According to the classical incretin concept, glucagon-like peptide (GLP)-1 is viewed as a hormone produced in the intestinal L cells and acting via the circulation on satiety in the brain, gut motility, and insulin and glucagon secretion in the pancreatic islet. However, in contrast to typical hormones, plasma levels of GLP-1 are relatively low with a very short half-life. Furthermore, GLP-1 is rapidly inactivated by dipeptidyl peptidase-4 (DPP-4) in the vicinity of L cells within <1 min from the secretion of the gut peptide (1,2). This rapid metabolism of GLP-1 raises questions about how its effects are mediated on target organs such as pancreatic β-cells. In this review, we will discuss possible alternative pathways for the incretin effect on pancreatic islet. These involve L cell–derived GLP-1 via neuronal activation and α-cell–derived GLP-1 via auto/paracrine effects. Of note, GLP-1 is not acting alone, and its effect can be modulated by other factors including GIP. Since no data are available yet in the present context, we have limited this review to GLP-1.
GLP-1 in the gut-to-brain-to-periphery axis for the control of glucose metabolism
Recent rodent data show that GLP-1 can induce its metabolic actions by interacting with its receptors in extrapancreatic locations such as the gut to activate the submucosal and the myenteric nervous plexi (3,4) and the brain, which then transmit the signal to peripheral tissues (5) within minutes from the absorption of glucose and lipids. The final aim of this axis is to anticipate the breakthrough of the nutrients into the blood and their better handling. Indeed, GLP-1 secreted from L cells can influence brain neuronal activities via an alternative neural pathway initiated by sensors in the hepatic portal region (6–8). Thereby, the vagus nerve transmits the metabolic information to the nucleus tractus solitarii in the brain stem, which relays the glucose signal to hypothalamic nuclei (9). This process is called the gut-to-brain-to-periphery axis. Seminal studies from our group showed that the direct infusion of glucose into the portal vein of mice at a low rate increased muscle glucose utilization through an insulin-independent mechanism (10–12). This process required the activation of the hepatoportal vein glucose sensor (6). The blockage of the portal GLP-1 receptor by exendin 9 directly into the hepatoportal vein or in GLP-1 receptor knockout mice (8) prevented the portal glucose sensor activation for the control of muscle glucose utilization (8) or insulin secretion (8,13). Furthermore, the inhibition of the enteric DPP-4 by small doses of DPP-4 inhibitor improved glucose tolerance without increasing the blood concentration of GLP-1 through a GLP-1 receptor–dependent manner (3). In such conditions, the vagus nerve activity was increased in response to oral DPP-4 inhibitors, whereas the intravenous administration of the drug had no therapeutic effect, further suggesting the important role of enteric GLP-1 on the control of glycemia through the activation of the gut brain axis. Furthermore, the direct administration of the DPP-4 inhibitor into the rat portal vein significantly increased portal (but not peripheral) GLP-1 and insulin levels and decreased glucose concentrations (14). However, despite the large amount of experimental evidence described above showing the important role of GLP-1 on the gut-to-brain axis, a recent observation in mice suggests that the circulating GLP-1 could also directly access the brain and the β-cells and induce insulin secretion (15). Transgenic mice that expressed the human GLP-1 receptor in islets and in pancreatic ductal cells within the background of the GLP-1 receptor knockout mice were characterized by increased glucose-induced insulin secretion that was sufficient to normalize glucose tolerance, whereas no effect on food intake, hindbrain c-fos expression, or gastric emptying was observed (15). This new set of data suggests that part of the gut-released GLP-1 may resemble some of the incretin effect through a process not involving the gut-brain axis. Alternatively or additionally, the release of GLP-1 from an intraislet processing may contribute to triggering glucose-induced insulin secretion (see below).
A further demonstration of the role played by the GLP-1–dependent gut-brain axis is the recent analysis of the therapeutic role of GLP-1 receptor agonists on neuropathy in mice with diabetes owing to streptozotocin (16). The authors showed the presence of the GLP-1 receptor on the lumbar dorsal root ganglion by immunohistochemical analyses and further demonstrated that exendin-4 increases the neurite outgrowth. Importantly, the delayed current perception threshold and motor and sensory nerve conduction velocity impaired by type 1 diabetes, was improved by the GLP-1 agonist (16). Hence, gut-released hormone would further favor the gut-brain axis by controlling the enteric neural development. Similarly, a therapeutic role of the gut-brain axis has been proposed regarding the therapeutic efficacy of gastric bypass. Obese and diabetic patients who underwent this type of bariatric surgery lose weight within months and reverse their diabetes status within weeks from the surgery. It has been proposed that a hormonal characteristic is that these patients secrete large amounts of GLP-1 (17) that could even lead to uncontrolled insulin secretion and hypoglycemic episodes (18), although this still needs to be confirmed. However, numerous other peptides such as peptide YY, oxyntomodulin, and GLP-2 may be responsible for the activation of the gut-brain axis in patients with bypass surgery (19).
Altogether, the role of GLP-1 on the gut-to-brain-to-periphery axis is now considered a major mechanism of action of the gut hormone for the control of glycemia. Many controversial debates are ongoing regarding its role in the control of food intake. Similarly, the production of GLP-1 into the brain and the colocalized receptors have also been important fields of investigation in explaining the metabolic effect of this insulinotropic peptide.
α-Cell as a source of GLP-1
GLP-1 is spliced from its precursor proglucagon in intestinal L cells through processing by the enzyme PC1/3 (20–23). In the pancreatic α-cell, proglucagon is processed by PC2 to yield glucagon (24,25). Adult α-cells are thought to produce little GLP-1. However, a switch of PC2 to PC1/3 is sufficient to convert the α-cell from a hyperglycemia-promoting cell to one that lowers blood glucose levels and promotes islet survival (26). Furthermore, several animal studies show that islet-derived GLP-1 may be stimulated by increased demand of insulin secretion, suggesting that locally produced GLP-1 may play a role in the long-term β-cell adaptation. Indeed, β-cell destruction with streptozotocin leads to hyperglycemia followed by β-cell regeneration associated with α-cell hyperplasia and pancreatic PC1/3 upregulation along with increased pancreatic and circulating levels of GLP-1 and GLP-1–to–glucagon ratio (27,28). Interestingly, blocking GLP-1 reduced β-cell regeneration. Similarly, α-cell hyperplasia along with PC1/3 upregulation and GLP-1 production was observed in mouse models of insulin resistance including pregnant, ob/ob, db/db, and prediabetic NOD mice (29). When considered together, these studies strongly suggest that α-cell hyperplasia and release of GLP-1 play an integral role in β-cell regeneration. In line with this concept, α-cell hyperplasia occurs early in response to high-fat diet and preceding β-cell expansion and is required for β-cell adaptation (30).
The next step was to uncover the molecular trigger of PC1/3 with subsequent GLP-1 production in α-cells. Two important studies point to a direct effect of elevated glucose concentrations. Thus, in the Psammomy obesus, high-energy diet induces diabetes along with a significant increase of GLP-1 in the portal vein and pancreas extract (31). Isolated islets from hyperglycemic animals released more GLP-1 than in controls. This ex vivo effect was confirmed in vitro by culture of normal P. obesus islets with high glucose, which induced GLP-1 release. Similarly, high glucose concentrations increased PC1/3 in rat islets, while in an α-cell line glucose increased GLP-1 and decreased glucagon secretion (32).
An additional trigger of GLP-1 is interleukin (IL)-6. Indeed, type 2 diabetes and obesity are characterized by an activation of the innate immune system reflected by an increase in various inflammatory markers including elevated plasma levels of IL-6 (33). It was shown in mice that elevated IL-6 levels in response to exercise (34), as well as acute and chronic IL-6 administration in mice, can stimulate GLP-1 secretion from intestinal L cells and pancreatic α-cells leading to improved glucose homeostasis (35). In vitro, IL-6 increased GLP-1 synthesis and secretion from entero-endocrine L cells both acutely and chronically. IL-6 also increased GLP-1 synthesis and secretion from human pancreatic α-cells in association with increased proglucagon and PC1/3 transcription. Similarly, the beneficial effects of elevated IL-6 were shown in animal models of type 2 diabetes and obesity. Furthermore, IL-6 neutralization deteriorated glycemic control and reduced pancreatic GLP-1 content. Hence, IL-6 mediates cross-talk between insulin-sensitive tissues, L cells, and pancreatic islets to adapt to changes in insulin demand by increasing L-cell GLP-1 secretion and reprogramming α-cells to process proglucagon to GLP-1.
The question remains of the importance of α-cell–derived compared with systemic GLP-1. An argument against a significant contribution by the α-cell is the islet architecture. Indeed, in most rodents, β-cells compose the core of the islets while the α-cells form the mantle region. Furthermore, it is believed that the blood flow goes from the center to the periphery, making it difficult to imagine that GLP-1 released from α-cells could act on β-cells. However, in animal models of diabetes in which insulin secretion is decreased, normal organization of islet cells was found to be perturbed so that β-cells were intermingled with non-β-cells (36). Moreover, a study describing the architecture in human islets reported direct intercellular contacts between α- and β-cells, supporting the notion that α-cell products can act in a paracrine manner to regulate the β-cell (37). In support of this, acetylcholine secreted by α-cells acts in a paracrine manner to prime the β-cell to respond optimally to subsequent increases in glucose (38). Hence, release of GLP-1 directly into the islets at the vicinity of the β-cells might be a potent way to trigger cell survival and insulin production. Another issue is the activity of the GLP-1 species released by α-cells. Indeed, under normal conditions α-cells do not process proglucagon to active GLP-1 (39). However, metabolic stress appears to reprogram the α-cells, allowing processing of active GLP-1. Indeed, islet-derived GLP-1 activity was demonstrated in human islets using a bioassay and for mouse islet experiments using GLP-1 receptor antagonists and Glp1r−/− mice (35). Furthermore, GLP-1 derived from human islets has been described ex vivo from patients with type 2 diabetes (31,40). Importantly, the presence of a functionally competent GLP-1 system was recently demonstrated (40). Finally, in human islets, after IL-6 stimulation, the GLP-1–to–glucagon molar ratio is >1.5 (35); therefore, it can be postulated that this is a meaningful phenomenon. Clearly, this hypothesis remains to be demonstrated by direct evidences.
Summary and proposed hypothesis
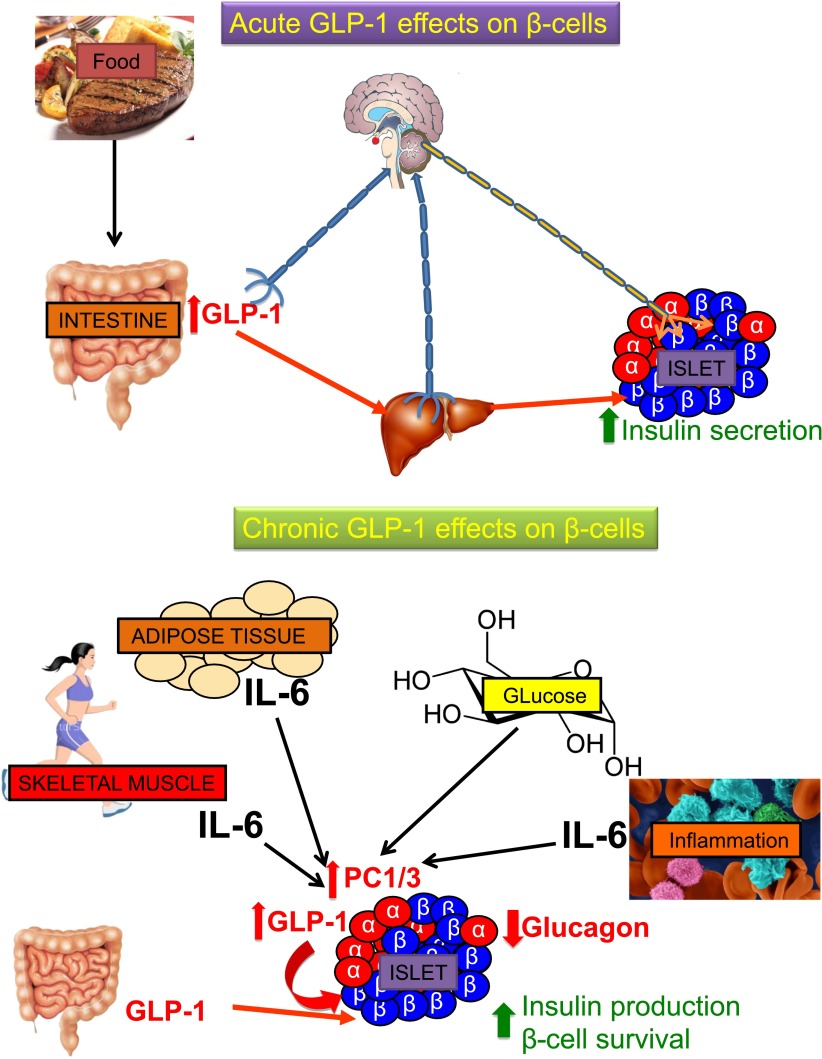
In summary, we propose the following working hypothesis for the GLP-1 effect on the pancreatic islet (Fig. 1). The acute postprandial-mediated incretin effect would be mediated mainly by GLP-1 released from L cells acting locally and via portal release into the liver on neuronal activation. Chronic trophic effects of GLP-1 on β-cells promoting survival and insulin production would be mediated via reprograming of the α-cell by increasing PC1/3. This would switch proglucagon processing from glucagon to GLP-1 allowing for auto/paracrine effects. The trigger for this long-term adaptation could be IL-6 released by insulin-resistant fat tissues by contracting muscles or by insulitis as well as hyperglycemia. Accordingly, the main action of DPP-4 inhibition may occur at the tissue rather than at the plasma level.
Figure 1.
Hypothetical model for the acute and chronic effects of GLP-1 on pancreatic islets.
We do realize that several aspects of this hypothesis are controversial or not sufficiently supported by experimental data. In particular, the relative contribution of circulating GLP-1 versus neuronal and auto/paracrine remains to be clarified. Finally, most of the findings described were obtained using rodents, isolated human islets, or animal models; therefore, the in vivo relevance for humans remains to be demonstrated.
Acknowledgments
No potential conflicts of interest relevant to this article were reported.
M.Y.D. and R.B. wrote the manuscript. M.Y.D. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This publication is based on the presentations from the 4th World Congress on Controversies to Consensus in Diabetes, Obesity and Hypertension (CODHy). The Congress and the publication of this supplement were made possible in part by unrestricted educational grants from Abbott, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Ethicon Endo-Surgery, Janssen, Medtronic, Novo Nordisk, Sanofi, and Takeda.
References
- 1.Hansen L, Deacon CF, Orskov C, Holst JJ. Glucagon-like peptide-1-(7-36)amide is transformed to glucagon-like peptide-1-(9-36)amide by dipeptidyl peptidase IV in the capillaries supplying the L cells of the porcine intestine. Endocrinology 1999;140:5356–5363 [DOI] [PubMed] [Google Scholar]
- 2.Deacon CF. Circulation and degradation of GIP and GLP-1. Horm Metab Res 2004;36:761–765 [DOI] [PubMed] [Google Scholar]
- 3.Waget A, Cabou C, Masseboeuf M, et al. Physiological and pharmacological mechanisms through which the DPP-4 inhibitor sitagliptin regulates glycemia in mice. Endocrinology 2011;152:3018–3029 [DOI] [PubMed] [Google Scholar]
- 4.Washington MC, Raboin SJ, Thompson W, Larsen CJ, Sayegh AI. Exenatide reduces food intake and activates the enteric nervous system of the gastrointestinal tract and the dorsal vagal complex of the hindbrain in the rat by a GLP-1 receptor. Brain Res 2010;1344:124–133 [DOI] [PubMed] [Google Scholar]
- 5.Cabou C, Burcelin R. GLP-1, the gut-brain, and brain-periphery axes. Rev Diabet Stud 2011;8:418–431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nakagawa A, Satake H, Nakabayashi H, et al. Receptor gene expression of glucagon-like peptide-1, but not glucose-dependent insulinotropic polypeptide, in rat nodose ganglion cells. Auton Neurosci 2004;110:36-43 [DOI] [PubMed]
- 7.Baraboi ED, St-Pierre DH, Shooner J, Timofeeva E, Richard D. Brain activation following peripheral administration of the GLP-1 receptor agonist exendin-4. Am J Physiol Regul Integr Comp Physiol 2011;301:R1011–R1024 [DOI] [PubMed] [Google Scholar]
- 8.Burcelin R, Da Costa A, Drucker D, Thorens B. Glucose competence of the hepatoportal vein sensor requires the presence of an activated glucagon-like peptide-1 receptor. Diabetes 2001;50:1720–1728 [DOI] [PubMed] [Google Scholar]
- 9.Shimizu N, Oomura Y, Novin D, Grijalva CV, Cooper PH. Functional correlations between lateral hypothalamic glucose-sensitive neurons and hepatic portal glucose-sensitive units in rat. Brain Res 1983;265:49–54 [DOI] [PubMed] [Google Scholar]
- 10.Burcelin R, Dolci W, Thorens B. Portal glucose infusion in the mouse induces hypoglycemia: evidence that the hepatoportal glucose sensor stimulates glucose utilization. Diabetes 2000;49:1635–1642 [DOI] [PubMed] [Google Scholar]
- 11.Burcelin R, Dolci W, Thorens B. Glucose sensing by the hepatoportal sensor is GLUT2-dependent: in vivo analysis in GLUT2-null mice. Diabetes 2000;49:1643–1648 [DOI] [PubMed] [Google Scholar]
- 12.Burcelin R, Crivelli V, Perrin C, et al. GLUT4, AMP kinase, but not the insulin receptor, are required for hepatoportal glucose sensor-stimulated muscle glucose utilization. J Clin Invest 2003;111:1555–1562 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Preitner F, Ibberson M, Franklin I, et al. Gluco-incretins control insulin secretion at multiple levels as revealed in mice lacking GLP-1 and GIP receptors. J Clin Invest 2004;113:635–645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fujiwara K, Gotoh K, Chiba S, et al. Intraportal administration of DPP-IV inhibitor regulates insulin secretion and food intake mediated by the hepatic vagal afferent nerve in rats. J Neurochem 2012;121:66–76 [DOI] [PubMed] [Google Scholar]
- 15.Lamont BJ, Li Y, Kwan E, Brown TJ, Gaisano H, Drucker DJ. Pancreatic GLP-1 receptor activation is sufficient for incretin control of glucose metabolism in mice. J Clin Invest 2012;122:388–402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Himeno T, Kamiya H, Naruse K, et al. Beneficial effects of exendin-4 on experimental polyneuropathy in diabetic mice. Diabetes 2011;60:2397–2406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Van der Schueren BJ, Homel P, Alam M, et al. Magnitude and variability of the glucagon-like peptide-1 response in patients with type 2 diabetes up to 2 years following gastric bypass surgery. Diabetes Care 2012;35:42–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Myint KS, Greenfield JR, Farooqi IS, Henning E, Holst JJ, Finer N. Prolonged successful therapy for hyperinsulinaemic hypoglycaemia after gastric bypass: the pathophysiological role of GLP1 and its response to a somatostatin analogue. Eur J Endocrinol 2012;166:951–955 [DOI] [PubMed]
- 19.Lee WJ, Chen CY, Chong K, Lee YC, Chen SC, Lee SD. Changes in postprandial gut hormones after metabolic surgery: a comparison of gastric bypass and sleeve gastrectomy. Surg Obes Relat Dis 2011;7:683–690 [DOI] [PubMed]
- 20.Lund PK, Goodman RH, Dee PC, Habener JF. Pancreatic preproglucagon cDNA contains two glucagon-related coding sequences arranged in tandem. Proc Natl Acad Sci USA 1982;79:345–349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bell GI, Santerre RF, Mullenbach GT. Hamster preproglucagon contains the sequence of glucagon and two related peptides. Nature 1983;302:716–718 [DOI] [PubMed] [Google Scholar]
- 22.Rouillé Y, Bianchi M, Irminger JC, Halban PA. Role of the prohormone convertase PC2 in the processing of proglucagon to glucagon. FEBS Lett 1997;413:119–123 [DOI] [PubMed] [Google Scholar]
- 23.Zhu X, Zhou A, Dey A, et al. Disruption of PC1/3 expression in mice causes dwarfism and multiple neuroendocrine peptide processing defects. Proc Natl Acad Sci USA 2002;99:10293–10298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rouillé Y, Martin S, Steiner DF. Differential processing of proglucagon by the subtilisin-like prohormone convertases PC2 and PC3 to generate either glucagon or glucagon-like peptide. J Biol Chem 1995;270:26488–26496 [DOI] [PubMed] [Google Scholar]
- 25.Furuta M, Zhou A, Webb G, et al. Severe defect in proglucagon processing in islet A-cells of prohormone convertase 2 null mice. J Biol Chem 2001;276:27197–27202 [DOI] [PubMed] [Google Scholar]
- 26.Wideman RD, Covey SD, Webb GC, Drucker DJ, Kieffer TJ. A switch from prohormone convertase (PC)-2 to PC1/3 expression in transplanted alpha-cells is accompanied by differential processing of proglucagon and improved glucose homeostasis in mice. Diabetes 2007;56:2744–2752 [DOI] [PubMed] [Google Scholar]
- 27.Nie Y, Nakashima M, Brubaker PL, et al. Regulation of pancreatic PC1 and PC2 associated with increased glucagon-like peptide 1 in diabetic rats. J Clin Invest 2000;105:955–965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Thyssen S, Arany E, Hill DJ. Ontogeny of regeneration of beta-cells in the neonatal rat after treatment with streptozotocin. Endocrinology 2006;147:2346–2356 [DOI] [PubMed] [Google Scholar]
- 29.Kilimnik G, Kim A, Steiner DF, Friedman TC, Hara M. Intraislet production of GLP-1 by activation of prohormone convertase 1/3 in pancreatic α-cells in mouse models of β-cell regeneration. Islets 2010;2:149–155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ellingsgaard H, Ehses JA, Hammar EB, et al. Interleukin-6 regulates pancreatic alpha-cell mass expansion. Proc Natl Acad Sci USA 2008;105:13163–13168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hansen AM, Bödvarsdottir TB, Nordestgaard DN, et al. Upregulation of alpha cell glucagon-like peptide 1 (GLP-1) in Psammomys obesus—an adaptive response to hyperglycaemia? Diabetologia 2011;54:1379–1387 [DOI] [PubMed] [Google Scholar]
- 32.Whalley NM, Pritchard LE, Smith DM, White A. Processing of proglucagon to GLP-1 in pancreatic α-cells: is this a paracrine mechanism enabling GLP-1 to act on β-cells? J Endocrinol 2011;211:99–106 [DOI] [PubMed] [Google Scholar]
- 33.Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol 2011;11:98–107 [DOI] [PubMed] [Google Scholar]
- 34.Febbraio MA, Pedersen BK. Muscle-derived interleukin-6: mechanisms for activation and possible biological roles. FASEB J 2002;16:1335–1347 [DOI] [PubMed] [Google Scholar]
- 35.Ellingsgaard H, Hauselmann I, Schuler B, et al. Interleukin-6 enhances insulin secretion by increasing glucagon-like peptide-1 secretion from L cells and alpha cells. Nat Med 2011;17:1481–1489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gannon M, Ray MK, Van Zee K, Rausa F, Costa RH, Wright CV. Persistent expression of HNF6 in islet endocrine cells causes disrupted islet architecture and loss of beta cell function. Development 2000;127:2883–2895 [DOI] [PubMed] [Google Scholar]
- 37.Bosco D, Armanet M, Morel P, et al. Unique arrangement of alpha- and beta-cells in human islets of Langerhans. Diabetes 2010;59:1202–1210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rodriguez-Diaz R, Dando R, Jacques-Silva MC, et al. Alpha cells secrete acetylcholine as a non-neuronal paracrine signal priming beta cell function in humans. Nat Med 2011;17:888–892 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holst JJ, Bersani M, Johnsen AH, Kofod H, Hartmann B, Orskov C. Proglucagon processing in porcine and human pancreas. J Biol Chem 1994;269:18827–18833 [PubMed] [Google Scholar]
- 40.Marchetti P, Lupi R, Bugliani M, et al. A local glucagon-like peptide 1 (GLP-1) system in human pancreatic islets. Diabetologia 2012;55:3262–3272 [DOI] [PubMed] [Google Scholar]