Abstract
Background
Irritable bowel syndrome (IBS) is the most common gastrointestinal disorder, affecting more than 10% of the general population worldwide. Although a genetic component is suspected, unambiguous susceptibility genes have so far not been identified. This study tested the hypothesis that genes contributing to epithelial barrier integrity, control of mucosal immune responses and interactions with bacteria in the gut are associated with IBS.
Methods
Single nucleotide polymorphisms (SNPs) corresponding to top signals of association with Crohn's disease at 30 known susceptibility loci were tested for their effect on IBS risk in 1992 individuals from two independent case–control cohorts from Sweden and the USA. Association tests included a conservative Bonferroni correction for multiple comparisons, and were also performed on specific subgroups of patients characterised by constipation (IBS-C), diarrhoea (IBS-D) or alternating constipation and diarrhoea (IBS-A).
Results
The Crohn's disease risk allele rs4263839 G in the TNFSF15 gene was significantly associated with an increased risk of both IBS (p=2.2×10–5; OR 1.37) and more pronouncedly, IBS-C (p=8.7×10–7; OR 1.79) in the entire sample. Similar associations and risk effects of the same magnitude were observed in the two cohorts analysed separately. A correlation between rs4263839 genotype and TNFSF15 mRNA expression was detected both in peripheral blood and in rectal mucosal biopsies from healthy individuals (combined p=0.0033).
Conclusions
TNFSF15 is a susceptibility gene for IBS and IBS constipation. As TL1A, the protein encoded by TNFSF15, contributes to the modulation of inflammatory responses, the results support a role of immune activation in IBS.
Irritable bowel syndrome (IBS) is a gastrointestinal disorder of unknown aetiology, affecting at least 10% of people worldwide. It presents predominantly in women, with a variety of symptoms including abdominal pain and bloating, constipation (IBS-C), diarrhoea (IBS-D) or alternating constipation and diarrhoea (IBS-A).1 Diet, stress, infections, defective mucosal immunity, intestinal dysmotility and altered gut–brain communication have all been proposed as contributing factors.2 Evidence is emerging that impaired barrier function, dysregulated interactions with intraluminal bacteria and alterations in local host immune responses may also play a role.3,4
Although a heritable component of IBS is predicted from epidemiological studies in families and twins, genetic investigations have so far failed to provide compelling evidence for specific markers of susceptibility.5 Earlier studies involved small sample sizes or failed to replicate findings in independent populations. Single nucleotide polymorphisms (SNP) in the TLR9, IL6 and CDH1 genes have recently been proposed as genetic risk factors for a strictly defined post-infectious IBS (PI-IBS) after waterborne epidemic diarrhoea in the Walkerton community of Ontario, suggesting that immune-mediated mechanisms may predispose to the development of PI-IBS.6 Promising results have also been obtained with alternative approaches through the analysis of intermediate phenotypes of disease, and we have shown that colonic transit in IBS is influenced by polymorphism in the neuropeptide S receptor gene (NPSR1), which is expressed by gastrointestinal enteroendocrine cells, and is involved in inflammation, anxiety and nociception.7
Our hypothesis is that susceptibility genes involved in the maintenance of the epithelial barrier, innate immunity and interactions with bacteria in the gut are associated with IBS. Specific variants in these genes confer an increased risk of inflammatory bowel diseases (IBD) such as Crohn's disease (CD) and ulcerative colitis.8 Therefore, to test our hypothesis initially, we selected 30 unequivocal CD risk loci recently identified in a meta-analysis of genome-wide association studies (GWAS),9 and assessed their potential association with IBS in 1992 individuals from two independent case–control cohorts from Sweden and the USA.
MATERIALS AND METHODS
Study populations
Two independent cohorts of IBS patients and controls from Sweden and the USA were included in this study. Their demographic and clinical characteristics are reported in table 1. Comorbidity with IBD, coeliac disease and microscopic colitis was excluded in all patients based on clinical evaluation. Swedish IBS patients were recruited at secondary and tertiary care centres and clinics in Stockholm, Gothenburg, Malmoe and Kalix–Haparanda, and have been, for the vast majority, described in detail in previous publications.10–13 Diagnosis of IBS was based on symptom phenotypes, consensus criteria and validated questionnaires of gastrointestinal and somatic symptoms. Swedish controls were healthy blood donors free of inflammatory disease, also described in previous studies.14,15 Informed consent was obtained from all patients and controls and local ethics committees approved the study protocol and genetic investigations. IBS patients and controls from the USA have been extensively described in previous publications,16 and consisted of 434 patients with Rome II-positive functional gastrointestinal disorders and 231 healthy volunteers recruited to previous studies of symptom phenotype and genotype from 2000 to 2007. All resided within 150 miles of Rochester, Minnesota, USA. The study was approved by the Mayo Clinic Institutional Review Board; all participants had provided written permission for research studies based on their medical records and DNA samples. A validated bowel symptom questionnaire, electronic medical record and information from direct physician interview and examination (MC) were used to char-acterise the subtype of functional gastrointestinal disorder.
Table 1.
Demographic and clinical characteristics of IBS patients and controls
| Sweden |
USA |
Total |
||||
|---|---|---|---|---|---|---|
| Control | IBS | Control | IBS | Control | IBS | |
| N | 900 | 427 | 231 | 434 | 1131 | 861 |
| Mean age, years | 45.6 | 42.2 | 36.7 | 44.5 | 43.8 | 43.4 |
| Men/women | 561/339 | 93/334 | 68/163 | 50/384 | 629/502 | 143/718 |
| Alternating (IBS-A) | 179 | 84 | 263 | |||
| Constipation (IBS-C) | 104 | 157 | 261 | |||
| Diarrhoea (IBS-D) | 144 | 174 | 318 | |||
| Chronic abdominal pain | 19 | 19 | ||||
IBS, irritable bowel syndrome.
Genotyping
SNPs to be genotyped were selected based on results previously published by Barrett et al,9 who identified 30 unequivocal CD risk loci through meta-analysis of previous GWAS. Based on this study, the SNP showing the strongest association signal at each CD risk locus was selected and genotyped in all individuals. Genotyping of the Swedish cohort and blood donors included in the analysis of TNFSF15 messenger RNA (mRNA) expression was performed at Karolinska Institutet's mutational analysis core facility using matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry and iPLEX chemistry (SEQUENOM Inc., San Diego, California, USA). TaqMan SNP genotyping assays and the 7300 real-time PCR system (Applied Biosystems, Foster City, California, USA) were used to genotype individuals recruited at the Mayo Clinic. The NOD2 SNP 2066844 was used on Mayo Clinic's material instead of SNP rs2066847 because no TaqMan SNP genotyping assay was available for the latter. SNPs were genotyped with average success rates of 99.8% and 100%, respectively, for Sweden and the USA, and none of the 1992 individuals included in the study had more than 5% missing genotypes. χ2 tests were used to verify that marker frequencies were within allelic Hardy–Weinberg equilibrium and statistically consistent in the two control populations. Measures of linkage disequilibrium (LD) between markers rs4263839 and rs6478109 in the two studied populations were obtained based on data available from Illumina custom bead chip arrays (The International IBD Genetics Consortium, unpublished) for 358 controls from the Swedish cohort, and from additional rs6478109 TaqMan genotyping of 231 US controls from the Mayo Clinic.
Analysis of TNFSF15 mRNA expression
Peripheral blood was obtained from 25 healthy donors (average age 44.2 years, 40% women), recruited at Örebro University Hospital, Örebro, Sweden, after written informed consent. Rectal mucosal biopsies were collected at flexible sigmoidoscopy from 15 healthy volunteers (average age 41 years, 93% women), recruited by public advertisement at the Mayo Clinic, Rochester, Minnesota, USA. Ethical approval was obtained from local responsible committees and the Mayo Clinic Institutional Review Board. Total RNA was extracted with commercially available kits (Qiagen, Hilden, Germany) and complementary DNA synthesised from 0.5 to 1 μg of RNA with SuperScript III reverse transcriptase (Invitrogen, Carlsbad, California, USA) according to the manufacturer's instructions. TNFSF15 mRNA expression levels were measured by performing quantitative real-time PCR in ABI Prism 7500 or 7900HT sequence detection systems (Applied Biosystems) with TaqMan gene expression assays on demand (Applied Biosystems), according to the manufacturer's instructions. Real-time PCR reactions were performed in triplicate on each sample with TaqMan assays Hs00270802_s1 for TNFSF15, and Hs99999905_m1 and Hs00193002_m1, respectively, for GAPDH (in peripheral blood) and SART1 (in rectal biopsies). After normalisation to internal endogenous controls (GAPDH for blood and SART1 for gut tissue), TNFSF15 mRNA expression levels in each sample were determined by the comparative CT method of relative quantification, and expressed in arbitrary units relative to a randomly chosen reference sample.
Functional analysis of the effect of the rs4263839 SNP on gene transcription
Production of luciferase reporter plasmids
To study the functional relevance of genetic variation at the rs4263839 SNP, DNA sequences corresponding to the two alleles A and G were inserted into reporter vectors suitable for transfection experiments and luciferase assays. For this purpose, sense and antisense oligonucleotides corresponding to 83 base pairs of DNA sequence surrounding TNFSF15 rs4263839 SNP alleles (GAGCTGCAATCATCCCCACACATCTGCTCTCCAGGAAGATGA[G/A]TTTAATGATAATGGAATGGATGGTGTCAGCCTTATTCATT) were synthesised (Eurofins MWG Operon, Ebersberg, Germany), annealed overnight after denaturation at 95°C for 5 min to produce duplexes, and cloned between KpnI and SacI restriction sites in the pGL4.23 (luc2/minP) luciferase reporter vector (Promega, Madison, Wisconsin, USA). The inserts of all clones were sequence verified, and one control vector (pGL4_minP, containing no insert) and two recombinant vectors (pGL4_minP-G and pGL4_minP-A, respectively, for the G and A alleles) were selected for further experiments.
Transient transfections and luciferase reporter assays
HEK293 epithelial cells (ATCC CRL-1573) were cultured in minimal essential medium supplemented with 10% fetal calf serum and 1% penicillin/streptomycin. Transfections were carried out with FUGENE 6 (Roche Diagnostics, Basel, Switzerland) with 1 μg of total DNA according to the manufacturer's instructions. HEK293 cells were co-transfected with 900 ng of either empty vector (pGL4_minP) or TNFSF15 rs4263839 SNP allelic constructs (pGL4_minP-G or pGL4_minP-A) in combination with 100 ng of pRL-TK Renilla luciferase reporter vector (Promega). Total cell lysates were prepared 18 h post-transfection, and luciferase activity was calculated using the dual-luciferase reporter assay system (Promega) on a Micro-plate Reader Infinite 200 (Tecan, Männedorf, Switzerland) according to the manufacturer's instructions. Experiments were performed in triplicate on two different plasmid preparations from each clone, and firefly luciferase activity was expressed in arbitrary units relative to the control empty vector (pGL4-minP) after normalisation for transfection efficiency based on values obtained for Renilla luciferase (pRL-TK vector).
Functional analysis of the effect of the rs4263839 SNP on DNA–protein interactions
Electrophoretic mobility shift assays (EMSA) were used to assess whether rs4263839 alleles A and G differentially affect nuclear proteins (transcription factors) binding to DNA at the SNP site. For this purpose, sense and antisense oligonucleotides corresponding to rs4263839 A and rs4263839 G allele sequences (TCCAGGAAGATGA[A/G]TTTAATGATAATG) were synthesised (Eurofins MWG Operon), annealed overnight after denaturation at 95°C for 5 min to produce duplexes, and 5′-end [32P]-labelled with T4 polynucleotide kinase (New England Biolabs, Ipswich, Massachusetts, USA). Nuclear extracts from HEK293 cells were prepared with the nuclear extract kit (Active Motif, Carlsbad, California, USA) following the manufacturer's protocol. DNAeprotein binding reactions were performed in 25 mM HEPES pH 7.9, 150 mM KCl, 10% glycerol and 5 mM DTT, with 3 μg of nuclear extract, approximately 0.5 ng of labelled probe and 1 μg of poly dI-dC in a total volume of 12 μl. Excess (50×) unlabelled probe or 1 μg of anti-Oct-1 or unspecific (anti-Sox5) antibody (Santa Cruz Biotechnology) were added to the binding reactions, respectively, for competition and super shift experiments, 10 min before the addition of the radiolabeled probe. Novex 6% DNA Retardation Gels (Invitrogen) were run in 0.5× TBE according to the manufacturer's protocols, and EMSA digital images produced with Quantity One 1-D software on a molecular imager FX (Bio-Rad, Hercules, California, USA).
Bioinformatic analyses
The potential effect of the rs4263839 SNP on transcription factor binding to DNA was evaluated by screening the TRANSFAC repository of transcription factor binding sites with MatInspector 4.3 software (http://www.genomatix.de). The Encyclopedia of DNA Elements (ENCODE) database linked to the Human Genome Browser at the University of California, Santa Cruz (http://genome.ucsc.edu/ENCODE) was interrogated to retrieve experimental data and identify DNA sequences associated with enhancers, promoters and regions of active transcription at the TNFSF15 locus. The Haploview 4.2 program (http://www.broadinstitute.org/haploview) was used with HapMap genotype data (http://hapmap.ncbi.nlm.nih.gov) from the CEU population (Utah residents with northern and western European ancestry from the CEPH collection) to characterise LD structure at the TNFSF15 locus in the Caucasian population.
Statistical analyses
Associations to disease were tested for each marker with a logistic regression, using gender and country of origin as covariates. A generic log-additive model was used to test allelic effects, and p values were estimated by a χ2 test over the deviance of the regression. Implementation of this method can be found in the package SNPassoc_1.6-0 in the statistical software suite R (http://www.r-project.org). A conservative Bonferroni correction was applied to take into account multiple tests. Under the most stringent calculation (30 loci × four phenotypes (IBS, IBS-C, IBS-D and IBS-A) × three groups (Sweden, USA and combined)) significance was thus set to p<1.4×10–4 (α=0.05/360) in our study. We estimated that our total sample had greater than 80% power to detect OR of 1.2 or more for 83.3% (25/30) SNP with minor allele frequency greater than 0.10.
The correlation between the rs4263839 genotype and TNFSF15 mRNA expression was determined by Spearman rank test applied separately to peripheral blood and rectal mucosal biopsies, while the Fisher's method was used to combine results for the meta-analysis of genotype expression data. A Mann–Whitney U test was used for the statistical analysis of luciferase reporter assays performed with rs4263839 variant clones.
RESULTS
Test of association of CD risk loci in IBS
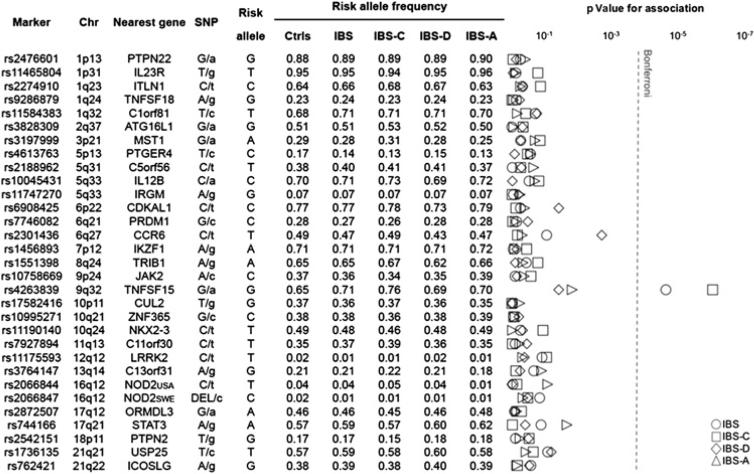
CD risk alleles at each locus were tested for association with IBS and its clinical subtypes IBS-C, IBS-D and IBS-A. The results of the analysis performed on the entire sample are reported in figure 1. For IBS, we detected significant association (withstanding Bonferroni correction) for the SNP rs4263839 at the TNFSF15 locus (p=2.2×10–5; OR 1.37; 95% CI 1.19 to 1.58). Nominal associations and risk effects of the same magnitude were also observed in the Swedish and US cohorts separately analysed (see supplementary table 1, available online only). In the analysis of IBS subtypes, we observed the most pronounced effect of the TNFSF15 locus in IBS constipation patients, in whom the rs4263839 risk allele (G) gave rise to the strongest association signal and nearly twofold increased risk of disease (p=8.7×10–7; OR 1.79; 95% CI 1.41 to 2.26). Also in this case, similar effects were observed after stratification for population (see supplementary table 2, available online only). Some other signals of nominal significance for IBS and its clinical subgroups were detected in the combined sample or in individual cohorts, but none of these withstood Bonferroni correction for multiple comparisons (figure 1 and supplementary tables 1–4, available online only).
Figure 1.
Association of Crohn's disease risk loci with irritable bowel syndrome (IBS) and its subtypes in a combined analysis of two cohorts of cases and controls from Sweden and the USA. Shown from left to right are: NCBI dbSNP database accession numbers (Marker), chromosomal positions (Chr), nearest gene at each locus (Nearest gene), type of polymorphism (single-nucleotide polymorphism (SNP); with minor allele in lowercase), alleles associated with Crohn's disease risk (Risk allele), and frequencies of risk alleles (Risk allele frequency) in controls (Ctrls), IBS, constipation-predominant IBS (IBS-C), diarrhoea-predominant IBS (IBS-D) and alternating IBS (IBS-A). The results of the statistical analysis are reported in the right most panel (p values for association), with p values on a logarithmic scale and the Bonferroni-corrected α level represented by a dashed line (Bonferroni).
Analysis of the effect of rs4263839 SNP variation on TNFSF15 expression
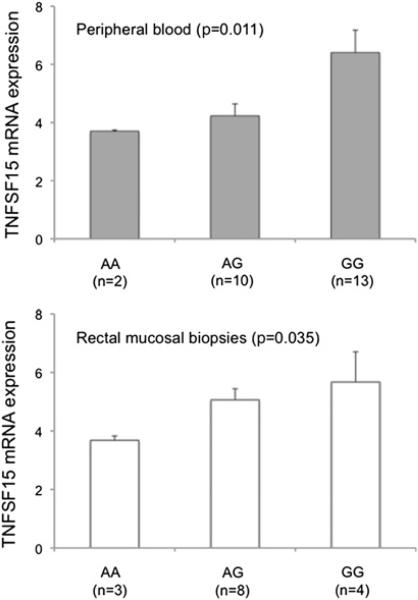
Analysis of correlation between rs4263839 genotype and gene expression (figure 2) revealed the risk allele G to be associated with higher TNFSF15 mRNA levels both in peripheral blood leucocytes and in rectal mucosal biopsies from healthy individuals (combined p=0.0033).
Figure 2.
Association between rs4263839 genotype and TNFSF15 mRNA expression in peripheral blood leucocytes and rectal mucosal biopsies from healthy individuals. TNFSF15 expression was quantified with TaqMan gene expression assays, and is reported in fold changes (average plus SEM) relative to a reference sample arbitrarily chosen in each set. Spearman correlation rank test p value is shown.
Functional characterisation of the rs4263839 SNP
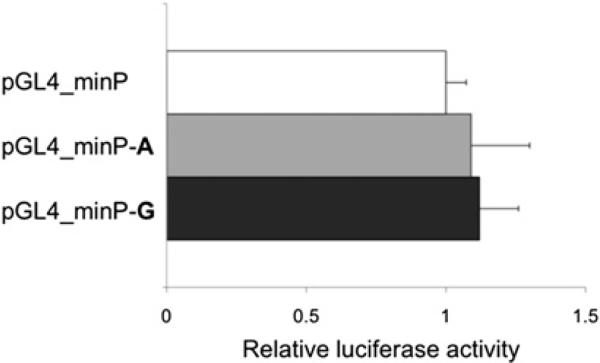
Experimental data retrieved from the ENCODE project (http://www.genome.gov/encode) indicate that the rs4263839 SNP maps within a DNA region associated with transcriptional regulation in several cell lines (see supplementary figure 1, available online only), and additional bioinformatic analyses predict the G allele to disrupt a consensus binding site for the ubiquitous transcription factor Oct-1. We cloned 83 bp of rs4263839 DNA surrounding sequence into a minimal promoter reporter vector, and compared individual alleles (A and G) for their potential direct involvement in the modulation of transcriptional activity in luciferase assays. As shown in figure 3, the presence of the rs4263839 extra sequence had no effect on luciferase transcription; in addition, no significant difference between the two variants could be detected in this experimental system. In EMSA experiments, we confirmed Oct-1 binding to the rs4263839 DNA sequence; however, this binding was not quantitatively different between the two alleles at the polymorphic site (see supplementary figure 2, available online only).
Figure 3.
Comparison of rs4263839 alleles for their effect on transcriptional activity in luciferase assays. HEK293 cells were transiently transfected with a minimal promoter vector (pGL4_minP) or identical plasmids containing allelic variants of the rs4263839 single-nucleotide polymorphism surrounding sequence (pGL4_minP–A and pGL4_minP–G), in two independent experiments performed in triplicate. Luciferase activity was measured 18 h after transfection, and is expressed in arbitrary units as fold changes (+SD) relative to pGL4_minP.
LD between rs4263839 and rs6478109 SNP
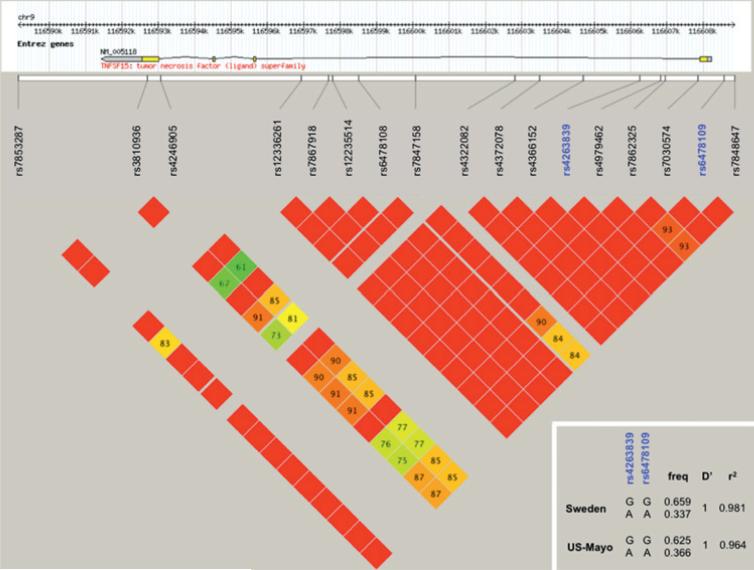
One TNFSF15 promoter polymorphism was recently identified (rs6478109), which associates with CD as part of a risk haplo-type and has functional effects on gene expression in vitro.17 Inspection of additional genotype data from our study cohorts (generated for 358 Swedish and 231 US–Mayo controls, respectively) revealed that the rs4263839 and rs6478109 SNPs are in complete LD and represent good proxies for each other. In particular, alleles associated with CD/IBS risk and higher mRNA expression at each SNP (rs4263839 G and rs6478109 G) were consistently found in cis on 1178 tested chromosomes (<0.6% non-cis combinations) (figure 4).
Figure 4.
Linkage disequilibrium (LD) structure of the TNFSF15 locus. Large panel: GOLD heatmap (chr9: 116 589 138–116 608 877) generated with Haploview (http://www.broadinstitute.org/haploview) on HapMap genotype data from the CEU (CEPH) population of European descent. The rs4263839 SNP, and the functional promoter single-nucleotide polymorphism (SNP) rs6478109 are highlighted in bold blue type. Insert: Specific LD values (D’, r2) and haplotype frequencies (freq) of the rs4263839 and rs6478109 SNP in the two studied populations.
DISCUSSION
Our results implicate the CD risk locus rs4263839 (TNFSF15) in IBS predisposition, and suggest that testing genetic variants associated with other intestinal conditions may be a good strategy to obtain novel clues on IBS pathogenesis, in the absence of GWAS data. As a result of large international consortia performing comprehensive GWAS meta-analyses, the number of confirmed risk loci for IBD (both CD and ulcerative colitis) and other gut disorders such as coeliac disease is increasing exponentially, and similar approaches may therefore lead to the identification of other genes that may also predispose to IBS. Detection of significant findings against a very conservative calculation of Bonferroni-corrected alpha and the observation of nominal associations and similar risk effects in two independent caseecontrol cohorts supports the assumption that TNFSF15 is a true IBS susceptibility locus. Although modest, the effect of TNFSF15 SNP rs4263839 on IBS risk (OR 1.37 and 1.79 for IBS and IBS constipation, respectively) is comparable to that observed in previous associations with CD, including the original meta-analysis by Barrett et al9 (OR 1.22). In addition, loci nominally associated with IBS in this study may be tested in future investigations on larger numbers of patients and controls.
The TNFSF15 gene is strongly associated with CD, and has recently been identified as a susceptibility locus also for anky-losing spondylitis and leprosy.18,19TNFSF15 codes for TL1A, a member of the TNF (ligand) superfamily expressed primarily in macrophages and Tcells, and in immune cells exposed to proinflammatory stimuli or microbes.20,21 Binding of TL1A to death receptor 3 (DR3) on activated CD4 T cells provides a co-stimulatory signal that amplifies the inflammatory response by inducing proliferation and differentiation of T-helper 17 cells, with production of interferon-γ and interleukin-17.22,23 These responses are particularly important in the defence against pathogens, and in homeostatic interactions with commensal bacteria in the gut. Therefore, it has been postulated that genetic alterations of TL1A signalling or expression may affect the regulation of such responses to microbes, and thereby modify the risk of CD.
Alterations in gut flora or immune activation in the gastrointestinal tract may lead to altered enteric nervous system signalling, and thus cause intestinal motor and sensory dysfunctions.3,4 Increased numbers of mast cells and activated CD4 T cells in the intestinal mucosa, as well as higher secretion of pro-inflammatory cytokines in unstimulated peripheral blood mononuclear cells, have been detected in IBS patients.24–30 The proximity of mast cells to mucosal nerves in colonic mucosa is associated with abdominal pain severity in IBS,31,32 and gut enterochromaffin cell numbers and secretory function can be modulated by CD4 T cells through the production of soluble mediators and cytokines.33 Immune activation may thus play an important role in IBS in genetically predisposed individuals. Our data are consistent with the hypothesis that TNFSF15 polymorphisms impacting TL1A function and/or expression may modulate inflammatory and neuroimmune responses persisting after infection, dietary triggers, or stress and thus influence individual susceptibility to IBS.
The rs4263839 G allele predisposing to CD is the same associated with increased IBS risk in our study, and similar genetic mechanism(s) may thus predispose to both conditions. Whether the rs4263839 polymorphism has direct functional effects had not been addressed previously and, although our in-vitro experiments did not support this hypothesis, we demonstrated a correlation between genotype at this SNP and TNFSF15 mRNA levels in peripheral blood leucocytes and rectal mucosal tissue. This is consistent with findings from previous studies, in which extended TNFSF15 haplotypes (spanning the entire coding region) have been shown to influence TNFSF15 protein (TL1A) or mRNA expression in, respectively, monocytes and stimulated T lymphocytes.17,34 Very strong LD exists throughout the entire TNFSF15 coding region (figure 4), and it is therefore conceivable that the CD/IBS risk allele rs4263839 G studied here only represents a marker for other, true causative variants of functional relevance. At least one functional SNP in the TNFSF15 promoter has recently been identified (rs6478109), which is part of the CD risk haplotype and whose risk allele G induces increased TNFSF15 expression in in-vitro experiments.17 Based on additional genotype data available for our study populations, rs4263839 G appears to be a perfect proxy for the functional allele rs6478109 G, thus providing support to the hypothesis that the latter may be a true causative variant also in IBS. Given the strong LD, and the presence of several other polymorphisms in the region, further analyses in much larger cohorts, and functional studies of several TNFSF15 SNPs may be needed to address this issue conclusively.
The finding that the TNFSF15 polymorphism exerts the strongest effect on IBS risk in the constipation-predominant subtype is intriguing. Immunological alterations and inflammatory responses may play a more pronounced role in post-infectious IBS (typically part of the IBS diarrhoea subtype). Unfortunately, the TNFSF15 locus was not tested in a recent elegant study of genetic risk factors of post-infectious IBS.6 In IBS constipation patients, reduced plasma levels have been reported for the neurotransmitter serotonin,35 which regulates gut motility and is synthesised from the amino acid tryptophan. Interferon-g, also produced by CD4 T cells upon TL1A-DR3 signalling, induces tryptophan conversion to kynurenine, and this can limit the amount of substrate available for serotonin synthesis.36 Therefore, although specific subtypes of disease were not investigated, it is noteworthy that increased kynurenine concentrations have been detected in IBS, and that interferon-γ levels positively correlate with kynurenine:tryptophan ratios in IBS patients but not in controls.37 Moreover, neuropeptides involved in the control of gut motility (such as substance P and neuropeptide Y) can also modulate inflammatory responses, and receptors for these peptides are expressed on immune cells and intestinal epithelial cells upon bacterial or immune stimulation.38,39 The intracerebroventricular administration of neuropeptide S inhibits distal colonic transit in mice,40 and we have shown that polymorphisms in the corresponding gene (NPSR1) impact both IBD risk and colonic transit in IBS.7,41 A key role in inflammation and the differentiation of T cells has also been demonstrated for the vasoactive intestinal peptide, which inhibits gastrointestinal contraction.42 It is therefore conceivable that persistent immune activation might affect gut motility leading to altered colonic function including constipation. Both the molecular mechanisms of symptom generation and the immunological and inflammatory profile of different IBS patient subgroups require better characterisation to explain the observed associations.
In summary, we have tested CD risk loci for their potential relevance in IBS, and have identified an association with TNFSF15 that may be mediated by genetic effects on gene expression. This finding supports a role of immune activation in IBS, and further investigations are warranted to reveal the precise mechanisms by which TNFSF15 and its polymorphisms contribute to IBS pathogenesis and affect disease risk.
Supplementary Material
Significance of this study.
What is already known about this subject?
▶ The aetiopathogenesis of irritable bowel syndrome (IBS) is poorly understood.
▶ Defects in the maintenance of the intestinal epithelial barrier and in the regulation of inflammation and immune interactions with bacteria in the gut are emerging as candidates playing an important role in IBS susceptibility.
▶ The existence of genetic predisposition is supported by the observation of familial clustering and higher incidence in monozygotic twins.
What are the new findings?
▶ Genetic variants known to affect the risk of inflammatory bowel diseases such as Crohn's disease may also be relevant to IBS pathogenesis.
▶ The risk of IBS and the constipation-predominant subtype is influenced by polymorphism in the TNFSF15 gene, which is involved in the regulation of immune and inflammatory responses.
▶ Genetic alterations of immune function(s) may contribute to IBS pathogenesis.
How might it impact on clinical practice in the foreseeable future?
▶ This information may help clinicians to identify individuals at risk in the future.
▶ The identification of key genetic and molecular players in IBS might ultimately lead to improved prognostic and therapeutic strategies.
Acknowledgements
The authors wish to thank all the study participants. The excellent technical assistance of Elisabeth Dungner (Karolinska Institutet) is greatly acknowledged.
Funding Supported by grants from the Swedish Research Council and the Prof Nanna Svartz Fond to MD. MC's research in IBS is supported by NIH grants R01-DK079866 and 1RC1-DK086182.
Footnotes
Competing interests None.
Patient consent Obtained.
Ethics approval The study was approved by the responsible local ethics committees at Swedish clinics and the Mayo Clinic Institutional Review Board.
Provenance and peer review Not commissioned; externally peer reviewed.
REFERENCES
- 1.Longstreth GF, Thompson WG, Chey WD, et al. Functional bowel disorders. Gastroenterology. 2006;130:1480–91. doi: 10.1053/j.gastro.2005.11.061. [DOI] [PubMed] [Google Scholar]
- 2.Camilleri M. Mechanisms in IBS: something old, something new, something borrowed. Neurogastroenterol Motil. 2005;17:311–16. doi: 10.1111/j.1365-2982.2004.00632.x. [DOI] [PubMed] [Google Scholar]
- 3.Ohman L, Simren M. Pathogenesis of IBS: role of inflammation, immunity and neuroimmune interactions. Nat Rev Gastroenterol Hepatol. 2010;7:163–73. doi: 10.1038/nrgastro.2010.4. [DOI] [PubMed] [Google Scholar]
- 4.Collins SM, Denou E, Verdu EF, et al. The putative role of the intestinal microbiota in the irritable bowel syndrome. Dig Liver Dis. 2009;41:850–3. doi: 10.1016/j.dld.2009.07.023. [DOI] [PubMed] [Google Scholar]
- 5.Saito YA, Mitra N, Mayer EA. Genetic approaches to functional gastrointestinal disorders. Gastroenterology. 2010;138:1276–85. doi: 10.1053/j.gastro.2010.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Villani AC, Lemire M, Thabane M, et al. Genetic risk factors for post-infectious irritable bowel syndrome following a waterborne outbreak of gastroenteritis. Gastroenterology. 2010;138:1502–13. doi: 10.1053/j.gastro.2009.12.049. [DOI] [PubMed] [Google Scholar]
- 7.Camilleri M, Carlson P, Zinsmeister AR, et al. Neuropeptide S receptor induces neuropeptide expression and associates with intermediate phenotypes of functional gastrointestinal disorders. Gastroenterology. 2010;138:98–107. e4. doi: 10.1053/j.gastro.2009.08.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009;361:2066–78. doi: 10.1056/NEJMra0804647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barrett JC, Hansoul S, Nicolae DL, et al. Genome-wide association defines more than 30 distinct susceptibility loci for Crohn's disease. Nat Genet. 2008;40:955–62. doi: 10.1038/NG.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aro P, Ronkainen J, Storskrubb T, et al. Valid symptom reporting at upper endoscopy in a random sample of the Swedish adult general population: the Kalixanda study. Scand J Gastroenterol. 2004;39:1280–8. doi: 10.1080/00365520410008141. [DOI] [PubMed] [Google Scholar]
- 11.Aro P, Talley NJ, Ronkainen J, et al. Anxiety is associated with uninvestigated and functional dyspepsia (Rome III criteria) in a Swedish population-based study. Gastroenterology. 2009;137:94–100. doi: 10.1053/j.gastro.2009.03.039. [DOI] [PubMed] [Google Scholar]
- 12.Ronkainen J, Talley NJ, Aro P, et al. Prevalence of oesophageal eosinophils and eosinophilic oesophagitis in adults: the population-based Kalixanda study. Gut. 2007;56:615–20. doi: 10.1136/gut.2006.107714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Truedsson M, Carlson J, Simren M, et al. Polymorphism in the oxytocin promoter region in patients with lactase non-persistence is not related to symptoms. BMC Gastroenterol. 2009;9:90. doi: 10.1186/1471-230X-9-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Torkvist L, Halfvarson J, Ong RT, et al. Analysis of 39 Crohn's disease risk loci in Swedish inflammatory bowel disease patients. Inflamm Bowel Dis. 2010;16:907–9. doi: 10.1002/ibd.21105. [DOI] [PubMed] [Google Scholar]
- 15.McGovern DP, Gardet A, Torkvist L, et al. Genome-wide association identifies multiple ulcerative colitis susceptibility loci. Nat Genet. 2010;42:332–7. doi: 10.1038/ng.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Camilleri M, Carlson P, Zinsmeister AR, et al. Mitochondrial DNA and gastrointestinal motor and sensory functions in health and functional gastrointestinal disorders. Am J Physiol Gastrointest Liver Physiol. 2009;296:G510–16. doi: 10.1152/ajpgi.90650.2008. [DOI] [PubMed] [Google Scholar]
- 17.Kakuta Y, Ueki N, Kinouchi Y, et al. TNFSF15 transcripts from risk haplotype for Crohn's disease are overexpressed in stimulated T cells. Hum Mol Genet. 2009;18:1089–98. doi: 10.1093/hmg/ddp005. [DOI] [PubMed] [Google Scholar]
- 18.Zinovieva E, Bourgain C, Kadi A, et al. Comprehensive linkage and association analyses identify haplotype, near to the TNFSF15 gene, significantly associated with spondyloarthritis. PLoS Genet. 2009;5:e1000528. doi: 10.1371/journal.pgen.1000528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang FR, Huang W, Chen SM, et al. Genomewide association study of leprosy. N Engl J Med. 2009;361:2609–18. doi: 10.1056/NEJMoa0903753. [DOI] [PubMed] [Google Scholar]
- 20.Croft M. The role of TNF superfamily members in T-cell function and diseases. Nat Rev Immunol. 2009;9:271–85. doi: 10.1038/nri2526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shih DQ, Kwan LY, Chavez V, et al. Microbial induction of inflammatory bowel disease associated gene TL1A (TNFSF15) in antigen presenting cells. Eur J Immunol. 2009;39:3239–50. doi: 10.1002/eji.200839087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pappu BP, Borodovsky A, Zheng TS, et al. TL1A-DR3 interaction regulates Th17 cell function and Th17-mediated autoimmune disease. J Exp Med. 2008;205:1049–62. doi: 10.1084/jem.20071364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Meylan F, Davidson TS, Kahle E, et al. The TNF-family receptor DR3 is essential for diverse T cell-mediated inflammatory diseases. Immunity. 2008;29:79–89. doi: 10.1016/j.immuni.2008.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chadwick VS, Chen W, Shu D, et al. Activation of the mucosal immune system in irritable bowel syndrome. Gastroenterology. 2002;122:1778–83. doi: 10.1053/gast.2002.33579. [DOI] [PubMed] [Google Scholar]
- 25.Cremon C, Gargano L, Morselli-Labate AM, et al. Mucosal immune activation in irritable bowel syndrome: gender-dependence and association with digestive symptoms. Am J Gastroenterol. 2009;104:392–400. doi: 10.1038/ajg.2008.94. [DOI] [PubMed] [Google Scholar]
- 26.Liebregts T, Adam B, Bredack C, et al. Immune activation in patients with irritable bowel syndrome. Gastroenterology. 2007;132:913–20. doi: 10.1053/j.gastro.2007.01.046. [DOI] [PubMed] [Google Scholar]
- 27.O'Sullivan M, Clayton N, Breslin NP, et al. Increased mast cells in the irritable bowel syndrome. Neurogastroenterol Motil. 2000;12:449–57. doi: 10.1046/j.1365-2982.2000.00221.x. [DOI] [PubMed] [Google Scholar]
- 28.Ohman L, Isaksson S, Lindmark AC, et al. T-cell activation in patients with irritable bowel syndrome. Am J Gastroenterol. 2009;104:1205–12. doi: 10.1038/ajg.2009.116. [DOI] [PubMed] [Google Scholar]
- 29.Spiller RC, Jenkins D, Thornley JP, et al. Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post-dysenteric irritable bowel syndrome. Gut. 2000;47:804–11. doi: 10.1136/gut.47.6.804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tornblom H, Lindberg G, Nyberg B, et al. Full-thickness biopsy of the jejunum reveals inflammation and enteric neuropathy in irritable bowel syndrome. Gastroenterology. 2002;123:1972–9. doi: 10.1053/gast.2002.37059. [DOI] [PubMed] [Google Scholar]
- 31.Barbara G, Stanghellini V, De Giorgio R, et al. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology. 2004;126:693–702. doi: 10.1053/j.gastro.2003.11.055. [DOI] [PubMed] [Google Scholar]
- 32.Barbara G, Wang B, Stanghellini V, et al. Mast cell-dependent excitation of visceralnociceptive sensory neurons in irritable bowel syndrome. Gastroenterology. 2007;132:26–37. doi: 10.1053/j.gastro.2006.11.039. [DOI] [PubMed] [Google Scholar]
- 33.Wang H, Steeds J, Motomura Y, et al. CD4+ T cell-mediated immunological control of enterochromaffin cell hyperplasia and 5-hydroxytryptamine production in enteric infection. Gut. 2007;56:949–57. doi: 10.1136/gut.2006.103226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Michelsen KS, Thomas LS, Taylor KD, et al. IBD-associated TL1A gene (TNFSF15) haplotypes determine increased expression of TL1A protein. PLoS One. 2009;4:e4719. doi: 10.1371/journal.pone.0004719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Camilleri M. Serotonin in the gastrointestinal tract. Curr Opin Endocrinol Diabetes Obes. 2009;16:53–9. doi: 10.1097/med.0b013e32831e9c8e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Heyes MP, Saito K, Crowley JS, et al. Quinolinic acid and kynurenine pathway metabolism in inflammatory and non-inflammatory neurological disease. Brain. 1992;115:1249–73. doi: 10.1093/brain/115.5.1249. [DOI] [PubMed] [Google Scholar]
- 37.Fitzgerald P, Cassidy Eugene M, Clarke G, et al. Tryptophan catabolism in females with irritable bowel syndrome: relationship to interferon-gamma, severity of symptoms and psychiatric co-morbidity. Neurogastroenterol Motil. 2008;20:1291–7. doi: 10.1111/j.1365-2982.2008.01195.x. [DOI] [PubMed] [Google Scholar]
- 38.Koon HW, Pothoulakis C. Immunomodulatory properties of substance P: the gastrointestinal system as a model. Ann NY Acad Sci. 2006;1088:23–40. doi: 10.1196/annals.1366.024. [DOI] [PubMed] [Google Scholar]
- 39.Vona-Davis LC, McFadden DW. NPY family of hormones: clinical relevance and potential use in gastrointestinal disease. Curr Top Med Chem. 2007;7:1710–20. doi: 10.2174/156802607782340966. [DOI] [PubMed] [Google Scholar]
- 40.Han RW, Chang M, Peng YL, et al. Central neuropeptide S inhibits distal colonic transit through activation of central neuropeptide S receptor in mice. Peptides. 2009;30:1313–17. doi: 10.1016/j.peptides.2009.03.012. [DOI] [PubMed] [Google Scholar]
- 41.D'Amato M, Bruce S, Bresso F, et al. Neuropeptide s receptor 1 gene polymorphism is associated with susceptibility to inflammatory bowel disease. Gastroenterology. 2007;133:808–17. doi: 10.1053/j.gastro.2007.06.012. [DOI] [PubMed] [Google Scholar]
- 42.Yadav M, Rosenbaum J, Goetzl EJ. Cutting edge: vasoactive intestinal peptide (VIP) induces differentiation of Th17 cells with a distinctive cytokine profile. J Immunol. 2008;180:2772–6. doi: 10.4049/jimmunol.180.5.2772. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.