Summary
Wnt signaling is a crucial aspect of the intestinal stem cell niche required for crypt cell proliferation and differentiation. Paneth cells or subepithelial myofibroblasts are leading candidate sources of the required Wnt ligands, but this has not been tested in vivo. To abolish Wnt-ligand secretion, we used Porcupine (Porcn) conditional-null mice crossed to strains expressing inducible Cre recombinase in the epithelium, including Paneth cells (Villin-CreERT2); in smooth muscle, including subepithelial myofibroblasts (Myh11-CreERT2); and simultaneously in both compartments. Elimination of Wnt secretion from any of these compartments did not disrupt tissue morphology, cell proliferation, differentiation, or Wnt pathway activity. Thus, Wnt-ligand secretion from these cell populations is dispensable for intestinal homeostasis, revealing that a minor cell type or significant and unexpected redundancy is responsible for physiologic Wnt signaling in vivo.
Graphical Abstract

Highlights
-
•
Porcn knockout in candidate intestinal niche cells eliminates Wnt secretion in vivo
-
•
Wnts from Paneth cells or intestinal subepithelial myofibroblasts are dispensable
-
•
Physiologic intestinal Wnt signaling is more complex than current views suggest
Wnt signaling is crucial in the intestinal stem cell niche, but the cell types producing essential Wnt ligands are unknown. Using Porcn conditional-null mice, Shivdasani and colleagues eliminated Wnt ligand secretion from intestinal epithelium, including Paneth cells; smooth muscle, including subepithelial myofibroblasts; and in both compartments. They found no defects in intestinal homeostasis or Wnt signaling, revealing unanticipated niche redundancy or complexity.
Introduction
Intestinal crypts house self-renewing stem cells and transit-amplifying progenitors that depend on Wnt signaling. Expression of endogenous pathway antagonists such as Dickkopf-1 reduces Wnt signaling, arrests stem and progenitor cell proliferation, and impairs secretory cell differentiation (Kuhnert et al., 2004; Pinto et al., 2003). Additionally, although it is unclear exactly when Wnt signaling begins in the developing intestine (Kim et al., 2007), mice lacking Tcf4, a transcriptional effector of the Wnt pathway, show marked epithelial defects (Korinek et al., 1998; van Es et al., 2012). Conversely, constitutive Wnt activity drives excessive cell replication and tumors, including human colorectal cancer (Korinek et al., 1997; Morin et al., 1997). Intestinal stem cells (ISCs) located at the base of mouse small-intestine crypts express the Wnt-responsive gene Lgr5 (Barker et al., 2007), which encodes a coreceptor for Wnt-agonist R-spondins (Carmon et al., 2011; de Lau et al., 2011; Glinka et al., 2011) and are the origin for epithelial tumors spurred by constitutive Wnt activity (Barker et al., 2009). Although Wnt signaling is thus an imperative aspect of intestinal homeostasis, the bona fide physiologic Wnt source within the ISC niche in vivo is unknown.
Current hypotheses for this source draw on diverse observations. Paneth cells are a favored epithelial source because they reside in intimate contact with Lgr5+ ISCs (Barker et al., 2007; Cheng and Leblond, 1974) and their coculture with the latter improves intestinal organoid formation (Sato et al., 2011). Wnt3, Wnt6, and Wnt9b are highly expressed at the crypt base, and Wnt3, in particular, is present at higher levels in Paneth cells than in ISCs (Gregorieff et al., 2005; Sato et al., 2011). However, partial (Bastide et al., 2007; Garabedian et al., 1997; Mori-Akiyama et al., 2007; Shroyer et al., 2005) or complete, irreversible (Durand et al., 2012; Kim et al., 2012) Paneth cell ablation preserves mouse-crypt homeostasis in vivo, suggesting extraepithelial Wnt sources. A likely mesenchymal source is the subepithelial myofibroblasts (SEMFs) that envelop intestinal crypts (Powell et al., 2011) and support intestinal epithelial growth ex vivo and in tissue xenografts, likely through Wnt secretion (Lahar et al., 2011). SEMFs and other subepithelial cells express several Wnts, including Wnt2b, Wnt4, and Wnt5a (Gregorieff et al., 2005). Non-SEMF mesenchymal cells, such as nonmuscle fibroblasts, endothelial cells, neurons, or blood leukocytes, could also provide the required Wnts. Finally, epithelial and subepithelial cells may serve as redundant sources, much as renal development and function reveal complex, overlapping sites of Wnt production (Kispert et al., 1998; Park et al., 2007).
We used Porcupine (Porcn) conditional knockout mice to identify the source of intestinal Wnts. Porcn encodes an O-acyltransferase that palmitoylates all vertebrate Wnts at a conserved serine residue and is necessary for their secretion (Chen et al., 2009; Najdi et al., 2012; Proffitt and Virshup, 2012). PORCN mutations cause focal dermal hypoplasia in humans (Grzeschik et al., 2007; Wang et al., 2007), and the tissue defects in Porcn-null mice resemble those observed upon loss of single Wnts, including Wnt3, Wnt3a, Wnt5a, and Wnt7b (Barrott et al., 2011; Biechele et al., 2011, 2013; Liu et al., 2012). Thus, our approach circumvented the problem of redundancy that afflicts studies of single-Wnt gene disruption. We used Villin-CreERT2 mice (el Marjou et al., 2004) to delete Porcn in the intestinal epithelium and Myh11-CreERT2 mice (Wirth et al., 2008) for deletion in subepithelial cells. Loss of Porcn from all epithelial or smooth-muscle (including SEMF) cells, alone or in combination, produced none of the defects in crypt cell proliferation, differentiation, or Wnt target-gene expression expected from Wnt deficiency. This rigorous genetic study therefore points to some other minor cell type as a source of Wnt ligands in the mammalian intestine.
Results and Discussion
To test if the Wnt ligand(s) required for intestinal homeostasis originate in epithelial (including Paneth) cells, we crossed Villin-CreERT2 and PorcnFl/Y mice (Figure 1A; Barrott et al., 2011; el Marjou et al., 2004). LoxP sites flank exons 2 and 3 in the PorcnFl allele, poised for Cre recombinase-mediated deletion of the first three transmembrane domains to produce a null allele. As Porcn is X-linked, hemizygote males carry a single null allele. Although Villin-Cre is active in Paneth cells (Kim et al., 2012), we examined PorcnFl/Y;Villin-CreERT2 (PorcnE-Del for epithelium-deleted) males at least 8 weeks after tamoxifen (TAM) exposure to ensure that Paneth cells were derived from Porcn-null ISCs. PCR on genomic DNA and quantitative RT-PCR (qRT-PCR) on mRNA isolated from the small intestine epithelium verified efficient Porcn deletion (Figure 1B) and loss of Porcn transcripts (Figure 1C; note logarithmic scale). PorcnE-Del mice thrived without weight loss or morbidity (Figure S1A available online), and tissue morphology and crypt cell replication were intact. Both Paneth cells and goblet cells, which require Wnt signaling to differentiate (Pinto et al., 2003; van Es et al., 2005), were undisturbed (Figures 1D, S1B, and S1C). Nuclear β-catenin was present in crypt base cells and the levels of well-characterized Wnt target mRNAs, including Axin2, Myc, Cyclin D1, Cd44, Sox9, and Lgr5, were equal in PorcnE−Del and wild-type small-intestine crypt epithelium (Figure 1E; Table S1). These data reveal that intestinal epithelium is not an essential source of Wnt ligands in vivo.
Figure 1.

Porcn Deletion in Epithelial Cells Spares Intestinal Epithelial Morphology and Function
(A) Strategy for Porcn deletion by TAM-inducible Cre recombinase driven by epithelium-specific Villin promoter. ER, estrogen receptor.
(B) Genomic PCR of epithelium isolated from four PorcnE-Del intestines indicates complete recombination of the floxed allele (485 bp band) compared to two Porcn+/Y control mice (685 bp band from the wild-type allele). For reference, DNA is also shown from single PorcnDel/X (Δ/+; 485 bp and 685 bp products) and PorcnFl/Y;Cre− (762 bp band from the unrecombined floxed allele) mice. WT, wild-type.
(C) qRT-PCR analysis of Porcn mRNA in isolated PorcnE-Del intestinal crypts (N = 4) reveals reduction by orders of magnitude compared to controls (N = 2).
(D) Histology and immunohistochemistry (IHC) of PorcnE-Del (N = 5) and control (Porcn+/Y;Villin-CreERT2; N = 3) mice reveals no abnormalities. Left to right: hematoxylin and eosin (H&E) staining, Ki67 IHC, lysozyme IHC, and Alcian blue staining. High-magnification images are shown to the right of low-magnification views.
(E) Left, β-catenin IHC in control and PorcnE-Del mice shows nuclear-staining crypt-base cells (arrows). Right, qRT-PCR analysis of Wnt target mRNAs in isolated control (N = 2) and PorcnE-Del (N = 4) crypt epithelium demonstrates unperturbed Wnt-pathway activity.
Bars represent mean ± SEM of biological replicates; all scale bars, 50 μm.
To determine if subepithelial cells, such as SEMFs, are a required physiologic Wnt source, we used Myh11-CreERT2 mice. Myh11-encoded smooth-muscle myosin heavy chain is expressed in much of the intestinal subepithelium (Wirth et al., 2008). To define the precise expression domain, we crossed Myh11-CreERT2 to Rosa26-lox-STOP-lox-YFP (Rosa26R) reporter mice and treated with TAM. Yellow fluorescent protein (YFP) expression was evident in circular and longitudinal muscle layers as well as in SEMF 5 days post-Cre induction, colocalizing with α-smooth muscle actin (Figures 2A and S2). Most subepithelial cells expressed YFP, but the lamina propria was not totally marked, indicating that Myh11-CreERT2 is expressed in most, but not all, subepithelial lineages. We then crossed Myh11-CreERT2 and PorcnFl/Y mice to ablate Wnt-ligand secretion specifically in SEMF and smooth muscle (Figure 2B), while preserving Porcn expression in the epithelium (Figure 2C). To estimate the mesenchymal cell fraction with recombined Porcn, we genotyped Porcn alleles in unfractionated PorcnFl/Y;Myh11-CreERT2 (PorcnM-Del) intestines, expecting no recombination in epithelial, serosal, endothelial, or blood cells, which lack Myh11 expression. In this light, a substantial contribution from recombined null Porcn DNA implied efficient recombination in most Myh11+ cells (Figure 2D), a point we demonstrate with greater confidence below (Figure 3B). Three weeks following Cre activation in PorcnM-Del males, we did not observe weight loss (Figure S1A), implying preserved intestinal function. Gross and microscopic intestinal anatomy were intact, and PorcnM-Del did not differ from control mice in the numbers of proliferating Ki67+ crypt cells; Paneth or other secretory cells (Figures 2E, S1B, and S1C); expression of nuclear β-catenin; or levels of Wnt target genes in crypt epithelium (Figure 2F). Thus, disruption of a subepithelial Myh11+ cell source of Wnts also did not perturb intestinal homeostasis.
Figure 2.
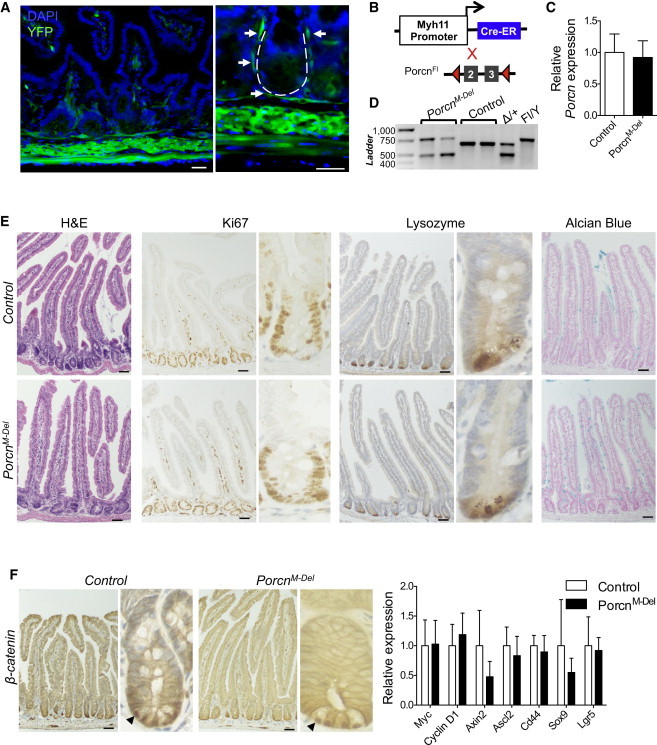
Porcn Loss in Intestinal Smooth Muscle Cells and SEMFs Does Not Adversely Affect Epithelial Morphology and Function
(A) Myh11-CreERT2;Rosa26R mice activate Cre recombinase in all muscle layers, including subepithelial myofibroblasts. The dotted line in the high-magnification image (right) outlines a crypt, and arrows point to the thin SEMF layer of YFP+ cells enveloping the crypt. The scale bars represent 30 μm. See also Figure S2.
(B) Strategy to induce muscle-cell-specific recombination in PorcnFl/Y;Myh11-CreERT2 mice.
(C) qRT-PCR analysis of isolated crypt epithelium shows no significant difference in Porcn mRNA expression in PorcnM-Del (N = 4) mice compared to control (Porcn+/Y;Myh11-CreERT2, N = 4) mice, p = 0.35.
(D) PCR from whole-intestine genomic DNA, revealing the expected proportion of recombined Porcn in smooth muscle, with residual unrecombined Porcn DNA contributed by epithelial and other cell types that lack Myh11-CreERT2 expression.
(E) Histology and immunostains on PorcnM-Del (N = 7) and control (Porcn+/Y;Myh11-CreERT2, N = 6) intestines reveal no abnormalities.
(F) Left, β-catenin IHC in control and PorcnM-Del mice shows nuclear staining in crypt-base cells (arrows). Right, qRT-PCR reveals intact Wnt target expression in crypts isolated from PorcnM-Del intestines, compared to controls (N = 4; statistics in Table S1).
Bars represent mean ± SEM of biological replicates; scale bars (E and F), 50 μm.
Figure 3.

Consequences of Simultaneous Loss of Porcn from Epithelial and Muscle Cells
(A) DNA from whole intestines in TAM-treated PorcnFl/Y;Villin-CreERT2;Myh11-CreERT2 mice shows significant recombination at the floxed Porcn allele, with a minimal contribution of unrecombined DNA from nonepithelial, nonmuscle cells.
(B) Left, qRT-PCR analysis of RNA from crypts of PorcnEM-Del mutants (N = 3) compared to controls (Porcn+/Y;Villin-CreERT2;Myh11-CreERT2; N = 3) shows total Porcn deficiency, p = 0.0013. Right, qRT-PCR of unfractionated PorcnM-Del (N = 2) intestines shows lower Porcn expression than controls (Porcn+/Y;Villin-CreERT2;Myh11-CreERT2 and Porcn+/Y;Myh11-CreERT2; N = 4), which is further reduced in PorcnEM-Del (N = 2) mice.
(C) Equivalent BrdU uptake in control and PorcnEM-Del intestinal crypts.
(D) Histology and immunostains from PorcnEM-Del and control intestines reveal intact morphology, cell proliferation, and differentiation (N = 3).
(E) Left, β-catenin IHC in control and PorcnEM-Del mice shows nuclear staining in crypt-base cells (arrows). Right, qRT-PCR analysis of control (N = 3) and PorcnEM-Del (N = 3) intestines indicates a statistically insignificant (Table S1) reduction in levels of Wnt target transcripts.
Bars in graphs represent mean ± SEM of biological replicates; all scale bars, 50 μm.
Intestinal organoid cultures from isolated Lgr5+ ISCs require supplemental WNT3A, which is thought to derive in vivo from Paneth cells (Sato et al., 2011). However, mouse intestines devoid of epithelial Wnt3 are normal and coculture of Lgr5+ ISCs with mesenchymal cells can substitute for WNT3A in organoid formation, suggesting redundant Wnt sources (Farin et al., 2012). To test this idea in vivo, we crossed PorcnFl/Y mice onto a compound Villin-CreERT2;Myh11-CreERT2 background and treated conditional null mice with TAM to force Porcn loss from both compartments (PorcnEM-Del). Three weeks post-Cre activation, we observed nearly complete recombination of the floxed mutant allele in unfractionated intestines (Figure 3A), producing total absence of Porcn mRNA in the epithelium (Figure 3B). Unfractionated intestines showed 40% less Porcn mRNA in PorcnM-Del mice and 85% less in compound PorcnEM-Del mice (Figure 3B). Together, these data reveal highly efficient Porcn depletion in both compartments, with the expected residual contribution from Villin−;Myh11− cells. Compound mutant mice showed no weight loss (Figure S1A) or clinical compromise, decrease in proliferation as assessed by Ki67 expression or bromodeoxyuridine (BrdU) uptake (Figures 3C and 3D), or defects in intestine morphology (Figures 3D, S1B, and S1C). Strong immunostaining for nuclear β-catenin provided direct evidence for sustained Wnt-signaling activity, and qRT-PCR analysis revealed subtle, statistically insignificant deficits in Wnt target transcripts (Figure 3E). Thus, combined loss of Porcn in the gut epithelium and the dominant subepithelial compartment, including SEMFs, preserves all measurable Wnt-dependent functions.
These findings conflict with previous results from forced expression of Wnt antagonists, which produced crypt atrophy, villus shortening, loss of Wnt target transcripts, and failure of secretory cell differentiation within 2–4 days (Kuhnert et al., 2004). There are several possible reasons for the absence of similar defects in PorcnDel intestines. First, Wnts necessary for intestinal homeostasis might circumvent the requirement for PORCN in secretion. This is unlikely because two independent studies reveal that PORCN seems necessary to palmitoylate all human Wnts; this in turn is required for Wnts to bind the carrier protein Wntless for secretory transport (Coombs et al., 2010; Liu et al., 2013) and to bind Frizzled receptors (Janda et al., 2012; Najdi et al., 2012). Second, the potent effect of Wnt antagonists on crypt functions might not reflect the native activity of Wnts per se but rather of R-spondin or another family of ligands. Available evidence, however, indicates that R-spondins act in conjunction with, and not separate from, Wnts (Niehrs, 2012). A third possibility is that Wnt reserves in the subepithelial basement membrane or elsewhere persisted for the duration of our experiments. Wnts can bind heparan sulfate proteoglycans present on the surface of Wnt-recipient cells, a proposed mechanism to prevent their diffusion and allow prolonged activity (Mikels and Nusse, 2006). However, even in the absence of information on intestinal Wnt concentrations, turnover, or reserves, secreted Wnts are unlikely to have persisted for the length of our studies. Porcn deletion was efficient, and we deliberately examined mice weeks after Cre activation, giving time for Wnt reserves to decay. Moreover, in a tissue that self-renews continually and responds quickly to injury or cell loss (Clevers, 2013), Wnts probably turn over rapidly to allow responsive homeostasis.
We therefore favor the final possibility: that a cell type that evaded Cre-mediated Porcn deletion is a sufficient source of essential, intestine-active Wnts. That cell is unlikely to reside in the epithelium, where Villin-CreERT2 mice drove efficient Cre expression, leaving no intact Porcn DNA in isolated epithelial cells. By contrast, although Myh11-expressing smooth-muscle cells or SEMF are not a required physiologic Wnt source, gut mesenchyme contains diverse additional cell types, including endothelium, nonmuscle fibroblasts, leukocytes, lymphocytes, and neurons (Powell et al., 2011). Endothelial and neuronal contributions cannot readily be assessed in mice because their deficiencies are lethal early (Dumont et al., 1994; Enomoto et al., 1998), and intestinal functions seem intact in Rag2-null mice (Shinkai et al., 1992), indicating that lymphocytes either provide no essential Wnts or act redundantly with other cells. This extent of specificity or redundancy in intestinal Wnt source(s) challenges the prevailing view and reveals unanticipated complexity in control of intestinal self-renewal.
Experimental Procedures
Mice
Villin-CreERT2, Myh11-CreERT2, Rosa26-lox-STOP-lox-YFP, and PorcnFl/Y mouse strains were described previously (Barrott et al., 2011; el Marjou et al., 2004; Srinivas et al., 2001; Wirth et al., 2008). To induce recombination of conditional alleles, mice were injected intraperitoneally with 1 to 2 mg TAM (Sigma) dissolved in sunflower oil (Sigma) on 5 consecutive days. All animal care and procedures were approved and monitored by an Institutional Animal Care and Use Committee.
Tissue Harvests
Small intestines were dissected and flushed with cold PBS. The proximal and distal 1/3 were fixed overnight in 4% paraformaldehyde and then washed in PBS and embedded in paraffin or frozen for immunohistochemical analysis. The first 1 cm of the middle 1/3 was reserved for whole intestine (unfractionated) DNA or mRNA analysis. The remainder was used for isolating epithelium by incubating in 5 mM EDTA, shaking by hand, and passage through 70 μm filters to separate crypts and villi.
DNA Isolation and Genotyping
Epithelium or whole intestine was agitated in SNET buffer (0.2% SDS, 0.2 M NaCl, 100 mM Tris [pH 8], and 5 mM EDTA) with 15 μg Proteinase K at 55°C for 2 hr (epithelium) or overnight (whole intestine), and DNA was isolated. Porcn gene recombination was assessed using primers specifically detecting the wild-type, floxed, and deleted allele, as described in Supplemental Information.
RNA Isolation and Gene-Expression Analysis
RNA was isolated using TRIzol reagent (Invitrogen) and RNeasy mini kits (QIAGEN) followed by treatment with DNaseI and reverse transcription using the Superscript III First-Strand Synthesis System (Invitrogen). Quantitative RT-PCR was performed using FastStart Universal SYBR Green Master (Roche) and gene-specific primers; data analysis is described in Supplemental Information.
Histochemistry and Immunohistochemistry
Some mice received 1 mg BrdU (Sigma) 1 hr before euthanasia. Five-micrometer paraffin tissue sections were stained with hematoxylin and eosin, Alcian blue, or specific antibodies (Abs) to Ki67, BrdU, Chromogranin A, β-catenin, cleaved caspase 3, and lysozyme. Ten-micrometer frozen tissue sections were stained with smooth muscle actin Ab, followed by incubation with Alexa546-conjugated anti-immunoglobulin G. See Supplemental Information for antibody sources and conditions.
Detailed descriptions of all materials and methods can be found in the Supplemental Information.
Acknowledgments
We thank Sylvie Robine for VillinCre-ERT2 mice. This work was supported by National Institute of Health grants R01DK081113 and R01DK082889 to R.A.S. and a National Science Foundation Graduate Research Fellowship to A.K.S.R.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial-No Derivative Works License, which permits non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
Supplemental Information
References
- Barker N., van Es J.H., Kuipers J., Kujala P., van den Born M., Cozijnsen M., Haegebarth A., Korving J., Begthel H., Peters P.J., Clevers H. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature. 2007;449:1003–1007. doi: 10.1038/nature06196. [DOI] [PubMed] [Google Scholar]
- Barker N., Ridgway R.A., van Es J.H., van de Wetering M., Begthel H., van den Born M., Danenberg E., Clarke A.R., Sansom O.J., Clevers H. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature. 2009;457:608–611. doi: 10.1038/nature07602. [DOI] [PubMed] [Google Scholar]
- Barrott J.J., Cash G.M., Smith A.P., Barrow J.R., Murtaugh L.C. Deletion of mouse Porcn blocks Wnt ligand secretion and reveals an ectodermal etiology of human focal dermal hypoplasia/Goltz syndrome. Proc. Natl. Acad. Sci. USA. 2011;108:12752–12757. doi: 10.1073/pnas.1006437108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bastide P., Darido C., Pannequin J., Kist R., Robine S., Marty-Double C., Bibeau F., Scherer G., Joubert D., Hollande F. Sox9 regulates cell proliferation and is required for Paneth cell differentiation in the intestinal epithelium. J. Cell Biol. 2007;178:635–648. doi: 10.1083/jcb.200704152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biechele S., Cox B.J., Rossant J. Porcupine homolog is required for canonical Wnt signaling and gastrulation in mouse embryos. Dev. Biol. 2011;355:275–285. doi: 10.1016/j.ydbio.2011.04.029. [DOI] [PubMed] [Google Scholar]
- Biechele S., Cockburn K., Lanner F., Cox B.J., Rossant J. Porcn-dependent Wnt signaling is not required prior to mouse gastrulation. Development. 2013;140:2961–2971. doi: 10.1242/dev.094458. [DOI] [PubMed] [Google Scholar]
- Carmon K.S., Gong X., Lin Q., Thomas A., Liu Q. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/beta-catenin signaling. Proc. Natl. Acad. Sci. USA. 2011;108:11452–11457. doi: 10.1073/pnas.1106083108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen B., Dodge M.E., Tang W., Lu J., Ma Z., Fan C.-W., Wei S., Hao W., Kilgore J., Williams N.S. Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer. Nat. Chem. Biol. 2009;5:100–107. doi: 10.1038/nchembio.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng H., Leblond C.P. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. V. Unitarian Theory of the origin of the four epithelial cell types. Am. J. Anat. 1974;141:537–561. doi: 10.1002/aja.1001410407. [DOI] [PubMed] [Google Scholar]
- Clevers H. The intestinal crypt, a prototype stem cell compartment. Cell. 2013;154:274–284. doi: 10.1016/j.cell.2013.07.004. [DOI] [PubMed] [Google Scholar]
- Coombs G.S., Yu J., Canning C.A., Veltri C.A., Covey T.M., Cheong J.K., Utomo V., Banerjee N., Zhang Z.H., Jadulco R.C. WLS-dependent secretion of WNT3A requires Ser209 acylation and vacuolar acidification. J. Cell Sci. 2010;123:3357–3367. doi: 10.1242/jcs.072132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Lau W., Barker N., Low T.Y., Koo B.-K., Li V.S.W., Teunissen H., Kujala P., Haegebarth A., Peters P.J., van de Wetering M. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature. 2011;476:293–297. doi: 10.1038/nature10337. [DOI] [PubMed] [Google Scholar]
- Dumont D.J., Gradwohl G., Fong G.H., Puri M.C., Gertsenstein M., Auerbach A., Breitman M.L. Dominant-negative and targeted null mutations in the endothelial receptor tyrosine kinase, tek, reveal a critical role in vasculogenesis of the embryo. Genes Dev. 1994;8:1897–1909. doi: 10.1101/gad.8.16.1897. [DOI] [PubMed] [Google Scholar]
- Durand A., Donahue B., Peignon G., Letourneur F., Cagnard N., Slomianny C., Perret C., Shroyer N.F., Romagnolo B. Functional intestinal stem cells after Paneth cell ablation induced by the loss of transcription factor Math1 (Atoh1) Proc. Natl. Acad. Sci. USA. 2012;109:8965–8970. doi: 10.1073/pnas.1201652109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- el Marjou F., Janssen K.-P., Chang B.H.-J., Li M., Hindie V., Chan L., Louvard D., Chambon P., Metzger D., Robine S. Tissue-specific and inducible Cre-mediated recombination in the gut epithelium. Genesis. 2004;39:186–193. doi: 10.1002/gene.20042. [DOI] [PubMed] [Google Scholar]
- Enomoto H., Araki T., Jackman A., Heuckeroth R.O., Snider W.D., Johnson E.M., Jr., Milbrandt J. GFR alpha1-deficient mice have deficits in the enteric nervous system and kidneys. Neuron. 1998;21:317–324. doi: 10.1016/s0896-6273(00)80541-3. [DOI] [PubMed] [Google Scholar]
- Farin H.F., Van Es J.H., Clevers H. Redundant sources of Wnt regulate intestinal stem cells and promote formation of Paneth cells. Gastroenterology. 2012;143:1518–1529.e7. doi: 10.1053/j.gastro.2012.08.031. [DOI] [PubMed] [Google Scholar]
- Garabedian E.M., Roberts L.J., McNevin M.S., Gordon J.I. Examining the role of Paneth cells in the small intestine by lineage ablation in transgenic mice. J. Biol. Chem. 1997;272:23729–23740. doi: 10.1074/jbc.272.38.23729. [DOI] [PubMed] [Google Scholar]
- Glinka A., Dolde C., Kirsch N., Huang Y.L., Kazanskaya O., Ingelfinger D., Boutros M., Cruciat C.M., Niehrs C. LGR4 and LGR5 are R-spondin receptors mediating Wnt/β-catenin and Wnt/PCP signalling. EMBO Rep. 2011;12:1055–1061. doi: 10.1038/embor.2011.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregorieff A., Pinto D., Begthel H., Destrée O., Kielman M., Clevers H. Expression pattern of Wnt signaling components in the adult intestine. Gastroenterology. 2005;129:626–638. doi: 10.1016/j.gastro.2005.06.007. [DOI] [PubMed] [Google Scholar]
- Grzeschik K.-H., Bornholdt D., Oeffner F., König A., del Carmen Boente M., Enders H., Fritz B., Hertl M., Grasshoff U., Höfling K. Deficiency of PORCN, a regulator of Wnt signaling, is associated with focal dermal hypoplasia. Nat. Genet. 2007;39:833–835. doi: 10.1038/ng2052. [DOI] [PubMed] [Google Scholar]
- Janda C.Y., Waghray D., Levin A.M., Thomas C., Garcia K.C. Structural basis of Wnt recognition by Frizzled. Science. 2012;337:59–64. doi: 10.1126/science.1222879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim B.M., Mao J., Taketo M.M., Shivdasani R.A. Phases of canonical Wnt signaling during the development of mouse intestinal epithelium. Gastroenterology. 2007;133:529–538. doi: 10.1053/j.gastro.2007.04.072. [DOI] [PubMed] [Google Scholar]
- Kim T.-H., Escudero S., Shivdasani R.A. Intact function of Lgr5 receptor-expressing intestinal stem cells in the absence of Paneth cells. Proc. Natl. Acad. Sci. USA. 2012;109:3932–3937. doi: 10.1073/pnas.1113890109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kispert A., Vainio S., McMahon A.P. Wnt-4 is a mesenchymal signal for epithelial transformation of metanephric mesenchyme in the developing kidney. Development. 1998;125:4225–4234. doi: 10.1242/dev.125.21.4225. [DOI] [PubMed] [Google Scholar]
- Korinek V., Barker N., Morin P.J., van Wichen D., de Weger R., Kinzler K.W., Vogelstein B., Clevers H. Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC-/- colon carcinoma. Science. 1997;275:1784–1787. doi: 10.1126/science.275.5307.1784. [DOI] [PubMed] [Google Scholar]
- Korinek V., Barker N., Moerer P., van Donselaar E., Huls G., Peters P.J., Clevers H. Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat. Genet. 1998;19:379–383. doi: 10.1038/1270. [DOI] [PubMed] [Google Scholar]
- Kuhnert F., Davis C.R., Wang H.-T., Chu P., Lee M., Yuan J., Nusse R., Kuo C.J. Essential requirement for Wnt signaling in proliferation of adult small intestine and colon revealed by adenoviral expression of Dickkopf-1. Proc. Natl. Acad. Sci. USA. 2004;101:266–271. doi: 10.1073/pnas.2536800100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lahar N., Lei N.Y., Wang J., Jabaji Z., Tung S.C., Joshi V., Lewis M., Stelzner M., Martín M.G., Dunn J.C.Y. Intestinal subepithelial myofibroblasts support in vitro and in vivo growth of human small intestinal epithelium. PLoS ONE. 2011;6:e26898. doi: 10.1371/journal.pone.0026898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu W., Shaver T.M., Balasa A., Ljungberg M.C., Wang X., Wen S., Nguyen H., Van den Veyver I.B. Deletion of Porcn in mice leads to multiple developmental defects and models human focal dermal hypoplasia (Goltz syndrome) PLoS ONE. 2012;7:e32331. doi: 10.1371/journal.pone.0032331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J., Pan S., Hsieh M.H., Ng N., Sun F., Wang T., Kasibhatla S., Schuller A.G., Li A.G., Cheng D. Targeting Wnt-driven cancer through the inhibition of Porcupine by LGK974. Proc. Natl. Acad. Sci. USA. 2013;110:20224–20229. doi: 10.1073/pnas.1314239110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikels A.J., Nusse R. Wnts as ligands: processing, secretion and reception. Oncogene. 2006;25:7461–7468. doi: 10.1038/sj.onc.1210053. [DOI] [PubMed] [Google Scholar]
- Mori-Akiyama Y., van den Born M., van Es J.H., Hamilton S.R., Adams H.P., Zhang J., Clevers H., de Crombrugghe B. SOX9 is required for the differentiation of paneth cells in the intestinal epithelium. Gastroenterology. 2007;133:539–546. doi: 10.1053/j.gastro.2007.05.020. [DOI] [PubMed] [Google Scholar]
- Morin P.J., Sparks A.B., Korinek V., Barker N., Clevers H., Vogelstein B., Kinzler K.W. Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science. 1997;275:1787–1790. doi: 10.1126/science.275.5307.1787. [DOI] [PubMed] [Google Scholar]
- Najdi R., Proffitt K., Sprowl S., Kaur S., Yu J., Covey T.M., Virshup D.M., Waterman M.L. A uniform human Wnt expression library reveals a shared secretory pathway and unique signaling activities. Differentiation. 2012;84:203–213. doi: 10.1016/j.diff.2012.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niehrs C. The complex world of WNT receptor signalling. Nat. Rev. Mol. Cell Biol. 2012;13:767–779. doi: 10.1038/nrm3470. [DOI] [PubMed] [Google Scholar]
- Park J.S., Valerius M.T., McMahon A.P. Wnt/beta-catenin signaling regulates nephron induction during mouse kidney development. Development. 2007;134:2533–2539. doi: 10.1242/dev.006155. [DOI] [PubMed] [Google Scholar]
- Pinto D., Gregorieff A., Begthel H., Clevers H. Canonical Wnt signals are essential for homeostasis of the intestinal epithelium. Genes Dev. 2003;17:1709–1713. doi: 10.1101/gad.267103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell D.W., Pinchuk I.V., Saada J.I., Chen X., Mifflin R.C. Mesenchymal cells of the intestinal lamina propria. Annu. Rev. Physiol. 2011;73:213–237. doi: 10.1146/annurev.physiol.70.113006.100646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proffitt K.D., Virshup D.M. Precise regulation of porcupine activity is required for physiological Wnt signaling. J. Biol. Chem. 2012;287:34167–34178. doi: 10.1074/jbc.M112.381970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato T., van Es J.H., Snippert H.J., Stange D.E., Vries R.G., van den Born M., Barker N., Shroyer N.F., van de Wetering M., Clevers H. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature. 2011;469:415–418. doi: 10.1038/nature09637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinkai Y., Rathbun G., Lam K.P., Oltz E.M., Stewart V., Mendelsohn M., Charron J., Datta M., Young F., Stall A.M. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell. 1992;68:855–867. doi: 10.1016/0092-8674(92)90029-c. [DOI] [PubMed] [Google Scholar]
- Shroyer N.F., Wallis D., Venken K.J.T., Bellen H.J., Zoghbi H.Y. Gfi1 functions downstream of Math1 to control intestinal secretory cell subtype allocation and differentiation. Genes Dev. 2005;19:2412–2417. doi: 10.1101/gad.1353905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivas S., Watanabe T., Lin C.S., William C.M., Tanabe Y., Jessell T.M., Costantini F. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 2001;1:4. doi: 10.1186/1471-213X-1-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Es J.H., Jay P., Gregorieff A., van Gijn M.E., Jonkheer S., Hatzis P., Thiele A., van den Born M., Begthel H., Brabletz T. Wnt signalling induces maturation of Paneth cells in intestinal crypts. Nat. Cell Biol. 2005;7:381–386. doi: 10.1038/ncb1240. [DOI] [PubMed] [Google Scholar]
- van Es J.H., Haegebarth A., Kujala P., Itzkovitz S., Koo B.K., Boj S.F., Korving J., van den Born M., van Oudenaarden A., Robine S., Clevers H. A critical role for the Wnt effector Tcf4 in adult intestinal homeostatic self-renewal. Mol. Cell. Biol. 2012;32:1918–1927. doi: 10.1128/MCB.06288-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X., Reid Sutton V., Omar Peraza-Llanes J., Yu Z., Rosetta R., Kou Y.-C., Eble T.N., Patel A., Thaller C., Fang P., Van den Veyver I.B. Mutations in X-linked PORCN, a putative regulator of Wnt signaling, cause focal dermal hypoplasia. Nat. Genet. 2007;39:836–838. doi: 10.1038/ng2057. [DOI] [PubMed] [Google Scholar]
- Wirth A., Benyó Z., Lukasova M., Leutgeb B., Wettschureck N., Gorbey S., Örsy P., Horváth B., Maser-Gluth C., Greiner E. G12-G13-LARG-mediated signaling in vascular smooth muscle is required for salt-induced hypertension. Nat. Med. 2008;14:64–68. doi: 10.1038/nm1666. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


