Abstract
Objectives:
The main purpose of this study was to determine the accuracy of cone beam CT (CBCT) in measuring the trabecular bone microstructure, in comparison with micro-CT. The subobjective was to examine to what extent bone quality assessment is influenced by X-ray tube current and voltage settings as well as soft tissue surrounding the bone.
Methods:
Eight human mandibular bone samples were scanned using three different clinical exposure protocol within water (W1–3) and without water (NW1–3) by a high-resolution (80 µm) CBCT machine (3D Accuitomo 170®; Morita, Kyoto, Japan). Subsequently, the samples underwent micro-CT scanning (SkyScan 1174®; SkyScan, Antwerp, Belgium). After image acquisition, similar volumes of interest of the trabecular structures captured with CBCT and micro-CT were aligned with each other. Segmentation was then performed, and the morphometric parameters were quantified within the volumes of interest by CTAn software (CTAnalyser®; SkyScan, Antwerp, Belgium). Descriptive statistical analyses and multiple comparisons between all protocols were applied in R software.
Results:
High positive Pearson's correlation coefficients were observed between CBCT and micro-CT protocols for all tested morphometric indices except for trabecular thickness. No significant differences were observed between all exposure protocols except for trabecular separation. When examining the soft-tissue effect on trabecular bone structures, no significant differences between NW (1–3) and W (1–3) protocols were observed for all variables.
Conclusions:
The present study demonstrated the potential of high-resolution CBCT imaging for in vivo applications of quantitative bone morphometry and bone quality assessment. However, the overestimation of morphometric parameters and acquisition settings in CBCT must be taken into account.
Keywords: cone beam CT, micro-computed tomography, bone architecture, quantitative bone morphometry
Introduction
Quantitative bone morphometry is the standard method to assess structural properties of trabeculae by means of morphometric indices.1 In the past, microarchitectural characteristics of trabecular and cortical bone have been intensively investigated by examining two-dimensional (2D) sections of bone biopsies, combined with calculation of morphometric parameters using stereological methods.2 Although such histological analysis offers a high spatial resolution and good image contrast, it remains a labour-intensive and thus time-consuming job. Moreover, it is a destructive technique that allows tissue quantification in only a limited number of 2D sections and, therefore, prevents the specimens from being used for other comparative measures. The latter is highly desirable because of the anisotropic nature of the cancellous bone. To overcome some of the limitations of 2D analyses, various three-dimensional (3D) imaging modalities of analysis techniques have been proposed. Recently, micro-CT (µCT) has been validated as a highly reliable tool to determine trabecular bone parameters, while being considered as the new reference method for ex vivo bone studies.3 Nevertheless, µCT has hardly any clinical value for structural bone analysis. This is where cone beam CT (CBCT) may come into play.
CBCT is a new emerging radiographic method possessing plenty of benefits regarding radiation dose, cost effectiveness, scanning time and 3D modalities in evaluating the trabecular bone structure in a clinically objective and quantitative way. Still, bone characterization using morphometry with CBCT has not yet been properly investigated. To establish CBCT as a method for 3D assessment and analysis of trabecular bone, the method needs proper validation by comparing the results with 3D μCT, serving as the reference (gold standard). For a long time, CBCT's main drawback, when compared with µCT, was its low spatial resolution (order of 200–300 µm).4 During the last decade, various advancements within the CBCT imaging chain have led to clear improvements in resolution. However, spatial resolution is highly variable between CBCT devices, with voxel sizes between 76 μm and 400 μm and actual sharpness showing wide ranges on both a clinical level and a technical level. A previous study has shown that images with voxel sizes higher than 300 μm would be unsuitable for imaging individual trabeculae.5 Given that various CBCT devices use voxel sizes well below that limit, it may be possible to use them to evaluate trabecular morphology. It remains to be investigated to what degree the resolution provided by CBCT scans allows correct computation of morphometric indices, considering that the smallest voxel sizes in CBCT are in the order of typical trabecular thicknesses. Additionally, X-ray tube settings and soft-tissue surrounding samples could affect the morphological parameters and the associated clinical evaluation of bone architecture.
The main purpose of this study was to determine the accuracy of CBCT for measuring the trabecular bone microstructure in comparison with μCT. Secondly, the effect of X-ray tube current and voltage settings, as well as soft tissue surrounding the bone, on bone quality assessment was investigated.
Material and methods
Image acquisition
Eight edentulous human bone samples from the left and right mandible (first premolar to third molar) were scanned using a high-resolution CBCT machine (80 μm), which was calibrated for the radiographic study (3D Accuitomo 170®; Morita, Kyoto, Japan). Each sample was placed in a sponge block to prevent any movement during the scanning process. The mandibles were scanned using three different clinical exposure protocols within water (W) and without water (NW). Scanning parameters were fixed at 360° rotation, 30.8 s exposure time, 80 µm voxel size, high resolution and a field of view of 40 × 40 mm. Tube voltage and current were varied using three combinations: (1) 90 kVp, 5 mA, 8.1 mGy CT dose index (CTDIvol); (2) 90 kVp, 2 mA, 3.4 mGy CTDIvol and (3) 70 kVp, 5 mA, 4.8 mGy CTDIvol. All CTDIvol values were predetermined by the manufacturer and served as a dose estimation.
The bone samples were subsequently scanned with the SkyScan 1174® μCT system (SkyScan, Antwerp, Belgium). The μCT parameters were 26 μm voxel size, 50 kVp, 800 µA, 1 mm aluminium filter, angular rotation step 0.8°, 360° scanning, 450 projections and an exposure time of 9 s with a total scan duration of 2 h 20 min.
Image processing and analysis
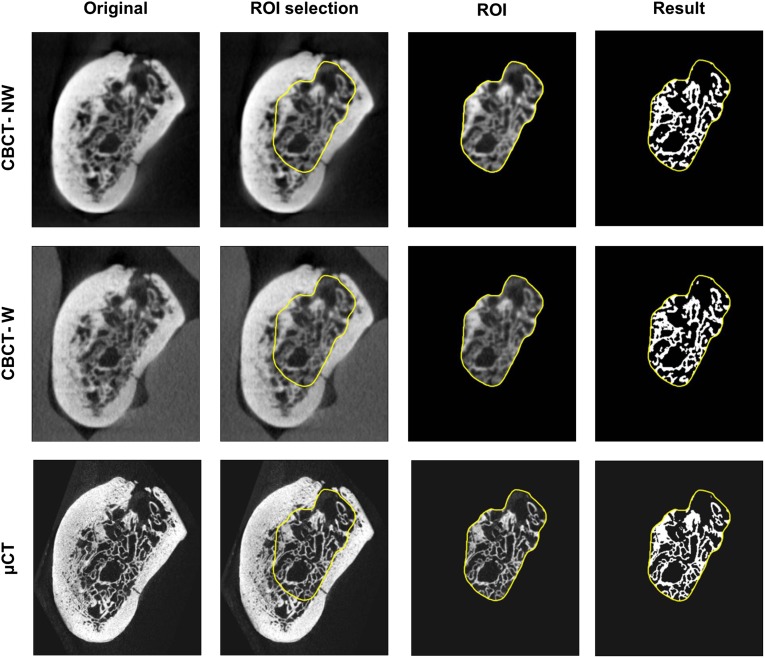
To evaluate the microarchitecture of the trabecular bone, a volume of interest (VOI) comprising trabecular bone only was delineated in the μCT image of each sample. To minimize errors generated by comparing different morphological structures, it is desirable to obtain a VOI comprising the exact same structures in the CBCT images of the respective samples. This was achieved by computing the rigid transformation that spatially aligns each CBCT scan of a sample with the μCT image of the corresponding sample, using registration software based on mutual information.6 This transformation was applied to the VOI of the μCT images, resulting in highly accurate delineation of the VOI in the coordinate system of the CBCT images. This approach does not alter the resolution of the μCT or CBCT images, allowing a fair comparison without introducing errors by unmatched VOIs. An impression of how the same VOI is applied to different images is given in Figure 1.
Figure 1.
Registered cone beam CT (CBCT) and micro-CT (μCT) images with the same volume of interest (VOI) containing the trabecular bone. For display purposes, images are transformed to the coordinate system of the CBCT scan with water. In our method, only the VOI is transformed to prevent loss of resolution of the images during transformation
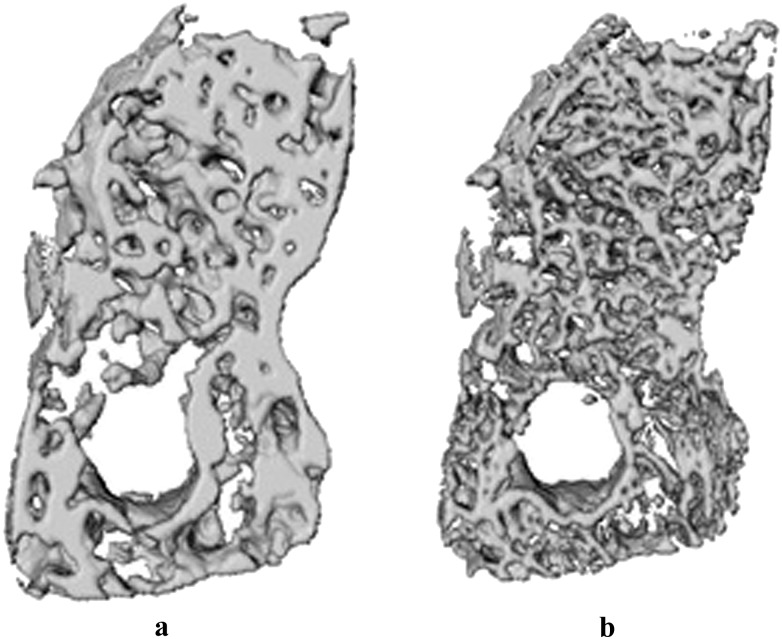
After precise registration, images were segmented to allow trabecular bone structure quantification with CTAn software (CTAnalyser®; SkyScan). Bone voxels were determined by using the adaptive thresholding algorithm, with a radius of four voxels for CBCT images and six voxels for μCT images, and a visually determined background threshold. Voxel radius values were visually determined in CTAnalyser to give the best segmentation overlap with the original image. The adaptive threshold approach, chosen as adaptive or local thresholding algorithms, has been shown to perform better than global thresholding on low-resolution images.7 From the resulting binary images, the trabecular morphometric parameters were computed for each scan within the corresponding VOI (Figure 1). Selected CBCT and µCT VOI were additionally visualized in 3D (MeVisLab®; MeVis Medical Solutions AG, Bremen, Germany) to facilitate the interpretation of the morphometric results (Figure 2).
Figure 2.
Visual three-dimensional representation from cone beam CT (CBCT) (a) and the micro-CT (µCT) (b) bone sample. The difference in morphology shown by the three-dimensional models was reflected by various morphometric parameters.
In the present study, it was opted to follow the recommendations of American Society of Bone and Mineral Metabolism for calculation of 3D bone parameters and structural indices.8 All dimension units were given in millimeters, and parameters were named according to the Parfitt system:1 bone volume (BV), total volume of interest (TV), bone volume fraction (BV/TV), bone surface (BS), bone surface density (BS/TV), trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), trabecular number (Tb.N), total porosity percentage [Po(tot)] and connectivity density (Conn.dn).
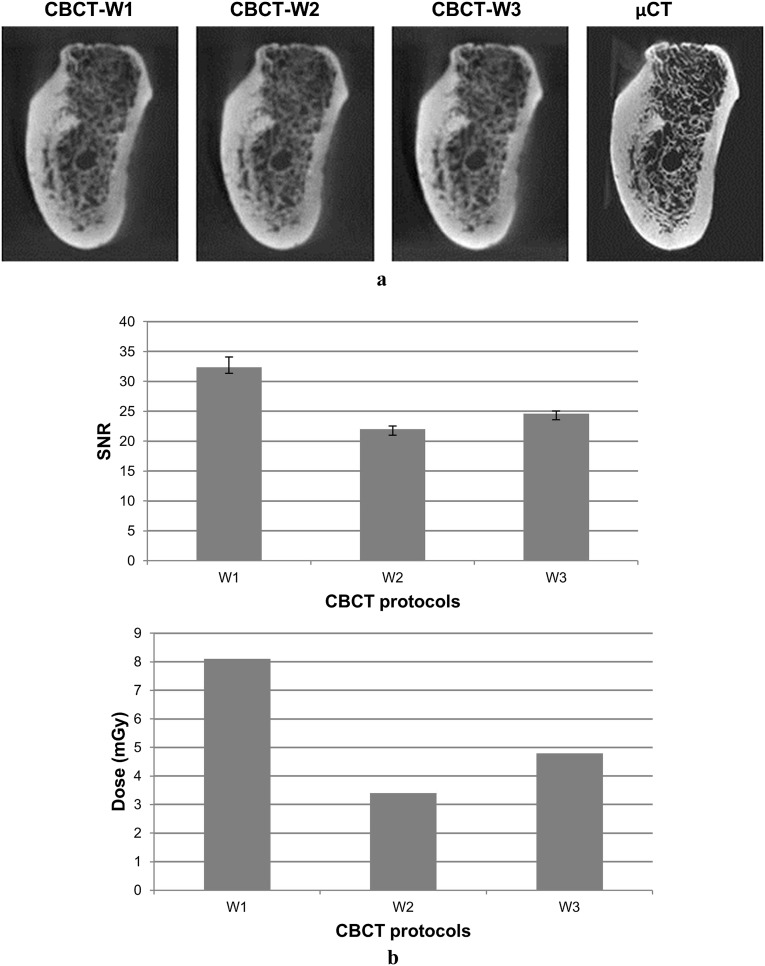
To investigate the effect of X-ray tube current and voltage settings on bone quality assessment, image quality was quantified by the signal-to-noise ratio (SNR), which is defined as the ratio of the mean intensity over the standard deviation of the intensity in a homogenous region. Higher SNR values indicate less noise in the image. The SNR was measured in four VOIs containing only water in the images of jaw samples surrounded by water (Figure 3).
Figure 3.
Visual and quantitative evaluation on image quality for three different cone beam CT (CBCT) acquisition protocols in water. (a) No obvious visual difference in image quality can be observed between different CBCT protocols; (b) higher signal-to-noise ratio values are obtained in higher dose protocols (W1)
Statistical analysis
Statistical analysis was performed in R 2.14.2 (http://www.R-project.org). Descriptive analysis expressed data as central tendency and dispersion measurements. The Fisher exact test was used to compare variance between the groups and the Tukey's honestly significant difference (HSD) test to compare variables. Non-parametric statistical methods were used when normality was not confirmed. Bivariate linear association between morphometric CBCT parameters was sought by calculating Pearson's or Spearman's coefficients. For all tests performed, the confidence level α was 5%.
Results
Descriptive comparison between different µCT and CBCT scanning protocols are shown in Table 1. The Tukey’s HSD test was used to compare mean values among groups. No significant differences between NW(1–3) and W(1–3) protocols were observed for all variables, indicating that all CBCT scan protocols result in similar morphometric indices. The influence of water and scan settings does not effect significant differences.
Table 1.
Descriptive statistics of three-dimensional morphometric parameters for different scanning protocols
| Morphometric parameters | Mean (SD) | Min–max | ||||||||||||
| NW1 | NW2 | NW3 | W1 | W2 | W3 | µCT | NW1 | NW2 | NW3 | W1 | W2 | W3 | µCT | |
| TV | 994.5 (449.9) | 994.5 (449.9) | 1014.0 (464.1) | 1030.0 (460.8) | 1030.0 (460.8) | 1030.0 (460.8) | 989.9 (448.2) | 355.0–1633.0 | 355.0–1633.0 | 371.0–1680.0 | 373.0–1685.0 | 373.0–1685.0 | 373.0–1685.0 | 353.5–1627.0 |
| BV | 520.2 (214.2) | 523.9 (217.1) | 519.5 (210.8) | 530.9 (225.6) | 553.2 (242.2) | 502.8 (202.3) | 323.9 (121.2) | 201.5–844.5 | 198.8–849.7 | 206.7–848.9 | 215.0–894.7 | 222.8–936.5 | 214.1–840.5 | 137.6–489.8 |
| BV/TV | 53.74 (5.69) | 53.98 (5.60) | 52.96 (6.46) | 52.77 (5.10) | 54.57 (3.90) | 50.63 (6.41) | 34.39 (5.41) | 45.36–60.66 | 45.64–60.63 | 44.93–61.14 | 45.24–58.86 | 49.33–59.72 | 43.09–58.80 | 28.90–42.99 |
| BS | 4302 (1877) | 4474 (1964) | 4327 (1869) | 4615 (2108) | 5341 (2498) | 4587 (1986) | 6104 (2044) | 1579–7269 | 1598–7550 | 1603–7310 | 1750–8216 | 1999–9534 | 1826–8019 | 2769–8720 |
| BS/TV | 4.40 (0.46) | 4.56 (0.51) | 4.36 (0.51) | 4.53 (0.40) | 5.19 (0.31) | 4.55 (0.47) | 6.61 (1.32) | 3.67–4.79 | 3.81–5.04 | 3.66–4.86 | 3.88–4.92 | 4.78–5.66 | 3.87–5.11 | 5.06–8.52 |
| Tb.Th | 0.42 (0.01) | 0.41 (0.01) | 0.42 (0.01) | 0.40 (0.01) | 0.38 (0.01) | 0.39 (0.01) | 0.19 (0.01) | 0.40–0.43 | 0.40–0.41 | 0.40–0.42 | 0.39–0.41 | 0.37–0.39 | 0.38–0.40 | 0.17–0.20 |
| Tb.Sp | 0.64 (0.20) | 0.61 (0.20) | 0.66 (0.21) | 0.68 (0.17) | 0.55 (0.12) | 0.72 (0.19) | 0.54 (0.18) | 0.44–0.94 | 0.41–0.92 | 0.44–0.95 | 0.47–0.91 | 0.42–0.69 | 0.47–0.92 | 0.28–0.77 |
| Tb.N | 1.29 (0.13) | 1.32 (0.14) | 1.27 (0.15) | 1.31 (0.13) | 1.44 (0.10) | 1.29 (0.16) | 1.85 (0.32) | 1.09–1.45 | 1.11–1.48 | 1.07–1.46 | 1.11–1.46 | 1.29–1.56 | 1.08–1.50 | 1.51–2.33 |
| Po(tot) | 46.26 (5.69) | 46.02 (5.60) | 47.04 (6.46) | 47.23 (5.10) | 45.43 (3.90) | 49.37 (6.41) | 65.61 (5.41) | 39.34–54.64 | 39.37–54.36 | 38.86–55.07 | 41.14–54.76 | 40.28–50.67 | 41.20–56.91 | 57.01–71.10 |
| Conn.dn | 5.45 (1.99) | 6.34 (2.35) | 5.43 (2.05) | 6.41 (1.53) | 9.89 (1.70) | 6.91 (1.17) | 54.27 (30.51) | 3.65–9.13 | 4.10–10.56 | 3.59–9.14 | 4.96–8.95 | 8.52–13.04 | 4.96–8.25 | 23.73–107.70 |
BS, bone surface in mm2 per mm3; BS/TV, bone surface density in mm2 per mm3; BV, bone volume in mm3; BV/TV, bone volume fraction in %; Conn.dn, connectivity density in %; max, maximum; µCT, micro-CT scanning protocol; min, minimum; NW1–3, CBCT scanning Protocols 1–3 without water; Po(tot), total porosity percentage in %; SD, standard deviation; Tb.N, trabecular number in 1 per mm; Tb.Sp, trabecular separation in mm; Tb.Th, trabecular thickness in mm; TV, total volume of interest in mm3; W1–3, CBCT scanning Protocols 1–3 in water.
Statistically significant differences (α < 0.05) were observed for the variable BV/TV, BS/TV, Po(tot), Tb.Th, Tb.N and Conn.dn between CBCT and µCT protocols. No significant differences were observed between all scanning protocols for the variable Tb.Sp, demonstrating that morphometric indices obtained by CBCT imaging differ from those obtained with µCT. These structural differences can also be observed on the 3D reconstructions of the VOI from both CBCT and µCT images (Figure 2).
Table 2 shows the direct relationship between all scanning protocols. High positive Pearson’s correlation coefficients were observed between CBCT and µCT protocols for all variables, except for Tb.Th.
Table 2.
Correlation of morphometric parameters between μCT and CBCT protocols
| Morphometric parameters | μCT—NW1 | μCT—NW2 | μCT—NW3 | μCT—W1 | μCT—W2 | μCT—W3 |
| BV/TVa | 0.80 | 0.76 | 0.78 | 0.86 | 0.82 | 0.89 |
| BS/TVa | 0.75 | 0.69 | 0.74 | 0.73 | 0.68 | 0.73 |
| Tb.Th | 0.29 | 0.21 | 0.43 | 0.43 | 0.57 | 0.32 |
| Tb.Spa | 0.82 | 0.80 | 0.84 | 0.62 | 0.61 | 0.63 |
| Tb.Na | 0.83 | 0.77 | 0.81 | 0.82 | 0.72 | 0.86 |
| Po(tot)a | 0.80 | 0.76 | 0.78 | 0.86 | 0.82 | 0.89 |
| Conn.dna | 0.73 | 0.70 | 0.73 | 0.74 | 0.70 | 0.73 |
Pearson and Spearman correlation coefficients have a p < 0.05.
BS/TV, bone surface density in mm2 per mm3; BV/TV, bone volume fraction in %; Conn.dn, connectivity density in %; µCT, micro-CT scanning protocol; NW1–3, CBCT scanning Protocols 1–3 without water; Po(tot), total porosity percentage in %; Tb.N, trabecular number in 1 per mm; Tb.Sp, trabecular separation in mm; Tb.Th, trabecular thickness in mm; W1–3: CBCT scanning Protocols 1–3 in water.
CBCT acquisition Protocol 2 has a lower correlation with µCT than Protocols 1 and 3, which is in accordance with low-obtained SNR values (Figure 3b). However, no visual difference in image quality can be observed between all scanning protocols (Figure 3a).
Discussion
Although the accuracy of µCT morphology measurements for the non-destructive assessment of trabecular bone quality has already been extensively studied in both animal7,9,10 and human3,11 specimens, these techniques are not used regularly in clinical routines. This study addresses the use of CBCT in evaluating the trabecular bone architecture by means of morphometric parameters, and the effect of X-ray tube current and voltage settings, as well as soft tissue surrounding the bone, on bone quality assessment was investigated. This has paved the way for clinical evaluation of bone healing (whether or not this is peri-implant) and for follow-up assessment studies.
In the present study, water was used to mimic the soft tissue surrounding the bone.12 Table 1 shows that CBCT yields similar results at the 3D level for all W- and NW-protocols. Therefore, it can be demonstrated that the soft tissue around the bone has limited influence on the CBCT and µCT comparison, making it applicable for clinical settings.
After a more detailed comparison of both analysis techniques, it seemed that radiographic CBCT analysis tended to slightly overestimate BV/TV, Tb.Sp and Tb.Th compared with µCT (Table 1 and Figure 2). The possible explanation for the overestimation of CBCT is its lower resolution. Waarsing et al7 showed using µCT that decreasing the image resolution resulted in overestimation of these parameters. Trabeculae that were thin with respect to the image resolution were smeared out and appeared thicker. The high spatial resolution of μCT has always been considered crucial for accurate assessment of individual trabecular characteristics using structural analysis. In particular, measurements such as Tb.Th are known to have a strong resolution dependency requiring very high resolutions for precise measurement,7 explaining the lower correlation between CBCT and µCT for this particular parameter (Table 2). The actual spatial resolution of a 3D image is determined by various factors, including the voxel size and the amount of noise. Early generation CBCT machines were characterized by small voxel sizes but a high degree of noise, limiting their spatial and contrast resolution. The present study used one of the best quality CBCT machines currently in the market (3D Accuitomo). When these overestimations persist, a correction factor can be calculated to reduce these inaccuracies. Despite the low number of bone samples, the present study demonstrates the potential of high-resolution CBCT imaging for in vivo applications of quantitative bone morphometry and bone quality assessment.
However, the present study was conducted in vitro and thus in the absence of motion artefacts. Patient movement may reduce CBCT spatial resolution substantially.13 Head-motion artefacts can be simulated by application of phantoms.14 However, in the present study, human jaw bones were used instead, as the primary focus was to simulate jaw bone structure as close to clinical reality as possible. Thus, it was opted to make the CBCT scans by applying clinical scanning protocols, soft-tissue simulation and evaluation of the human trabecular bone structures. Though, we realize that this choice hampered us to further include motion artefacts. In the future, an integrated phantom with a human trabecular bone might offer the possibility to include this variable as well.
There is a common belief that greyscale values in the reconstructed CBCT database do not allow for bone density assessment.15,16 However, bone density is only one factor representing bone quality. Bone structure also largely contributes to bone quality. It is noteworthy to stress that CBCT images may enable bone structural analysis by thresholding CBCT images to a binary format, provided that high enough image resolution is attained.17 With further advancements in CBCT resolution, morphological analysis of bone structure is most promising for clinical implementation. A morphometric analysis of mandibular trabecular bone reported that BV/TV obtained using CBCT images was highly correlated with CT values obtained using multislice CT images, suggesting that trabecular bone morphometry can be used to evaluate the density of mandibular cancellous bone.18 The resolution of currently clinically used CBCT equipment also allows for detailed observation of the peri-implant bone,19 which is consistent with research evaluated in the zygomatic bone.20 Furthermore, morphometric indices of the trabecular bone structure have been proven to have evident correlation with the physical property of the bone in many histology and µCT studies.2,11,21 In the present study, the correlation among morphometric parameters has been proven between CBCT as well. The occurrence of positive significant correlations indicates that there are definitely strong relations among CBCT parameters for all the scanning protocols (Table 2).
Table 2 shows that CBCT acquisition Protocol 2 has a lower correlation with µCT than Protocols 1 and 3 for all parameters. This indicates that image quality is hardly affected by altering the peak voltage between 90 kVp (Protocol 1) and 70 kVp (Protocol 3) compared with the effect of lowering the tube current from 5 mA (Protocols 1 and 3) to 2 mA (Protocol 2). The drop in correlation because of the lower tube current can be explained by an inverse relationship between tube current and image noise.22 Consequently, lower tube currents yield noisier images (Figure 3b), hampering accurate segmentation.
In conclusion, the present study demonstrates the potential of high-resolution CBCT imaging for in vivo applications of quantitative bone morphometry and bone quality assessment. This is especially true for repetitive follow-up measurements, which cannot be performed using histological sections. Additionally, the method offers reliable easy access to the 3D structure of trabecular bone, which is mandatory for the analyses of the anisotropic mechanical behaviour of the cancellous bone. Therefore, the development of this technique seems promising for clinical use of low-dose CBCT and offers an ideal accurate alternative to multislice CT to determine the 3D trabecular bone structure during the pre-operative phase and also, if necessary, during the bone healing phase. However, one must still take into account the overestimation of CBCT while measuring BV/TV, Tb.Sp and Tb.Th and the used acquisition settings.
Acknowledgments
The authors thank Ms Laura Nicolielo (University of São Paulo, Brazil) for her help during the scanning process and Professor Dr Marco Duarte (University of São Paulo, Brazil) for kindly enabling the scanning on the SkyScan 1174 μCT system.
References
- 1.Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, et al. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphomerty Nomenclature Committee. J Bone Miner Res 1997; 2: 595–610 10.1002/jbmr.5650020617 [DOI] [PubMed] [Google Scholar]
- 2.Parfitt AM, Mathews CH, Villanueva AR, Kleerekoper M, Frame B, Rao DS. Relationships between surface, volume, and thickness of iliac trabecular bone in aging and in osteoporosis. Implications for the microanatomic and cellular mechanisms of bone loss. J Clin Invest 1983; 72: 1396–1409 10.1172/JCI111096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Müller R, Van Campenhout H, Van Damme B, Van Der Perre G, Dequeker J, Hildebrand T, et al. Morphometric analysis of human bone biopsies: a quantitative structural comparison of histological sections and micro-computed tomography. Bone 1998; 23: 59–66 [DOI] [PubMed] [Google Scholar]
- 4.Kothari M, Keaveny TM, Lin JC, Newitt DC, Genant HK, Majumdar S. Impact of spatial resolution on the prediction of trabecular architecture parameters. Bone 1998; 22: 437–443 [DOI] [PubMed] [Google Scholar]
- 5.Issever AS, Link TM, Kentenich M, Rogalla P, Burghardt AJ, Kazakia GJ, et al. Assessment of trabecular bone structure using MDCT: comparison of 64- and 320- slice CT using HR-pQCT as the reference standard. Eur Radiol 2010; 20: 458–468 10.1007/s00330-009-1571-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Maes F, Collignon A, Vandermeulen D, Marchal G, Suetens P. Multimodality image registration by maximization of mutual information. IEEE Trans Med Imaging 1997; 16: 187–198 10.1109/42.563664 [DOI] [PubMed] [Google Scholar]
- 7.Waarsing JH, Day JS, Weinans H. An improved segmentation method for in vivo microCT imaging. J Bone Miner Res 2004; 19: 1640–1650 10.1359/JBMR.040705 [DOI] [PubMed] [Google Scholar]
- 8.Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Müller R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res 2010; 25: 1468–1486 10.1002/jbmr.141 [DOI] [PubMed] [Google Scholar]
- 9.Kapadia RD, Stroup GB, Badger AM, Koller B, Levin JM, Coatney RW, et al. Applications of micro-CT and MR microscopy to study pre-clinical models of osteoporosis and osteoarthritis. Technol Health Care 1998; 6: 361–372 [PubMed] [Google Scholar]
- 10.Bonnet N, Laroch N, Vico L, Dolleans E, Courteix D, Benhamou CL. Assessment of trabecular bone microarchitecture by two different X-ray microcomputed tomographs: a comparative study of the rat distal tiba using Skyscan and Scanco devices. Med Phys 2009; 36: 1286–1297 [DOI] [PubMed] [Google Scholar]
- 11.Chappard D, Retailleau-Gaborit N, Legrand E, Baslé MF, Audran M. Comparison insight bone measurements by histomorphometry and microCT. J Bone Miner Res 2005; 20: 1177–1184 10.1359/JBMR.050205 [DOI] [PubMed] [Google Scholar]
- 12.Sandborg M, Alm Carlsson G, Persliden J, Dance DR. Comparison of different materials for test phantoms in diagnostic radiology. Radiat Prot Dosimetry 1993; 49: 345–347 [Google Scholar]
- 13.Schulze R, Heil U, Gross D, Bruellmann DD, Dranischnikow E, Schwanecke U, et al. Artefacts in CBCT: a review. Dentomaxillofac Radiol 2011; 40: 265–273 10.1259/dmfr/30642039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ens S, Bruder R, Ulrici J, Hell E, Buzug TM. A validation framework for head-motion artifacts in dental cone-beam CT. In: World Congress on Medical Physics and Biomedical Engineering, 7–12 September 2009; Munich, Germany. Munich, Germany: IFMBE Proceedings Volume 25/2; 2009. [Google Scholar]
- 15.Katsumata A, Hirukawa A, Noujeim M, Okumura S, Naitoh M, Fujishita M, et al. Image artifact in dental cone-beam CT. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2006; 101: 652–657 10.1016/j.tripleo.2005.07.027 [DOI] [PubMed] [Google Scholar]
- 16.Pauwels R, Stamatakis H, Manousaridis G, Walker A, Michielsen K, Bosmans H, et al. Development and applicability of a quality control phantom for dental cone-beam CT. J Appl Clin Med Phys 2011; 12: 3478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kothari M, Keaveny TM, Lin JC, Newitt DC, Genant HK, Majumdar S. Impact of spatial resolution on the prediction of trabecular architecture parameters. Bone 1998; 22: 437–443 [DOI] [PubMed] [Google Scholar]
- 18.Naitoh M, Aimiya H, Hirukawa A, Ariji E. Morphometric analysis of mandibular trabecular bone using cone beam computed tomography: an in vitro study. Int J Oral Maxillofac Implants 2010; 25: 1093–1098 [PubMed] [Google Scholar]
- 19.Huang Y, Van Dessel J, Liang X, Depypere M, Zhong W, Ma G, et al. Effects of immediate and delayed loading on peri-implant trabecular structures: a cone beam CT evaluation. Clin Implant Dent Relat Res Apr 2013. Epub ahead of print. 10.1111/cid.12063 [DOI] [PubMed] [Google Scholar]
- 20.Liu SM, Zhang ZY, Li JP, Liu DG, Ma XC. A study of trabecular bone structure in the mandibular condyle of healthy young people by cone beam computed tomography. Zhonghua Kou Qiang Yi Xue Za Zhi 2007; 42: 357–360 [PubMed] [Google Scholar]
- 21.de Oliveira RC, Leles CR, Lindh C, Ribeiro-Rotta RF. Bone tissue microarchitectural characteristics at dental implant sites. Part 1: identification of clinical-related parameters. Clin Oral Implants Res 2012; 23: 981–986 10.1111/j.1600-0501.2011.02243.x [DOI] [PubMed] [Google Scholar]
- 22.Goldman LW. Principles of CT: radiation dose and image quality. J Nucl Med Technol 2007; 35: 213–225 10.2967/jnmt.106.037846 [DOI] [PubMed] [Google Scholar]