEM; n=4). *Amount of NFκB nuclear colocalization was significantly lower in echinochrome treated group than untreated EIU controls (p<0.05).
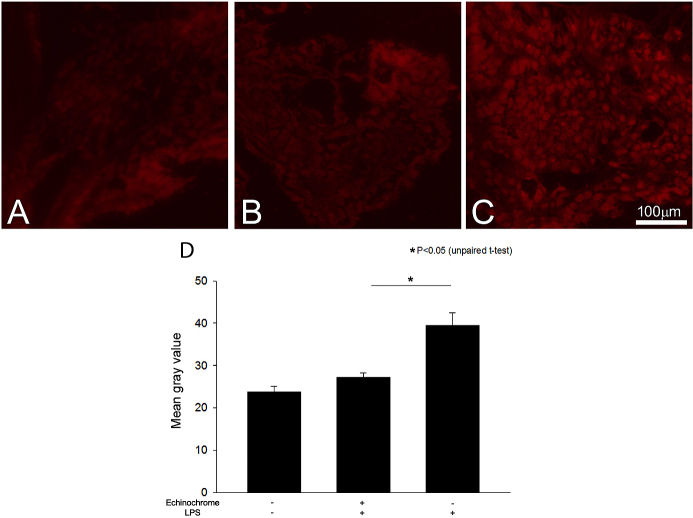
Detection of reactive oxygen species
Confocal microscopic images of ROS production were examined in the ICB tissues. All of the images were made in parallel at identical exposure settings (Figure 5). Only a background ROS signal was detected in the ICB of the naïve rats (Figure 5A). ROS production was lower in the echinochrome-treated EIU rats (Figure 5B) than in the untreated EIU rats, in which a strong ROS signal was detected (Figure 5C). The color of the corneal epithelium on the DHE-stained slides was transformed into grayscale; then, gray values were evaluated by Image J software and summarized (Figure 5D). The mean gray value in the ICB of the naïve rats was 23.8±1.4. The mean gray value of the EIU rats treated with 10 mg/kg of echinochrome (27.2±1.0) was significantly lower (p<0.05) than that of the untreated EIU rats (39.4±3.0).
Figure 5.
Reactive oxygen species (ROS) signal expression 24 h after lipopolysaccharide injection is shown. ROS signal (red) was weak in the A: naïve eyes. B: ROS signal was reduced by echinochrome treatment compared with C: iris/ciliary body (ICB) tissue of untreated endotoxin induced uveitis (EIU) rats, in which ROS signal was strongly expressed. D: Mean gray values of the corneal epithelium in dihydroethidium (DHE)-stained slides of the ICB area were evaluated. Mean gray values were significantly lower in the ICB of rats treated with echinochrome than in the eyes of untreated EIU controls (p<0.05). Data are shown as mean ± standard error of the mean (SEM; n=4). *Mean gray value of echinochrome treated eyes was significantly lower than untreated EIU controls (p<0.05).
Discussion
In this study, we examined the therapeutic effects of the antioxidant compound echinochrome on EIU, an animal model for acute ocular inflammation. Echinochrome is a type of pigment present in sea urchin shells and spines. Small-scale research studies of its biologic effects have been conducted locally in the Russian Far East, which is rich in northern marine resources. However, because many of the studies were reported in Russian, the data from the basic research regarding echinochrome are quite limited outside the area. In the present study, a single intravenous administration of echinochrome significantly ameliorated EIU. Inflammatory cell infiltration and protein levels in the aqueous humor of EIU rats treated with echinochrome were significantly lower than those of untreated EIU rats. Proinflammatory cytokines and ROS activate NFκB, which is a key transcription factor that regulates various inflammatory processes [22]. NFκB is found in the cytoplasm and is bound to inhibitor of kappa B alpha (IκBα) and IκBβ. When cells are stimulated, NFκB dissociates from IκB and migrates into the nucleus, where it binds to specific sequences in the promoter regions of the target genes [23]. The activation of NFκB promotes the expression of several genes, including TNF-α, which mediate inflammation and active immune responses. These cytokines sometimes activate—and on other occasions are activated by—NFκB, thereby inducing a positive feedback loop in inflammatory response [24].
The present study shows that NFκB p65 nuclear translocation in the ICB was significantly suppressed by the single administration of echinochrome. Immunohistochemical findings indicate that NFκB inhibition by echinochrome is achieved via significant reduction of ROS production in ocular tissue, which supports previous data obtained in vivo [7] and in vitro [11,25]. However, this kind of study has some limitations, including the following: (1) the number of test subjects was limited, (2) EIU is an acute model so duration of the study was not long enough to fully access all biological effect of the echinochrome administration, (3) evaluation at different time points were lacking during the course of the experiments. Further studies are needed in the near future.
Several other substances, including resveratrol and astaxanthin, have been reported to have ameliorative effects on EIU by suppressing ROS production [26,27]. However, most of these antioxidant substances are poorly soluble in water, and therefore can only be applied orally or subcutaneously; this reduces the amount of substances that effectively reaches the ocular tissue and delays the onset of the drug’s effect. Echinochrome is water soluble and can be administered intravenously, thereby reaching the eyes within seconds after administration. Echinochrome has already been approved as an experimental treatment for acute infarction in humans, and it has been used as a human dietary supplement in Russia with no reported serious side effects [14]. This supports the findings of the present study. We also checked the systemic side effects of echinochrome injection, and no serious effects were observed in rats at concentrations of up to 25 mg/kg (undiluted solution), as long as the substance was administered slowly (data not shown). Echinochrome can be chemically synthesized [28], but the shells and spines of several sea urchin species provide a cheaper source of spinochromes [29-31]. In Japan and other countries, these parts of sea urchins are usually disposed as industrial trash in food-processing factories after gonads are harvested [32]. The use of sea urchin shells and spines (echinochrome) in health foods and medical products is meaningful both economically and environmentally. As such, the naturally derived pigment echinochrome appears to be a promising candidate as an agent for the safe treatment of ocular inflammatory disorders.
Acknowledgments
This work was supported by grants from the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant Number: 24791826), and Grant-in-aid for the 2012–2013 Research Project of the Research Institute of Personalized Health Sciences, Health Sciences University of Hokkaido. Patent pending (AL, NK, SO, SI)
References
- 1.Kuhn R, Wallenfels K.Über die chemische Natur des Stoffes, den die Eier des Seeigels (Arbacia pustulosa) absondern, um die Spermatozoen anzulocken. Berichte der deutschen chemischen Gessellschaft. 2006:72:1407-1413 [Google Scholar]
- 2.Lebedev AV, Ivanova MV, Ruuge EK. How do calcium ions induce free radical oxidation of hydroxy-1,4-naphthoquinone? Ca2+ stabilizes the naphthosemiquinone anion-radical of echinochrome A. Arch Biochem Biophys. 2003;413:191–8. doi: 10.1016/s0003-9861(03)00111-5. [DOI] [PubMed] [Google Scholar]
- 3.Ageenko NV, Kiselev KV, Odintsova NA. Expression of Pigment Cell-Specific Genes in the Ontogenesis of the Sea Urchin Strongylocentrotus intermedius. Evid-Based Compl Alt 2011:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bay SM, Oshida PS, Jenkins KD. A Simple New Bioassay Based on Echinochrome Synthesis by Larval Sea-Urchins. Mar Environ Res. 1983;8:29–39. [Google Scholar]
- 5.Rhoades GJ. Evaluating Sea Otter Urchin Kelp Relationship Using NASA-LANDSAT Data. J Phycol. 1977;13:57–8. [Google Scholar]
- 6.Pearse JS, Timm RW. Juvenile Nematodes (Echinocephalus pseudouncinatus) in Gonads of Sea Urchins (Centrostephanus coronatus) and Their Effect on Host Gametogenesis. Biol Bull. 1971;140:95–103. doi: 10.2307/1540029. [DOI] [PubMed] [Google Scholar]
- 7.Tsybul'skii AV, Popov AM, Artiukov AA, Kostetskii E, Krivoshapko ON, Mazeika AN, Kozlovskaia EP. The comparative study of the medical action of lyuteolin, rosmarinic acid and echinochrom A at experimental stress-induced cardiopathology. Biomed Khim. 2011;57:314–25. [PubMed] [Google Scholar]
- 8.Tsybul'skii AV, Popov AM, Artiukov AA, Mazeika AN, Kostetskii E, Sanina NM, Krivoshapko ON. Enhancing the immunogenic activity of influvac vaccine in the use of adjuvant TI complexes modified by echinochrome A. Vopr Virusol. 2012;57:23–7. [PubMed] [Google Scholar]
- 9.Shvilkin AV, Serebriakov LI, Tskitishvili OV, Sadretdinov SM, Kol'tsova EA, Maksimov OB, Mishchenko NP, Novikov VL, Levitskii DO, Ruda M. Kardiologiia. 1991;31:79–81. Effect of echinochrom on experimental myocardial reperfusion injury. [PubMed] [Google Scholar]
- 10.Anufriev VPh, Novikov VL, Maximov OB, Elyakov GB, Levitsky DO, Lebedev AV, Sadretdinov SM, Shvilkin AV, Afonskaya NI, Ruda M, Cherpachenko NM. Synthesis of some hydroxynaphthazarins and their cardioprotective effects under ischemia-reperfusion in vivo. Bioorg Med Chem Lett. 1998;8:587–92. doi: 10.1016/s0960-894x(98)00075-4. [DOI] [PubMed] [Google Scholar]
- 11.Lebedev AV, Ivanova MV, Levitsky DO. Echinochrome, a naturally occurring iron chelator and free radical scavenger in artificial and natural membrane systems. Life Sci. 2005;76:863–75. doi: 10.1016/j.lfs.2004.10.007. [DOI] [PubMed] [Google Scholar]
- 12.Zakirova AN, Ivanova MV, Golubiatnikov VB, Mishchenko NP, Kol'tsova EA, Fedoreev SA, Krasnovid NI, Lebedev AV. Pharmacokinetics and clinical efficacy of histochrome in patients with acute myocardial infarction. Eksp Klin Farmakol. 1997;60:21–4. [PubMed] [Google Scholar]
- 13.Buĭmov GA, Maksimov IV, Perchatkin VA, Repin AN, Afanas'ev SA, Markov VA, Karpov RS. Effect of the bioantioxidant histochrome on myocardial injury in reperfusion therapy on patients with myocardial infarction. Ter Arkh. 2002;74:12–6. [PubMed] [Google Scholar]
- 14.Artiukov AA, Popov AM, Tsybul'skiĭ AV, Krivoshapko ON, Poliakova NV. Pharmacological activity echinochrome A singly and consisting of BAA “Timarin”. Biomed Khim. 2012;58:281–90. doi: 10.18097/pbmc20125803281. [DOI] [PubMed] [Google Scholar]
- 15.Rosenbaum JT, McDevitt HO, Guss RB, Egbert PR. Endotoxin-induced uveitis in rats as a model for human disease. Nature. 1980;286:611–3. doi: 10.1038/286611a0. [DOI] [PubMed] [Google Scholar]
- 16.Yadav UC, Srivastava SK, Ramana KV. Aldose reductase inhibition prevents endotoxin-induced uveitis in rats. Invest Ophthalmol Vis Sci. 2007;48:4634–42. doi: 10.1167/iovs.07-0485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baeuerle PA, Henkel T. Function and activation of NF-kappa B in the immune system. Annu Rev Immunol. 1994;12:141–79. doi: 10.1146/annurev.iy.12.040194.001041. [DOI] [PubMed] [Google Scholar]
- 18.de Vos AF, van Haren MA, Verhagen C, Hoekzema R, Kijlstra A. Kinetics of intraocular tumor necrosis factor and interleukin-6 in endotoxin-induced uveitis in the rat. Invest Ophthalmol Vis Sci. 1994;35:1100–6. [PubMed] [Google Scholar]
- 19.Planck SR, Huang XN, Robertson JE, Rosenbaum JT. Cytokine mRNA levels in rat ocular tissues after systemic endotoxin treatment. Invest Ophthalmol Vis Sci. 1994;35:924–30. [PubMed] [Google Scholar]
- 20.Lennikov A, Kitaichi N, Noda K, Ando R, Dong Z, Fukuhara J, Kinoshita S, Namba K, Mizutani M, Fujikawa T, Itai A, Ohno S, Ishida S. Amelioration of endotoxin-induced uveitis treated with an IkappaB kinase beta inhibitor in rats. Mol Vis. 2012;18:2586–97. [PMC free article] [PubMed] [Google Scholar]
- 21.Lennikov A, Kitaichi N, Fukase R, Murata M, Noda K, Ando R, Ohguchi T, Kawakita T, Ohno S, Ishida S. Amelioration of ultraviolet-induced photokeratitis in mice treated with astaxanthin eye drops. Mol Vis. 2012;18:455–64. [PMC free article] [PubMed] [Google Scholar]
- 22.Barnes PJ. Nuclear factor-kappa B. Int J Biochem Cell Biol. 1997;29:867–70. doi: 10.1016/s1357-2725(96)00159-8. [DOI] [PubMed] [Google Scholar]
- 23.Baldwin AS., Jr The NF-kappa B and I kappa B proteins: new discoveries and insights. Annu Rev Immunol. 1996;14:649–83. doi: 10.1146/annurev.immunol.14.1.649. [DOI] [PubMed] [Google Scholar]
- 24.Barnes PJ, Karin M. Nuclear factor-kappaB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med. 1997;336:1066–71. doi: 10.1056/NEJM199704103361506. [DOI] [PubMed] [Google Scholar]
- 25.Glazunov VP, Berdyshev DV, Novikov VL. 7-Ethyl-2,3,5,6,8-pentahydroxy-1,4-naphthoquinone (echinochrome A): a DFT study of the antioxidant mechanism. 3. The structures of dianions and disodium salts of echinochrome A and their reactions with the hydroperoxyl radical. Russ Chem Bull. 2010;59:2185–202. [Google Scholar]
- 26.Kubota S, Kurihara T, Mochimaru H, Satofuka S, Noda K, Ozawa Y, Oike Y, Ishida S, Tsubota K. Prevention of ocular inflammation in endotoxin-induced uveitis with resveratrol by inhibiting oxidative damage and nuclear factor-kappaB activation. Invest Ophthalmol Vis Sci. 2009;50:3512–9. doi: 10.1167/iovs.08-2666. [DOI] [PubMed] [Google Scholar]
- 27.Suzuki Y, Ohgami K, Shiratori K, Jin XH, Ilieva I, Koyama Y, Yazawa K, Yoshida K, Kase S, Ohno S. Suppressive effects of astaxanthin against rat endotoxin-induced uveitis by inhibiting the NF-kappaB signaling pathway. Exp Eye Res. 2006;82:275–81. doi: 10.1016/j.exer.2005.06.023. [DOI] [PubMed] [Google Scholar]
- 28.Peña-Cabrera E, Liebeskind LS. Squaric acid ester-based total synthesis of echinochrome A. J Org Chem. 2002;67:1689–91. doi: 10.1021/jo016034m. [DOI] [PubMed] [Google Scholar]
- 29.Nishibori K. Isolation of Echinochrome A from Spines of Sea Urchin, Stomopneustes variolaris (Lamarck). Nature. 1961;192:1293–4. doi: 10.1038/1921293a0. [DOI] [PubMed] [Google Scholar]
- 30.Nishibori K. Isolation of Echinochrome A from the Spines of the Sea Urchin, Diadema setosum (Leske). Nature. 1959;184:1234. doi: 10.1038/1921293a0. [DOI] [PubMed] [Google Scholar]
- 31.Zhou DY, Qin L, Zhu BW, Wang XD, Tan H, Yang JF, Li DM, Dong XP, Wu HT, Sun LM, Li XL, Murata Y. Extraction and antioxidant property of polyhydroxylated naphthoquinone pigments from spines of purple sea urchin Strongylocentrotus nudus. Food Chem. 2011;129:1591–7. [Google Scholar]
- 32.Garau G, Castaldi P, Deiana S, Campus P, Mazza A, Deiana P, Pais A. Assessment of the use potential of edible sea urchins (Paracentrotus lividus) processing waste within the agricultural system: Influence on soil chemical and biological properties and bean (Phaseolus vulgaris) and wheat (Triticum vulgare) growth in an amended acidic soil. J Environ Manage. 2012;109:12–8. doi: 10.1016/j.jenvman.2012.05.001. [DOI] [PubMed] [Google Scholar]